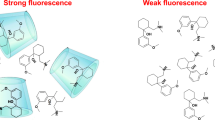
An inclusion complex was formed with methyl-β-cyclodextrin (Me-β-CD) and the smallest unit of the macrocyclic family of bathocuproine, 2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline. With this newly formed fluorescent probe, the quenching effect of heavy metals was investigated. Spectral characterization of the complex was performed by UV-visible and fluorescence spectroscopy techniques. The complex formation constant (Kb), Gibbs free energy (ΔG0), and approximate quenching activation energy (Ea) values were calculated. Lifetime (τ), bimolecular quenching rate constant (kq) values, and diffusion rate constant (kd) calculations were made, and the mechanism of quenching by the effect of heavy metals was clarified.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R. Balgley, M. Drees, T. Bendikov, M. Lahav, A. Facchetti, and M. E. Van Der Boom, J. Mater. Chem. C, 4, 4634–4639 (2016).
Y. Kijima, N. Asai, and S. I. Tamura, Jpn. J. Appl. Phys., Regul. Pap. Short Notes Rev. Pap., 38, 5274–5277 (1999).
D. F. O’Brien, M. A. Baldo, M. E. Thompson, and S. R. Forrest, Appl. Phys. Lett., 74, 442–444 (1999).
H. Tang, H. Liao, and L. Zhu, Chem. Phys. Lett., 381, 605–608 (2003).
S. Ohno, M. Tanaka, N. Teshima, and T. Sakai, Anal. Sci., 20, 171–175 (2004).
A. C. F. De Brito, R. S. Correa, A. A. Pinto, M. J. S. Matos, J. C. Tenorio, J. G. Taylor, and T. Cazati, J. Mol. Struct., 1163, 197–204 (2018).
U. Ay, Z. Dogruyol, and N. Arsu, Supramol. Chem., 26, 66–70 (2014).
U. Ay and S. E. Sarli, J. Fluoresc., 28, 1371–1378 (2018).
A. Ray, S. Das, and N. Chattopadhyay, ACS Omega, 4, 15–24 (2019).
S. Barman, B. K. Barman, and M. N. J. Roy, Mol. Struct., 1155, 503–512 (2018).
S. E. Sarli and U. Ay, Inorg. Chem. Commun., 114, 107820 (2020).
H. Yang, G. Ran, J. Yan, H. Zhang, and X. Hu, Luminescence, 33, 349–355 (2018).
F. A. Carey and R. J. Sundberg, Advanced Organic Chemistry: Structure and Mechanisms, Plenum Press, New York and London, 469 (1995), https://doi.org/10.1007/978-1-4615-8881-5.
A. H. Al-Marzouqi, I. Shehatta, B. Jobe, and A. Dowaidar, J. Pharm. Sci., 95, 292–304 (2006).
G. S. Jadhav and P. R. Vavia, Int. J. Pharm., 352, 5–16 (2008).
D. D. Chow and A. H. Karara, Int. J. Pharm., 28, 95–101 (1986).
K. Uekama, T. Fujinaga, F. Hirayama, M. Otagiri, and M. Yamasaki, Int. J. Pharm., 10, 1–15 (1982).
N. Rajagopalan, S. C. Chen, and W.-S. Chow, Int. J. Pharm., 29, 161–168 (1986).
K. Uekama, S. Narisawa, F. Hirayama, and M. Otagiri, Int. J. Pharm., 16, 327–338 (1983).
G. Smulevich, A. Feis, G. Mazzi, and F. F. Vincieri, J. Pharm. Sci., 77, 523–526 (1988).
X. Tian-Xiang and B. D. Anderson, Int. J. Pharm., 59, 45–55 (1990).
R. Singh, N. Bharti, J. Madan, and S. N. Hiremath, J. Pharm. Sci. Technol., 2, 171–183 (2010).
Slideplayer.biz.tr/slide/2790521/ (visited 17.04.2020).
R. A. Alberty and G. G. Hammes, J. Phys. Chem., 62, 154–159 (1958).
Y. Cui, G. Li, N. Zhang, X. Wang, J. Xie, and L. Deng, IEEE Trans. Magn., 51, 1–4 (2015).
R. M. Melavanki, R. A. Kusanur, M. V. Kulakarni, and J. S. Kadadevarmath, J. Lumin., 128, 573–577 (2008).
R. M. Melavanki, N. R. Patil, H. D. Patil, R. A. Kusanur, and J. S. Kadadevaramath, Indian J. Pure Appl. Phys., 49, 748–753 (2011).
R. M. Melavanki, R. A. Kusanur, J. S. Kadadevaramath, and M. V. Kulkarni, J. Fluoresc., 20, 1175–1180 (2010).
R. M. Melavanki, R. A. Kusanur, J. S. Kadadevaramath, and M. V. Kulakarni, J. Lumin., 129, 1298–1303 (2009).
N. R. Patil, R. M. Melavanki, D. Nagaraja, H. D. Patil, F. M. Sanningannavar, and S. B. Kapatakar, J. Mol. Liq., 180, 112–120 (2013).
N. R. Patil, R. M. Melavanki, S. B. Kapatkar, K. Chandrashekhar, H. D. Patil, and S. Umapathy, Spectrochim. Acta A: Mol. Biomol. Spectrosc., 79, 1985–1991 (2011).
H. R. Deepa, J. Thipperudrappa, and H. M. Suresh Kumar, J. Lumin., 132, 1382–1388 (2012).
J. S. Kadadevarmath, G. H. Malimath, R. M. Melavanki, and N. R. Patil, Spectrochim. Acta A: Mol. Biomol. Spectrosc., 117, 630–634 (2014).
J. S. Kadadevarmath, T. P. Giraddi, and G. C. Chikkur, J. Photosci., 4, 105–112 (1997).
J. Keizer, J. Phys. Chem., 86, 5052–5067 (1982).
J. Keizer, J. Am. Chem. Soc., 105, 1494–1498 (1983).
J. Keizer, J. Am. Chem. Soc., 107, 5319 (1985).
J. Keizer, Chem. Rev., 87, 167–180 (1987).
H. Zeng and G. Durocher, J. Lumin., 63, 75–84 (1995).
Author information
Authors and Affiliations
Corresponding author
Additional information
Abstract of article is published in Zhurnal Prikladnoi Spektroskopii, Vol. 88, No. 4, p. 661, July–August, 2021.
Rights and permissions
About this article
Cite this article
Ay, U. Effect of Heavy Metals on Dynamic and Static Quenching of the Fluorescence of the Host-Guest Inclusion Complex Methyl-β-Cyclodextrin by 2,9-Dimethyl-4,7-Diphenyl-1,10-Phenanthroline in Aqueous Media. J Appl Spectrosc 88, 838–846 (2021). https://doi.org/10.1007/s10812-021-01248-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-021-01248-7