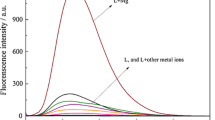
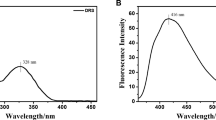
A new fluorescent “turn-on” sensor for Hg2+, N-allyl-4-(ethylenediamine-5-methylsalicylidene)-1,8-naphthalimide (HL) has been designed by combining a 1,8-naphthalimide moiety as a fluorophore and a Schiff base as a recognition group. As expected, HL displays high selectivity for Hg2+ over other ions (Na+, K+, Ca2+, Mg2+, Al3+, Pb2+, Fe3+, Ni2+, Zn2+, Hg2+, Ag+, Co2+, Cr3+, Mn2+, and Cd2+) with obvious fluorescence enhancement in solution (DMF/Tris–HCl buffer, 1:1, v/v, pH 7.2). Moreover, the fluorescence intensity of HL has shown good linearity with a correlation coefficient (R2) of 0.99, confirming that HL could be applied to quantitatively detect mercury ions in the range of 0.5–4.0 μm, whereby the detection limit reaches 0 .26 μm. Meanwhile, the association constant (Ka) between Hg2+ and HL achieves 7.35 × 1011 M–1. Based on the fluorescence titration and Job’s plot analysis, the formation of a complex between HL and Hg2+ is by 2:1 complex ratio.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Nendza, T. Herbst, C. Kussatz, and A. Gies, Chemosphere, 35, 1875–1885 (1997).
I. Hoyle and R. D. Handy, Aquat. Toxicol., 72, 147 (2005).
A. Renzoni, F. Zino, and E. Franchi, Environ. Res., 77, 68–72 (1998).
L. Patrick, Altern. Med. Rev., 7, 456–471 (2002).
P. Grandjean, P. Weihe, R. F. White, and F. Debes, Environ. Res., 77, 165–172 (1998).
T. Takeuchi, N. Morikawa, H. Matsumoto, and Y. Shiraishi, Acta Neuropathol., 2, 40–57 (1962).
M. Harada, Crit. Rev. Toxicol., 25, 1–25 (1995).
I. V. Boevski, N. Daskalova, and I. Havezov, Spectrochim. Acta B, 55, 1643–1657 (2000).
G. C. Li, G. Q. Gao, J. Y. Cheng, X. P. Chen, Y. F. Zhao, and Y. Ye, Luminescence, 31, 992–996 (2016).
Y. L. Liu, X. Lv, Y. Zhao, M. L. Chen, J. Liu, P. Wang, and W. Guo, Dyes Pigments, 92, 909–915 (2012).
F. Ye, X. M. Liang, K. X. Xu, X. X. Pang, Q. Chai, and Y. Fu, Talanta, 200, 494–502 (2019).
C. B. Huang, H. R. Li, Y. Y. Luo, and L. Xu, Dalton Trans., 43, 8102–8108 (2014).
J. H. Hu, J. B. Li, J. Qi, and Y. Sun, Sensor. Actuat. B: Chem., 208, 581–587 (2015).
W. K. Dong, X. L. Li, L. Wang, Y. Zhang, and Y. J. Ding, Sensor. Actuat. B: Chem., 229, 370–378 (2016).
W. K. Dong, S. F. Akogun, Y. Zhang, Y. X. Sun, and X. Y. Dong, Sensor. Actuat. B: Chem., 238, 723–734 (2017).
Y. L. Xu, S. S. Mao, H. P. Peng, F. Wang, H. Zhang, S. O. Aderinto, and H. L. Wu, J. Lumin., 192, 56–63 (2017).
S. O. Aderinto, Y. L. Xu, H. P. Peng, F. Wang, H. L. Wu, and X. Y. Fan, J. Fluoresc., 27, 79–87 (2017).
D. Zhang, M. Li, Y. Jiang, C. Wang, Z. Wang, Y. Ye, and Y. Zhao, Dye Pigment, 99, 607–612 (2013).
J. Wang and B. Liu, Chem. Commun., 39, 4759–4761 (2008).
Z. Q. Zhu, Y. Y. Su, J. Li, D. Li, J. Zhang, S. P. Song, Y. Zhao, G. X. Li, and C. H. Fan, Anal. Chem., 81, 7660–7666 (2009).
S. O. Aderinto, H. Zhang, H. L. Wu, C. Y. Chen, J. W. Zhang, H. P. Peng, Z. H. Yang, and F. Wang, Color. Technol., 133, 40–49 (2017).
W. K. Dong, Y. X. Sun, C. Y. Zhao, X. Y. Dong, and L. Xu, Polyhedron, 29, 2087–2097 (2010).
Y. L. Xu, S. O. Aderinto, H. L. Wu, H. P. Peng, H. Zhang, J. W. Zhang, and X. Y. Fan, Z. Naturforsch. B, 72, 35–41 (2017).
H. L. Wu, S. O. Aderinto, Y. L. Xu, H. Zhang, and X. Y. Fan, J. Appl. Spectrosc., 84, 25–30 (2017).
Y. Qu, C. Wang, Y. C. Wu, K. Zhao, and H. L. Wu, J. Appl. Spectrosc., 87, 429–436 (2020).
Y. Qu, Y. C. Wu, C. Wang, K. Zhao, and H. L. Wu, J. Chem. Res., 44, 121–127 (2020).
H. Zhang, Y. Qu, K. Zhao, C. Wang, Y. C. Wu, and H. L. Wu, J. Chin. Chem. Soc., 67, 1062–1069 (2020).
Y. Qu, Y. C. Wu, C. Wang, K. Zhao, and H. L. Wu, Z. Naturforsch. B, 74, 665–670 (2019).
G. Z. Huang, C. Li, X. T. Han, S. O. Aderinto, K. S. Shen, S. S. Mao, and H. L. Wu, Luminescence, 33, 660–669 (2018).
K. S. Shen, S. S. Mao, X. K. Shi, F. Wang, Y. L. Xu, S. O. Aderinto, and H. L. Wu, Luminescence, 33, 54–63 (2018).
C. Li, X. T. Han, S. S. Mao, S. O. Aderinto, X. K. Shi, K. S. Shen, and H. L. Wu, Color. Technol., 134, 230–239 (2018).
H. P. Peng, K. S. Shen, S. S. Mao, X. K. Shi, Y. L. Xu, S. O. Aderinto, and H. L. Wu, J. Fluoresc., 27, 1191–1200 (2017).
F. Wang, Y. L. Xu, S. O. Aderinto, H. P. Peng, H. Zhang, and H. L. Wu, J. Photochem. Photobiol. A, 332, 273–282 (2017).
H. L. Wu, H. P. Peng, F. Wang, H. Zhang, C. G. Chen, J. W. Zhang, and Z. H. Yang, J. Appl. Spectrosc., 83, 931–937 (2017).
S. O. Aderinto, H. Zhang, H. L. Wu, C. Y. Chen, J. W. Zhang, H. P. Peng, Z. H. Yang, and F. Wang, Color. Technol., 133, 40–49 (2017).
M. H. Lim, B. A. Wong, W. H. Pitcock, Jr., D. Mokshagundam, M. H. Baik, and S. J. Lippard, J. Am. Chem. Soc., 128, 14364–14373 (2006).
N. I. Georgiev and V. B. Bojinov, J. Lumin., 132, 2235–2241 (2012).
S. Roy, P. Gayen, R. Saha, T. K. Mondal, and C. Sinha, Inorg. Chim. Acta, 410, 202–213 (2014).
K. A. Alamry, N. I. Georgiev, S. A. EI-Daly, L. A. Taib, and V. B. Bojinov, J. Lumin., 158, 50–59 (2015).
Y. F. Liu, M. Deng, X. S. Tang, T. Zhu, Z. G. Zang, X.F. Zeng, and S. Han, Sens. Actuat. B: Chem., 233, 25–30 (2016).
W. K. Dong, J. C. Ma, Y. J. Dong, L. Zhao, L. C. Zhu, Y. X. Sun, and Y. Zhang, J. Coord. Chem., 69, 3231–3241 (2016).
B. McLaughlin, E. M. Surender, G. D. Wright, B. Daly, and A. P. de Silva, Chem. Commun., 54, 1319–1322 (2018).
W. Shen, L. Q. Yan, W. W. Tian, X. Cui, Z. J. Qi, and Y. M. Sun, J. Lumin., 177, 299–305 (2016).
C. Y. Li, X. B. Zhang, L. Qiao, Y. Zhao, C. M. He, S. Y. Huan, L M. Lu, L. X. Jian, G. L. Shen, and R. Q. Yu, Anal. Chem., 81, 9993–10001 (2009).
M. Liu, L. N. Dong, A. J. Chen, Y. Zheng, D. Z. Sun, X. Wang, and B. Q. Wang, Spectrochim. Acta A, 115, 854–860 (2013).
Z. J. Chen, L. M. Wang, G. Zou, X. M. Cao, Y. Wu, and P. J. Hu, Spectrochim. Acta A, 114, 323–329 (2013).
L. Zhao, G. Wang, J. Chen, L. Zhang, B. Liu, J. Zhang, Q. Zhao, and Y. Zhou, J. Fluorine Chem., 158, 53–59 (2014).
US EPA. EPA-452/R-05-003. Res. Triangle Park, NC: US EPA (2005).
M. J. Culzoni, A. Muñoz de la Peña, A. Machuca, H. C. Goicoechea, R. Brasca, and R. Babiano, Talanta, 117, 288–296 (2013).
H. L. Tan, B. X. Liu, and Y. Chen, ACS Nano, 6, 10505–10511 (2012).
H. S. Lee, H. S. Lee, J. H. Reibenspies, and R. D. Hancock, Inorg. Chem., 51, 10904–10915 (2012).
M. Shellaiah, Y. C. Rajan, P. Balu, and A. Murugan, New J. Chem., 39, 2523–2531 (2015).
Y. M. Shen, Y. Y. Zhang, X. Y. Zhang, C. X. Zhang, L. L. Zhang, J. L. Jin, H. T. Li, and S. Z. Yao, Anal. Methods, 6, 4797–4802 (2014).
L. Kang, Y. T. Liu, N. N. Li, Q. X. Dang, Z. Y. Xing, J. L. Li, and Y. Zhang, J. Lumin., 186, 48–52 (2017).
Q. C. Su, Q. F. Niu, T. Sun, and T. D. Li, Tetrahedron Lett., 57, 4297–4301 (2016).
Z. J. Chen, L. M. Wang, G. Zou, J. Tang, X. F. Cai, M. S. Teng, and L. Chen, Spectrochim. Acta A, 105, 57–61 (2013).
Y. L. Fu, C. B. Fan, G. Liu, and S. Z. Pu, Sens. Actuat. B: Chem., 239, 295–303 (2017).
R. H. Shen, J. J. Yang, H. Luo, B. X. Wang, and Y. L. Jiang, Tetrahedron, 73, 373–377 (2017).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Zhurnal Prikladnoi Spektroskopii, Vol. 89, No. 3, pp. 386–393, May–June, 2022.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, HL., Dong, JP., Sun, FG. et al. New Selective Fluorescent “Turn-On” Sensor for Detection of Hg2+ Based on a 1,8-Naphthalimide Schiff Base Derivative. J Appl Spectrosc 89, 487–494 (2022). https://doi.org/10.1007/s10812-022-01384-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-022-01384-8