Abstract
Purpose
Several clinical trials showed inconsistent results of the effect of isolating all versus arrhythmogenic pulmonary veins (PVs) on long-term control of atrial fibrillation (AF). We hypothesized that isolation of arrhythmogenic veins had a comparable success rate to the empiric isolation of all PVs.
Methods
PUBMED, EMBASE, and the Cochrane Library were searched for randomized controlled trials and nonrandomized, observational studies. The efficacy and adverse events of isolating all versus arrhythmogenic PVs were presented as risk ratio (RR) with 95 % confidence intervals (CIs), and weighted mean differences and 95 % CIs were calculated to compare the procedure time and fluoroscopic time between the isolation all PVs and arrhythmogenic PVs.
Results
Six trials with 658 patients were included in the analysis. Isolation of arrhythmogenic PVs was as efficacious as empiric isolation of all PVs in achieving long-term AF control (RR, 0.96; 95 % CI, 0.87–1.05; p = 0.36). Isolation of arrhythmogenic PVs group had shorter procedure time, fluoroscopic time and fewer adverse events than the isolation of all PVs group.
Conclusions
The present analysis suggests that isolation of arrhythmogenic veins had a comparable long-term success rate, shorter procedure time, fluoroscopic time, and fewer adverse events than the empiric isolation of all PVs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Since its original description in 1998 [1], isolation of all pulmonary veins (PVs) has become the routine approach for ablation of atrial fibrillation (AF). Although this strategy has shown good AF control rates, it involves extensive left atrium ablation [2]. Extensive ablation would increase the risk of post-ablation gap-related reentrant tachycardia, post-ablation pro-arrhythmias, collateral damage, and so on. Therefore, selective isolation of only the arrhythmogenic PVs instead of all PVs may be appropriate because of less atrial scarring, fewer post-ablation pro-arrhythmias, and lower radiation exposure and procedure time [3].
The procedures of indentifying the arrhythmogenic PVs are time-consuming because PV triggers of AF can be evanescent, and no established protocol has been consistently shown to reproducibly elicit them [4]. Therefore, selective isolation of arrhythmogenic PVs has not gained wide acceptance. Several randomized, controlled trials (RCTs) and nonrandomized, observational studies (nROSs) tried to compare the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF. Some studies found that isolation of arrhythmogenic PVs was as efficacious as empiric isolation of all PVs in achieving long-term AF control [5–8]. However, some other studies found that elective isolation of arrhythmogenic PVs was associated with a relatively high recurrence rate in the study population or the subgroup [9, 10]. Therefore, it is necessary to conduct a meta-analysis and to assess the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF, and it is also necessary to compare the difference in efficacy of isolating all versus arrhythmogenic PVs between RCTs and nROSs.
2 Materials and methods
2.1 Literature search
A literature search was performed on the PUBMED, EMBASE, Cochrane Library, and Google Scholar databases to identify all the studies that compared the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF. The following search headings were used: AF, PVs isolation, arrhythmogenic PV or selective PVs isolation. For all relevant publications, the records retrieved with the “related articles” link in PUBMED were reviewed; reference lists were checked for other relevant studies. The final literature search was finished on 31th July 2013 and all the literature were limited to English language publication. The major inclusion criteria were: (1) clinical trials published in peer-reviewed journals with full available text in English; (2) clinical trials comparing the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF; and (3) follow-up duration of ≥6 months. The major reasons for exclusion of studies were: (1) overlapping data; (2) data published in the form of abstracts without peer-reviewed publication of manuscripts; and (3) studies in which it was not possible to extract data from the published results as well as those studies that did not report appropriate outcomes.
2.2 Data collection and quality assessment
Two investigators independently reviewed all potentially eligible studies using predefined eligibility criteria and collected data from the included trials. Both RCTs and nROSs comparing the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF were included in the analysis. Any discrepancy was resolved by consensus. Baseline characteristics of patients were extracted as well as data about each trial’s intervention and outcomes assessed. The quality of each study was assessed by evaluating specific elements of each study design, with Jadad quality scale and Newcastle-Ottawa scales for RCTs and nROSs, respectively [11, 12].
2.3 Statistical analysis
Statistical analyses were performed using STATA 11.0 (StataCorp LP, College Station, TX, USA). The efficacy and adverse events of isolating all versus arrhythmogenic PVs were presented as risk ratio (RR) with 95 % confidence intervals (CIs), and weighted mean differences (WMD) and 95 % CIs were calculated to compare the procedure time and fluoroscopic time between the isolating all and arrhythmogenic PVs groups. Heterogeneity was evaluated with Cochran’s Q statistic and quality by I 2 statistic. As there was a significant heterogeneity with p < 0.1, random-effects model was used; otherwise, fixed-effects model was used. Publication bias was evaluated by Begg’s and Egger’s methods. Results were considered statistically significant if p < 0.05. Sensitivity analyses were undertaken by omitting one study at a time to examine influence of one study on the overall summary estimate, and fixed or random effect models described above were used. We explored possible explanations for heterogeneity according to a prior hypothesis, which included differences in the baseline characteristics of patients, designs of studies, types of AF, and the ablation strategies.
3 Results
3.1 Search results
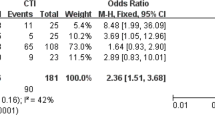
Figure 1 shows the detailed study selection process. A total of 81 potential literature citations were identified through systematic search, with 46 citations being excluded during title and abstract review. The full-text documents of the remaining 35 articles were retrieved. Of theses 35 articles, 29 articles were excluded because of review articles, case report, without inappropriate control group or with no relevant outcomes. Six trials (3 RCTs and 3 nROSs) with 658 patients (348 patients assigned to selective arrhythmogenic PVs arm and 310 patients assigned to the empirical all PVs arm) were included in the analysis.
In the study by Dilling-Boer et al., there were 21 arrhythmogic PVs were targeted, and the acute procedure success rate was 81 %. At the end of follow-up, there was nine patients was free from AF. In the study by Dixit, there was a mean of 2.9 ± 0.9 PVs were isolated in the arrhythmogenic group. Over a mean of 17 ± 5 months, long-term AF control was achieved in 47 (90 %) patients. A total of 2.1 ± 1.0 arrhythmogenic PVs per patients were isolated in the study by Fichtner et al. There were 56 (53 %) patients in sinus rhythms after 12 months. There were 101 arrhythmogenic PVs were isolated in the study by Walczak et al., and the long–term AF control was achieved in 54 (90 %) patients. In the study by Park et al., long-term AF control was achieved in 37 (88.1 %) patients in the arrhythmogenic group. In the study by Hu et al., there were 44 (70 %) patients in the arrhythmogenic group were free from AF at the end of follow-up. The characteristics of included studies were shown in Table 1.
3.2 Baseline characteristics of patients and quality of the included studies
The baseline characteristics of patients enrolled were summarized in Table 2. There was no significant difference on the baseline characteristics of patients between the two groups. The mean patient age ranged from 46 to 61 years. The mean proportion of male ranged from 60 to 75 %. The mean duration of AF ranged from 4 to 6 years. All nonrandomized observational studies were of high quality, ranging from 5 to 9 points on the Newcastle-Ottawa Scale. The quality of included RCTs was moderate, ranging from 2 to 4 points on Jadad score scale.
3.3 The efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF
Figure 1 shows the forest plot comparing the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF. I 2 statistic and Q test showed that there was no significant heterogeneity among the studies included (I 2 = 0 %, p = 0.514), so the fixed effect model was used to pool the data. Isolation of arrhythmogenic PVs was as efficacious as empiric isolation of all PVs in achieving long-term AF control (RR, 0.96; 95 % CI, 0.87–1.05; p = 0.36). Subgroup analysis, again performed despite the absence of heterogeneity, showed that there was a comparable long-term success rate between the two methods of PVs isolation in the RCTs (RR, 0.95; 95 % CI, 0.85–1.07; p = 0.39) or nROSs (RR, 0.96; 95 % CI, 0.80–1.15; p = 0.68, Fig. 2).
3.4 Comparison of the procedure time, fluoroscopic time, and adverse events between the two groups
The pooled data showed that the isolation of arrhythmogenic PVs group had shorter procedure time (WMD, −50.1; 95 % CI, −79.61 to −20.42; p = 0.001; Fig. 3a), fluoroscopy time (WMD, −50.1; 95 % CI, −79.61 to −20.42; p = 0.001; Fig. 3b) and fewer adverse events (RR, 0.43; 95 % CI, 0.18 to 0.99; p = 0.047; Fig. 3c) than the isolation of all PVs group.
3.5 Subgroup analysis
Subgroup analyses were separately performed to investigate heterogeneity. The results showed that the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF were not affected by the mean age of patients, design of studies, ablation strategy, and types of AF (Table 3).
3.6 Sensitivity analysis
Firstly, sensitivity analyses were performed by omitting one study at a time and calculating the pooled RR for the remaining studies. Secondly, the pooled RR was estimated using fixed effect model and random effect model, respectively. Sensitivity analysis indicated that the results of the meta-analysis were reliable and stable.
3.7 Risk of publication bias
Funnel plots were performed to assess the risk of publication bias for six studies with outcome of long-term control of AF. Results showed that the funnel plot was symmetrical. The Egger and Begg’s tests showed no potential publication bias existed among the included trials (Egger’s test, p = 0.75; Begg’s test, p = 0.85).
4 Discussion
We performed this meta-analysis of three RCTs and three nROS with 658 patients to investigate efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF. The results showed that isolation of arrhythmogenic PVs was as efficacious as empiric isolation of all PVs in achieving long-term AF control. In addition, the isolation of arrhythmogenic PVs group might have shorter procedure time, fluoroscopy time and fewer adverse events. For assurance, we performed Egger’s and Begg’s tests to exclude the influence of publication bias on the analysis.
It has been demonstrated that PVs play an important role in the initiation and perpetuation of AF. In the expert consensus statement on catheter and surgical ablation of AF, ablation strategies that target the PVs and/or PV antrum are recommended as the cornerstone for most AF ablation procedures [13]. Operators of this approach do not seek the proof of arrthymogenic PV but isolate all the four PVs empirically. Recent studies have demonstrated that this strategy had a high successful rate but might lead to more adverse events after the procedure [14]. This disadvantage may be related to the extensive LA ablation. EP studies in patients with AF showed that there were one or more arrhythogenic PVs and isolation of the arrhythogenic PVs might prevent patients from AF [3]. The present meta-analysis of RCTs and nROSs indicated that isolation of arrhythmogenic PVs was as efficacious as empiric isolation of all PVs in achieving long-term AF control. In addition, the isolation of arrhythmogenic PVs group might have shorter procedure time, fluoroscopy time and fewer adverse events.
Although the result of our meta-analysis showed that isolation of arrhythmogenic PVs had comparable efficacy with isolation of all PVs in patients with AF, most operators around the world have found that isolation of all PVs is the optimal approach whether cryoballoon or RF is used. There might be some problems to be resolved before isolation of arrhythmogenic PVs was popularly accepted. The first problem is the selection of patients. The population in our study had a high proportion of paroxysmal AF. Whether the patients with persistent AF could get benefits from the strategy is unclear. Studies showed that electrical and electroanatomic remodeling in the left atrium and PVs was more extensive in patients with persistent AF [15]. There might be more arrhythmogenic veins and other potential non-PV triggers in patients with persistent AF [16]. Therefore, whether isolation of arrhythmogenic PVs still had advantages compared with the isolation of all PVs is unclear. In addition, the efficacy of isolating ipsilateral versus bilateral PVs on long-term control of AF depended on the age and left atrium diameter of the patients [10]. Older age and lager LAD might be related to more potential arrhythmogenic PVs which were difficult to identify during the first ablation procedure [17]. Therefore, age and LAD might be factors needed to be considered when the ablation strategy was employed.
How to identify the arrhythmogenic PVs is another problem. There was no established protocol to identify the arrhythmogenic PVs presently. The methods used to identify the arrhythmogenic PVs at present relied on the spontaneous or provocative electric activity from a given PV and on the chance that a recording catheter was placed in the right PV at the right moment. However, this approach is laborious, and it is difficult to localize all the arrhythmogenic PVs during an electrophysiological study [18]. A previous electrophysiological study found the PVs of patients with AF exhibited distinctive electrophysiological properties [19]. Decremental conduction properties at the PV ostium were more frequent and greater in patients with AF, and were the essential properties of the arrhythmogenity of PVs. In the study by Fichtner et al., the researchers indentified the arrhythmogenic PVs by measuring the degree of decremental conduction at PV ostia and had a high successful rate [10]. Another electrophysiological study by Suenari et al. found that the arrhythmogenic PVs were related with high dominant frequency and continuous complex fractionated electrograms in the surrounding atrial substrate [20]. Therefore, evaluation of the atrial substrate properties might be useful for locating the arrhythmogenic PVs.
In addition, which method employed to isolate the PVs also should be considered. Isolation of each individual PV and isolation of ipsilateral PVs are mainly two methods used to isolate the PVs presently. Previous study showed that isolation of ipsilateral PVs was more efficient on long-term control of AF and had shorter fluoroscopy time that the isolation of each individual PV [21]. In the study by Hu et al., isolation of ipsilateral PVs was employed, and isolation of each individual PV was used in the other study. The result of our sub-group analysis showed no difference between the two subgroups. However, there were only six studies in our meta-analysis. This point also should be considered in the clinical trials and practice.
5 Limitations
There are limitations of the present study. First, out of total six studies included, there are only three RCTs. Despite the fact that much effort has been exerted to control biases, residual confounding factors may exist. Therefore, we should interpret the results with caution. CPVI is the most favourable ablation procedure for catheter ablation of AF presently. However, only patients in the study by Hu et al. and proportion patients in the study by Pak et al. received CPVI as the ablation procedure. This would also affect the practical values of our meta-analysis. In addition, the endpoints were arbitrary in the studies included in this analysis. However, in the expert consensus statement, 1-year success is defined as freedom from AF/AFL/AT off antiarrhythmic drug therapy as assessed from the end of the 3 months blanking period to 12 months following the ablation procedure. Long term success is defined as freedom from AF/AFL/AT recurrences through a minimum of 36 months follow-up in the absence of antiarrhythmic drug therapy [13]. Further study with this definition is needed to assess the efficacy of isolating all versus arrhythmogenic PVs on long-term control of AF.
6 Conclusions
The present analysis suggests that isolation of arrhythmogenic PVs had a comparable long-term success rate, shorter procedure time, fluoroscopic time, and fewer adverse events than the empiric isolation of all PVs.
References
Haissaguerre, M., Jais, P., Shah, D. C., et al. (1998). Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. New England Journal of Medicine, 339, 659–666.
Pappone, C., Manguso, F., Vicedomini, G., et al. (2004). Prevention of iatrogenic atrial tachycardia after ablation of atrial fibrillation: a prospective randomized study comparing circumferential pulmonary vein ablation with a modified approach. Circulation, 110, 3036–3042.
Gerstenfeld, E. P., Sauer, W., Callans, D. J., et al. (2006). Predictors of success after selective pulmonary vein isolation of arrhythmogenic pulmonary veins for treatment of atrial fibrillation. Heart Rhythm, 3, 165–170.
Dixit, S., Gerstenfeld, E. P., Callans, D. J., et al. (2004). Mechanisms underlying sustained firing from pulmonary veins: evidence from pacing maneuvers and pharmacological manipulation. Pacing and Clinical Electrophysiology, 27, 1120–1129.
Walczak, F., Szumowski, L., Urbanek, P., et al. (2006). Selective ablation or isolation of all pulmonary veins in atrial fibrillation—when and for whom? Kardiologia Polska 64:26–35.
Dixit, S., Gerstenfeld, E. P., Ratcliffe, S. J., et al. (2008). Single procedure efficacy of isolating all versus arrhythmogenic pulmonary veins on long-term control of atrial fibrillation: a prospective randomized study. Heart Rhythm, 5, 174–181.
Pak, H. N., Kim, J. S., Shin, S. Y., et al. (2008). Is empirical four pulmonary vein isolation necessary for focally triggered paroxysmal atrial fibrillation? Comparison of selective pulmonary vein isolation versus empirical four pulmonary vein isolation. Journal of Cardiovascular Electrophysiology, 19, 473–479.
Fichtner, S., Hessling, G., Ammar, S., et al. (2013). A prospective randomized study comparing isolation of the arrhythmogenic vein versus all veins in paroxysmal atrial fibrillation. Clinical Cardiology, 36, 422–426.
Dilling-Boer, D., Van Der Merwe, N., Adams, J., et al. (2004). Ablation of focally induced atrial fibrillation: selective or extensive? Journal of Cardiovascular Electrophysiology, 15, 200–205.
Hu, J. Q., Ma, J., Ouyang, F., et al. (2012). Is selective ipsilateral PV isolation sufficient for focally triggered paroxysmal atrial fibrillation? Comparison of selective ipsilateral pulmonary vein isolation versus bilateral pulmonary vein isolation. Journal of Cardiovascular Electrophysiology, 23, 130–136.
Jadad, A. R., Moore, R. A., Carroll, D., et al. (1996). Assessing the quality of reports of randomized clinical trials: is blinding necessary? Controlled Clinical Trials, 17, 1–12.
Zhuang, J., Lu, Y., Tang, K., et al. (2013). Influence of body mass index on recurrence and quality of life in atrial fibrillation patients after catheter ablation: a meta-analysis and systematic review. Clinical Cardiology, 36, 269–275.
Calkins, H., Kuck, K. H., Cappato, R., et al. (2012). HRS/EHRA/ECAS Expert Consensus Statement on Catheter and Surgical Ablation of Atrial Fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design. Europace, 14, 528–606.
Satomi, K. (2010). Electrophysiological characteristics of atrial tachycardia after pulmonary vein isolation of atrial fibrillation. Circulation Journal, 74, 1051–1058.
Teh, A. W., Kistler, P. M., Lee, G., et al. (2011). Electroanatomic properties of the pulmonary veins: slowed conduction, low voltage and altered refractoriness in AF patients. Journal of Cardiovascular Electrophysiology, 22, 1083–1091.
Pak, H. N., Hwang, C., Lim, H. E., et al. (2006). Electroanatomic characteristics of atrial premature beats triggering atrial fibrillation in patients with persistent versus paroxysmal atrial fibrillation. Journal of Cardiovascular Electrophysiology, 17, 818–824.
Hsieh, M. H., Tai, C. T., Lee, S. H., et al. (2006). The different mechanisms between late and very late recurrences of atrial fibrillation in patients undergoing a repeated catheter ablation. Journal of Cardiovascular Electrophysiology, 17, 231–235.
Haissaguerre, M., Jais, P., & Shah, D. C. (2000). Electrophysiological end point for catheter ablation of atrial fibrillation initiated from multiple pulmonary venous foci. Circulation, 101, 1409–1417.
Jais, P., Hocini, M., Macle, L., et al. (2002). Distinctive electrophysiological properties of pulmonary veins in patients with atrial fibrillation. Circulation, 106, 2479–2485.
Suenari, K., Lin, Y. J., Chang, S. L., et al. (2011). Relationship between arrhythmogenic pulmonary veins and the surrounding atrial substrate in patients with paroxysmal atrial fibrillation. Journal of Cardiovascular Electrophysiology, 22, 405–410.
Arentz, T., Weber, R., Burkle, G., et al. (2007). Small or large isolation areas around the pulmonary veins for the treatment of atrial fibrillation? Results from a prospective randomized study. Circulation, 115, 3057–3063.
Acknowledgments
We thank all of our colleagues working in the department of Cardiology, the affiliated People’s Hospital of Jiangsu University.
Disclosures
The authors have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Baowei Zhang and Ya Zhen are co-first authors.
Rights and permissions
About this article
Cite this article
Zhang, B., Zhen, Y., Tao, A. et al. Efficacy of selective arrhythmogenic pulmonary veins isolation versus empirical all pulmonary veins isolation for atrial fibrillation: a meta-analysis of randomized and observational studies. J Interv Card Electrophysiol 39, 233–240 (2014). https://doi.org/10.1007/s10840-013-9865-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-013-9865-8