Abstract
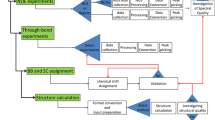
We present two new databases of NMR-derived distance and dihedral angle restraints: the Database Of Converted Restraints (DOCR) and the Filtered Restraints Database (FRED). These databases currently correspond to 545 proteins with NMR structures deposited in the Protein Databank (PDB). The criteria for inclusion were that these should be unique, monomeric proteins with author-provided experimental NMR data and coordinates available from the PDB capable of being parsed and prepared in a consistent manner. The Wattos program was used to parse the files, and the CcpNmr FormatConverter program was used to prepare them semi-automatically. New modules, including a new implementation of Aqua in the BioMagResBank (BMRB) software Wattos were used to analyze the sets of distance restraints (DRs) for inconsistencies, redundancies, NOE completeness, classification and violations with respect to the original coordinates. Restraints that could not be associated with a known nomenclature were flagged. The coordinates of hydrogen atoms were recalculated from the positions of heavy atoms to allow for a full restraint analysis. The DOCR database contains restraint and coordinate data that is made consistent with each other and with IUPAC conventions. The FRED database is based on the DOCR data but is filtered for use by test calculation protocols and longitudinal analyses and validations. These two databases are available from websites of the BMRB and the Macromolecular Structure Database (MSD) in various formats: NMR-STAR, CCPN XML, and in formats suitable for direct use in the software packages CNS and CYANA.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BMRB:
-
BioMagResBank
- CCPN:
-
Collaborative Computing Project for NMR
- DOCR:
-
Database Of Converted Restraints
- DR:
-
Distance Restraints
- EBI:
-
European Bioinformatics Institute
- FRED:
-
Filtered REstraints Database
- MSD:
-
Macromolecular Structure Database
- PDB:
-
Protein Data Bank
- RDC:
-
residual dipolar coupling
- s.d.:
-
standard deviation.
References
H.M. Berman J. Westbrook Z. Feng G. Gilliland T.N. Bhat H. Weissig I.N. Shindyalov P.E. Bourne (2000) Nucleic Acids Res. 28 235–242 Occurrence Handle10.1093/nar/28.1.235 Occurrence Handle10592235
BMRB NMR-STAR data dictionary (2004), http://www.bmrb.wisc.edu/dictionary/htmldocs/nmr_star/dictionary.html.
Brünger, A.T. (1996) in X-PLOR Manual (Version 4.0). Dep of Molecular Biophysics and Biochemistry, Yale University.
A.T. Brünger P.D. Adams G.M. Clore W.L. DeLano P. Gros R.W. Grosse-Kunstleve J.S. Jiang J. Kuszewski M. Nilges N.S. Pannu R.J. Read L.M. Rice T. Simonson G.L. Warren (1998) Acta Cryst. D54 905–921
J.F. Doreleijers S. Mading D. Maziuk K. Sojourner L. Yin J. Zhu J.L. Markley E.L. Ulrich (2003) J. Biomol. NMR 26 139–146 Occurrence Handle10.1023/A:1023514106644 Occurrence Handle12766409
J.F. Doreleijers M.L. Raves T. Rullmann R. Kaptein (1999a) J. Biomol. NMR 14 123–132 Occurrence Handle10.1023/A:1008335423527
J.F. Doreleijers J.A.C. Rullmann R. Kaptein (1998) J. Mol. Biol. 281 149–164 Occurrence Handle10.1006/jmbi.1998.1808 Occurrence Handle9680482
J.F. Doreleijers G. Vriend M.L. Raves R. Kaptein (1999b) Proteins 37 404–416 Occurrence Handle10.1002/(SICI)1097-0134(19991115)37:3<404::AID-PROT8>3.0.CO;2-2
R.H. Fogh J. Ionides E.L. Ulrich W. Boucher W. Vranken J. Linge M. Habeck W. Rieping T.N. Bhat J. Westbrook K. Henrick G. Gilliland H. Berman J.M. Thornton M. Nilges J.L. Markley E. Laue (2002) Nat. Struct. Biol. 9 416–418 Occurrence Handle10.1038/nsb0602-416 Occurrence Handle12032555
P. Güntert C. Mumenthaler K. Wüthrich (1997) J. Mol. Biol. 273 283–298 Occurrence Handle10.1006/jmbi.1997.1284 Occurrence Handle9367762
R.W.W. Hooft G. Vriend C. Sander E.E. Abola (1996) Nature 381 272 Occurrence Handle8692262
R.A. Laskowski (2001) Nucleic Acids Res. 29 221–222 Occurrence Handle10.1093/nar/29.1.221 Occurrence Handle11125097
R.A. Laskowski (2003) Methods Biochem. Anal. 44 273–303 Occurrence Handle12647391
R.A. Laskowski E.G. Hutchinson A.D. Michie A.C. Wallace M.L. Jones J.M. Thornton (1997) Trends. Biochem. Sci. 22 488–490 Occurrence Handle10.1016/S0968-0004(97)01140-7 Occurrence Handle9433130
R.A. Laskowski J.A.C. Rullmann M.W. MacArthur R. Kaptein J.M. Thornton (1996) J. Biomol. NMR 8 477–486 Occurrence Handle10.1007/BF00228148 Occurrence Handle9008363
J.P. Linge S.I. O’Donoghue M. Nilges (2001) Methods Enzymol. 339 71–90 Occurrence Handle11462826
J.L. Markley A. Bax Y. Arata C.W. Hilbers R. Kaptein B.D. Sykes P.E. Wright K. Wüthrich (1998) J. Biomol. NMR 12 1–23 Occurrence Handle10.1023/A:1008290618449 Occurrence Handle9729785
S.B. Nabuurs A.J. Nederveen W. Vranken J.F. Doreleijers A.M. Bonvin G.W. Vuister G. Vriend C.A. Spronk (2004a) Proteins 55 483–486 Occurrence Handle10.1002/prot.20118
S.B. Nabuurs C.A. Spronk G. Vriend G.W. Vuister (2004b) Concep. Magnetic Res. 22 90–105 Occurrence Handle10.1002/cmr.a.20016
Nederveen, A.J., Doreleijers, J.F., Vranken, WF., Miller, Z., Spronk, C.A.E.M., Nabuurs, S.B., Güntert, P., Livny, M., Markley, J.L., Nilges, M., Ulrich, E.L., Kaptein, R. and Bonvin, A.M.J.J. (2005) accepted.
M. Nilges (1993) Proteins 17 297–309 Occurrence Handle10.1002/prot.340170307 Occurrence Handle8272427
J.L. Sussman D. Lin J. Jiang N.O. Manning J. Prilusky O. Ritter E.E. Abola (1998) Acta. Cryst. D54 1078–1084
Vranken, WF., Boucher, W., Stevens, T., Fogh, R.H., Pajon, A., Llinás, M., Ulrich, E.L., Markley, J.L., Ionides, J. and Laue, E. (2005) accepted.
G. Vriend (1990) J. Mol. Graph. 8 52–56 Occurrence Handle10.1016/0263-7855(90)80070-V Occurrence Handle2268628
J. Westbrook Z. Feng K. Burkhardt H.M. Berman (2003) Methods Enzymol. 374 370–385 Occurrence Handle14696382
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Doreleijers, J., Nederveen, A., Vranken, W. et al. BioMagResBank databases DOCR and FRED containing converted and filtered sets of experimental NMR restraints and coordinates from over 500 protein PDB structures. J Biomol NMR 32, 1–12 (2005). https://doi.org/10.1007/s10858-005-2195-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10858-005-2195-0