Abstract
Purpose
The posterior wall of left atrium plays an important role in atrial fibrillation (AF) recurrence, but the benefit of left atrial posterior wall isolation (PWI) remains still unclear. The objective was to evaluate the benefit of PWI in radiofrequency ablation.
Methods
PubMed and the Web of Science were searched in September 2015. Studies comparing catheter ablation with PWI [PWI(+)] vs. ablation without PWI [PWI(−)] were included. We performed a meta-analysis to assess atrial arrhythmia recurrence, procedure-related complications, and procedural time.
Results
Five studies with 594 AF patients were included. Compared with PWI(−), PWI(+) resulted in a significantly lower atrial arrhythmia recurrence rate (relative risk [RR] 0.81, 95 % confidence interval [CI] 0.68–0.97, p = 0.02), which was largely driven by the decreased AF recurrence (RR 0.55, 95 % CI 0.35–0.86, p = 0.009). Recurrence rates of atrial tachycardia/flutter (AT/AFL) were comparable between two groups (RR 1.16, 95 % CI 0.85–1.58, p = 0.34). There were no significant differences in procedure-related complications (RR 1.07, 95 % CI 0.45–2.53, p = 0.89) and procedural times (weight mean difference 0.88, 95 % CI −7.29–9.06, p = 0.83).
Conclusions
This meta-analysis shows that with comparable procedure-related complications and procedural time, ablation with PWI reduces AF recurrence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Atrial fibrillation (AF) is one of the most common arrhythmias and plays an important role in stroke and death [1, 2]. Catheter ablation has proven to be an effective treatment for drug-refractory AF, and pulmonary vein isolation (PVI) is the cornerstone for radiofrequency ablation [3]. However, recurrences are quite common even after successful PVI [4].
Studies have reported non-pulmonary vein (PV) ectopic beats on left atrial posterior wall that can initiate AF [5]. Therefore, left atrial posterior wall isolation (PWI) may be able to prevent the arrhythmogenic effect of these triggers. However, the benefit of PWI in radiofrequency catheter ablation remains unclear.
Although several trials comparing radiofrequency ablation with PWI [PWI(+)] vs. ablation without PWI [PWI(−)] have been reported, the results are conflicting [6–8]. Therefore, we performed a meta-analysis of all available data to assess the effect of PWI for patients with AF.
2 Methods
2.1 Search strategy
We performed an electronic search in September 2015 in PubMed and the Web of Science. No restrictions were applied. The following terms were searched in [Title/Abstract]: atrial fibrillation, isolation, posterior wall, and posterior atrium. We also examined the bibliographies of all identified studies and reviews for potential additional reports. When several studies describing the same population were published, the most recent or complete one was selected.
2.2 Inclusion criteria
Randomized controlled trials (RCTs) or non-randomized trials that compared PWI(+) with PWI(−) in the primary catheter ablation were included. To be included, studies were required to report the recurrence of any atrial arrhythmia with a follow-up period of at least 3 months. There was no restriction pertaining to AF type. Any ablation technique was accepted as long as PWI was achieved in the intervention group. Except for PWI, studies had to adopt comparable ablation in both groups. Conference abstracts were excluded because they did not provide sufficient data.
2.3 Outcomes
The primary outcome was recurrence of any atrial arrhythmia during follow-up. If sufficient data were available, atrial arrhythmia was subdivided into AF and atrial tachycardia/flutter (AT/AFL). Secondary outcomes were procedure-related complication rate and procedural time.
2.4 Data extraction
Two reviewers (Xin He and Yue Zhou) independently extracted the data from the included studies into a standard form. A senior reviewer (Jiangui He) was responsible for adjudication of disagreement.
2.5 Quality assessment
The methodological quality of RCTs was assessed by the components recommended by the Cochrane Collaboration [9]. As described in previous published article [10], we used the modified Newcastle-Ottawa scale for quality assessment of non-randomized trials. The quality of each trial except RCTs was quantified by a score of 0–9.
2.6 Data analysis
All meta-analyses were performed using Review Manager Version 5.3 (The Cochrane Collaboration, 2014). The weighted mean difference and the relative risk (RR) were used to compare continuous and dichotomous variables, respectively. Ninety-five percent confidence intervals (CIs) and p values were also reported.
The statistical heterogeneity noted among the studies was assessed using the I 2 test. The heterogeneity among studies was defined significant if I 2 > 50 %. We used a random-effects model if there was significant heterogeneity among the studies; otherwise, we used a fixed-effects model [9]. Sensitivity analysis including only RCTs was performed. Potential publication bias was tested using funnel plots.
3 Results
3.1 Search results
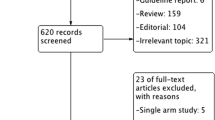
Our initial search identified 421 references, of which 100 were duplications and 306 were excluded by screening based on titles and abstracts. Six conference abstracts, one editorial, and one sub-study of a larger RCT were excluded by full-text reading. Two trials aiming to compare PWI(+) with PWI(−) were excluded because PWI was not achieved in most of the patients in the intervention group. Finally, five studies were included (Fig. 1).
3.2 Study characteristics
Table 1 summarizes the baseline characteristics of each study. Three studies involved both paroxysmal and persistent AF patients [8, 11, 12], while the other two studies involved only patients with persistent AF [6, 7]. Of the included studies, three were RCTs [7, 8, 11], one was prospective observational trial [6], and one used matched historical control [12]. None of the studies reported any patients with previous ablation. Table 2 summarizes the methodological characteristics of each study. To achieve PWI, two studies adopted single-ring ablation technique [11, 12], two studies connected two PV-circling lesions by two linear lesions [7, 8], and one study performed extensive ablation on the left atrial posterior wall [6]. PWI was defined as entrance block and complete electrical silence in one study [6], entrance block and exit block in three studies [7, 8, 11], and only entrance block confirmed by mapping in one study [12].
3.3 Study quality
The quality assessments of included studies are shown in Supplementary Material. Of the three RCTs, two mentioned the detailed method of randomization [7, 8] and only one provided information on blinding [8]. None of the three studies provided details about allocation concealment. As for non-randomized studies, one scored 8 in the assessment [6] and the other scored 7 [12] (Online Resource Table S1 and S2).
3.4 Primary outcomes
Five studies reported the recurrence rates of atrial arrhythmia. Compared with PWI(−), PWI(+) showed a significantly lower atrial arrhythmia recurrence rate (RR 0.81, 95 % CI 0.68–0.97, p = 0.02). There was no significant heterogeneity among studies (I 2 = 41 %, p = 0.15) (Fig. 2).
Further analyses showed that AF recurrence was significantly reduced in PWI(+) group (RR 0.55, 95 % CI 0.35–0.86, p = 0.009) (Fig. 3), while AT/AFL recurrence rates were comparable between two groups (RR 1.16, 95 % CI 0.85–1.58, p = 0.34) (Fig. 4). The heterogeneity among studies was significant for AF recurrence (I 2 = 62 %, p = 0.03), but not for AT/AFL recurrence (I 2 = 29 %, p = 0.24).
3.5 Secondary outcomes
3.5.1 Procedure-related complications
Five studies reported the procedure-related complications. The rates of procedure-related complications were comparable between two groups (RR 1.07, 95 % CI 0.45–2.53, p = 0.89). Heterogeneity among studies was not significant (I 2 = 0, p = 0.89) (Online Resource Fig. S1).
3.5.2 Procedural time
Pooling data from six studies showed that there was no significant difference in procedural time between two groups (weight mean difference 0.88, 95 % CI −7.29–9.06, p = 0.83). There was no significant heterogeneity among studies (I 2 = 43 %, p = 0.14) (Online Resource Fig. S2).
3.6 Sensitivity analysis
Table 3 summarizes the results of sensitivity analysis. The reduction of atrial arrhythmia recurrence in PWI(+) was no longer statistically significant. However, AF recurrence rate remained significantly lower in PWI(+) compared with PWI(−).
3.7 Publication bias
Figure 5 depicts a funnel plot of included studies that reported atrial arrhythmia recurrence rates. All the studies lay inside the triangle and distributed symmetrically around the central axis, indicating no significant publication bias.
4 Discussion
In this meta-analysis, we found that PWI significantly reduced the recurrence of atrial arrhythmia after the procedure. The reduction was mainly driven by the reduced AF recurrence. There were no significant differences in procedure-related complications and procedural times between PWI(+) group and PWI(−) group. To our knowledge, this is the first meta-analysis to assess the effect of PWI.
Although PVI is now the cornerstone of treatment for AF, additional ablation has been advocated to increase the long-term success rate of catheter ablation. Animal studies have suggested the role of posterior wall in the maintenance of AF [13, 14]. Electrophysiological study has demonstrated non-PV ectopic beats in left atrial posterior wall [5]. The posterior wall has proven to be an important site initiating and maintaining AF [15, 16]. Satisfactory clinical results have been achieved by ablation strategies with PWI in both catheter ablation and surgical treatment for AF [17–20]. Our meta-analysis further demonstrated the benefit of PWI(+) ablation in patients with AF compared with PWI(−) ablation.
Patients with persistent AF tend to have greater fibrosis extent, and left atrial posterior wall is one of the remodeling sites [21, 22]. While PV firing is considered to be the main trigger of paroxysmal AF, the non-PV ectopic foci play an important role in persistent AF. Therefore, PWI may have greater beneficial effect in persistent AF than in paroxysmal AF. In fact, two included studies [6, 7] that involved only persistent AF reported the most significant reductions in atrial arrhythmia and AF recurrence. Two studies [8, 11] reported trends of better clinical outcomes in PWI(+) than in PWI(−) in persistent AF patients. Although a subgroup analysis assessing the effect of PWI in paroxysmal AF and persistent AF respectively will be ideal, insufficient data limited the opportunity for this analysis.
While the AF recurrence was reduced significantly in PWI(+) group, the difference in AT/AFL recurrence was not statistically significant. A previous study has suggested that the PWI with the single-ring ablation technique may be predisposed to AT/AFL recurrence, and one of the mechanisms is reentry through two gaps in the single-ring lesion [23]. This is not unexpected because long lines of lesions may increase the risk of reconnections. Although there was a trend of higher AT/AFL recurrence rate in PWI(+) group in our analysis, it did not reach statistical significance. As the number of involved patients was limited, the effect of PWI on AT/AFL recurrence remains to be further tested by future high-volume trial.
5 Study limitations
This meta-analysis was characterized by several limitations that had to be taken into account.
First, we included two non-randomized studies in the analysis, which might increase the risk of bias. However, the sensitivity analysis including only RCTs demonstrated that the results were robust, except for atrial arrhythmia recurrence. Although there was still a trend of less atrial arrhythmia recurrence in PWI(+), it did not reach statistical significance in sensitivity analysis. It might be attributed to the limited number of involved patients. Therefore, more clinical trials are needed to confirm the finding.
Second, the included studies adopted different ablation techniques to achieve PWI. Although heterogeneities were not significant in most of the variables, different ablation techniques might result in different clinical outcomes. However, our analysis at least demonstrated the importance of PWI in AF ablation.
Third, the included studies used different definitions for PWI and PVI. Some studies confirmed both entrance block and exit block, and the others confirmed only entrance block. It is believed that checking for block in both directions is of great importance, because there can exist exit conduction after entrance block is confirmed [24]. The difference in definition for PWI and PVI may affect the clinical outcomes and introduce bias into the analysis.
Finally, there was significant variation in lengths of follow-up. The follow-up periods of included studies ranged from 6 months to more than 3 years. Although we demonstrated the additional benefit of PWI by pooling the data from these studies, we could not figure out whether the benefit was long-term or short-term.
6 Conclusion
This meta-analysis demonstrates that ablation with PWI results in a significant reduction in AF recurrence, but not in AT/AFL recurrence. The recurrence of atrial arrhythmia is also significantly reduced. Procedure-related complications and procedural time do not change significantly. This analysis suggests that PWI may be beneficial to AF ablation. Because of several inherent limitations, more large-scale RCTs are needed to evaluate the benefits of PWI.
Abbreviations
- AF:
-
Atrial fibrillation
- PWI:
-
Posterior wall isolation
- PVI:
-
Pulmonary vein isolation
- AT:
-
Atrial tachycardia
- AFL:
-
Atrial flutter
References
Benjamin, E. J., Wolf, P. A., D'Agostino, R. B., Silbershatz, H., Kannel, W. B., & Levy, D. (1998). Impact of atrial fibrillation on the risk of death: the Framingham Heart Study. Circulation, 98(10), 946–52.
Wolf, P. A., Abbott, R. D., & Kannel, W. B. (1991). Atrial fibrillation as an independent risk factor for stroke: the Framingham Study. Stroke, 22(8), 983–8.
Camm, A. J., Lip, G. Y., De Caterina, R., Savelieva, I., Atar, D., Hohnloser, S. H., et al. (2012). 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation—developed with the special contribution of the European Heart Rhythm Association. Europace, 14(10), 1385–413.
Oral, H., Knight, B. P., Tada, H., Ozaydin, M., Chugh, A., Hassan, S., et al. (2002). Pulmonary vein isolation for paroxysmal and persistent atrial fibrillation. Circulation, 105(9), 1077–81.
Lee, S. H., Tai, C. T., Hsieh, M. H., Tsao, H. M., Lin, Y. J., Chang, S. L., et al. (2005). Predictors of non-pulmonary vein ectopic beats initiating paroxysmal atrial fibrillation: implication for catheter ablation. Journal of the American College of Cardiology, 46(6), 1054–9.
Bai, R., Biase L.D., Mohanty P., Trivedi C., Russo A.D., Themistoclakis S., et al. (2015). Proven isolation of the pulmonary vein antrum with or without left atrial posterior wall isolation in patients with persistent atrial fibrillation. Heart Rhythm.
Kim, J. S., Shin, S. Y., Na, J. O., Choi, C. U., Kim, S. H., Kim, J. W., et al. (2015). Does isolation of the left atrial posterior wall improve clinical outcomes after radiofrequency catheter ablation for persistent atrial fibrillation?: a prospective randomized clinical trial. International Journal of Cardiology, 181, 277–83.
Tamborero, D., Mont, L., Berruezo, A., Matiello, M., Benito, B., Sitges, M., et al. (2009). Left atrial posterior wall isolation does not improve the outcome of circumferential pulmonary vein ablation for atrial fibrillation: a prospective randomized study. Circulation. Arrhythmia and Electrophysiology, 2(1), 35–40.
Green, S., & Higgins, J. P. T. (2008). Cochrane handbook for systematic reviews of interventions. Chichester: Wiley-Blackwell.
Taggart, D. P., D'Amico, R., & Altman, D. G. (2001). Effect of arterial revascularisation on survival: a systematic review of studies comparing bilateral and single internal mammary arteries. Lancet, 358(9285), 870–5.
Lim, T. W., Koay, C. H., See, V. A., McCall, R., Chik, W., Zecchin, R., et al. (2012). Single-ring posterior left atrial (box) isolation results in a different mode of recurrence compared with wide antral pulmonary vein isolation on long-term follow-up longer atrial fibrillation-free survival time but similar survival time free of any atrial arrhythmia. Circulation. Arrhythmia and Electrophysiology, 5(5), 968–77.
Thomas, S. P., Lim, T. W., McCall, R., Seow, S. C., & Ross, D. L. (2007). Electrical isolation of the posterior left atrial wall and pulmonary veins for atrial fibrillation: feasibility of and rationale for a single-ring approach. Heart Rhythm, 4(6), 722–30.
Morillo, C. A., Klein, G. J., Jones, D. L., & Guiraudon, C. M. (1995). Chronic rapid atrial pacing. Structural, functional, and electrophysiological characteristics of a new model of sustained atrial fibrillation. Circulation, 91(5), 1588–95.
Mandapati, R., Skanes, A., Chen, J., Berenfeld, O., & Jalife, J. (2000). Stable microreentrant sources as a mechanism of atrial fibrillation in the isolated sheep heart. Circulation, 101(2), 194–9.
Schmitt, C., Ndrepepa, G., Weber, S., Schmieder, S., Weyerbrock, S., Schneider, M., et al. (2002). Biatrial multisite mapping of atrial premature complexes triggering onset of atrial fibrillation. American Journal of Cardiology, 89(12), 1381–7.
Ndrepepa, G., Weber, S., Karch, M. R., Schneider, M. A., Schreieck, J. J., Schomig, A., et al. (2002). Electrophysiologic characteristics of the spontaneous onset and termination of atrial fibrillation. American Journal of Cardiology, 90(11), 1215–20.
Sanders, P., Hocini, M., Jais, P., Sacher, F., Hsu, L. F., Takahashi, Y., et al. (2007). Complete isolation of the pulmonary veins and posterior left atrium in chronic atrial fibrillation. Long-term clinical outcome. European Heart Journal, 28(15), 1862–71.
Kumagai, K., Muraoka, S., Mitsutake, C., Takashima, H., & Nakashima, H. (2007). A new approach for complete isolation of the posterior left atrium including pulmonary veins for atrial fibrillation. Journal of Cardiovascular Electrophysiology, 18(10), 1047–52.
Todd, D. M., Skanes, A. C., Guiraudon, G., Guiraudon, C., Krahn, A. D., Yee, R., et al. (2003). Role of the posterior left atrium and pulmonary veins in human lone atrial fibrillation—electrophysiological and pathological data from patients undergoing atrial fibrillation surgery. Circulation, 108(25), 3108–14.
Voeller, R. K., Bailey, M. S., Zierer, A., Lall, S. C., Sakamoto, S., Aubuchon, K., et al. (2008). Isolating the entire posterior left atrium improves surgical outcomes after the Cox maze procedure. The Journal of Thoracic and Cardiovascular Surgery, 135(4), 870–7.
Platonov, P. G., Mitrofanova, L. B., Orshanskaya, V., & Ho, S. Y. (2011). Structural abnormalities in atrial walls are associated with presence and persistency of atrial fibrillation but not with age. Journal of the American College of Cardiology, 58(21), 2225–32.
Rohr, S. (2012). Arrhythmogenic implications of fibroblast-myocyte interactions. Circulation. Arrhythmia and Electrophysiology, 5(2), 442–52.
Lim, T. W., Koay, C. H., McCall, R., See, V. A., Ross, D. L., & Thomas, S. P. (2008). Atrial arrhythmias after single-ring isolation of the posterior left atrium and pulmonary veins for atrial fibrillation mechanisms and management. Circulation. Arrhythmia and Electrophysiology, 1(2), 120–6.
Riley, M. P. (2015). Techniques to improve and confirm pulmonary vein isolation. Future Cardiology, 11(6), 655–62.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None
Rights and permissions
About this article
Cite this article
He, X., Zhou, Y., Chen, Y. et al. Left atrial posterior wall isolation reduces the recurrence of atrial fibrillation: a meta-analysis. J Interv Card Electrophysiol 46, 267–274 (2016). https://doi.org/10.1007/s10840-016-0124-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-016-0124-7