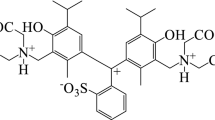
Colorimetry and spectrophotometry with chemometric data processing were used to study the acid–base properties of azo dyes in aqueous solution. The capabilities of both methods were compared. Ionization constants of all the functional groups of the azo compounds studied could be determined relative to the change in the specific color difference depending on the acidity of the medium. The colorimetric functions of ion-molecular forms of azo compounds used as an analytical signal allow us to obtain complete information on the acid–base equilibrium in a wide acidity range.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
V. P. Antonovich and A. O. Stoyanov, Aspects of the History and Methodology of Analytical Chemistry [in Russian], Astroprint, Odessa (2015), pp. 57–60.
M. I. Bulatov and I. P. Kalinkin, Practical Handbook of Photometric Analysis Methods [in Russian], Khimiya, Moscow (1985), pp. 239–280.
V. S. Shubina and Yu. V. Shatalin, Zh. Prikl. Spektrosk., 80, No. 5, 771–776 (2013) [V. S. Shubina and Yu. V. Shatalin, J. Appl. Spectrosc., 80, No. 5, 761–766 (2013)].
Yu. É. Zevatskii, D. V. Samoilov, and N. O. Mchedlov-Petrosyan, Zh. Obshch. Khim., 79, No. 9, 1504–1532 (2009) [Yu. É. Zevatskii, D. V. Samoilov, and N. O. Mchedlov-Petrosyan, Russ. J. Gen. Chem., 79, No. 9, 1859–1889 (2009)].
Yu. É. Zevatskii and D. V. Samoilov, Zh. Org. Khim., 47, No. 10, 1423–1444 (2011) [Yu. É. Zevatskii and D. V. Samoilov, Russ. J. Org. Chem., 47, No. 10, 1445–1467 (2011)].
J. Reijenga, A. Hoof, A. Loon, and B. Teunissen, Anal. Chem. Ins., 8, 53–71 (2013).
Y. Gonen and G. Rytwo, Anal. Chem. Ins., 4, 21–27 (2009).
A. N. Chebotarev and D. V. Snigur, Zh. Analit. Khim., 70, No. 1, 53–57 (2015) [A. N. Chebotarev and D. V. Snigur, J. Analyt. Chem., 70, No. 1, 55–59 (2015)].
V. M. Ivanov, E. M. Adamova, and V. N. Figurovskaya, Zh. Analit. Khim., 65, No. 5. 488–496 (2010) [V. M. Ivanov, E. M. Adamova, and V. N. Figurovskaya, J. Analyt. Chem., 65, 473–481 (2010)].
A. N. Chebotarev and D. V. Snigur, Zh. Obshch. Khim., 86, No. 4, 627–633 (2016) [A. N. Chebotarev and D. V. Snigur, Russ. J. Gen. Chem., 86, No. 4, 815–820 (2016)].
J. Barbosa and C. Bosch, Talanta, 38, No. 11, 1297–1302 (1991).
A. N. Chebotarev, D. V. Snigur, Yu. P. Zhukova, E. V. Bevzyuk, Ya. I. Studenyak, and Ya. R. Bazel′, Zh. Obshch. Khim., 87, No. 2, 216–224 (2017) [A. N. Chebotarev, D. V. Snigur, Yu. P. Zhukova, E. V. Bevzyuk, Ya. I. Studenyak, and Ya. R. Bazel′, Russ. J. Gen. Chem., 87, No. 2, 196–203 (2017)].
V. M. Ivanov, O. V. Monogarova, and K. V. Oskolok, Zh. Analit. Khim., 70, No. 10, 1011–1025 (2015) [V. M. Ivanov, O. V. Monogarova, and K. V. Oskolok, J. Analyt. Chem., 70, No. 10, 1165–1178 (2015)].
A. N. Chebotarev, D. V. Snigur, E. V. Bevzyuk, and I. S. Efimova, Metody i Ob′ekty Khim. Analiza, 9, No. 1, 4–11 (2014).
A. Shokrallahi and E. Zare, J. Mol. Liq., 219, 1165–1171 (2016).
O. R. Sovin and I. O. Patsai, Metody i Ob′ekty Khim. Analiza, 7, No. 2, 74–80 (2012).
K. Poy and P. Popeliel, J. Phys. Org. Chem., 22, No. 3, 186–196 (2009).
R. W. Sabnis, Handbook of Acid–Base Indicators, CRC Press, Boca Raton (2008).
E. Bishop, Indicators, Pergamon Press (1972).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Prikladnoi Spektroskopii, Vol. 85, No. 1, pp. 27–32, January–February, 2018.
Rights and permissions
About this article
Cite this article
Snigur, D.V., Chebotarev, A.N. & Bevziuk, K.V. Acid–Base Properties of Azo Dyes in Solution Studied Using Spectrophotometry and Colorimetry. J Appl Spectrosc 85, 21–26 (2018). https://doi.org/10.1007/s10812-018-0605-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-018-0605-9