Abstract
Forests support high concentrations of species and beetles in particular are often used to evaluate forest biodiversity. Ancient pasture woodlands are facing a major decline in Europe mainly due to the abandonment of traditional management and subsequent succession. We studied click beetles (Coleoptera: Elateridae) in one of the largest central-European remnants of pasture woodland in Lány Game Park (Czech Republic) using flight interception traps placed at standing veteran trees. The gradient of sun-exposure, circumference of stem, height and vitality of tree and tree species were studied in relation to the species richness of click beetles and their ecological groups. Total species richness reached nearly one half of the recently documented fauna in the study area and species accumulations showed us that the majority of species were represented. Most species preferred solitary trees in sun-exposed habitats and avoided shaded trees in closed canopies. The same results were obtained for ecological groups, such as saproxylic and non-saproxylic species, functional groups and guilds. Our results showed that the species richness of one of the most ecologically diverse beetle families, click beetles, benefits from a high level of sun exposure. Thus, the long spatial and temporal continuity of sun-exposed veteran trees could be a good predictor for sustainable forest management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The history of forest management in Europe is well documented (Rackham 2006). Authors have written about the ecosystem changes and impacts caused by forestry (Grove 2002; Lindbladh et al. 2007). Human activities have been the main factors influencing the condition of European forests after most of the large herbivores died out. The degree of openness is predicted to be one of the most important factors in forest management because many forest species are dependent on sunny and warm conditions, in particular saproxylic (dead-wood dependent) beetle species (Jonsell et al. 1998; Alexander 2008; Horák et al. 2012).
Ancient woodland pastures are facing a major decline in European countries, mainly due to the abandonment of traditional management and subsequent succession. These woodland pastures are of great importance for biodiversity because of their special semi-natural characteristics that increase landscape variability and connectivity (Hofmeister et al. 2004; Goldberg et al. 2007; Spitzer et al. 2008; Moga et al. 2009).
Beetles are among the most intensively studied taxa of insects (New 2007; Novotny et al. 2010). Saproxylic beetles, in particular, are key natural elements. Thus, this group of beetles is often used to evaluate forest biodiversity (Kaila et al. 1994, 1997; Økland 1996; Sverdrup-Thygeson and Ims 2002; Foit 2010). Modern studies have shown that the quantity and quality of dead wood, including tree species, decay stage, stem diameter and position, sun-exposure and microhabitat characteristics, contribute towards explaining the species richness of forest beetles (Jonsell et al. 1998; McGeoch et al. 2007).
Much of the current research on saproxylic beetles has been carried out in Fennoscandia, although this region is on the margin of the ranges of many European saproxylic beetles (Komonen 2007; Baselga 2008; Vodka et al. 2009; Nieto and Alexander 2010). Most of studies were focused on wider groups (Siitonen 1994; Sverdrup-Thygeson and Ims 2002) or single species (Siitonen and Saaristo 2000; Ranius and Hedin 2001; Horák et al. 2010).
The click beetles (Elateridae) are one of the most ecologically diverse families of beetles (Laibner 2000; Bouchard et al. 2009). Forests and woodlands of mixed successional stages and forest-grassland ecotonal areas harbour the greatest local diversity of click beetles (Johnson 2002). Adult click beetles are generally active in the afternoon and evening and most fly well and can be collected in various types of traps. The ecology of click beetles has been poorly studied (Johnson 2002) and most studies have focused on pest management (Parker and Howard 2001; Hicks and Blackshaw 2008) or single species requirements (Tolasch et al. 2007; Mertlik 2010).
We studied the response of one of the most ecologically diverse and abundant families of click beetles (Elateridae) and the component ecological groups in relation to tree level habitat parameters in one of the largest central European pasture woodland areas in the Czech Republic. We specifically focused on the influence of sun-exposure on click beetle fauna. Furthermore, we also evaluated the responses of disparate ecological groups, comprising non-saproxylic and saproxylic (total and guilds) species and functional groups.
Materials and methods
Study area
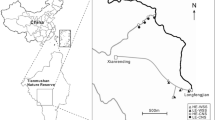
The study area was located in Lány Game Park (50°05′N; 13°55′E; Fig. 1), which is a part of Křivoklátsko UNESCO Biosphere Reserve and Landscape Protected Area. The Lány Game Park is also protected in Natura 2000 and it is a site for four Natura 2000 saproxylic beetles—the violet click beetle (Limoniscus violaceus Müller, 1821), the great Capricorn beetle (Cerambyx cerdo Linnaeus, 1758), the hermit beetle (Osmoderma eremita Scopoli, 1763) and the stag beetle (Lucanus cervus Linnaeus, 1758) (AOPK ČR 2010).
The study area was historically established for game hunting with long temporal continuity of disparate levels of canopy openness (oldmaps.geolab.cz). The first references to hunting for nobility are from the early Middle Ages (Škoudlínová 2000). The area is 30 km2 in size and the recent shape of the park dates back to 1713. The Game Park managers have recently been keeping red, fallow and sika deer, mouflon and wild boar (Škoudlínová 2000).
Most of the areas of within the Lány Game Park are pastured beechwood and oakland areas in a mosaic with pastures and 89 % of the area is forested land (Škoudlínová 2000; AOPK ČR 2010). The Lány Game Park is probably the largest remnant of former open canopy woodlands showing the impact of large herbivores in central Europe.
Study group
There is a long-standing tradition for studies of the response of selected taxa to disparate ecological processes (Niemelä et al. 1993; Oliver and Beattie 1996) and identifying species or other taxonomic units that reliably indicate changes in the environment and reflect the responses of other species or the overall biodiversity to such changes (Bohac 1999; Rainio and Niemelä 2003).
The click beetles (Elateridae) are one of the most ecologically and taxonomically diverse and abundant families of beetles (Hanzák et al. 1973; Laibner 2000; Johnson 2002; Löbl and Smetana 2007; Konstantinov et al. 2009). The range of functional groups and guilds in this taxon is very high. There are known negative economic effects of some click beetles (Hicks and Blackshaw 2008), and some are efficient predators of potential pests (Johnson 2002). Some species have been used as tools for biological control, and some are pollinators (Schatz 2006). They have a wide diversity of larval feeding habits, including feeding on dead wood, herbivory, fungivory and carnivory (Laibner 2000; Johnson 2002).
Sampling method
Flight interception traps were used as passive tree-trunk traps on standing high-dimension (i.e., veteran) trees. We used large traps consisting of three transparent plastic panes (one part 400 mm × 500 mm and two parts 200 mm × 500 mm), a protective top cover (Ø 450 mm) and a funnel leading down into a container holding a solution of water and salt with a small amount of detergent to reduce the surface tension of the liquid. This solution preserved the insects but did not attract them. Each trap was fixed to a trunk with small nail and wire on the top and fastened by wire around the trunk and trap. Each trap was placed on the trunk of a tree at a height of 2 m facing the south (Fig. 2). All traps (n = 16; minimum inter-trap distance = 50 m) were placed in the north-western part of the Game Park which comprises a mosaic of solitary trees in pasture, open canopy and dense pasture woodland. The traps were activated at the middle of May and deactivated at the end of August 2009, which resulted in eight sampling efforts that were made fortnightly. Thus, each trap was working for a period of 106 days (i.e., 1,696 days for our trapping design).
Study variables
To avoid over-fitting and resulting biases we included no more than n/3 predictor variables, where n is the total sample size (Crawley 2002). All study predictor variables were recorded at the tree level and species richness of click beetles was the dependent variable and five predictor variables used. One predictor variable was continuous – the circumference of the trunk (circumference) measured in centimetres in 1.3 m height (mean = 344.25; min = 177.00; max = 486.00 cm). One predictor variable was semi-continuous – tree height (height), rounded up in meters (mean = 10.6; min = 3.00; max = 18.00 m). Besides these two predictors, we used three categorical predictor variables. These were recorded on an ordinal scale, as in previous studies (Buse et al. 2007; Allen et al. 2010). The first was tree species (tree species) and two species were used for this study—beech (Fagus sylvatica; coded as 1; n = 11) and oak (Quercus petraea; coded as 0; n = 5). The vitality of tree (vitality) was divided into three categories; dead (2; n = 5), withered (1; n = 8) and living (0; n = 3). In order to depict the sun-exposure gradient (sun-exposure) we distinguished the following three categories related to the light incidence of the trees—sun-exposed (solitary) trees (2; n = 6), semi-shaded trees (1; n = 5) and shaded trees (0; n = 5). Most of the study trees were formerly grown in open canopy or planted as solitary trees, which was indicated by the higher dimensions of their stems compared to the surrounding trees (i.e., in the case of semi-shaded and shaded trees) and the presence of thick branches and limbs in the lower canopy. Solitary trees were sampled in the pasture area, shaded trees in the forest stand (growing under the recent closed forest canopy) and semi-shaded trees in groups of trees between the pasture and the forest. The range of selected trees approximately reflected the conditions of the study area.
Statistical analyses
For the analysis of total species richness we computed species accumulations with confidence intervals using sample-based rarefaction (Mao Tau function with 95 % CI; Gotelli and Colwell 2001; Colwell 2006) and the Chao function (Chao 1984). Analyses were computed using EstimateS 8.2. The number of randomizations was set at 1,000 with strong hash encryption and the randomization of samples without replacements. We used the classic formulae for Chao for bias correction (Colwell 2006).
Statistical analyses to evaluate the response of click beetles to the study predictor variables were carried out using Statistica 7.0 (StatSoft Inc., Tulsa, OK, USA). We used species richness (i.e., the number of species trapped) as the dependent variable. Normality was tested with the Shapiro-Wilks test (Hill and Lewicki 2007).
To reduce the effect of multicolinearity, the covariance matrix was examined and predictor variables with significant Pearson’s correlation coefficients (p < 0.05) were removed step by step from the following analyses using variance inflation factor (VIF ≥ 2) of predictors (Graham 2003; Allen et al. 2010). General linear model (GLM), for data with a normal distribution, was chosen as an appropriate method of statistical analysis (Hill and Lewicki 2007). The study variables were then analysed using generalized linear model (GLZ) with the backwards model selection procedure of the predictors (Hill and Lewicki 2007; Horák et al. 2010) as implemented in Statistica. The first approach used in the GLZ with the Logit link function was with all effects included, followed by final model building (p < 0.01).
An analysis aimed at identifying species composition and species responses to the classes of predictor variables derived from the final model was carried out CANOCO for Windows version 4.5 (ter Braak and Šmilauer 2002). Redundancy analysis (RDA), a constrained linear ordination method, was used to resolve the response of click beetles to the study variables. We used focused scaling on inter-species correlations and species scores divided by SD. Square-root transformation of species data and Monte-Carlo permutation tests with a test of the significance of the first ordination axis and canonical axes together and with the number of permutations set at 9,999 in the full model were also used (Lepš and Šmilauer 2003). The resulting ordination diagrams were created in CanoDraw 4.14 (ter Braak and Šmilauer 2002). The projection the centroids of individual classes (in right-angle direction) on individual species arrow show the average value of species in individual classes. The species with the longest distance from the zero point [0; 0] (in the positive direction) had the largest average abundance in particular class. Long distance in opposite (negative) direction show that the species had a lower average predicted abundance (Lepš and Šmilauer 2003). Statistical significance of associations between species and categories of variable were tested using T value biplots with Van Dobben circles as implemented in CANOCO (Lepš and Šmilauer 2003).
We also separated the study species into ecological groups, which is a method often used in beetle studies (Johansson et al. 2007; Goßner et al. 2008). The groups analysed were made up of (1) non-saproxylic and saproxylic species (Schmidl and Bußler 2004), which reflected the larval development strategy (i.e., non-saproxylic in soil and saproxylic in wood; Laibner 2000) (2) guilds (mould and old dead wood) of saproxylic species (Schmidl and Bußler 2004) and (3) functional groups separated into phytophages, predators and scavengers (Laibner 2000). Descriptive analyses of subgroups were done with the parametric ANOVA (d.f. = 2) and finally compared using Fisher’s LSD post hoc comparisons in Statistica (Hill and Lewicki 2007).
Results
During the study we trapped 33 species of click beetles (301 individuals; see Table 2), which comprised 49.3 % of the known fauna in the area from the recent decade (Rébl 2010).
Species accumulation
The species accumulation curve for the entire dataset did not reach an asymptote, but it approached the Chao 1 estimate of the total number of species (Fig. 3), suggesting that the majority of the species in the study site were represented in the analysis and the number of traps was sufficient.
Total species accumulation and estimate of total species richness of the trapped click beetles (Elateridae) in Lány Game Park pasture woodland. Complete data for the click beetle species from all traps are included. The solid line shows the species accumulation curve, a sample-based rarefaction of assemblages, the two surrounding dashed lines are Mao Tau estimates with 95 % CI and the upper dashed line is the Chao 1 estimate of the total number of species
Species richness of click beetles and response to study predictor variables
The final approach built with p < 0.01 was used to create a multivariate model for the species richness of click beetles and their response to the study variables. This model considered sun-exposure as the only responsible predictor variable of the trapping success of click beetles in the study area with more than 60 % of adjusted explained variability (Table 1). All of the other study variables were removed due to multicolinearity (i.e., circumference with VIF = 2.14) and during the selection procedure (i.e., vitality, height and tree species).
Click beetle species richness and sun-exposure gradient
The trapped click beetle species preferred sun-exposed (solitary) trees and avoided shaded trees under the closed canopy. Therefore, we tested the differences between categories of sun exposure. Sun-exposed trees supported a greater number of species than either semi-shaded (p = 0.0004) and shaded (p = 0.0001) trees. No difference was observed between semi-shaded and shaded trees, as illustrated in Fig. 4, using sample-based rarefaction.
Species composition of click beetles and gradient of sun-exposure
The first axis (explained variance = 22.8 %; F = 3.83; p = 0.0001) in redundancy analysis of the species data represented a sun-exposure gradient. All canonical axes together explained 26.9 % of the variance (F = 2.40; p = 0.0004) and showed that most of the trapped click beetles preferred sun-exposed trees, whereas a minority preferred semi-shaded or shaded trees (Fig. 5; Table 2).
Upper RDA ordination diagram resulting from the analysis of species composition and sun-exposure gradient, species indicated by full name were statistically significantly associated with the closest category of sun-exposure. The projection the centroids of individual classes (in right-angle direction) on individual species arrow show the average value of species in individual classes. The species with the longest distance from the [0; 0] point of upper figure (in the positive direction) had the largest mean abundance in particular class. Long distance in opposite (negative) direction show that the species had a lower mean predicted abundance. Particular species are denoted by arrows with abbreviations: Agrypnus murinus (Ag_mur), Ampedus brunnicornis (Am_bru), Ampedus cinnaberinus (Am_cin), Ampedus nigroflavus (Am_nig), Ampedus pomorum (Am_pom), Ampedus praeustus (Am_pra), Ampedus rufipennis (Am_ruf), Ampedus sanguinolentus (Am_sgl), Ampedus sanquineus (Am_sqn), Athous bicolor (At_bic), Athous haemorrhoidalis (At_hae), Athous vittatus (At_vit), Athous zebei (At_zeb), Brachygonus megerlei (Br_meg), Calambus bipustulatus (Cl_bip), Cardiophorus gramineus (Cr_gra), Cardiophorus ruficollis (Cr_ruf), Dalopius marginatus (Da_mar), Denticollis linearis (De_lin), Hypoganus inunctus (Hy_inu), Lacon querceus (La_que), Limonius aeneoniger (Li_aen), Procraerus tibialis (Pc_tib), Prosternon tesselatum (Ps_tes), Selatosomus aeneus (Se_aen) and Stenagostus rhombeus (St_rho). Lower Pie symbols plot visualising distribution of species with different response to the level of sun-exposure. White is for sun-exposed, grey for semi-shaded and black for shaded habitats
Sun-exposure and diversity of ecological groups
The response of saproxylic species to sun-exposure was significantly positive (F = 4.69; p = 0.0292). Species diversity was higher on sun-exposed trees than shaded trees (p = 0.0098). The other categories did not differ. Non-saproxylic species also responded in a significant way to the sun-exposure gradient (F = 8.85; p = 0.0038). Sun-exposed habitats supported a greater number of species than shaded (p = 0.0033) and even semi-shaded (p = 0.0033) trees. No difference was observed between semi-shaded and shaded trees (Fig. 6).
Species richness of saproxylic and non-saproxylic click beetles in relation to the gradient of sun-exposure in Lány Game Park pasture woodland. Significant differences are abbreviated by 1 for sun-exposed, 2 for semi-shaded and 3 for shaded trees. Black squares show means, boxes show SE and whiskers show SD
The functional group of phytophagous click beetles responded in a significant way to the level of sun-exposure (F = 18.74; p = 0.0002). We trapped a greater number of species on sun-exposed trees than on semi-shaded (p = 0.0003) or shaded (p = 0.0001) trees, and no difference was observed between semi-shaded and shaded trees. The response of scavengers and predators was not significant. The species richness of scavengers was higher on sun-exposed trees than on shaded trees (p = 0.0404; Fig. 7).
Species richness of phytophagous, predator and scavenger functional groups of click beetles in relation to the gradient of sun-exposure in Lány Game Park pasture woodland. Significant differences are abbreviated by 1 for sun-exposed, 2 for semi-shaded and 3 for shaded trees. Black squares show means, boxes show SE and whiskers show SD
We also evaluated the response of two different saproxylic guilds. The species inhabiting wood mould in cavities did not respond to the sun-exposure gradient but the species dependent on old and rotten dead wood responded in a significant way (F = 7.74; p = 0.0061). We found that old dead wood species reached a higher richness on sun-exposed trees than on shaded trees (p = 0.0019), and the same results were observed for sun-exposed versus semi-shaded trees (p = 0.0321). We did not observe any differences between semi-shaded and shaded trees, similar to the previous cases (Fig. 8).
Species richness of saproxylic click beetles dependent on mould and old dead wood in relation to the gradient of sun-exposure in Lány Game Park pasture woodland. Significant differences are abbreviated by 1 for sun-exposed, 2 for semi-shaded and 3 for shaded trees. Black squares show means, boxes show SE and whiskers show SD
Discussion
Křivoklátsko is known for its high level of biodiversity (Hůla 2009). A relatively high proportion—62.9 % (83 species) of the click beetle fauna known from Bohemia (Czech Republic) was previously observed there (Jelínek 1993; Dušánek and Mertlik 2010; Rébl 2010), although only 67 species have been observed during the last decade (Rébl 2010). During our study we trapped 33 species, which comprised 39.8 % of the known fauna in the area (Rébl 2010), thus we trapped nearly one half of the recently known fauna of Křivoklátsko during one season. Most of the other (i.e., not trapped) species are not connected with forest habitats (e.g., stream habitats), occur with low population densities or are highly sedentary (e.g., in cavities; Laibner 2000; Rébl 2010).
Our results showed that the sun-exposure gradient was highly important for click beetle species richness and that the majority of the species with significant responses preferred solitary trees in fully sun-exposed habitats. These results support those of other studies, which predicted (Vera 2000; Sutherland 2002) and evaluated (Lindhe et al. 2005; Franc and Götmark 2008; Horák et al. 2012) the openness of forests as being one of the main factors supporting a high level of forest biodiversity.
The former natural factors that created the heterogeneous mosaic of large areas with sun-exposed living, withering and dead trees have almost been eliminated. Forest fires, as the leading factor of forest disturbances, are nowadays limited to only a few forest reserves and in most landscapes they are avoided (Lindhe et al. 2005; Lindenmayer et al. 2008). The same situation is found with forest pasture in most temperate forest areas (Ranius and Jansson 2000; Vera 2000). Thus, as our study showed, game parks probably provide one of the last refugees of former mosaics of forests and woodlands with mixed successional stages and forest-grassland ecotonal areas.
Closed canopies in commercial forests were cultivated since they were thought to be highly stable with such stands being predicted to be more resistant to abiotic and biotic agents such as windstorms, snow breakages and insect outbreaks (Křístek et al. 2002; Mason 2007; Jonasova et al. 2010). However, in the Czech Republic (78,863 km2 total area; 34 % is forested land), for example, more than 14 million m3 of salvage cutting in closed canopy commercial stands was taken after the windstorms Kyrill in 2007 and Emma in 2008 (Příhoda 2008), thus approximately 0.5 m3 of salvage cutting per hectare was lost in only 2 years. In addition, recent studies showed that open canopies compared to closed canopies support greater species diversity and do not seem to supported fatal pest densities (e.g., Martikainen et al. 1996; Wermelinger et al. 2007).
As beetles are ectothermic their activity is dependent on temperature. Thus, it has been predicted that their trapping success almost reflects insect activity rather than their requirements—i.e., greater numbers of individuals occur in warm, sun-exposed sites than in cooler, less sun-exposed sites (e.g., Sverdrup-Thygeson and Ims 2002). Our results show that the species richness (not abundance) of click beetles, which are thought to be highly adapted to forest habitats (i.e., saproxylics with larval development in wood and old dead wood associates), was significantly higher in the sun than in the shade. This is in agreement with a recent study on the larval habitats of early arriving saproxylic beetles (Vodka et al. 2009).
No other habitat parameter significantly influenced the species richness of click beetles compared to the sun-exposure gradient—even if tree species is one of the traditionally studied habitat parameters (Lindhe and Lindelöw 2004; Horák 2011). Our study was undertaken in an area where oak and beech dominate the tree species composition with long temporal continuity. Both tree species (oak and beech) are thought to be key species for the biodiversity of temperate forests (Jonsell et al. 1998; Lindbladh et al. 2007; Vodka et al. 2009) and veteran oaks and beeches in particular support a rich fauna (Schiegg 2001; Ranius 2002; Müller and Goßner 2007; Buse et al. 2008; Sverdrup-Thygeson et al. 2010).
It has been shown that tree species with a long temporal continuity and high level of sun-exposure are key factors for the occurrence of large diameter (i.e., veteran) trees and thus of many of their associated species (Lindbladh et al. 2007; Alexander 2008; Sverdrup-Thygeson et al. 2010) many of which are often dispersal limited (e.g., Ranius and Hedin 2001). Thus the long spatial and temporal continuity of sun-exposed veteran trees (Nilsson and Baranowski 1994), as in our study area, could be a good predictor for sustainable forest management.
We hope that we were able to fill the knowledge gap in selected taxonomical group research using flying interception traps. We used click beetles rather than repeating the experiments of others on wider groups. Our results showed that diversified family of click beetles seems to be applicable in similar research activities.
Our results indicate that click beetles, surveyed by flight interception traps may be a useful bioindicator group and appropriate sampling methodology (see Niemelä et al. 1993; Rainio and Niemelä 2003 for pitfall traps and ground beetles). Furthermore, species accumulation rates and comparisons with other beetle sampling in the same location (Rébl 2010) also indicate that mass trapping designs are not necessary for this type of study (e.g., Martikainen and Kaila 2004).
In conclusion, solitary (sun-exposed) veteran trees seem to be an essential part of the maintenance of high species richness of click beetles. The spatial and temporal mosaic of open and closed stands created by the pasturing of large herbivores in woodlands can restore a forest habitat, which is beneficial to many organisms. Our results show that one of the most ecologically diverse beetle families, click beetles, and its species richness, benefits from a high level of sun exposure. Thus, modern forest management nowadays should have a component that focuses on opening forest stands rather than on generalized conservation activities through protecting forests by non-interventional (hands-off) practices that may often lead to canopy closure. Management which focuses on open stands will promote those forest taxa that are dependent on the activity of large herbivores, a natural component present in the past (Vera 2000) that is now missing.
References
Alexander KNA (2008) Tree biology and saproxylic Coleoptera: issues of definitions and conservation language. Rev Ecol 63:1–5
Allen KA, Le Duc MG, Thompson DJ (2010) Habitat and conservation of the enigmatic damselfly Ischnura pumilio. J Insect Conserv 14:689–700
AOPK ČR (2010) Lánská obora. http://www.nature.cz/natura2000-design3/web_lokality.php?cast=1805&akce=karta&id=1000002673. Accessed on 26 Oct 2010
Baselga A (2008) Determinants of species richness, endemism and turnover in European longhorn beetles. Ecography 31:263–271
Bohac J (1999) Staphylinid beetles as bioindicators. Agr Ecosyst Environ 74:357–372
Bouchard P, Grebennikov VV, Smith ABT, Douglas H (2009) Biodiversity of Coleoptera. In: Footit RG, Adler PH (eds) Insect biodiversity: science and society. Wiley-Blackwell, UK, pp 265–302
Buse J, Schröder B, Assmann T (2007) Modelling habitat and spatial distribution of an endangered longhorn beetle—A case study for saproxylic insect conservation. Biol Conserv 137:372–381
Buse J, Ranius T, Assmann T (2008) An endangered longhorn beetle associated with old oaks and its possible role as an ecosystem engineer. Conserv Biol 22:329–337
Chao A (1984) Non-parametric estimation of the number of classes in a population. Scand J Stat 11:265–270
Colwell RK (2006) EstimateS: statistical estimation of species richness and shared species from samples. Version 8. http://viceroy.eeb.uconn.edu/estimates. Accessed on 5 May 2010
Crawley MJ (2002) Statistical computing—an introduction to data analysis using S-Plus. Wiley, UK
Dušánek V, Mertlik J (2010) Elateridae. Click beetles of the Palearctic region. http://www.elateridae.com. Accessed on 26 Oct 2010
Farkač J, Král D, Škorpík M (2005) Red list of threatened species in the Czech Republic. Invertebrates. Agentura ochrany přírody a krajiny ČR, Praha
Foit J (2010) Distribution of early-arriving saproxylic beetles on standing dead Scots pine trees. Agr For Entomol 12:133–141
Franc N, Götmark F (2008) Openness in management: hands-off vs partial cutting in conservation forests, and the response of beetles. Biol Conserv 141:2310–2321
Goldberg E, Kirby K, Hall J, Latham J (2007) The ancient woodland concept as a practical conservation tool in Great Britain. J Nat Conserv 15:109–119
Goßner M, Engel K, Jessel B (2008) Plant and arthropod communities in young oak stands: are they determined by site history? Biodiv Conserv 17:3165–3180
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4:379–391
Graham MH (2003) Confronting multicollinearity in ecological multiple regression. Ecology 84:2809–2815
Grove SJ (2002) Saproxylic insect ecology and the sustainable management of forests. Annu Rev Ecol Syst 33:1–23
Hanzák J, Moucha J, Zahradník J (1973) Světem zvířat V. díl (2. část). Bezobratlí. Albatros, Praha
Hicks H, Blackshaw RP (2008) Differential responses of three Agriotes click beetle species to pheromone traps. Agr For Entomol 10:443–448
Hill T, Lewicki P (2007) Statistics methods and applications. StatSoft, Tulsa
Hofmeister J, Mihaljevič M, Hošek J (2004) The spread of ash (Fraxinus excelsior) in some European oak forests: an effect of nitrogen deposition or successional change? For Ecol Manag 203:35–47
Horák J (2011) Response of saproxylic beetles to tree species composition in a secondary urban forest area. Urb For Urb Gree 10:213–222
Horák J, Vávrová E, Chobot K (2010) Habitat preferences influencing populations, distribution and conservation of the endangered saproxylic beetle Cucujus cinnaberinus at the landscape level. Eur J Entomol 107:81–88
Horák J, Chumanová E, Hilszczański J (2012) Saproxylic beetle thrives on the openness in management: a case study on the ecological requirements of Cucujus cinnaberinus from Central Europe. Insect Conserv Divers. doi:10.1111/j.1752-4598.2011.00173.x
Hůla P (2009) Křivoklátsko Protected Landscape Area. Ochr Přír 1:2–5
Jelínek J (1993) Check-list of Czechoslovak insects IV (Coleoptera). Fol Heyrovsk 1:1–172
Johansson T, Hjältén J, Gibb H, Hilaszczański J, Stenlid J, Ball JP, Alnvi O, Danell K (2007) Variable response of different functional groups of saproxylic beetles to substrate manipulation and forest management: implications for conservation strategies. For Ecol Manag 242:496–510
Johnson PJ (2002) Family Elateridae Leach, 1815. In: Arnett RH, Thomas MC, Skelley PE, Frank JH (eds) American beetles, vol 2., Polyphaga: Scarabeoidea through Curculionoidea. CRC Press, Boca Raton, pp 160–173
Jonasova M, Vavrova E, Cudlin P (2010) Western Carpathian mountain spruce forest after a windthrow: natural regeneration in cleared and uncleared areas. For Ecol Manag 259:1127–1134
Jonsell M, Weslien J, Ehnström B (1998) Substrate requirements of red-listed saproxylic invertebrates in Sweden. Biodiv Conserv 7:749–764
Kaila L, Martikainen P, Puntilla P, Yakovlec E (1994) Saproxylic beetles (Coleoptera) on dead birch trunks decayed by different polypore species. Ann Zool Fenn 31:97–107
Kaila L, Martikainen P, Punttila P (1997) Dead trees left in clearcuts benefit saproxylic Coleoptera adapted to natural disturbances in boreal forest. Biodiv Conserv 6:1–18
Komonen A (2007) Are we conserving peripheral populations? An analysis of range structure of longhorn beetles (Coleoptera: Cerambycidae) in Finland. J Insect Conserv 11:281–285
Konstantinov AS, Korotyaev BV, Volkovitsh MG (2009) Insect biodiversity in the Palaearctic region. In: Footit RG, Adler PH (eds) Insect biodiversity: science and society. Wiley-Blackwell, UK, pp 107–162
Křístek J et al (2002) Ochrana lesů a přírodního prostředí. Matice Lesnická, Písek
Laibner S (2000) Elateridae of the Czech and Slovak Republics. Kabourek, Zlín
Lepš J, Šmilauer P (2003) Multivariate analysis of ecological data using CANOCO. Cambridge University Press, UK
Lindbladh M, Brunet J, Hannon G, Niklason M, Eliasson P, Eriksson G, Ekstrand A (2007) Forest history as a basis for ecosystem restoration—A multidisciplinary case study in a south Swedish temperate landscape. Restor Ecol 15:284–295
Lindenmayer DB, Franklin JF, Fischer J (2008) General management principles and a checklist of strategies to guide forest biodiversity conservation. Biol Conserv 131:433–445
Lindhe A, Lindelöw Å (2004) Cut high stumps of spruce, birch, aspen and oak as breeding substrates for saproxylic beetles. For Ecol Manag 203:1–20
Lindhe A, Lindelow A, Asenblad A (2005) Saproxylic beetles in standing dead wood density in relation to substrate sun-exposure and diameter. Biodiv Conserv 14:3033–3053
Löbl I, Smetana A (2007) Catalogue of Palaearctic Coleoptera, vol. 4. Elateroidea, Derontoidea, Bostrichoidea, Lymexyloidea, Cleroidea and Cucujoidea. Apollo Books, Svenstrup, Denmark
Martikainen P, Kaila L (2004) Sampling saproxylic beetles: lessons from a 10-year monitoring study. Biol Conserv 120:171–181
Martikainen P, Siitonen J, Kaila L, Punttila P (1996) Intensity of forest management and bark beetles in non-epidemic conditions: a comparison between Finnish and Russian Karelia. J Appl Entomol 120:257–264
Mason WL (2007) Changes in the management of British forests between 1945 and 2000 and possible future trends. Ibis 149:41–52
McGeoch MA, Schroeder M, Ekbom B, Larsson S (2007) Saproxylic beetle diversity in a managed boreal forest: importance of stand characteristics and forestry conservation measures. Divers Distrib 13:418–429
Mertlik J (2010) A review of findings of click-beetle Agriotes gallicus Lacordaire, 1835 and jewel-beetle Anthaxia candens (Panzer, 1792), known from eastern Bohemia (Czech Republic). Elateridarium 4:33–67
Moga CI, Hartel T, Öllerer K (2009) Ancient oak wood-pasture as a habitat for the endangered tree pipit Anthus trivialis. Biologia 64:1011–1015
Müller J, Goßner M (2007) Single host trees in a closed forest canopy matrix: a highly fragmented landscape? J Appl Entomol 131:613–620
New TR (2007) Beetles and conservation. J Insect Conserv 11:1–4
Niemelä J, Langor D, Spence JR (1993) Effects of clear-cut harvesting on boreal ground-beetle assemblages (Coleoptera: Carabidae) in western Canada. Conserv Biol 7:551–561
Nieto A, Alexander K (2010) European red list of saproxylic beetles. Publication Office of the EU, Luxembourg
Nilsson SG, Baranowski R (1994) Indicators of megatree continuity—Swedish distribution of click beetles (Coleoptera: Elateridae) dependent on hollow trees. Ent Tidskr 115:81–97
Novotny V, Miller SE, Baje L, Balagawi S, Basset Y, Cizek L, Craft KJ, Dem F, Drew RAI, Hulcr J, Leps J, Lewis O, Pokon R, Stewart AJA, Weiblen GD (2010) Guild-specific patterns of species richness and host specialization in plant-herbivore food webs from a tropical forest. J Anim Ecol 79:1193–1203
Økland B (1996) A comparison of three methods of trapping saproxylic beetles. Eur J Entomol 93:195–209
Oliver I, Beattie AJ (1996) Designing a cost-effective invertebrate survey: a test of methods for rapid assessment of biodiversity. Ecol Appl 6:594–607
Parker E, Howard JJ (2001) The biology and management of wireworms (Agriotes spp.) on potato with particular reference to the UK. Agr For Entomol 3:85–98
Příhoda J (2008) Odhady škod po orkánu Kyrill (2007) a vichřici Emma (2008) v České Republice. Les Práce 4:6
Rackham O (2006) Woodlands. Collins, London
Rainio J, Niemelä J (2003) Ground beetles (Coleoptera: Carabidae) as bioindicators. Biodiv Conserv 12:487–506
Ranius T (2002) Osmoderma eremita as an indicator of species richness of beetles in tree hollows. Biodiv Conserv 11:931–941
Ranius T, Hedin J (2001) The dispersal rate of a beetle, Osmoderma eremita, living in tree hollows. Oecologia 126:363–370
Ranius T, Jansson N (2000) The influence of forest regrowth, original canopy cover and tree size on saproxylic beetles associated with old oaks. Biol Conserv 95:85–94
Rébl K (2010) Results of faunistic survey of beetles (Coleoptera) in the territory of Protected Landscape Area and Biospheric Reservation Křivoklátsko (Czech Republic). Elateridarium 4:1–253
Schatz B (2006) Fine scale distribution of pollinator explains the occurrence of the natural orchid hybrid × Orchis bergonii. Ecoscience 13:111–118
Schiegg K (2001) Saproxylic insect diversity of beech: limbs are richer than trunks. For Ecol Manag 149:295–304
Schmidl J, Bußler H (2004) Ökologische Gilden xylobionter Käfer Deutschlands. Naturschutz Landschaftsplan 36:202–218
Siitonen J (1994) Decaying wood and saproxylic Coleoptera in two old spruce forests: a comparison based on two sampling methods. Ann Zool Fenn 31:89–95
Siitonen J, Saaristo L (2000) Habitat requirements and conservation of Pytho kolwensis a beetle species of old-growth boreal forest. Biol Conserv 94:211–220
Škoudlínová A (2000) Lánská obora. Okresní úřad Rakovník a Okresní úřad Kladno
Spitzer L, Konvicka M, Benes J, Tropek R, Tuf IH, Tufova J (2008) Does closure of traditionally managed open woodlands threaten epigeic invertebrates? Effects of coppicing and high deer densities. Biol Conserv 141:827–837
Sutherland WJ (2002) Openness in management. Nature 418:834–835
Sverdrup-Thygeson A, Ims RA (2002) The effect of forest clearcutting in Norway on the community of saproxylic beetles on aspen. Biol Conserv 106:347–357
Sverdrup-Thygeson A, Skarpaas O, Ødegaard F (2010) Hollow oaks and beetle conservation: the significance of the surroundings. Biodiv Conserv 19:837–852
ter Braak CJF, Šmilauer P (2002) CANOCO reference manual and CanoDraw for windows user’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
Tolasch T, Von Vragstein M, Steidle JLM (2007) Sex pheromone of Elater ferrugineus L. (Coleoptera: Elateridae). J Chem Ecol 33:2156–2166
Vera FWM (2000) Grazing ecology and forest history. CABI Publishing, Oxford
Vodka S, Konvicka M, Cizek L (2009) Habitat preferences of oak-feeding xylophagous beetles in a temperate woodland: implications for forest history and management. J Insect Conserv 13:553–562
Wermelinger B, Flückinger PF, Obrist MK, Duelli P (2007) Horizontal and vertical distribution of saproxylic beetles (Col., Buprestidae, Cerambycidae, Scolytinae) across sections of forest edges. J Appl Entomol 131:104–114
Zicha O (2010) Biological library—BioLib. http://www.biolib.cz/. Accessed on 18 Dec 2010
Acknowledgments
We would like to thank the zoologist of PLA Křivoklátsko A. Hoffmannová, and all of the forest and game managers in the Lány Game Park (LS Lány), especially J. Janda, M. Štrunc, L. Frank and Š. Stanický for their deeply kind support during our study and the permission to access the study sites. E. Chumanová helped with the statistical analyses, A. Kohutka helped in the field, Z.F. Fric had many good ideas for improving this manuscript, T. Rose reviewed the English, two anonymous reviewers provided many good suggestions and editor provided a very careful final revision of the text. This project was funded by the contract PPK-2a/24/10 of non-governmental organization Lesák, o.s. (www.lesak.eu) and a grant from the CZ Ministry of Environment MSM 6293359101 of VÚKOZ, v.v.i. (www.vukoz.cz).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horák, J., Rébl, K. The species richness of click beetles in ancient pasture woodland benefits from a high level of sun exposure. J Insect Conserv 17, 307–318 (2013). https://doi.org/10.1007/s10841-012-9511-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-012-9511-2