Abstract
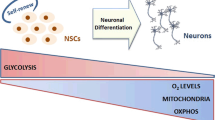
Energy metabolism in the adult brain consumes large quantities of glucose, but little is known to date regarding how glucose metabolism changes during neuronal differentiation, a process that is highly demanding energetically. We studied changes in glucose metabolism during neuronal differentiation of P19 mouse embryonal carcinoma cells, E14Tg2A embryonic stem cells as well as during brain development of BLC57 mice. In all these models, we find that neurogenesis is accompanied by a shift from oxidative to fermentative glucose metabolism. This shift is accompanied by both a decrease in mitochondrial enzymatic activities and mitochondrial uncoupling. In keeping with this finding, we also observe that differentiation does not require oxidative metabolism, as indicated by experiments demonstrating that the process is preserved in cells treated with the ATP synthase inhibitor oligomycin. Overall, we provide evidence that neuronal differentiation involves a shift from oxidative to fermentative metabolism, and that oxidative phosphorylation is not essential for this process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bain G, Ray WJ, Yao M, Gottlieb DI (1994) Bioessays 16:343–348
Bain G, Kitchens D, Yao M, Huettner JE, Gottlieb DI (1995) Dev Biol 168:342–357
Bernstein BW, Bamburg JR (2003) J Neurosci 23:1–6
Bittar PG, Charnay Y, Pellerin L, Bouras C, Magistretti PJ (1996) J Cereb Blood Flow Metab 16:1079–1089
Bolaños JP, García-Nogales P, Vega-Agapito V, Delgado-Esteban M, Cidad P, Almeida A (2004) J Neurochem 63:910–916
Budd SL, Nicholls DG (1996) J Neurochem 67:2282–2291
Castilho RF, Hansson O, Ward MW, Budd SL, Nicholls DG (1998) J Neurosci 18:10277–10286
Civitarese AE, Carling S, Heilbronn LK, Hulver MH, Ukropcova B, Deutsch WA, Smith SR, Ravussin E, CALERIE Pennington Team (2007) PLoS Med 4:e76
Cox DWG, Bachelard HS (1982) Brain Res 239:527–534
Dinsmore J (1996) Cell Transpl 5:131–141
Fiskum G, Craig SW, Decker GL, Lehninger AL (1980) Proc Natl Acad Sci USA 77:3430–3434
Fraichard A, Chassande O, Bilbaut G, Dehay C, Savatier P, Samarut J (1995) J Cell Sci 108:3181–3188
Hertz L (2007) J Cereb Blood Flow Metab 27:219–249
Hooper M, Hardy K, Handyside A, Hunter S, Monk M (1987) Nature 326:292–295
Hyder F, Patel AB, Gjedde A, Rothman DL, Behar KL, Shulman RG (2006) J Cereb Blood Flow Metab 26:865–877
Jones-Villeneuve EM, McBurney MW, Rogers KA, Kalnins VI (1982) J Cell Biol 94:253–262
Kasischke KA, Vishwasrao HD, Fisher PJ, Zipfel WR, Webb WW (2004) Science 305:99–103
Keller G (2005) Genes Dev 19:1129–1155
Kowaltowski AJ, Cosso RG, Campos CB, Fiskum G (2002) J Biol Chem 277:42802–42807
Magistretti PJ (2006) J Exp Biol 209(Pt 12):2304–2311
Martins AH, Resende RR, Majumder P, Faria M, Casarini DE, Tárnok A, Colli W, Pesquero JB, Ulrich H (2005) J Biol Chem 280:19576–19586
Mcllwain H, Bachelard HS (1985) In: Biochemistry and the central nervous system. Churchilll Livinstone, Edinburg. pp 54–83
Nicholls DG, Budd SL (1998) Biochim Biophys Acta 1366:97–112
Nicholls DG, Ferguson SJ (2002) In: Bioenergetics 3, Academic, London
Pauwels PJ, Opperdoes FR, Trouet A (1985) J Neurochem 44:143–148
Pellerin L, Magistretti PJ (1994) Proc Natl Acad Sci USA 91:10625–10629
Rehman J (2010) J Mol Med 88:981–986
Resende RR, Majumder P, Gomes KN, Britto LR, Ulrich H (2007) Neurosci 146:1169–1181
Resende RR, Alves AS, Britto LR, Ulrich H (2008a) Exp Cell Res 314:1429–1443
Resende RR, Britto LR, Ulrich H (2008b) Int J Dev Neurosci 26:763–777
Shepherd D, Garland PB (1966) Biochem Biophys Res Commun 22:89–93
Sibson NR, Dhankhar A, Mason GF, Rothman DL, Behar KL, Shulman RG (1998) Proc Natl Acad Sci USA 95:316–321
Soprano DR, Teets BW, Soprano KJ (2007) Vitam Horm 75:69–95
Tárnok A, Ulrich H (2001) Cytometry 43:175–181
Tsacopoulos M, Magistretti PJ (1996) J Neurosci 16:877–885
Ulrich H, Majumder P (2006) Cell Prolif 39:281–300
Véga C, Poitry-Yamate CL, Jirounek P, Tsacopoulos M, Coles JA (1998) J Neurochem 71:330–337
Wada H, Okada Y, Uzuo H, Nakamura H (1998) Brain Res 788:144–150
Wang VY, Zoghbi HY (2001) Nat Rev Neurosci 2:484–491
Warburg O (1956) Science 124:269–270
Yamane K, Yokono K, Okada Yasuhiro O (2000) J Neurosci Methods 103:163–171
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.

Supplementary Figure 1
Viability of undifferentiated (UND) P19 cells and cells following completion of neuronal differentiation (DIF). Cell viability was detected by staining for 1 min with 0.004% Trypan Blue as stated in Experimental Procedures. (JPEG 961 kb)

Supplementary Figure 2
O2 consumption of P19 cells differentiated in the absence of cytosine-arabinoside. Cells were incubated with Krebs buffer and O2 consumption was estimated as described in Experimental Procedures. Cells were differentiated in the absence of 5 μg/ml cytosine arabinoside which was used to eliminate proliferating glial cells. (JPEG 972 kb)
Rights and permissions
About this article
Cite this article
Fornazari, M., Nascimento, I.C., Nery, A.A. et al. Neuronal differentiation involves a shift from glucose oxidation to fermentation. J Bioenerg Biomembr 43, 531–539 (2011). https://doi.org/10.1007/s10863-011-9374-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-011-9374-3