Abstract
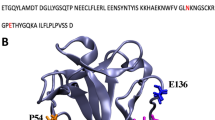
The binding site and backbone dynamics of a bioactive complex formed by the acidic fibroblast growth factor (FGF-1) and a specifically designed heparin hexasaccharide has been investigated by HSQC and relaxation NMR methods. The comparison of the relaxation data for the free and bound states has allowed showing that the complex is monomeric, and still induces mutagenesis, and that the protein backbone presents reduced motion in different timescale in its bound state, except in certain points that are involved in the interaction with the fibroblast growth factor receptor (FGFR).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abraham A. (1961) The Principles of Nuclear Magnetism. Clarendon Press, Oxford
Akke M., Brüschweiler R. and Palmer A.G., III (1993a) J. Am. Chem. Soc. 115:9832–9833
Akke M., Skelton N.J., Kordel J., Palmer A.G., III and Chazin W.J. (1993b). Biochemistry 32:9832–9844
Atwell S., Ultsch M., De Vos A.M., Wells J.A. (1997). Science 278:1125–1128
Angulo J., Ojeda R., de Paz J.L., Lucas R., Nieto P.M., Lozano R.M., Redondo-Horcajo M., Giménez-Gallego G. and Martín-Lomas M. (2004). Chem. Bio. Chem. 5:55–61
Arunkumar A.I., Srisailam S., Krishnaswamy T., Kumar S., Kathir K.M., Chi Y.-H., Wang H.-M., Chang G.-G., Chiu I.-M. and Yu C. (2002a). J. Biol. Chem. 277: 46424–46432
Arunkumar A.I., Kumar T.K., Kathir K.M., Srisailam S., Wang H.-M., Leena P.S., Chi Y.H., Chen H.C., Wu C.H., Wu R.T., Chang G.G., Chiu I.M. and Yu C. (2002b). Protein Sci. 11:1050–61
Capila I. and Lindhardt R.J. (2002). Angew. Chem. 114:428–451
Cavanagh J., Fairbrother W.J., Palmer A.G., III and Skelton N.J (1996). Protein NMR Spectroscopy: Principles and Practice. Academic Press, San Diego, CA
Cavanagh J. and M. Akke (2000). Nat. Struct. Biol.7:11–3
Chi Y., Kumar K.S., Chiu I.-M. and Yu C. (2000). J. Biol. Chem. 275:39444–50
Chi Y.-H., Kumar T., Kathir K., Lin D.-H., Zhu G., Chiu I.- M and Yu C. (2002). Biochemistry 41:15350–15359
Clackson T. and Wells J.A. (1995). Science 267:383–386
Cobas J.C. and Sardina F.J. (2003). Concepts Magn. Reson. 19A:80–96
Conrad H.E. (1998) Heparin-Binding Proteins. Academic Press, San Diego
DiGabriele A.D., Lax I., Chen D.I., Svahn C.M., Jaye M., Schlessinger J. and Hendrickson W.A. (1998). Nature 393: 812–817
Dosset P., Hus J.C., Blackledge M. and Marion D. (2000). J. Biomol. NMR 16:23–28
Dyson H.J. and Wright P.E. (2001). Methods Enzymol. 339:258–70
Epstein D.M., Benkovic S.J. and Wright P.E. (1995). Biochemistry 34:11037–48
Faham S., Hileman R.E., Fromm J.R., Lindhardt R.J. and Rees D.C. (1996). Science 271:1116–1120
Faham S., Lindhardt R.J. and Rees D.C. (1998) Curr. Opin Struct. Biol. 8:578–586
Farrow N.A., Muhandiram R., Singer A.U., Pascal S.M., Kay C.M., Gish G., Shoelson S.E., Pawson T., Forman-Kay J.D. and Kay L.E. (1994). Biochemistry 33:5984–6003
Farrow N.A., Zhang O., Szabo A., Torchia D.A. and Kay L.E. (1995). J. Biomol. NMR 6:153–62
Favier A., Brutscher B., Blackledge M., Galinier A., Deutscher J., Penin F. and Marion D. (2002). J. Mol. Biol. 317:131–44
Fayos R., Melacini G., Newlon M.G., Burns L., Scott J.D and Jennings P.A. (2003). J. Biol. Chem. 278:18581–7
Feher V.A. and Cavanagh J. (1999). Nature 400: 221–222
Forman-Kay J.D. (1999). Nat. Struct. Biol. 6:1086–1087
Giménez- Gallego G., Rodkey J., Bennett C., Rios-Candelore M., DiSalvo J. and Thomas K. (1985). Science 230:1385–1388
Giménez-Gallego G. and Cuevas P. (1994). Neurolog. Res. 16:313–316
Guignard L., Padilla A., Mispelter J., Yang Y.S., Stern M.H., Lhoste J.M. and Roumestand C. (2000). J. Biomol. NMR 17: 215–230
Harmer N.J., Pellegrini L., Chirgadze D., Fernández Recio J. and Blundell T.L. (2004a). Biochemistry 43:629–640
Harmer N.J., Ilag L.L., Mulloy B., Pellegrini L., Robinson C.V., and Blundell T.L. (2004b). J. Mol. Biol. 339:821–834
Hiyama Y., Niu C.-H., Silverton J.V., Bavoso A. and Torchia D.A. (1988). J. Am. Chem. Soc. 110:2378–2383
Itoh N. and Ornitz D.M. (2004). Trend. Gen. 20:563–569
Korzhnev D.M., Billeter M., Arseniev A.S. and Orekhov V.Y. (2001). Prog. Nucl. Magn. Reson. Spectrosc. 38:197–266
Krishnan V.V., Sukumar M., Gierasch L.M. and Cosman M (2000). Biochemistry 39:9119–9129
Lefèvre J.-F., Dayie K.T., Peng J.W. and Wagner G. (1996). Biochemistry 35:2674–2686
Lipari G. and Szabo A. (1982). J. Am. Chem. Soc. 104:4546–4570
McLachlan A.D. (1979). J. Mol. Biol. 133:557–563
Mohammadi M., Olsen S.K., and Goetez R. (2005a). Curr. Opin. Struct. Biol. 15:506–516
Mohammadi M., Olsen S.K. and Ibrahimi O.A. (2005b). Cytok. Growth. Fact. Rev. 16:107–137
Morton A. and Matthews B.W. (1995). Biochemistry 34:8576–8588
Moy F., Seddon A., Campell E., Bohlen P., and Powers R. (1995). J. Biomol. NMR 6:245–254
Nicholson L.K., Kay L.E., Baldisseri D.M., Arango J., Young P.E., Bax A. and Torchia D.A. (1992). Biochemistry 31:5253–63
Nishimura T., Nakatake Y., Konishi M. and Itoh N. (2000). Biochim. Biophys. Acta 1492:203–206
Ogura K., Nagata K., Hatanaka H., Habuchi H., Kimata K., Tate S.-I., Ravera M., Jaye M., Schlessinger J. and Inagaki K. (1999). J. Biomol. NMR 13:11–24
Ornitz D., Yayon A., Flanagan J., Svahn C., Levi E. and Leder P. (1992). Mol. Cell. Biol. 12:240–247
Ornitz. D.M., Xu J., Colvin J.S., Mc Ewen D.G., Mac Arthur C.A., Coulier F., Gao G. and Goldfarb M. (1996). J. Biol. Chem. 271:15292–15297
Osborne M.J., Schnell J., Benkovic S.J., Dyson H.J. and Wright P.E. (2001). Biochemistry 40:9846–59
Palmer A.G. (1993). Curr. Opin. Biotechnol. 4:385–391
Pellegrini L., Burke D.F., von Delft F., Mulloy B. and Blundell T.L. (2000). Nature 407:1029–1034
Pellegrini L. (2001). Curr. Opin. Struct. Biol. 11:629–634
Peng J.W. and Wagner G. (1992a). J. Magn. Reson. 98:308–332
Peng J.W. and Wagner G. (1992b). Biochemistry 31:8571–8586
Peränen J., Rikkonen M., Hyvönen M. and Kääriänien L. (1996). Anal. Biochem. 236:371–373
Pineda-Lucena A., Jiménez M.A., Nieto J.L., Santoro J., Rico M. and Giménez-Gallego G. (1994). J. Mol. Biol. 242:81–98
Pineda-Lucena A., Jiménez M.A., Lozano R.M., Nieto J.L., Santoro J., Rico M. and Giménez-Gallego G. (1996) J. Mol. Biol. 264:162–78
Powers C.J., Mc Leskey S.W. and Wellstein A. (2000). Endocr. Relat. Cancer 7:65–97
Rapraeger A., Krufka A. and Olwin B. (1991). Science 252:1705–1708
Schlessinger J., Plotnikov A.N., Ibrahimi O.A., Eliseenkova O.A., Yeh B.K., Yayon A., Lindhardt R.J. and Mohammadi M. (2000). Mol. Cell. 6:743–750
Slichter C. (1990) Principles of Magnetic Resonance. New York, Springer Verlag
Spivak-Kroizman T., Lemmon M.A., Dikic I., Ladbury J.E., Pinchsi D., Huang J., Jaye M., Crumley G., Schlessinger J. and Lax I. (1994). Cell 79:1015–1024
Springer B.A., Pantoliano M.W., Barbera F.A., Gunyuzla P.L., Thompson L.D., Herblin W.F., Rosendeld S.A. and Book G.W. (1994). J. Biol. Chem. 269:26879–26884
Stauber D.J., Digabriele A.D. and Hendrickson W.A. (2000). Proc. Natl. Acad. Sci. USA 97:49–54
Stivers J.T., Abeygunawardana C., Mildvan A.S. (1996). Biochemistry 35:16036–16047
Stone M.J., Gupta S. and Snyder N. (2001). J. Am. Chem. Soc. 123:185–186
Venkataraman G., Shriver Z., Davis J.C. and Sasisekharan R. (1999). Proc. Natl. Acad. Sci. USA 96:1892–1897
Waksman G. and Herr A.B. (1998). Nat. Struct. Biol. 5:527–530
White K.E., Evans W.E., O’Riordan J.L., Speer M.C., Econs M.J., Lorenz-Depiereux B., Grabowski M., Meitinger T., Strom T.M. (2000). Nat. Genet. 26:345–348
Wu Z.L., Zhang L., Yabe T., Kuberan B., Beeler D.L., Love A. and Rosenberg R.D. (2003). J. Biol. Chem. 278:17121–17129
Yayon A., Klagsbrun M., Esko J.D., Leder P. and Ornitz D.M (1991). Cell 64: 841–848
Zhang P., Dayie K.T. and Wagner G. (1997). J. Mol. Biol. 272:443–55
Zhu X., Hsu B.T. and Rees D.C. (1993). Structure 1:27–34
Acknowledgements
We thank Mrs. Mercedes Zazo for her help during the expression and purification of labelled FGF-1. This work was supported by the Dirección General de Investigación Científica y Técnica (Grants BQU2000-1501-C02-01, BQU2002-0374, and BQU2003-03550-C03-01). We are indebted to Dr Dosset and Dr Guenneugues for kindly providing the programmes TENSOR2 and anal_roe, respectively. We also thank Comunidad de Madrid, Fundación Ramón Areces, and Fundación Francisco Cobos for fellowships to R.F., J.A., and R.O., respectively. NMR time from the Parc Cientific of Barcelona (Dr. M. Gairí), University of Santiago de Compostela, and CAI-NMR of Universidad Complutense is warmly thanked.
Author information
Authors and Affiliations
Corresponding author
Additional information
Angeles Canales-Mayordomo and Rosa Fayos have contributed equally to this research.
Rights and permissions
About this article
Cite this article
Canales-Mayordomo, A., Fayos, R., Angulo, J. et al. Backbone dynamics of a biologically active human FGF-1 monomer, complexed to a hexasaccharide heparin-analogue, by 15N NMR relaxation methods. J Biomol NMR 35, 225–239 (2006). https://doi.org/10.1007/s10858-006-9024-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10858-006-9024-y