Abstract
Purpose
The aim of this study is to evaluate the frequency and nature of chromosomal abnormalities in Moroccan couples with recurrent spontaneous miscarriage (RSM). In addition, the data were compared with those reported elsewhere in order to give a global estimation of chromosomal abnormalities frequencies.
Methods
The study was performed for all couples with RSM who were referred to the cytogenetic department, Pasteur Institute of Morocco, from different hospitals in Morocco between 1996 and 2016. Cytogenetic analysis was performed according to the standard method.
Results
Among 627 couples with RSM, the chromosomal abnormalities were identified in 11.00% of couples, with chromosomal inversions in 4.30%, reciprocal translocations in 2.71%, Robertsonian translocations in 1.43%, and deletion, isochromosome, and insertion in 0.15% each. The insertion identified [46,XX,ins(6)(p24q21q27)] is new, and is the fourth reported in association with RSM. The mosaic karyotypes were observed in 0.64%, polymorphic variants were identified in 1.27%, and numerical aneuploidy was observed in 0.15%.
In regrouping our results with those in 27 other studies already published in 21 different countries, we obtained the frequency of chromosomal abnormalities in couple with RSM to be 5.16% (991/19197 couples). The reciprocal translocation was the most frequent with 2.50%, followed by Robertsonian translocation 0.83% and inversions 0.77%. The other types of chromosomal abnormalities were present with 0.98% in the world.
Conclusion
This data showed that the frequency of chromosomal abnormalities in Moroccan couples with RSM is 11.00%, and in regrouping our results with other studies, the frequency changes to 5.16%.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recurrent spontaneous miscarriage (RSM) also called recurrent spontaneous abortion and recurrent pregnancy loss has been traditionally defined as the loss of consecutive pregnancies before 20 weeks’ gestation; this definition has been widely adopted [1,2,3,4,5,6,7]. Indeed, for more than 30 years, controversy exists on the number of pregnancy loss required to define the RSM and when diagnostic testing is needed [8]. The American Society for Reproductive Medicine (ASRM), International Committee for Monitoring Assisted Reproductive Technology (ICMART), and World Health Organization (WHO) have defined the RSM as the loss of two or more pregnancies, not necessarily consecutive [9, 10]. According to this definition, approximately 3% of all couples trying to conceive have RSM, but considering the definition of the European Society for Human Reproduction and Embryology (ESHRE) and the Royal College of Obstetricians and Gynecologists (RCOG) who defined the RSM as the loss of three or more consecutive pregnancies (not necessarily intra-uterine), the incidence of RSM in couples desiring a baby is 1% [4, 11].
Until today, multiple potential etiologies for RSM have been described, such as genetic factors (chromosomal abnormalities, embryonic aneuploidy, genetic polymorphisms, and gene mutations); anatomical factors (congenital uterine malformations, acquired anatomic disorders); cervical incompetence; infective factors; endocrine factors (thyroid dysfunction, diabetes mellitus, luteal phase deficiency, PCOS); autoimmune factors/APS; and environmental, occupational, and personal habits [12].
Chromosomal abnormalities (CA) are one of the principal genetic factors in RSM. The rate of chromosomal abnormalities in the general population is less than 1% [12]. Reciprocal translocation (RT) is one of the most frequent structural rearrangements observed in humans [13]. The individuals carrying this type of translocation are clinically normal, but they do have an increased risk of having children with unbalanced karyotypes. Carriers of Robertsonian (Rob) translocations may also develop RSM and offspring with birth defects and mental retardation. Indeed, Robertsonian translocations are more present in infertile men with a frequency of 3% comparing with the general population in which this incidence is about 0.1% [14]. To the best of our knowledge, no retrospective study of chromosomal abnormalities has been performed so far on Moroccan couples with RSM. Thus, this is the first study conducted in Morocco to determine the frequency and nature of chromosomal abnormalities in Moroccan couples with RSM, in order to compare them with those reported elsewhere and give a global estimation of frequency of chromosomal abnormalities in these couples worldwide.
Materials and methods
Study population
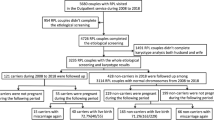
In this retrospective study, we reviewed the cytogenetic analysis database of all couples with RSM (loss of two or more pregnancies before 20 weeks’ gestation) who were referred to the cytogenetic department, Pasteur Institute of Morocco, from different hospitals in Morocco between 1996 and 2016.
All patients with intellectual disabilities, Down syndrome, polymalformative syndrome, and disorder of sex development and those without clinical information were excluded. Clinical information of all patients was collected and analyzed. The present study was approved by the local committee on research ethics of the Pasteur Institute of Morocco and performed following the STROBE guidelines.
Cytogenetic analysis
For routine cytogenetic analysis, peripheral blood (2–3 ml) was collected in heparin vacutainers and all samples were incubated in an RPMI-1640 solution in the presence of phytohemagglutinin (PHA-E) for 72 h at 37 °C. Two hours before the end of the culture, colchicine was added. After centrifugation, the pellet recovered was treated with a hypotonic solution (0.075 M KCl). The samples were fixed by Carnoy’s fixative (acetic acid/methanol 1/3 acid). Fixed cell suspensions were spread on glass slides using a Pasteur pipette. These slides were immersed in the fixative Berger. The slides were immersed in the denaturing medium Earle. Then, they underwent Giemsa staining, and finally reading slides by G-banding technique using a microscope connected to a computer through a camera. At least 20 metaphases were counted for each sample. The chromosomes were classified into several groups and numbered according to the International System for Human Cytogenomic Nomenclature.
Statistical analysis
The comparison of percentages was performed using the exact Fisher test. P values less than 0.05 were considered as statistically significant. All statistical analyses were performed using the R programming language.
Results
A total of 1254 individual cases comprising 627 couples with RSM were included in this study. All these patients had been referred to the cytogenetic department of the Pasteur Institute of Morocco, between 1996 and 2016, and they came from different parts of Morocco. The karyotype analysis revealed that 69 couples (11.00%) showed chromosome aberration, 22 men (3.50%) and 47 women (7.50%) (Table 1).
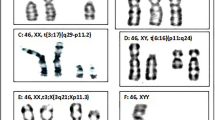
Structural chromosome abnormalities, which had been found in 56 cases (8.93%) (20 men and 36 women), are the most common chromosomal abnormalities. Twenty-seven cases (4.30%) of inversion (9 men and 18 women), 17 cases (2.71%) of reciprocal translocation (7 men and 10 women), 9 cases (1.43%) of Robertsonian translocation (3 men and 6 women), 1 case (0.15%) of deletion (1 man), 1 case (0.15%) of insertion (1 woman), and 1 case (0.15%) of isochromosome (1 woman) were found (Fig. 1). Mosaic karyotypes were observed in 4 women (0.64%); polymorphic variants in 8 cases (1.28%) (3 men and 5 women); and numerical aneuploidy in 1 case (0.16%) (1 man).
Frequency of various chromosomal abnormalities and distribution of chromosomes involved in RSM. a Frequency of various chromosomal abnormalities in couples with RSM. b Gender distribution of various chromosomal abnormalities in couples with RSM. c Distribution of p and q arm chromosome involved in RSM. Inv, inversion; RT, reciprocal translocation; PV, polymorphic variation; Rob, Robertsonian translocation; Mos, mosaicism; Del, deletion; Ins, insertion; Iso, isochromosome; NA, numerical aneuploidy
Reciprocal translocations
Reciprocal translocations were identified in 7 men and 10 women (2.71%). The chromosomes 1, 8, and 10 were found altered by reciprocal translocation in three cases each (2 men and 1 woman, 3 men, 3 women respectively). Chromosomes 6, 11, 14, 15, and 21 were also found altered in two women, while chromosome 18 showed reciprocal translocation in two men. Chromosomes 2, 3, 7, 9, and 13 were found altered in two cases (1 man and 1 woman).
Unlike one man who presented with reciprocal translocation on the 19th chromosome, two women showed this structural alteration on the 5th and 17th chromosomes respectively. No reciprocal translocation was identified on chromosomes 4, 12, 16, 20, 22, X, and Y (Fig. 1).
It should be noted that chromosomes cited above do not have the same translocation points. Thus, both chromosomes 2 and 5 were found altered in the short arm (p) while chromosomes 3, 10, 11, 15, 17, and 19 were found affected in the long arm (q), whereas chromosome 1, 6, 7, 8, 9, 13, 14, 18, and 21 were found affected in both arms (p and q) (Fig. 1).
Robertsonian translocations
Robertsonian translocations were seen in 3 men and 6 women (1.43%). Only chromosomes 13, 14, 15, 21, and 22 were found altered. Robertsonian translocations between chromosomes 13 and 14 were the most frequent. Translocations involving both the homologues of chromosome 13 and the homologues of chromosome 21 were observed in three women. Translocation between chromosomes (13, 22) and (15, 21) was identified in one woman and one man respectively.
Inversions, insertions, deletions, and isochromosomes
Inversions were identified in 9 men and 18 women (4.30%) on chromosome 7 (1 woman) and chromosome 9 (17 women and 9 men). Chromosome 7 was found affected in only one woman inv(7)(p1q21), while on the chromosome 9, the inv(9)(p12q13) was observed in one man, and all the others carried the similar break point (inv(9)(p11q13)).
A new insertion that has not been previously reported was determined in one woman (0.15%). It is an insertion of the q21q27 fragment of chromosome 6 at the p24 of the same chromosome ins(6)(p24q21q27).
Deletion and isochromosome were observed in one patient each (man and woman). The man (0.15%) showed a deletion in chromosome Yq12 and the woman (0.15%) had isochromosome (9) (p10) and isochromosome (9) (q10) instead of normal chromosome 9.
Numerical chromosomal abnormalities and mosaicism
47,XYY is the only numerical chromosomal abnormality identified in this study (0.15%), whereas the mosaic forms were identified in 4 cases (0.64) (all of them women). Two women with 46,XX/45,X0, one woman with 46,XX/47,XX,+mar, and one woman with 46,XX[89%]/46,XX,fra(16)(q22)[7%]/46,XX,del(16)(q22)[4%].
Polymorphic variations
Heterochromatin polymorphic variants were observed in chromosome 9 (9qh+) in two women (0.32%), whereas the satellite polymorphic variant was most common in our patients with a frequency of 0.96%. This polymorphic variant touched chromosome 14 (14ps+) in one man, chromosome 21(21ps+) in three women and one man, and chromosome 22 (22ps+) in one man.
Discussion
The present study determines the frequency of chromosomal abnormalities in Moroccan couples with RSM and compares these frequencies with other studies, which had followed the same method and gave a global estimation of frequency of chromosomal abnormalities in these couples worldwide.
During the 20-year study period (1996–2016), the cytogenetic department of the Pasteur Institute in Morocco received 627 couples with RSM for cytogenetic analysis. Therefore, we identified that 69 couples (11.00%) had chromosomal abnormalities. This frequency is comparable to that reported in Iran (11.68%) [15], Italy (9.66%) [16], the Netherlands (9.46%) [16], Sultanate of Oman (8.68%) [17], Switzerland (7.29%) [16], Saudi Arabia (7.10%) [18, 19], Egypt (6.40%) [20], Mexico (5.83%) [16, 21], Belgium (5.34%) [22], Pakistan (5.33%) [23], Argentina (5.28%) [24], Brazil (5.09%) [25], the Philippines (5.06%) [26], India (4.90%) [27,28,29], Japan (4.54%) [30], Turkey (4.21%) [16, 31], France (4.14%) [16], Tunisia (4.07%) [32], and the UK (3.52%) [33]. But it is significantly higher than that identified in China (2.98%, P = 0.048) [34] and in the USA (2.96%; P = 0.048) [35]. Table 2 regroups cytogenetic results of all these countries.
After regrouping the countries and the results obtained by regions, the highest frequency of chromosomal abnormalities in couples with RSM is that of the Middle East (6.84%; 281/4111 couples) varying between 4.21% in Turkey and 11.68% in Iran, followed by North Africa, Europe, Asia, and America. Indeed, in North Africa, the frequency of CA in couples with RSM is 6.28% (134/2152 couples) varying between 4.07% in Tunisia and 11.00% in Morocco (present study). In Europe, the frequency of CA is 5.15% (178/3495 couples) varying between 3.52% in the UK and 9.66% in Italy. In Asia, the frequency of CA is 4.22% (236/5587 couples) varying between 2.98% in China and 5.33% in Pakistan. In addition, in America, the frequency of CA is 4.17% (162/3888 couples) varying between 2.69% in the USA and 5.83% in Mexico (Fig. 2).
Frequency of various chromosomal abnormalities observed in different studies by region. a Distribution of chromosomal abnormalities frequencies in couples with RSM by region. b Distribution of reciprocal translocation frequencies in couples with RSM by region. c Distribution of Robertsonian translocation frequencies in couples with RSM by region. d Distribution of chromosome inversion frequencies in couples with RSM by region. e Distribution of various chromosomal abnormalities in the world. Blue, highest frequency; brown, mean frequency; gray, lowest frequency
In this study, the most common structural abnormalities were inversions. They were observed in 27 couples with 4.32%, making it the most frequent in North Africa and in the world. In the Sultanate of Oman, pericentric inversion (9)(p13q12) was identified in 6 cases; all of them had a history of RSM [17]. Other chromosomal inversions have been identified in different populations, such as inversion in chromosomes 8 and 4 which have been identified in Saudi Arabia [18], inversion in chromosomes 1, 5, 7, 12, 18, and Y in Turkey [31], and inversion in chromosomes 1, 5, 7, and 10 in India [36]. The results of all these studies indicate that chromosomal inversions may have a role in the etiology of RSM, which must be confirmed by other studies.
Even if the inversions are the most frequent in our study (North Africa), they are the least frequent when compared to other studies. Indeed, in the Middle East, the frequency of inversions is 1.26% varying between 0.63% in Turkey and 2.88% in Iran. Furthermore, in Europe, the frequency of inversions is 0.72% varying between 0% in Switzerland and 2.76% in Italy. In America, the frequency of inversions is 0.28% varying between 0% in Argentina and 0.46% in Brazil. Furthermore, in Asia, the frequency of inversions is 0.29% varying between 0.16% in Japan and 1.67% in Pakistan (Fig. 2).
In the present work, reciprocal translocation was identified in all chromosomes except chromosomes 4, 12, 16, 20, 22, X, and Y. However, this does not exclude their involvement in RSM. In the Sultanate of Oman, Goud et al. identified a reciprocal translocation in chromosomes 4, 12, 20, and 22 and chromosome X [17]. Additionally, in India, Saxena observed a reciprocal translocation in chromosomes 4, 12, 16, 22, and 22 [36]. In Turkey, Tunç et al. found a reciprocal translocation in chromosomes 4 and 12 (Fig. 3) [31]. However, in the Middle East, the frequency of RT in couples with RSM is 3.28% varying between 1.95% in Turkey and 5.08% in Iran. In Europe, the frequency of RT is 2.92% varying between 1.32% in France and 4.17% in Switzerland. In North Africa, the frequency of RT is 2.47% varying between 2.07% in Tunisia and 5.60% in Egypt. In America, the frequency of RT is 2.29% varying between 1.39% in Brazil and 3.96% in Argentina. In Asia, the frequency of RT is 2.13% varying between 1.88% in India and 2.97% in Japan (Fig. 2).
Human chromosome ideograms showing regions of reciprocal translocation identified in different studies. The red indicates regions of reciprocal translocation identified in Morocco, the pink in the Sultanate of Oman [17], the aqua in Turkey [31], the green in India [27, 36], the yellow in India and Turkey [27, 31, 36], the orange in India and the Sultanate of Oman [17, 27, 36], and finally the brown in the Sultanate of Oman and Turkey [17, 31]
In this study, Robertsonian translocations were observed in 5 couples with a frequency of 0.8%. These observations agreed with previews reports (0.78%) [17], (0.66%) [31], and (0.94%) [36].
In North Africa, the frequency of Robertsonian translocation in couples with RSM is 1.02% varying between 0.80% in Egypt and 1.44% in Morocco (the present study). In America, the frequency is 0.90% varying between 0.63% in the USA and 1.32% in Argentina. In Europe, the frequency is 0.87% varying between 0.38% in the UK and 2.76% in Italy. In the Middle East, the frequency is 0.85% varying between 0.71% in Saudi Arabia and 0.96% in Iran. In Asia, the frequency of Robertsonain translocation in couples with RSM is 0.66% varying between 0% in Pakistan and 1.69% in the Philippines (Fig. 2).
The global frequency of chromosomal abnormalities in couples with RSM is 5.16% (991/19197 couples). The reciprocal translocation was the most frequent with 2.50%, followed by Robertsonian translocations 0.83% and inversions 0.77%. The other types of chromosomal abnormalities were present with 0.99% in the world (Fig. 2).
Pericentric inversion of the chromosome 9, Inv(9)(p11q13), is considered as the most commonly observed structurally balanced rearrangement of chromosome involving the heterochromatic region. It is frequently found in normal individuals, and its frequency in the general population is expected to be approximately 1–3% [37]. Most cytogeneticists believe that this variant is a chromosomal polymorphism of the normal human karyotype without any clinical significance. Contradictorily, many clinical investigators have suggested several associations of inv(9) with clinical diagnosis, particularly with idiopathic reproductive failure. Various reports on its association with infertility, recurrent miscarriages, hydatidiform moles, azoospermia, congenital anomalies, growth retardation, and rarely abnormal phenotype have been published [38]. Sasiadek et al. reported inv(9) in 2.3% of all couples presenting with infertility and recurrent abortions [39]. Šípek et al. published the largest study on inv(9) and found a higher frequency among females than in males, especially among those who suffer from infertility [40]. In the present study, we have identified that 4.14% of couples with RSM showed inversions of chromosome 9. This indicates that inversion of chromosome 9 may play an unknown significant role in RSM.
Intrachromosomal insertion in couples with RSM is very rare. Indeed, three couples with RSM were described until today. The first couple was reported in India by Rao L et al., who noticed an insertion in women with karyotype 46,XX,ins(12:6)(12;6)(q24.2;q23q25) [41]. The second was reported by Iyer P, who identified an insertion in men with karyotype 46,XY,ins(1;13)(q22;q31q33) [42], and the third was reported by Fuente-Cortés BED, who found an insertion in women with karyotype 46,XX,ins(15;8)(q26;p11p23) [16]. In our study, we identified a new insertion, 46,XX,ins(6)(p24q21q27), in one woman with RSM, and to the best of our knowledge, this is the fourth insertion reported in the literature.
Limitations of the present study include the lack of questionnaire data (e.g., history of diagnosis, obstetric history, and previous treatment received) from the couples who were included in this study and the lack of cytogenetic analysis of miscarriages.
Conclusion
Our data clearly illustrates that the frequency of chromosomal abnormalities in Moroccan couples is 11.00%, while in regrouping our results with other studies, the frequency changes to 5.16%. Cytogenetic analysis should be part of the investigation of any couple who has experienced at least three consecutive pregnancies of unknown origin. Genetic counseling is important in the management of couples with RSM because of the clear relationship between a chromosomal abnormality in a parent and subsequent miscarriages and a malformed child. Thus, the couples with RSM and normal karyotype should be evaluated with molecular techniques, such as chromosome microarray analysis (CMA) and next-generation sequencing (NGS), which have opened new possibilities for discovery of genetic changes that are undetectable by constitutional chromosome analysis.
References
McNamee K, Dawood F, Farquharson R. Recurrent miscarriage and thrombophilia: an update. Curr Opin Obstet Gynecol. 2012;24:229–34.
Duckitt K, Qureshi A. Recurrent miscarriage. BMJ Clin Evid. 2011;2011.
Cohn DM, Goddijn M, Middeldorp S, Korevaar JC, Dawood F, Farquharson RG. Recurrent miscarriage and antiphospholipid antibodies: prognosis of subsequent pregnancy. J Thromb Haemost. 2010;8:2208–13.
Jauniaux E, Farquharson RG, Christiansen OB, Exalto N. Evidence-based guidelines for the investigation and medical treatment of recurrent miscarriage. Hum Reprod. 2006;21:2216–22.
Christiansen OB, Nybo Andersen A-M, Bosch E, Daya S, Delves PJ, Hviid TV, et al. Evidence-based investigations and treatments of recurrent pregnancy loss. Fertil Steril. 2005;83:821–39.
Coulam CB. Epidemiology of recurrent spontaneous abortion. Am J Reprod Immunol. 1991;26:23–7.
Stirrat GM. Recurrent miscarriage. Lancet. 1990;336:673–5.
Jaslow CR, Kutteh WH. Effect of prior birth and miscarriage frequency on the prevalence of acquired and congenital uterine anomalies in women with recurrent miscarriage: a cross-sectional study. Fertil Steril. 2013;99:1916–1922.e1.
Practice Committee of American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril. 2013;99:63.
Zegers-Hochschild F, Adamson GD, de Mouzon J, Ishihara O, Mansour R, Nygren K, et al. The International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary on ART terminology, 2009. Hum Reprod. 2009;24:2683–7.
Green-top Guideline No.17. Recurrent miscarriage, investigation and treatment of couples (Green-top Guideline No. 17) [Internet]. Royal College of Obstetricians & Gynaecologists. 2011 [cited 2017 Nov 16]. Available from: https://www.rcog.org.uk/en/guidelines-research-services/guidelines/gtg17/
Diejomaoh MFE. Recurrent spontaneous miscarriage is still a challenging diagnostic and therapeutic quagmire. Med Princ Pract. 2015;24:38–55.
Mierla D, Malageanu M, Tulin R, Albu D. Prevalence of chromosomal abnormalities in infertile couples in Romania. Balkan J Med Genet. 2015;18:23–30.
Morin SJ, Eccles J, Iturriaga A, Zimmerman RS. Translocations, inversions and other chromosome rearrangements. Fertil Steril. 2017;107:19–26.
Ghazaey S, Keify F, Mirzaei F, Maleki M, Tootian S, Ahadian M, et al. Chromosomal analysis of couples with repeated spontaneous abortions in Northeastern Iran. Int J Fertil Steril. 2015;9:47–54.
De la Fuente-Cortés BE, Cerda-Flores RM, Dávila-Rodríguez MI, García-Vielma C, Alvarado RMD la R, Cortés-Gutiérrez EI. Chromosomal abnormalities and polymorphic variants in couples with repeated miscarriage in Mexico. Reprod BioMed Online. 2009;18:543–8.
Goud TM, Mohammed Al Harassi S, Khalfan Al Salmani K, Mohammed Al Busaidy S, Rajab A. Cytogenetic studies in couples with recurrent miscarriage in the Sultanate of Oman. Reprod BioMed Online. 2009;18:424–9.
Al-Hussain M, Al-Nuaim L, Abu ZT, Zaki OK. Cytogenetic study in cases with recurrent abortion in Saudi Arabia. Ann Saudi Med. 2000;20:233–6.
Awartani KA, Al Shabibi MS. Description of cytogenetic abnormalities and the pregnancy outcomes of couples with recurrent pregnancy loss in a tertiary-care center in Saudi Arabia. Saudi Med J. 2018;39:239–42.
Gaboon NEA, Mohamed AR, Elsayed SM, Zaki OK, Elsayed MA. Structural chromosomal abnormalities in couples with recurrent abortion in Egypt. Turk J Med Sci. 2015;45:208–13.
Meza-Espinoza JP, Anguiano LO, Rivera H. Chromosomal abnormalities in couples with reproductive disorders. Gynecol Obstet Investig. 2008;66:237–40.
Fryns JP, Van Buggenhout G. Structural chromosome rearrangements in couples with recurrent fetal wastage. Eur J Obstet Gynecol Reprod Biol. 1998;81:171–6.
Azim M, Khan AH, Khilji ZL, Pal JA, Khurshid M. Chromosomal abnormalities as a cause of recurrent abortions: a hospital experience. J Pak Med Assoc. 2003;53:117–9.
Gadow EC, Lippold S, Otano L, Serafin E, Scarpati R, Matayoshi T. Chromosome rearrangements among couples with pregnancy losses and other adverse reproductive outcomes. Am J Med Genet. 1991;41:279–81.
Kiss A, Rosa RFM, Dibi RP, Zen PRG, Pfeil JN, Graziadio C, et al. Anormalidades cromossômicas em casais com história de aborto recorrente. Rev Bras Ginecol Obstet. 2009;31:68–74.
Cutiongco-de la Paz E, Berboso AG, Salonga EAG, Padilla C. Chromosomal structural abnormalities among Filipino couples with recurrent pregnancy losses. Acta Med Philipp. 2011;45:58–62.
Dutta UR, Rajitha P, Pidugu VK, Dalal AB. Cytogenetic abnormalities in 1162 couples with recurrent miscarriages in southern region of India: report and review. J Assist Reprod Genet. 2011;28:145–9.
Dubey S, Chowdhury MR, Prahlad B, Kumar V, Mathur R, Hamilton S, et al. Cytogenetic causes for recurrent spontaneous abortions – an experience of 742 couples (1484 cases). 2005 [cited 2018 Aug 8]; Available from: https://tspace.library.utoronto.ca/handle/1807/5942
Sudhir N, Kaur T, Beri A, Kaur A. Cytogenetic analysis in couples with recurrent miscarriages: a retrospective study from Punjab, north India. J Genet. 2016;95:887–94.
Makino T, Tabuchi T, Nakada K, Iwasaki K, Tamura S, Iizuka R. Chromosomal analysis in Japanese couples with repeated spontaneous abortions. Int J Fertil. 1990;35:266–70.
Tunç E, Tanrıverdi N, Demirhan O, Süleymanova D, Çetinel N. Chromosomal analyses of 1510 couples who have experienced recurrent spontaneous abortions. Reprod BioMed Online. 2016;32:414–9.
Elghezal H, Hidar S, Mougou S, Khairi H, Saâd A. Prevalence of chromosomal abnormalities in couples with recurrent miscarriage. Fertil Steril. 2007;88:721–3.
Flynn H, Yan J, Saravelos SH, Li T-C. Comparison of reproductive outcome, including the pattern of loss, between couples with chromosomal abnormalities and those with unexplained repeated miscarriages. J Obstet Gynaecol Res. 2014;40:109–16.
Fan H-T, Zhang M, Zhan P, Yang X, Tian W-J, Li R-W. Structural chromosomal abnormalities in couples in cases of recurrent spontaneous abortions in Jilin Province, China. Genet Mol Res. 2016;15.
Stephenson MD, Sierra S. Reproductive outcomes in recurrent pregnancy loss associated with a parental carrier of a structural chromosome rearrangement. Hum Reprod. 2006;21:1076–82.
Saxena SG, Desai K, Shewale L, Ranjan P, Saranath D. Chromosomal aberrations in 2000 couples of Indian ethnicity with reproductive failure. Reprod BioMed Online. 2012;25:209–18.
Amiel A, Sardos-Albertini F, Fejgin MD, Sharony R, Diukman R, Bartoov B. Interchromosomal effect leading to an increase in aneuploidy in sperm nuclei in a man heterozygous for pericentric inversion (inv 9) and C-heterochromatin. J Hum Genet. 2001;46:245–50.
Yamada K. Population studies of INV(9) chromosomes in 4,300 Japanese: incidence, sex difference and clinical significance. Jpn J Hum Genet. 1992;37:293–301.
Sasiadek M, Haus O, Lukasik-Majchrowska M, Slezak R, Paprocka-Borowicz M, Busza H, et al. Cytogenetic analysis in couples with spontaneous abortions. Ginekol Pol. 1997;68:248–52.
Šípek A, Panczak A, Mihalová R, Hrčková L, Suttrová E, Sobotka V, et al. Pericentric inversion of human chromosome 9 epidemiology study in Czech males and females. Folia Biol (Praha). 2015;61:140–6.
Rao L, Murthy K, Babu A, Venkata P, Deenadayal M, Singh L. Chromosome inversions and a novel chromosome insertion associated with recurrent miscarriages in South India. Arch Gynecol Obstet. 2005;272:273–7.
Iyer P, Wani L, Joshi S, Lakshmi J, Dalvi R, Chavan D, et al. Cytogenetic investigations in couples with repeated miscarriages and malformed children: report of a novel insertion. Reprod BioMed Online. 2007;14:314–21.
Acknowledgments
We acknowledge and greatly appreciate the excellent technical assistance of all the staff at the Genomics and Human Genetics laboratory, and we thank all patients for their cooperation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The present study was approved by the local committee on research ethics of the Pasteur Institute of Morocco.
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Elkarhat, Z., Kindil, Z., Zarouf, L. et al. Chromosomal abnormalities in couples with recurrent spontaneous miscarriage: a 21-year retrospective study, a report of a novel insertion, and a literature review. J Assist Reprod Genet 36, 499–507 (2019). https://doi.org/10.1007/s10815-018-1373-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-018-1373-4