Abstract
Species from natural communities show different capacities for moving across contrasting habitats, and they may gradually respond along the transition between forests and anthropogenic areas. Because beetles are effective bioindicators, we analyzed the Coleoptera assemblage structure in the transition between sugarcane matrices and forest fragment. The study was conducted in an Atlantic rainforest fragment and sugarcane matrices after 2 and 6 months of replantation. Beetles were sampled in linear transects that were 50, 100, and 200 m from the forest edge and toward both forest core and matrices. We analyzed beetle abundance, species richness and composition, and diversity numbers. The abundance and species richness were higher in the forest, and species richness was higher at the sampling site 100 m inside the forest than at the gradients within the sugarcane matrices. The species composition differed between the forest and matrices, but not between the matrices with different replantation ages. Alpha diversity based on the number of species was higher in the forest, and alpha diversity based on the Shannon index was higher in the forest and matrix after 6 months of replantation. Our results suggest that the sugarcane matrices, chiefly the matrix after 2 months of replantation, sustain an impoverished beetle assemblage when compared with the forest fragment. Despite the strong habitat distinction, the beetle fauna in the sugarcane matrices was not affected by the horizontal distance from the forest. Here, we have highlighted the importance of forest fragments embedded in harsh matrices for the maintenance of biodiversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Environmental disturbances (e.g., habitat fragmentation and transformation) are a threat to the natural environment and one of the main causes of biodiversity loss (Naeem et al. 1999; Myers et al. 2000; Slingenberg et al. 2009). The disturbances arise mainly because of anthropogenic activities that modify landscapes, transforming continuous portions of native forests into remnants with heterogeneous configurations (Whitmore 1997; Laurance and Peres 2006). In the current fragmentation scenario, forest patches are surrounded by different anthropogenic matrices (Laurance and Peres 2006; Franklin and Lindenmayer 2009; Lizée et al. 2012).
Depending on the type of anthropogenic matrix and natural ecosystems embedded in them, the forest-matrix transition can affect the capacity of native species to move across such disturbed habitats (Castellon and Sieving 2006). The matrix may represent an alternative or secondary habitat for forest organisms, favoring the displacement of native populations among the fragments (Prevedello and Vieira 2010). In contrast, the matrix may represent a selective barrier, constraining the movement of animals and plants (Gascon et al. 1999). The dispersal of native species through the landscape is favored when both the conserved habitat and anthropogenic matrix have a similar vegetation structure (e.g., canopy cover) (Gascon et al. 1999; Heinrichs and Pauchard 2015; Williams-Guillén et al. 2015). The habitat becomes inhospitable for forest-specialist species when the matrix and original environment have a different vegetation structure, limiting or disrupting their movement among remnants (Gascon et al. 1999; Castellon and Sieving 2006).
Forest edges represent a barrier that limits anthropogenic and natural habitats. The effects of the edge on biodiversity are gradual through horizontal distances from the forest edge to the core; thus, forest-specialist species show higher success near the forest core (Oosterhoon and Kappelle 2000; Eggleton et al. 2002; Carpio et al. 2009). Similarly, in horizontal gradients from the forest edge toward anthropogenic matrices, some forest species successfully occupy the first few meters of the disturbed areas, drastically reducing, or even disappearing, with the spacing from forest remnants (Oosterhoon and Kappelle 2000; Karem et al. 2006; Filgueiras et al. 2015). Fragment portions near the forest edge have lower abundances of secondary succession trees and a lower canopy (Oosterhoon and Kappelle 2000), affecting climatic conditions and, consequently, the ecological communities in such sites. In cloud forests of Costa Rica, for example, forest core presents a high abundance of tree species, as Quercus copeyensis C.H. Mull. and Q. seemannii Liebm., which are negatively affected by edge effect, being replaced toward forest edges by secondary shrubs and tree species, as Comarostaphylis arburoides Lindley and Cornus disciflora Moc. & Sessé ex DC. (Oosterhoon and Kappelle 2000). Another example are the wasps in temperate forests of the United States, being the species Aptesis incompta Townes and Cratichneumon sp. favored by the forested habitats, while Phanerotoma sp. and Dusona sp. benefits from forest edges (Karem et al. 2006). Previous studies of horizontal gradients from disturbed to conserved environments have shown that it is possible to estimate distances at which the species from ecological communities have their distribution positively or negatively affected by the edge effects.
To evaluate how habitat transformation affects biodiversity, indicators group are frequently used to reflect the condition of the community structure (Gardner et al. 2008). Among insects, some families of edaphic beetles (e.g., Carabidae, Cicindelidae, and Scarabaeidae) represent effective indicator groups, and they are widely used in monitoring studies because of their standardized sampling, high species richness, well-defined taxonomy, and sensitivity to habitat changes (Brown 1991; Rodríguez et al. 1998; Gardner et al. 2008; Maleque et al. 2009). Soil and vegetation characteristics are determinant factors for edaphic beetle assemblages because of microclimatic conditions (e.g., temperature, light intensity, and soil and air moisture) that regulate the distribution of edaphic beetles (Reddy and Venkataiah 1990; Larsen et al. 2006; Fagundes et al. 2011; Wardhaugh et al. 2013).
After the sixteenth century, the native vegetation of the Atlantic rainforest, which is distributed throughout the coastal region of northeastern Brazil, was affected by deforestation processes throughout the colonial and post-colonial period of the country (Tabarelli et al. 2006). Currently, the region is urbanized and the agricultural landscapes are mainly dominated by sugar-cane monocultures (Ranta et al. 1998; Tabarelli et al. 2006; Enedino et al. 2018), which is in contrast with the vegetation structure of the native Atlantic rainforest. The two ecosystems share almost no plant or animal species; the forest remnants of the Atlantic rainforest are composed mainly of tree species belonging to the families Fabaceae, Euphorbiaceae, Lauraceae, Moraceae, and Sapotaceae, whereas sugarcane is grown in a monospecific habitat in which only sugarcane plants are found (Ranta et al. 1998; Pôrto et al. 2005). The forest fragments have a closed-canopy structure with lower light incidence and milder microclimatic conditions than the sugarcane ecosystem (Pinto et al. 2010). Furthermore, burning practices are outdated but still common in sugarcane plantations in Brazil (ELLA 2012). Because of such activities, edaphic beetles that successfully inhabit sugarcane matrices are constantly exposed to harsh microclimatic transformations, as fire that affects the soil and vegetation.
The objective of the current study was to evaluate the diversity of edaphic beetles in the transition from anthropogenic matrix (i.e., sugarcane agricultural lands with two plantation ages) to forest fragment in the Atlantic rainforest. We tested the following hypotheses: (1) Habitat type will affect beetle assemblage structure in the fragmented landscape of the Atlantic rainforest. (2) The horizontal gradient among habitats affects beetle abundance and species richness. (3) Age of the plantation affects beetle assemblages. For these hypotheses, we presented the following predictions: (1) the habitat types will present distinct beetle species compositions. (2) The forest fragment will present higher species richness and abundance of beetles than the sugarcane matrix. (3) The beetle species richness will increase towards the forest core and abundance will increase towards the edges of the environments analyzed. (4) The older sugarcane plantation will present higher species richness and abundance of beetles than the recently planted sugarcane plantation.
Materials and methods
Study site
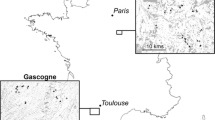
The study was performed in an Atlantic forest fragment of 3500 ha and the surrounding sugarcane matrices within the Serra Grande sugar processing plant located in the municipalities of Ibateguara and São José da Laje (8°30′S, 35°50′W) in the state of Alagoas, northeastern Brazil (Fig. 1). The processing plant has 9000 ha of conservation units, which are distributed over a highly fragmented landscape. The forest remnants in the region (n = 109) range from 1.67 to 3500 ha and are surrounded by a homogeneous sugarcane matrix. The Serra Grande landscape is located on a low-altitude plateau (300–400 m asl) with shallow and wide valleys and two dystrophic soils (i.e., podsoil and pozolic) (IBGE 1985). The climate is warm and wet, with a mean annual temperature of 23 °C and mean annual rainfall of 2000 mm; the rains are concentrated between April and August, with 3 months of dry season (< 60 mm per month) from November to January (IBGE 1985; Veloso et al. 1991a, b; Oliveira et al. 2004). The native vegetation of the region is ombrophilous tropical rainforest with elements from both evergreen and semi-deciduous forests. The region is composed mainly of plants belonging to the families Lauraceae, Sapotaceae, Moraceae, and Fabaceae (Veloso et al. 1991a, b).
Map of South America (a) showing the sampling sites in the forest fragment (n = 20) and sugarcane matrices (n = 20) in northeastern Brazil (b). Scheme of the trap locations in the gradients in the forest and matrix habits, with pitfall traps indicated using dark circles (c). The sampled sugarcane matrices surrounded the forest fragment. The surrounding fragments in the landscape are indicated using light gray. Circles = forest fragment; Triangles = sugarcane matrix after 2 months of replantation; squares = sugarcane matrix after 6 months of replantation
Beetle surveys
Between January and May 2011, beetles were sampled from three habitats: forest fragment; sugarcane plantation 2 months after replantation (M2), and sugarcane plantation 6 months after replantation (M6). The replantation, in both cases, was performed after the scorch. The beetles were collected using pitfall traps, which consisted of plastic cylinders (20 × 15 cm) with a solution of 100 mL of water (80%) and detergent (20%; to conserve the sampled material and break superficial tension of the water). The traps were installed in transects, which were composed of six sets located at the transition of the sugarcane matrix to the forest fragment: 50, 100, and 200 m from the edge to the interior of the habitats. In each set, six traps were installed transversally to the transect, 10 m apart from one another (Fig. 1). Forty transects were used: 20 in the forest fragment, 10 in M2, and 10 in M6. In total, 720 pitfall traps were used, and each habitat sampled once during the fieldwork. All traps were installed in the morning (between 06:00 and 12:00) and opened right after their installation. According to the methodology of a previous study that successfully surveyed Coleoptera assemblages in Brazil (see Fagundes et al. 2011), the traps were left for 48 h in the field, and the collected material was maintained in 70% ethanol. The specimens were identified at least to the family level, using identification key (Casari and Ide 2012), and morphotyped based on morphological characteristics that are used in each family for distinguishing species. The voucher specimens were deposited in the Coleção Entomológica da UFPE (CE-UFPE).
Statistical analysis
To compare the species richness and abundance of the beetles in the forest fragment and sugarcane matrices (M2 and M6), generalized linear models (GLMs) with Poisson error distribution were used. Furthermore, beetle abundance was also included as a predictor in the analysis of species richness, to clarify whether abundance may be affecting species richness. Abundance and richness were the response variables, and habitat type (forest fragment, M2, and M6) and gradient of the habitats (50, 100, and 200 m) were the predictive variables. Analyses of beetle species richness and abundance were fitted with the predictor variables and the interactions between them. Akaike information Criterion (AIC) values were used to obtain the best model for the data (Johnson and Omland 2004). The predictor variables were ranked based on the p-value obtained from likelihood ratio tests. The ranked variables were tested, and those with p < 0.05 were kept for the best model (see Zuur et al. 2009). To compare the models, stepAIC in MASS library was also used, and the models with lower AIC values were selected. The best models obtained using the Zuur et al. (2009) criteria were compared to the ones obtained by stepAIC through likelihood ratio tests. If there was a significant difference between models, the one with the lowest AIC was selected. When there was no difference, the most parsimonious model (i.e. with less variables) was selected (Crawley 2013). The assumptions of normality of the residuals were visually analyzed with normal q-q plots; outliers were evaluated through Cook’s distance, but none was found (Cook’s distance < 1). The data were analyzed using R software version 3.2.0 (R Core Team 2015).
To compare the species composition of the beetles sampled from the forest remnant and matrices, non-metric multidimensional scaling (NMDS) was performed for a visual and explorative evaluation. Analysis of similarities (ANOSIM) was performed to check for statistical differences in the species compositions of the habitats. Habitat type was used as the factor, and species composition, as the attribute. The Jaccard index was calculated for the samples, and NMDS ordination was performed with 2,500 repetitions. Both ANOSIM and NMDS were performed using Primer software version 6.0 (Clarke and Gorley 2006).
To estimate beetle diversity in the forest fragment and matrices, analyses of alpha (Dα), beta (Dβ), and gamma (Dγ) diversities were performed using diversity number (Jost 2006). The diversity was evaluated using the number of species (0D) and Shannon diversity (1D) (Hill 1973; Jost 2006). 0Dα and 1Dα indicate the mean number of species in the assemblages and number of abundant species in the assemblages, respectively. 0Dβ and 1Dβ indicate the number of distinct assemblages based on species richness and abundance of species, respectively, and represent the species turnover among the samples. 0Dγ and 1Dγ indicate the total number of species and number of abundant species in the landscape, respectively (Jost 2006; Molina and Farinós 2012).
Results
A total of 505 individuals were captured, and 71 morphotypes and 17 families of Coleoptera were identified. The forest fragment encompassed 77.42% of the beetles collected, and of the 56 morphotypes identified, 42 were exclusively obtained from this habitat. However, 24 morphotypes from the forest fragment were singletons, and 20 were doubletons. M2 encompassed 9.50% of the total beetles sampled, and of the 20 morphotypes identified, five were exclusive to this habitat. Thirteen morphotypes in M2 were singletons, and four were doubletons. M6 encompassed 13.06% of the beetles sampled, and of the 17 morphotypes identified, four were exclusive to this habitat (see supplementary material). Three morphotypes in M6 were singletons, and five were doubletons.
Of the families that showed representative abundance (n > 10), three were mainly collected from the forest: Chrysomelidae (68.75% in forest), Curculionidae (88.23%), and Scarabaeidae (96.15%). Carabidae and Nitidulidae showed an almost equal distribution among the forest and matrices, and Cicindelidae was mainly collected from the matrices (60.78% obtained in matrices). Both species richness and abundance of the beetles were affected by habitat type (X2 = 25.902, d.f. = 2, p < 0.001 and X2 = 87.002, d.f. = 2, p < 0.001, respectively). The beetle species richness and abundance were significantly higher in the forest fragment than in the matrices (Fig. 2). In addition, beetle species richness presented a significantly positive relation with beetle abundance (X2 = 25.390, d.f. = 1, p < 0.001, R2 = 0.53) (see Fig. S1). The gradients of the habitats and interaction of habitat type and gradients were not retained by the best-supported models to explain the beetle species richness and abundance.
General linear models presenting mean ± 95% confidence intervals of beetle species richness (a) and abundance (b) sampled from sugarcane matrices after 2 and 6 months of replantation (M2 and M6, respectively) and forest fragment (F) in the Atlantic rainforest, northeastern Brazil. Different letters indicate significant differences
Among the families sampled, Scarabaeidae and Carabidae showed a higher number of morphotypes (S = 18 and 15, respectively), which together accounted for 47.14% of the species richness. The forest fragment yielded 14 families, of which seven were collected exclusively from this habitat (see supplementary material). In the matrices, eight families were recorded (six in M2 and seven in M6), and one was restricted to the matrix habitat (Eucnemidae, found in only M2) (Table 1). Morphotype dominance was distinct among the forest fragment and matrices, and Dichotomius iannuzziae Valois, Vaz-de-Mello and Silva and Trichillum sp.1 were dominant in the forest but not abundant in the matrices. Nitidulidae sp.1 was dominant in all three habitats, and Niditulidae sp.1, Cicindelidae sp.1 and Cicindelidae sp.2, were the dominant morphotypes in both M2 and M6 (Fig. 3). The species composition exhibited significant segregation among the habitat types (Global-R: 0.613, p = 0.001) (Fig. 4).
The forest fragment showed a mean of approximately eight morphotypes per transect, which is higher than M2 (three) and M6 (nearly five) (Table 2). Both forest fragment and M6 yielded approximately five abundant morphotypes in the samplings, whereas M2 yielded almost three abundant morphotypes per sampling (Table 2). In the landscape of the study site, about 18 abundant morphotypes were present, with six morphotypes per sampling and five abundant morphotypes per sampling (Table 2). With respect to species richness, the landscape encompassed approximately 11 distinct beetle assemblages; according to Shannon diversity, four distinct assemblages were present (Table 2).
Discussion
In this study, we assessed the effects of habitat type and horizontal transition between the sugarcane matrix and forest fragment with respect to edaphic beetle assemblage. Our results suggest that the sugarcane matrix sustains an impoverished beetle assemblage when compared with the forest fragment. The abundance, species richness, species composition, dominant species, and diversity numbers were different between the forest fragment and matrices. In addition, the replantation age of the sugarcane matrix did not have any clear effects on the beetle assemblage that was able to live in such a harsh environment.
The beetle abundance, species richness, and diversity numbers were lower in the sugarcane matrices than in the forest fragment. A similar pattern was observed in a previous study in which forest remnants and Pinus plantations were compared, with the anthropogenic site presenting a lower diversity of beetles than the forested habitat (Fagundes et al. 2011). Because of the environmental conditions generated by the vegetation structure within forests, such habitats provide higher microclimatic stability and soil moisture when compared with Pinus areas and favor beetle diversity (Fagundes et al. 2011). The annual sugarcane cycle includes slash-and-burn practices, a common method in this system that results in drastic changes in the landscape (D’Anunciação et al. 2013). Although there are insect species that flourish within sites after burning, most of the species are negatively affected, which reduces their abundance in the months after burning (Araújo et al. 2005). Therefore, we suggest that the practices used in sugarcane plantations result in inhospitable environments for beetles, and few species and individuals can successfully occupy this type of matrix.
The beetle species composition in the forest remnant was distinct when compared with that in the sugarcane matrices. The species composition exhibits subtle changes to habitat modification, whereas species richness and abundance are parameters that sometimes do not show clear responses to landscape transformation (Uehara-Prado et al. 2006; Barlow et al. 2007; Nichols et al. 2007). In this study, we could observe potential indicator groups that may be associated with conserved or disturbed conditions. For example, beetles from the families Chrysomelidae, Curculionidae, and Scarabaeidae were collected mostly from the forest fragment. The microclimatic requirement is an important characteristic for bioindicators, and Scarabaeidae has species with high habitat specificity (Noss 1990; Larsen et al. 2006; Nichols et al. 2007; Gardner et al. 2008). Chrysomelidae and Curculionidae are herbivorous beetles with strict or generalist food habits, and some species are considered as crop plagues (Kubo and Ando 1989; Ávila and Parra 2003; Hulcr et al. 2007; Martínez et al. 2013; Bozsik 2014). The high plant diversity presented by the forest, when compared with the sugarcane matrix, may favor the presence of a high number of Chrysomelidae and Curculiondae species because of the assortment of different food resources. Notwithstanding, M2 and M6 did not present clear distinctions in the beetle species composition. Our result indicates that sugarcane plantations, irrespective of replantation age, act as hard edges and present a barrier that constrains the dispersal of beetles that inhabit Atlantic rainforest remnants.
Three families dominated the beetle assemblage in the studied habitats: Scarabaeidae, Nitidulidae, and Cicindelidae. The beetles that belonged to Scarabaeidae were the most abundant in the forest samplings, chiefly because of D. iannuzziae. In tropical rainforests, dung beetles are a representative group among Coleoptera and are found in higher abundances in more conserved habitats (Nichols et al. 2007; Audino et al. 2014; Filgueiras et al. 2015). Dichotomius beetles are commonly found in the forest remnants of the Atlantic forest, and D. iannuzziae (reported as D. sericeus in previous studies of the region) is dominant in many dung beetle assemblages (Filgueiras et al. 2009, 2015; Costa et al. 2013). Interestingly, nitidulid beetles were dominant in all three habitats, although they showed higher abundance in the forest fragment than in the matrices. Nitidulid beetles show a wide variety of feeding habits (e.g., predator, saprophagous, detritivorous) and successfully occupy environments with different habitat configurations and conservation status (Elzen et al. 1999; Barbosa et al. 2002; Aguirre and Dirzo 2008). Habitat modification and fragmentation negatively affect Nitidulidae diversity, and this was evident in our study (a lower abundance of this family in the matrices) (Goehring et al. 2002; Aguirre and Dirzo 2008; Fagundes et al. 2011). However, among the beetle families obtained in the current study, Nitidulidae apparently was one of the most resilient to the harsh conditions of the sugarcane matrices. Tiger beetles (Cicindelidae) were also dominant in the matrices. Such beetles are considered excellent bioindicators, and vegetal structure acts as a strong selective barrier for their distribution (Pearson and Cassola 1992). However, this family has species that are adapted to occupy different habitat configurations (Pearson and Cassola 1992; Rodríguez et al. 1998). In the current study, one morphotype of Cicindelidae was collected chiefly from the forest, and the others were exclusive to the sugarcane matrices and exhibited a plastic pattern of habitat occupation.
Studies that consider family richness instead of species and genus richness may present less robust predictions and responses of biodiversity to changes in the environment (Kallimanis et al. 2012; Monk et al. 2012). The high diversity of Coleoptera in tropical forests, associated with the relatively low number of taxonomists in this region, is a crucial limiting factor for identification at the species level. Notwithstanding, in this study, we considered the taxonomic unit of the recorded morphospecies. Studies that compare biodiversity response at the species level and broader taxonomic groups consider the higher taxonomic levels as a unit of diversity (Mandelik et al. 2007; Monk et al. 2012; Kallimanis et al. 2012). The ecological importance of each species was maintained in this study; thus, we could analyze the effects of how Coleoptera species richness and diversity responded to the different habitat types and transition between the sugarcane matrices and forest fragment.
We expected the abundance to increase towards the edge of the habitats and the richness to gradually increase towards the forest core. However, this pattern was not observed through the gradients towards the forest and matrices. The forest edges represented the biological barriers of two distinct environments (Ries et al. 2004). This habitat has intermediate abiotic conditions (e.g., light intensity and temperature) when compared with the forest core and anthropogenic matrices (Murcia 1995), affecting biological elements in these sites. In natural communities, some beetle species benefit from edges and open areas (Didham et al. 1998; Karen et al. 2008; Costa et al. 2013). However, even species that are adapted to disturbances have limits. Hard edges cause strong selection of biodiversity in an area (López-Barrera et al. 2007). We suggest that, because of the strong distinction between the studied habitats, the impoverished beetle fauna in the sugarcane plantations was not affected by the distance from the forest fragment. Although edge effects have a mean penetration distance of 100 m (Broadbent et al. 2008), our data suggest that the beetles do not show clear effects in the gradient from the forest edge to the forest core. The northeastern region of the Atlantic rainforest is highly fragmented, and previous studies of this landscape have presented a scenario of biotic simplification and homogenization (Ranta et al. 1998; Oliveira et al. 2004). With respect to beetle abundance and species richness, beetle fauna present a homogenous distribution through the forest edge to the forest core.
We have highlighted the importance of maintenance of forest fragments in agricultural landscapes that have harsh matrices, as in sugarcane plantations. Hard edge effects are present in the fragment/matrix transition, which negatively affect the distribution of beetles with impoverished assemblages that can occupy anthropogenic habitats. Governmental policies are urgently required to maintain the ecological communities and ecosystems in this landscape because the prospects for the conservation of this ecosystem are alarming.
References
Aguirre A, Dirzo R (2008) Effects of fragmentation on pollinator abundance and fruit set of an abundant understory palm in a Mexican tropical forest. Biol Conserv 141:375–384
Araújo RA, Araújo MS, Gonring AHR, Guedes RNC (2005) Impact of controlled sugarcane straw burning on local insect community. Neotrop Entomol 34:649–658
Audino LD, Louzada J, Comita L (2014) Dung beetles as indicators of tropical forest restoration success: is it possible to recover species and functional diversity? Biol Conserv 169:248–257
Ávila CJ, Parra JRP (2003) Leaf consumption by Diabrotica speciosa (Coleoptera:Chrysomelidae) adults on different host plants. Sci Agr 60:789–792
Barbosa MGV, Fonseca CRV, Hammond PMH, Stork NE (2002) Diversity and similarity between habitats based on the leaf litter Coleoptera fauna from the terra firme forest of Central Amazonia. Mongr Terc Milenio 2:69–83
Barlow J, Gardner TA, Araujo IS et al (2007) Quantifying the biodiversity value of tropical primary, secondary, and plantation forests. Proc Natl Acad Sci USA 104:18555–18560
Bozsik A (2014) Food preference of Chrysolina fastuosa adults (Coleoptera: Chrysomelidae). Acta Zool Acad Sci Hung 60:239–245
Broadbent EN, Asner GP, Keller M, Knapp DE, Oliveira PJC, Silva JN (2008) Forest fragmentation and edge effects from deforestation and selective logging in the Brazilian Amazon. Biol Conserv 141:1745–1757
Brown KS Jr (1991) Conservation of Neotropical environments: insects as indicators. In: Collins NM (ed) The conservation of insects and their habitats, 1st edn. Academic Press, Cambridge, pp 349–404
Carpio C, Donoso DA, Ramón G, Dangles O (2009) Short term response of dung beetle communities to disturbance by road construction in the Ecuadorian Amazon. Ann Soc Entomol Fr 45:455–469
Casari S, Ide S (2012) Coleoptera. In: Rafael JA, Melo GAR, Carvalho CJB, Casari SA, Constantino R (eds) Insetos do Brasil. Holos Editora, Ribeirão Preto, pp 359–368
Castellon TD, Sieving KE (2006) An experimental test of matrix permeability and corridor use by an endemic understory bird. Conserv Biol 20:135–145
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. PRIMER-E, Plymouth
Costa FC, Pessoa KKT, Liberal CN, Filgueiras BKC, Salomão RP, Iannuzzi L (2013) What is the importance of open habitat in a predominantly closed forest area to the dung beetle (Coleoptera, Scarabaeinae) assemblage? Rev Bras Entomol 57:329–334
Crawley MJ (2013) The R book, 2nd edn. Aptara Inc., New Delhi
D’Anunciação PER, Silva MFV, Ferrante L, Assis DS, Casagrande T, Coelho AZG, Amâncio BCS, Pereira TR, da Silva VX (2013) Forest fragments surrounded by sugar cane are more inhospitable to terrestrial amphibian abundance than fragments surrounded by pasture. Int J Ecol. https://doi.org/10.1155/2013/183726
Didham R, Lawton J, Hammond P, Eggleton P (1998) Trophic structure stability and extinction dynamics of beetles (Coleoptera) in tropical forest fragments. Philos Trans R Soc B 353:437–451
Eggleton P, Bignell DE, Hauser S, Dibog L, Norgrove L, Madong B (2002) Termite diversity across an anthropogenic disturbance gradient in the humid forest zone of West Africa. Agr Ecosyst Environ 90:189–202
Elzen PJ, Baxter JR, Westervelt D, Randall C, Delaplane KS, Cutts L, Wilson WT (1999) Field control and biology studies of a new pest species, Aethinatumida Murray (Coleoptera, Nitidulidae), attacking European honey bees in the Western Hemisphere. Apidologie 30:361–366
Enedino TR, Loures-Ribeiro A, Santos BA (2018) Protecting biodiversity in urbanizing regions: the role of urban reserves for the conservation of Brazilian Atlantic Forest birds. Perspect Ecol Conserv 16:17–23
Evidences and Lessons from Latin America (ELLA) (2012) From manual to mechanical harvesting: reducing environmental impacts and increasing cogeneration potential. http://ella.practicalaction.org/knowledge-brief/from-manual-to-mechanical-harvesting-reducing-environmental-impacts-and-increasing-cogeneration-potential. Accessed 11 Jan 2018
Fagundes C, Di Mare R, Wink C, Manfio D (2011) Diversity of the families of Coleoptera captured with pitfall traps in five different environments in Santa Maria, RS, Brazil. Braz J Biol 71:381–390
Filgueiras BKC, Liberal CN, Aguiar CDM, Hernández MIM, Iannuzzi L (2009) Attractivity of omnivore, carnivore and herbivore mammalian dung to Scarabaeinae (Coleoptera, Scarabaeidae) in a tropical Atlantic rainforest remnant. Rev Bras Entomol 53:422–427
Filgueiras BKC, Tabarelli M, Leal IR, Vaz-de-Mello FZ, Iannuzzi L (2015) Dung beetle persistence in human-modified landscapes: combining indicator species with anthropogenic land use and fragmentation-related effects. Ecol Indic 55:65–73
Franklin JF, Lindenmayer DB (2009) Importance of matrix habitats in maintaining biological diversity. PNAS 106:349–350
Gardner TA, Barlow J, Araújo IS et al (2008) The cost-effectiveness of biodiversity surveys in tropical forests. Ecol Lett 11:139–150
Gascon C, Lovejoy TE, Bierregaard RO Jr, Malcolm JR, Stouffer PC, Vasconcelos HL, Laurance WF, Zimmerman B, Tocher M, Borges S (1999) Matrix habitat and species richness in tropical forest remnants. Biol Conserv 91:223–229
Goehring DM, Daily GC, Sekercioglu CH (2002) Distribution of ground-dwelling arthropods in tropical countryside habitats. J Insect Conserv 6:83–91
Heinrichs S, Pauchard A (2015) Struggling to maintain native plant diversity in a peri-urban reserve surrounded by a highly anthropogenic matrix. Biodivers Conserv 24:2769–2788
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–431
Hulcr J, Mogia M, Isua B, Novotny V (2007) Host specificity of ambrosia and bark beetles (Col., Curculionidae: Scolytinae and Platypodinae) in a New Guinea rainforest. Ecol Entomol 32:762–772
Instituto Brasileiro de Geografia e Estatisticas (IBGE) (1985) Atlas nacional do Brasil: região nordeste. IBGE, Rio de Janeiro
Johnson JB, Omland KS (2004) Model selection in ecology and evolution. Trends Ecol Evol 19:101–108
Jost L (2006) Entropy and diversity. Oikos 113:363–375
Kallimanis AS, Mazaris AD, Tsakanikas D, Dimopoulos P, Pantis JD, Sgardelis SP (2012) Efficient biodiversity monitoring: which taxonomic level to study? Ecol Indic 15:100–104
Karem J, Woods SA, Drummond F, Stubbs C (2006) Sampling native wasps along both vertical and horizontal gradients in the maine lowbush blueberry landscape. Environ Entomol 35:1083–1093
Karen M, John O, John B, Paul G, Josephine P, Thomas K (2008) Distribution and composition of carabid beetle (Coleoptera, Carabidae) communities across the plantation forest cycle—implications for management. Forest Ecol Manag 256:624–632
Kubo T, Ando Y (1989) Food preference of a polyphagous leaf beetle, Atrachya menetriesi Faldermann (Coleoptera:Chrysomelidae). Jpn J Appl Entomol Z 33:231–237
Larsen TH, Lopera A, Forsyth A (2006) Extreme trophic and habitat specialization by peruvian dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae). Coleop Bul 60:315–324
Laurance WF, Peres CA (2006) Emerging threats to tropical forests. University of Chicago Press, Chicago
Lizée M, Manel S, Mauffrey J, Tatoni T, Deschamps-Cottin M (2012) Matrix configuration and patch isolation influences override the species-area relationship for urban butterfly communities. Landsc Ecol 27:159–169
López-Barrera F, Armesto JJ, Williams-Linera G, Smith-Ramírez C, Manson RH (2007) Fragmentation and edge effects on plant-animal interactions, ecological processes and biodiversity. In: Newton AC (ed) Biodiversity loss and conservation in fragmented forest landscapes: the forests of montane Mexico and temperate South America. Cabi Publishing, Wallingford, pp 69–101
Maleque MA, Maeto K, Ishii HT (2009) Arthropods as bioindicators of sustainable forest management, with a focus on plantation forests. Appl Entomol Z 44:1–11
Mandelik Y, Dayan T, Chikatunov V, Kravchenko V (2007) Reliability of a higher-taxon approach to richness, rarity, and composition assessments at the local scale. Conserv Biol 21:1506–1515
Martínez FS, Franceschini MC, Poi A (2013) Food preference Neochetina eichhorniae (Coleoptera: Curculionidae) by aquatic plants of different nutritional value. Rev Colomb Entomol 39:81–87
Molina MF, Farinós HBM (2012) Los componentes alfa, beta y gamma de la biodiversidad. Aplicación al estudio de comunidades vegetales. Universitat Politècnica de València. https://riunet.upv.es/handle/10251/16285. Accessed 1 July 2017
Monk WA, Wood PJ, Hannah DM, Extence CA, Chadd RP, Dunbar MJ (2012) How does macroinvertebrate taxonomic resolution influence ecohydrological relationships in riverine ecosystems. Ecohydrology 5:36–45
Murcia C (1995) Edge effects in fragmented forests: implications for conservation. Trends Ecol Evol 10:58–62
Myers N, Mittermeier RA, Mittermeier CG, Da Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Naeem S, Chapin CFS, Costanza R, Ehrlich PR, Golley FB, Hooper DU, Lawton JH, O’Neill RV, Mooney HA, Sala OE, Symstad AJ, Tilman D (1999) Biodiversity and ecosystem functioning: maintaining natural life support processes. Issues Ecol 4:1–11
Nichols E, Larsen T, Spector D, Davis AL, Escobar F, Favila M, Vulinec K (2007) Global dung beetle response to tropical forest modification and fragmentation: a quantitative literature review and meta-analysis. Biol Conserv 137:1–19
Noss RF (1990) Indicators for monitoring biodiversity: a hierarchical approach. Conserv Biol 4:355–364
Oliveira MA, Grillo AS, Tabarelli M (2004) Forest edge in the Brazilian Atlantic forest: drastic changes in tree species assemblages. Oryx 38:389–394
Oosterhoon M, Kappelle M (2000) Vegetation structure and composition along an interior-edge-exterior gradient in a Costa Rican montane cloud forest. Forest Ecol Manag 126:291–307
Pearson DL, Cassola F (1992) World-wide species richness patterns of tiger beetles (Coleoptera: Cicindelidae): indicator taxon for biodiversity and conservation studies. Conserv Biol 6:376–391
Pinto SRR, Mendes G, Santos AMM, Dantas M, Tabarelli M, Melo FPL (2010) Landscape attributes drive complex spatial microclimate configuration of Brazilian Atlantic forest fragments. Trop Conserv Sci 3:389–402
Pôrto KC, Almeida-Cortez JS, Tabarelli M (2005) Diversidade Biológica e Conservação da Floresta Atlântica ao Norte do Rio São Francisco. Ministério do Meio Ambiente, Brasília
Prevedello JA, Vieira MV (2010) Does the type of matrix matter? A quantitative review of the evidence. Biodivers Conserv 19:1205–1223
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ranta P, Blom T, Niemela J, Joensuu E, Siitonen M (1998) The fragmented Atlantic rainforest of Brazil: size, shape and distribution of forest fragments. Biodivers Conserv 7:385–403
Reddy MV, Venkataiah B (1990) Seasonal abundance of soil-surface arthropods in relation to some meteorological and edaphic variables of the grassland and tree-planted areas in a tropical semi-arid savanna. Int J Biometeorol 34:49–59
Ries L, Fletcher RJ Jr, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Annu Rev Ecol Evol Syst 35:491–522
Rodríguez JP, Pearson DL, Barrera RR (1998) A test for the adequacy of bioindicator taxa: are tiger beetles (Coleoptera: Cicindelidae) appropriate indicators for monitoring the degradation of tropical forests in Venezuela? Biol Conserv 83:69–76
Slingenberg A, Braat L, Windt H, Rademaekers K, Eichler L, Turner K (2009) Study on understanding the causes of biodiversity loss and the policy assessment framework. Ecorys, Research and Consulting, Rotterdan
Tabarelli M, Melo M, Lira O (2006) A mata Atlântica do nordeste - Mata Atlântica uma rede pela floresta. Rede de Ongs da Mata Atlântica, Brasília
Uehara-Prado M, Brown KS, Freitas AVL (2006) Species richness, composition and abundance of fruit-feeding butterflies in the Brazilian Atlantic forest: comparison between a fragmented and a continuous landscape. Global Ecol Biogeogr 16:43–54
Veloso HP, Rangel Filho ALR, Lima JCA (1991a) Classificação da vegetação brasileira, adaptada a um sistema universal. Fundação Instituto Brasileiro de Geografia e Estatística, Brasília
Veloso HP, Rangel-Filho ALR, Lima JCA (1991b) Classificação da vegetação brasileira adaptado a um sistema universal. IBGE, Rio de Janeiro
Wardhaugh CW, Edwards W, Stork NE (2013) Variation in beetle community structure across five microhabitats in Australian tropical rainforest trees. Insect Conserv Diver 6:463–472
Whitmore T (1997) Tropical forest disturbance, disappearance, and species loss. In: Laurance WF, Bierregaard RO (eds) Tropical forest remnants: ecology, management, and conservation of fragmented communities. University of Chicago Press, Chicago, pp 3–12
Williams-Guillén K, Olimpi E, Maas B, Taylor PJ, Arlettaz R (2015) Bats in the anthropogenic matrix: challenges and opportunities for the conservation of Chiroptera and their ecosystem services in agricultural landscapes. In: Voigt C, Kingston T (eds) Bats in the anthropocene: conservation of bats in a changing world. Springer, Cham, pp 151–186
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
We thank to Serra Grande sugar processing plant for infrastructure, logistic support, and for authorizing our fieldwork. We also thank “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” (CAPES) for a doctoral scholarship to AFA Lira. We also thank “Consejo Nacional de Ciencia y Tecnología” (CONACYT) for a doctoral scholarship to RP Salomão.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest (financial and non-financial).
Ethical approval
The experiment was not invasive and complied with Brazilian law (SISBIO/ICMBIO Permit Number: 1982628). At the end of the experiment, the specimens were deposited in the Entomological Collection of the Federal University of Pernambuco, according to standard procedures. This study had no human participants; thus, no “informed consent” was required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10841_2018_79_MOESM1_ESM.tiff
Fig. S1—Significant relation between species richness and abundance of edaphic beetles sampled in in the Atlantic rainforest, northeastern Brazil (TIFF 38217 KB)
Rights and permissions
About this article
Cite this article
Salomão, R.P., Pordeus, L.M., de Araujo Lira, A.F. et al. Edaphic beetle (Insecta: Coleoptera) diversity over a forest-matrix gradient in a tropical rainforest. J Insect Conserv 22, 511–519 (2018). https://doi.org/10.1007/s10841-018-0079-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-018-0079-3