Abstract
Nanocrystalline (NC) tin dioxide thin films have been synthesized via sol–gel spin coating method. The sol solution produces thin films that suffer from cracking at room temperature and at different annealing temperatures. Hence, the determination of the smallest value of glycerin is necessary to solve the cracking problem. Glycerin was added to the sol solutions with different volume ratios (0:1, 1:12, 1:8) at low temperature to enhance film porosity and to eliminate cracks. Temperature affects on the characteristics of NC SnO2 thin films, particularly particle size where the crystallization of SnO2 thin films obtained after annealing at 400 °C is a NC tetragonal rutile structure. With the increase of annealing temperature the crystallinity has enhanced, the crystallite size increased, and it was observed that both of the intensity and a blue shift of the A1g phonon peak increased at constant volume ratio of glycerin. Hence, these results indicated that (1:12) of glycerin volume ratio to sol solution volume ratio represents the optimum value for the fabrication of SnO2 thin films without cracks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Recently, the present studies on metal oxide semiconductors have been attracted a lot of attention in various applications, due to their wide range of physical and chemical properties. Among of metal oxides, tin dioxide (SnO2) is an important material for many applications especially for the detection of inflammable gases and for toxic gases. SnO2 has a tetragonal rutile structure and is an n-type semiconductor with a large band gap (3.6 eV) [1, 2].
Various techniques have been used to prepare SnO2 thin films such as sol–gel [3, 4], thermal evaporation [5], chemical vapor deposition [6], solvothermal synthesis [7], spray pyrolysis [8], and RF magnetron sputtering [9]. The sol–gel method has been selected to prepare SnO2 thin films because it has various advantages over other methods. It can be done at low reaction temperatures, easy processes, and is not costly [3, 10]. Cracks appear in thin films synthesized via the sol–gel method, which have adverse effects on the response for any device. Thus, cracks in thin films are undesirable [11]. The previous used glycerin during the preparation of the precursor solution to solve this problem [12]. However, other factors were concerned to enhance the quality of the thin films. For example temperature which is affected in particle size [13]. An amorphous structure appears for as-deposited SnO2 thin films, whereas a tetragonal rutile structure is obtained after annealing films to 400 °C for 30 min. Moreover, the crystallinity and grain size increase with increasing annealing temperature [14]. Furthermore, the electrical resistance of SnO2 thin films increases with decreasing film thickness [15].
The aim of this study is to fabricate high porosity and crack-free NC SnO2 thin films at low temperature using of low-cost method, and to find out the best conditions for enhancement of uniform NC SnO2 thin films grown via sol–gel spin coating method on p-Type (100) Si substrates. The experimental results show that both glycerin volume ratio and annealing temperature are most probably in charge of the development of structural properties of the grown SnO2.
2 Experimental procedures
Prior to deposition, Si substrates were cleaned using the Radio Corporation of America (RCA) method. Nanocrystalline SnO2 thin films were synthesized using the sol–gel spin coating method according to previous studies [3, 4]. The sol solution was synthesized by dissolving 0.1 M of tin chloride dehydrate (SnCl2·2H2O) into 70 mL of pure ethanol (C2H5OH). Different volumes of glycerin (C3H8O3) were then added to the sol solutions according to the following ratios (0:1, 1:12, 1:8) [12]. The resulted sol solutions placed in a closed flasks and stirred by a magnetic stirrers for 3 h and kept at 70 °C for 8 h. The process of preparing the sol solutions was completed for the remainder of the 24 h at room temperature. Thereafter, the solutions were spin coated on p-Type (100) Si substrates at a rotation speed of 900 rpm for the first 6 s and then for 3000 rpm for the succeeding 30 s to create film layers that are uniformly distributed. The as-deposited films were oven-dried at 100 °C for 10–12 min. Spin-coating and drying was repeated ten times for all films. After that the completed films were annealed at 400, 500, and 600 °C in air for 2 h.
In the present work, the crystal structure of the fabricated samples characterized by using high resolution XRD PANalytical X’ pert Pro MRD equipped with a Cu Kα radiation of (λ = 0.154060 nm). The morphologies were characterized using a FESEM (model Leo-Supra 50VP, Carl Zeiss,Germany) equipped with an energy-dispersive X-ray spectroscopy (EDX, Oxford instrument Analytical Ltd). Raman backscattering measurements were achieved at room temperature using Jobin–Yvon HR800UV.
3 Results and discussion
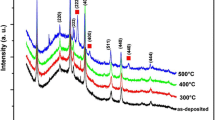
Figure 1 shows XRD patterns for the as-deposited and annealed SnO2 thin films without adding glycerin to sol solutions, which were grown using sol–gel spin coating technique on p-Type (100) Si substrates. The as-deposited films are amorphous and does not appear any diffraction peaks related to SnO2 [3, 14, 16]. All annealed thin films show different reflection peaks that match a very well with standard bulk SnO2 having a tetragonal rutile structure (JCPDS card No. 041-1445). Table 1 shows the comparison of the 2θ—values obtained from the measurement XRD patterns for the samples annealed at 600 °C. The diffraction peaks for the samples annealed at 400 °C which correspond to (110), (101), and (211) planes. It was observed that with increasing annealing temperature, the intensity of the diffraction peaks is becoming stronger and sharper [3, 14, 16], whereas the diffraction peaks obtained with high and sharp intensities that correspond to the (110), (101), (200), (211), (220), and (310) planes for films annealed at 600 °C.
The lattice parameters were calculated from peak positions of the three main peaks using Eq. (1) given by [17] as follows:
where d is the lattice plane spacing of a tetragonal rutile structure, and h, k, and l are the Miller.
Moreover, it was found that the lattice parameters at glycerin volume ratio 1:12, and it was very close to the standard value. While with increase of the volume ratio of glycerin to sol solution beyond (1:12) is lead to decrease the crystallinity. This result was attributed to the sharp decreasing in nanoparticles size, which causes the broadening of the diffraction peaks as shown from Fig. 2 [11, 18].
Figure 3 shows diffraction peaks with glycerin volume ratio (1:12), enhanced crystallinity and the diffraction peaks are became an intensive and more sharper. While increasing the volume ratio of glycerin to sol solution beyond (1:12) is lead to decrease the crystallinity. This result occurred due to the sharp decreasing in nanoparticles size, after increasing the volume ratios of glycerin, which causes the broadening of the diffraction peaks, as shown from Fig. 3 [11, 18].
The average crystallite size D for NC SnO2 thin films was calculated for the annealed samples from (110) the first and major diffraction peak using well-known Debye–Sherrer formula, [16]:
where D is the average crystallite size, β is the full width at half-maximum of the major peak, λ is the X-ray wavelength of the radiation, and θ is the diffraction angle.
It was found a significant increasing in crystallite size for films with increasing annealing temperature at constant glycerin volume ratio. The heat in the annealing process improves the crystallization of films, increases crystallite size, and reduces the amount of defects [14, 16]. While with increasing glycerin volume ratio beyond (1:12) all of the mentioned above decreased, as presented in Table 2.
However, the crystallite size of the films with glycerin volume ratio (1:8) shows a decrease in size as compared to films grown with glycerin volume (1:12) for the three annealing temperatures, indicating a reduction in crystallinity.
Low- and high magnification FESEM images showing the evolution of the morphology of SnO2 grown on Si substrates are shown in Figs. 4, 5, and 6. The NC SnO2 thin films were successfully fabricated via the sol–gel spin coating method. Figure 4 shows the surface morphologies of as-deposited and annealed thin films without adding glycerin, whereas the films are suffered from cracks. The surface morphology of as-deposited films, have a smooth surface which is consistent with the amorphous nature of SnO2 [3, 14, 16]. After annealing to 400 °C, small nanoparticles were observed, and the thin films were polycrystalline with uniform structures. The particles size increased and became more homogenous with increasing annealing temperature at 500 and 600 °C, respectively. These results occurred because of increasing annealing temperature are lead to improve the crystallinity and reduced defects for films [3, 14]. Figure 5 shows the surface morphologies of films when glycerin was added with a volume ratio of (1:12). The as-deposited SnO2 thin films were amorphous and the nanoparticles size increased with increasing annealing temperature. Agglomeration was observed after the film annealing at 600 °C. Figure 6 shows that with the addition of glycerin at a volume ratio (1:8), the nanoparticle size decreased at all annealing temperatures as compared to films with glycerin ratio (1:12) and no agglomerations were observed with increasing annealing temperature at 600 °C [11, 18].
These FESEM results correspond with the XRD measurements. The glycerin-free sol solution produces films that suffered from cracks for as-deposited and annealed at different annealing temperatures, as shown in Fig. 4. These cracks have a negative effects on the performance of any device [11, 12]. From Figs. 5, and 6, the problem of cracks was solved by adding a suitable volume of glycerin. Beyond glycerin volume ratio (1:12) there was a decrease in the nanoparticles size and reduction in crystallinity of the SnO2 thin films [11, 18].
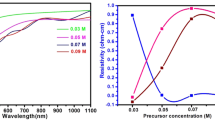
Raman backscattering geometry is a useful tool to verify phonon excitation in semiconductors. It can offer important information about impurity, defects, porosity, strain and stress in the lattice of materials. Raman spectroscopy measurements have been achieved. Figure 7 illustrates Raman spectra of NC SnO2 grown on Si substrates. The spectrum of the as-deposited SnO2 films, no Raman mode associated with SnO2 was observed because of the amorphous nature for SnO2 [19]. The major Raman peak at 520 cm−1 belongs to the Si (100) substrate. After the films were annealed the Raman peaks corresponding to the A1g phonon mode started to emerge, and the A1g phonon mode peaks are found at 611.96, 627.15, and 630.06 cm−1 for thin films annealed at 400, 500, and 600 °C, respectively. These values are less than the bulk value of 638 cm−1 for SnO2 [14]. The A1g phonon mode is affected by grain size and shifts to a higher wavenumber when the grain size is increased [1]. From Figs. 8, and 9 observed the wavenumber of the A1g phonon mode decreases with increasing glycerin volume ratio because of a decrease in nanoparticles size of the SnO2 thin films.
4 Conclusions
Nanocrystalline SnO2 thin films were successfully synthesized using a simple cost-effective via sol–gel spin coating method on p-Type (100) Si substrates. The sol solution produces thin films that suffer from cracking at room temperature and at different annealing temperatures. These cracks have negative effects on the films performance in various applications. This problem was solved by adding glycerin with a volume ratio (1:12) and (1:8). Adding glycerin concentration leads to increase the crystallite size of the SnO2 thin films, to enhance crystallinity and to reduce defects whenever the annealing temperature was increased from 400 to 600 °C. These obtained results are clearer in the (1:12) glycerin concentration SnO2 thin films. Moreover, we observed a blue shift for the A1g phonon peaks with increasing annealing temperature. Results draw a clear picture about glycerin effects and showed that (1:12) glycerin volume ratio is a good candidate for the fabrication of thin films without cracks.
References
A. Dieguez et al., The complete Raman spectrum of nanometric SnO2 particles. J. Appl. Phys. 90(3), 1550–1557 (2001)
M. Aziz, S.S. Abbas, W.R.W. Baharom, Size-controlled synthesis of SnO2 nanoparticles by sol–gel method. Mater. Lett. 91, 31–34 (2013)
J.S. Jeng, The influence of annealing atmosphere on the material properties of sol–gel derived SnO2: Sb films before and after annealing. Appl. Surf. Sci. 258(16), 5981–5986 (2012)
J. Liu, S. Gong, L. Quan, Z. Deng, H. Liu, D. Zhou, Influences of cooling rate on gas sensitive tin oxide thin films and a model of gradient distributed oxygen vacancies in SnO2 crystallites. Sens. Actuators B 145(2), 657–666 (2010)
S.H. Luo et al., Vacuum electron field emission from SnO2 nanowhiskers synthesized by thermal evaporation. Nanotechnology 15(11), 1424 (2004)
Y. Liu, E. Koep, M. Liu, A highly sensitive and fast-responding SnO2 sensor fabricated by combustion chemical vapor deposition. Chem. Mater. 17(15), 3997–4000 (2005)
X. Liu, J. Iqbal, Z. Wa, B. He, R. Yu, Structure and room-temperature ferromagnetism of Zn-doped SnO2 nanorods prepared by solvothermal method. J. Phys. Chem. C 114(11), 4790–4796 (2010)
S. Chacko, M.J. Bushiri, V.K. Vaidyan, Photoluminescence studies of spray pyrolytically grown nanostructured tin oxide semiconductor thin films on glass substrates. J. Phys. D Appl. Phys. 39(21), 4540 (2006)
M.A. Gubbins, V. Casey, S.B. Newcomb, Nanostructural characterisation of SnO2 thin films prepared by reactive rf magnetron sputtering of tin. Thin Solid Films 405(1), 270–275 (2002)
S. Gnanam, V. Rajendran, Luminescence properties of EG-assisted SnO2 nanoparticles by sol–gel process. Dig. J. Nanomater. Biostruct. 5(3), 699–704 (2010)
H. Köse, A.O. Aydin, H. Akbulut, Sol–gel synthesis of nanostructured SnO2 thin film anodes for Li-ion batteries. Acta Phys. Pol. A 121(1), 227–229 (2012)
S. Gong, J. Liu, J. Xia, L. Quan, H. Liu, Gas sensing characteristics of SnO2 thin films and analyses of sensor response by the gas diffusion theory. Mater. Sci. Eng. B 164(2), 85–90 (2009)
R. Adnan, N.A. Rohana, I.A. Rahman, Synthesis and characterization of high surface area tin oxide nanoparticles via the sol–gel method as a catalyst for the hydrogenation of styrene. J. Chin. Chem. Soc. 57(2), 222–229 (2010)
Y. Li, W. Yin, R. Deng, R. Chen, J. Chen, Q. Yan, Realizing a SnO2-based ultraviolet light-emitting diode via breaking the dipole-forbidden rule. NPG Asia Mater. 4(11), e30 (2012)
G. Sakai, N.S. Baik, N. Miura, N. Yamazoe, Gas sensing properties of tin oxide thin films fabricated from hydrothermally treated nanoparticles: dependence of CO and H2 response on film thickness. Sens. Actuators B 77(1), 116–121 (2001)
C. Ke, W. Zhu, J.S. Pun, Z. Yang, Annealing temperature dependent oxygen vacancy behavior in SnO2 thin films fabricated by pulsed laser deposition. Curr. Appl. Phys. 11(3), S306–S309 (2011)
L.D. Khanh, N.T. Binh, L.T. Binh, N.N. Liong, SnO2 nanostructures synthesized by using a thermal evaporation method. J. Korean Phys. Soc. 52, 1689–1692 (2008)
P. Lian, X. Zhu, S. Liang, Z. Li, W. Yang, H. Wang, High reversible capacity of SnO2 graphene nanocomposite as an anode material for lithium-ion batteries. Electrochim. Acta 56(12), 4532–4539 (2011)
M. Ristić, M. Ivanda, S. Popović, S. Musić, Dependence of nanocrystalline SnO2 particle size on synthesis route. J. Non-Cryst. Solids 303(2), 270–280 (2002)
Acknowledgments
This work was conducted under Research University (RU) Grant: 203/PFIZIK/6711197 the support from Universiti Sains Malaysia gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kadhim, I.H., Abu Hassan, H. Effects of glycerin volume ratios and annealing temperature on the characteristics of nanocrystalline tin dioxide thin films. J Mater Sci: Mater Electron 26, 3417–3426 (2015). https://doi.org/10.1007/s10854-015-2851-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-015-2851-4