Abstract
This study aimed to identify the optimal doses of the macroalgae Asparagopsis taxiformis and Oedogonium sp., individually and in combination, which would decrease the in vitro production of methane while minimizing adverse effects on fermentation, using rumen inoculant from Bos indicus steers. The dose-response experiment evaluated ten doses of Asparagopsis [ranging from 0 to 16.7 % of the organic matter (OM) incubated] and seven doses of Oedogonium (ranging from 0 to 100 % OM) using Rhodes grass hay as a basal substrate. Asparagopsis was highly effective in decreasing the production of methane with a reduction of 99 % at doses as low as 2 % OM basis. However, a dose of 2 % OM also decreased the production of volatile fatty acids (VFA). Oedogonium was less effective with doses ≥50 % OM significantly decreasing the production of methane. A combination of Asparagopsis (2 % OM) and Oedogonium (25 and 50 % OM) continued to suppress the production of methane, independent of the inclusion rate of Oedogonium. The effectiveness of Asparagopsis demonstrates its potential for the mitigation of methane emissions from ruminants at inclusion rates of ≤2 % OM. Oedogonium is a potential feed supplement due to its nutritional value, but supplements ≤25 % OM are recommended to avoid adverse effects on apparent in vitro fermentation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Animal agriculture, and in particular ruminant enteric methane (CH4) production, is the major contributor to greenhouse gas (GHG) emissions from the agricultural sector (Reay et al. 2010). Symbiotic microorganisms in the rumen ferment feed and release energy in the form of metabolic substrate molecules, particularly volatile fatty acids (VFA), which are absorbed by the host ruminant. Concomitantly, methanogens in the rumen microbial consortium use fermentation end-products to generate CH4 as a hydrogen (H2) sink within the rumen (Morgavi et al. 2010). This production of enteric CH4 has economic costs representing a substantial loss of over 5 % of the total gross dietary energy consumed by the animal (Beauchemin and McGinn 2006; Hristov et al. 2013). Given this loss and the high global warming potential of CH4, considerable efforts are being made to develop methodologies to mitigate CH4 from ruminant production systems.
The management of nutrition in ruminants using traditional or novel plants/forages is a target strategy for the mitigation of CH4 emissions from livestock (Meale et al. 2012; Patra 2012). This strategy focuses on using the nutritional and biochemical properties of feeds, including secondary metabolites, to manipulate ruminal microbial populations and metabolism to reduce the production of enteric CH4, enhance the efficiency of energy use, and consequently the productivity of livestock. Secondary metabolites from terrestrial plants have been widely targeted for their antimethanogenic potential (Bodas et al. 2012; Goel and Makkar 2012; Patra 2011). Secondary metabolites may suppress methanogenesis by directly reducing or inhibiting the population of methanogens, and indirectly through the reduction of methanogenic substrate or populations of ruminal protozoa that maintain symbiotic relationships with methanogens (Cieslak et al. 2013). However, high concentrations of secondary metabolites may be required to effectively decrease rumen methanogenesis, which often impairs the fermentation of feed and the overall productivity of the ruminant (Goel and Makkar 2012). Additionally, the effects of secondary metabolites are often variable and contradictory due to the differences in extracts, doses, and the type and quality of basal diet (Martínez-Fernández et al. 2013; Cieslak et al. 2014; Mateos et al. 2013).
Macroalgae and their secondary metabolites have been shown to effectively decrease in vitro methane production (Wang et al. 2008; Machado et al. 2014b; Kinley and Fredeen 2014; Dubois et al. 2013). However, studies to date have been fundamental in their approach utilizing a screening methodology to identify key target species for further investigation. Additionally, the biochemical profile of macroalgae is variable between species and their positive or adverse effects on animal health, and productivity will depend on doses of inclusion in the diet. Of the limited diversity of macroalgae assessed for effects on in vitro methane production, the red macroalga Asparagopsis taxiformis shows the greatest potential to inhibit the production of CH4 (Machado et al. 2014b). This species has high antimicrobial activity, and this may have an effect on the in vitro fermentation of substrates. In contrast, other species of macroalgae have high nutritional value and can improve the production of fermentation products but have lower antimethanogenic potency (Kinley and Fredeen 2014; Machado et al. 2014b; Dubois et al. 2013). Although the antimethanogenic activity of Oedogonium is considerably lower than Asparagopsis, this green macroalga ameliorates in vitro anaerobic fermentation resulting in higher concentration of metabolizable end-products when added to a low-quality hay, typical of forages available across northern Australia (Machado et al. 2014b).
Asparagopsis and Oedogonium are two macroalgae with distinct biochemical profiles suitable for use in ruminant production systems. Additional information is required to evaluate the potential of these macroalgae as novel feed supplements for ruminants, namely the antimethanogenic capability and effects on in vitro fermentation. The aim of this study was to evaluate the dose-response effects of Asparagopsis and Oedogonium individually, and in combination, on the production of CH4 and main fermentation parameters in vitro. A wide range of doses of each macroalga was assessed to identify an optimal dose to achieve the inhibition of methanogenesis while having minimal adverse effects on overall in vitro fermentation.
Methods
Substrates and biochemical analyses
The macroalgae Asparagopsis taxiformis (Rhodophyta) and Oedogonium sp. [Chlorophyta, Tsv1 GenBank Accession No KC701473 (Lawton et al. 2013)] were acquired from the culture collection of the Centre for Macroalgal Resources and Biotechnology (MACRO) at James Cook University (JCU), Townsville, Australia. Macroalgal biomass was rinsed in freshwater to remove epiphytes, detritus, and sand. The biomass was centrifuged and freeze-dried (VirTis 2 K benchtop freeze-drier) at −55 °C and 120 μbar for 48 h. Rhodes grass hay [Chloris gayana, neutral detergent fiber (NDF) = 750 g kg−1 dry matter (DM); acid detergent fiber (ADF) = 401 g kg−1 DM] was used as the basal diet of the donor steers and as substrate for in vitro incubations. Freeze-dried macroalgae and air-dried hay samples were ground to 1 mm and stored in airtight containers at −20 °C until incubation.
Dry matter (DM), organic matter (OM), total lipids, carbohydrates, and gross energy (GE) analyses were carried out as previously described by Machado et al. (2014b). Crude protein (CP) fraction was estimated using the total nitrogen content (wt %) of the biomass, which was determined through elemental analysis performed by OEA labs (www.oealabs.com, UK). The CP content was based on the nitrogen factors of 4.7 for Oedogonium (Neveux et al. 2014), 4.59 for Asparagopsis (Lourenço et al. 2002), and 6.25 for Rhodes grass hay.
In vitro incubation
Rumen fluid was collected from rumen-fistulated Brahman (Bos indicus) steers maintained at the College of Public Health, Medical and Veterinary Sciences, JCU, according to experimental guidelines approved by CSIRO Animal Ethics Committee (A5/2011) and in accordance with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (NHMRC 2004). The steers were fed Rhodes grass hay (Chloris gayana) ad libitum throughout the study to ensure consistency of microbial activity in the inoculums. Rumen sampling and preparation of rumen-buffer medium were as described by Machado et al. (2014b). In brief, collected rumen fluid was transported in insulated flasks to the laboratory, where they were pooled, blended for 30 s to ensure complete detachment of microbes from the substrate biofilm, and strained through a 0.5-mm mesh under continuous flow of high purity N2 to maintain anaerobic conditions. Rumen medium was prepared using the strained rumen fluid and a preheated buffer solution (Goering and Van Soest 1970) in a 1:4 (v/v) ratio.
The Rhodes grass hay substrate and macroalgae were weighed into 250-mL Schott bottles, maintaining a total of 1.0 g OM in 125 mL of rumen medium. A blank consisting of the rumen medium only was included in each incubator. Batch culture incubations were conducted using an automated incubation system with RF receivers (Ankom Technology RF, Macedon, NY). Bottles containing substrate, alga, and rumen incubation medium were maintained in dedicated incubators with oscillation platforms (Ratek OM11, Boronia, Australia) at 39 °C and 85 rpm. Each bottle was fitted with an Ankom RF module and monitored for 72 h with gas pressure being measured every 60 s and cumulative pressure recorded every 20 min to generate total gas production (TGP) curves. Incubations were replicated three times, and bottles were randomly allocated between and within incubators.
Dose-response of individual macroalgae
The effects of Asparagopsis and Oedogonium were tested individually at a range of doses. Rhodes grass hay was used as a basal substrate to identify the optimum doses of each macroalga for the reduction of methane production while minimizing the adverse effects on anaerobic fermentation. Asparagopsis was tested at 0, 0.07, 0.125, 0.25, 0.5, 1, 2, 5, 10, and 16.7 % OM; Oedogonium was tested across a broader range of 0, 10, 16.7, 25, 50, 75, and 100 % OM (Table 2), with Rhodes grass constituting the remaining % of OM required to achieve a total 1 g OM incubated. Each treatment consisted of four replicates over three incubation periods. Head space gas samples were collected at 72 h. Apparent degradability of organic matter (OMdeg), production of VFA, which is the main source of energy for the animal (Russell et al. 1992), and pH were also measured at the end of each 72-h incubation period to characterize the effects of macroalgae on in vitro fermentation.
Dose-response of macroalgae combinations
Based on results of the dose-response effects of macroalgae on fermentation parameters, an experiment was designed to quantify the effects of combining the two macroalgae on VFA production, pH, OMdeg, and total CH4. The combinations of Asparagopsis (no addition of Asparagopsis vs addition of Asparagopsis 2 % OM basis) and Oedogonium [(0 %, low (25 %), and high (50 % OM)] resulted in six diet treatments, each consisting of a total of four replicates over three incubation periods. Head space gas samples were analyzed for CH4 at 2, 4, 6, 8, 10, 12, 24, 48, and 72 h. For each treatment, bottles were removed after 72 h to determine the effects on OMdeg and production of VFA.
Gas and fermentation parameter analysis
Head space gas samples were collected in to pre-evacuated 10-mL vials (Exetainer® vials, Labco Ltd., UK). Head space gas samples (0.5 mL) were used to determine CH4 concentrations using gas chromatography (Varian CP-3800), equipped with a BR Q-Plot 30 m × 0.53 mm ID column and a flame ionization detector (FID). The injector temperature was 200 °C, column temperature ramped from 65 to 89 °C at a rate of 6.0 °C min−1 over 4 min. Helium was used as the carrier gas at 2.8 mL min−1. Gas standards used for construction of standard curves and quality control during analysis were as described by Machado et al. (2014b). Peak areas were determined by manual integration and CH4 reported as mL g−1 OM incubated.
For both experiments (doses of individual and combinations of macroalgae), the fermentation liquor of each replicate was measured for pH, filtered through sintered glass crucibles (porosity 1), and the solid phase dried at 100 °C for 48 h. Organic matter residues of the solid phase were then combusted in a muffle furnace for 8 h at 530 °C for the determination of OMdeg. Filtrates were analyzed for VFA concentrations, which were corrected for the blank as described by Machado et al. (2014b).
Data analysis
Total gas production (TGP) data were corrected for the blank and fitted to a modified nonlinear sigmoidal model of Gompertz as described by Machado et al. (2014b). The gas production parameters A, B, and C were calculated using a nonlinear procedure (JMP 10, SAS Institute, USA).
Results from the dose-response effects of macroalgae on individual fermentation parameters were analyzed separately for each species of macroalga. One-factor permutational analyses of variance (PERMANOVA) were used to test for significant differences in the mean between doses for each species (fixed factor) on TGP, CH4 production, VFA, OMdeg, and pH at 72 h of incubation. Pair wise a posteriori comparisons were used (α = 0.05), where applicable. All analyses were performed using PRIMER 6 [v. 6.1.13; Clarke and Gorley 2006] and PERMANOVA+ [v. 1.0.3; Anderson et al. 2008]. Bray-Curtis similarity matrices were produced using the untransformed raw data, and a dummy variable (0.0001) was used to account for zero values. P values of PERMANOVA analyses were calculated from 9999 random permutations of raw data.
Results from the effects of combinations of macroalgae on fermentation parameters were analyzed using two-factor PERMANOVA (Table S1). The effects of the fixed factor dose of Oedogonium (0, 25, and 50 % OM) and addition of Asparagopsis (0 and 2 % OM) on TGP, CH4 production, VFA, OMdeg, and pH were tested. Bray-Curtis matrices and P values of PERMANOVA analyses were calculated as described above.
Results
Biochemical parameters of substrates
Organic matter content varied between substrates and was highest for Asparagopsis (936.0 g kg−1 DM) followed by Oedogonium (885.6 g kg−1) and was lowest for Rhodes grass hay (859.4 g kg−1 DM, Table 1). The carbohydrate content of Oedogonium was 16 % lower than that of Rhodes grass hay (666.7 g kg−1 DM), while the carbohydrate content of Asparagopsis was similar to that of Rhodes grass hay. The crude protein content of Asparagopsis (254.7 g kg−1) and Oedogonium (231.2 g kg−1) was 52.6 and 38.5 % higher than Rhodes grass hay (166.9 g kg−1 DM), respectively. The lipid contents of Asparagopsis (33.3 g kg−1) and Oedogonium (79.4 g kg−1) were 30. 4 and 205 % higher than Rhodes grass hay, respectively.
Dose-response of individual macroalgae
Asparagopsis and Oedogonium significantly decreased in vitro gas parameters with increasing doses (Fig. 1, Table 2). Asparagopsis decreased TGP by 31.5 to 46.5 % compared with the control at doses ranging from 1 to 16.7 % OM (Fig. 1a). The production of CH4 decreased significantly by 84.7 % at a dose of 1 % OM Asparagopsis, and it was virtually eliminated at doses ≥2 % OM, with a decrease of over 99 % compared with the control (Fig. 1b). Although Oedogonium was not as effective as Asparagopsis in inhibiting methanogenesis, TGP and CH4 production steadily decreased as the dose of Oedogonium increased (Fig. 1c, d). At doses ≥50 % OM, Oedogonium significantly decreased TGP by at least 20 % compared with the control (Fig. 1c). At dose ≥75 % OM, Oedogonium decreased CH4 production at least 50 % compared with the control (Fig. 1d), and at a dose of 100 % Oedogonium, CH4 production was reduced by 61.6 %.
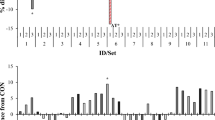
The degradability of organic matter (OMdeg) and the production of total VFA, both measures of efficiency of ruminal fermentation, decreased significantly as the dose of macroalgae increased for both Asparagopsis and Oedogonium (Fig. 2, Table 2). At doses ≤5 % OM, Asparagopsis had equal or higher OMdeg compared to the control, with OMdeg significantly decreasing at doses ≥10 % OM (Fig. 2a). Although the degradation of substrate was not affected by the addition of low doses of Asparagopsis, the production of total VFA was significantly decreased for doses ≥0.5 % OM. At doses of 1 and 2 % OM, Asparagopsis decreased the concentration of total VFA by 16.6 and 25 %, respectively, compared with the control (Fig. 2b, Table 2), and at doses ≥5 %, OM total VFA was decreased by 39.5 %. Conversely, the molar proportions of the VFAs propionate, butyrate, valerate, and isovalerate increased significantly for a dose of 2 % OM Asparagopsis compared with the control (Table 2) while the proportion of acetate and isobutyrate decreased. Acetate to propionate ratio decreased significantly by 63 % for the 2 % OM dose compared with the control (Table 2).
Dose-response of the macroalgae Asparagopsis and Oedogonium analyzed individually on degradability of organic matter (a, c, respectively) and total production of volatile fatty acids (b, d, respectively) after 72 h of in vitro incubation. Note that algal doses (X-axis) vary among species. Error bars represent ±SE (n = 4)
For Oedogonium, OMdeg and total VFA decreased as dose increased, with an average decrease of 11.3 % in OMdeg (Fig. 2c) and 17.4 % in the production of total VFA (Fig. 2d) for the dose of 25 % OM, compared with the control (Table 2). However, only doses of Oedogonium ≥50 % OM significantly decreased OMdeg and VFA compared with the control. The acetate to propionate ratio varied among doses but not from the control (Table 2). Doses of Oedogonium ≥25 % OM significantly increased the pH compared with the control after 72 h (Table 2).
Dose-response of macroalgae combinations
Oedogonium significantly decreased the TGP as the dose increased, and the addition of Asparagopsis at 2 % OM further decreased TGP (Fig. 3a, Table 3), showing a significant interaction between dose of Oedogonium and addition of Asparagopsis. Oedogonium alone decreased TGP by 8.5 and 26.9 % at 25 and 50 % OM, respectively, compared with the control (Oedogonium 0 % and Asparagopsis 0 %). When Asparagopsis 2 % OM was added in combination with Oedogonium at 25 or 50 % OM, the TGP was decreased by 42 and 59 %, respectively, compared with the control (Fig. 3a). Conversely, Oedogonium alone did not affect the production of CH4 at any dose, and no significant interaction among dose of Oedogonium and addition of Asparagopsis was detected. The addition of 2 % Asparagopsis individually, or in combination with Oedogonium, significantly decreased the production of CH4 by over 99 % independent of Oedogonium (Fig. 3b, Table 3).
The effects of combinations of the macroalgae Asparagopsis and Oedogonium on the production of total gas (a) and methane (b) after 72 h of in vitro incubation. Note that in b, all treatments with addition of Asparagopsis fall within the x-axis since the production of methane was near zero or below detection levels. −A, no addition of Asparagopsis; +A, addition of 2 % of Asparagopsis (OM basis). Error bars represent ±SE (n = 4). The control mentioned within the text refers to Oedogonium 0 % and no addition of Asparagopsis
Oedogonium significantly decreased the OMdeg and the production of total VFA as the dose increased (Fig. 4a, Table 3). Oedogonium decreased OMdeg by 14 and 24.8 % while VFA was decreased by 10 and 31 %, for the doses 25 and 50 % OM, respectively, compared with the control (Oedogonium 0 % and Asparagopsis 0 %). The addition of 2 % OM Asparagopsis also significantly decreased OMdeg by 7 % and production of total VFA by 12 %, compared with the control (Fig. 4, Table 3). When Asparagopsis was added at 2 % OM in combination with Oedogonium at 25 or 50 % OM, the production of total VFA decreased by 19.6 and 40 %, respectively, compared with the control. However, no significant interaction between dose of Oedogonium and the addition of Asparagopsis could be detected for OMdeg or total production of VFA. The addition of Asparagopsis individually, or in combination with Oedogonium, significantly decreased the ratio of acetate to propionate to less than half that of treatments without Asparagopsis, independent of the dose of Oedogonium.
The effects of combinations of the macroalgae Asparagopsis and Oedogonium on degradability of organic matter (a) and total production of volatile fatty acids (b) after 72 h of in vitro incubation. Note that in b, all treatments with addition of Asparagopsis fall within the x-axis since the production of methane was near zero or below detection levels. −A, no addition of Asparagopsis; +A, addition of 2 % of Asparagopsis (OM basis). Error bars represent ±SE (n = 4). The control mentioned within the text refers to Oedogonium 0 % and no addition of Asparagopsis
Discussion
The effects of Asparagopsis and Oedogonium on in vitro gas and fermentation parameters are dose-dependent. This study clearly defines the minimum effective dose required to decrease the production of CH4 in vitro while also identifying the doses at which the production of fermentation products, and potentially animal production, is affected. Asparagopsis has potent antimethanogenic activity with doses as low as 2 % OM decreasing the production of CH4 by more than 99 %, providing similar levels of reduction to that reported for other potent antimethanogens such as bromochloromethane (Goel et al. 2009) and 2-nitroethanol (Zhou et al. 2011). Asparagopsis produces more than 100 low molecular weight metabolites containing bromine, iodine, and chlorine that have antimicrobial activity (Woolard et al. 1979; Paul et al. 2006). Notably, bromoform is the most abundant metabolite produced by Asparagopsis (Paul et al. 2006). Halogenated analogs are known to inhibit methanogenesis by reacting with a vitamin B12 cofactor, thereby inhibiting the enzymatic reaction that decreases cobamide-dependent methane formation (Wood et al. 1968). Similar halogenated compounds have demonstrated long-lasting effects on methanogenesis in vivo with limited effects on animal production (Tomkins et al. 2009).
The organic matter ingested by ruminants is degraded through anaerobic fermentation by the rumen microbial consortium generating VFA, the main source of energy for ruminants (Russell et al. 1992), and both OMdeg and production of VFA are indicators of fermentation efficiency. In this study, OMdeg and the production of total VFA decreased as the dose of macroalgae increased for both Asparagopsis and Oedogonium. However, only doses of Asparagopsis ≥10 % OM significantly affected the OMdeg. Nevertheless, the final concentration of total VFA decreased by 12 to 25 % at a dose of 2 % OM (see Tables 2 and 3), due to a decrease in the production of acetate lowering the ratio of acetate to propionate. Nevertheless, the proportion of propionate, butyrate, valerate, and isovalerate increased significantly for the Asparagopsis dose of 2 % OM, suggesting that alternative fermentation processes take place when methanogenesis is inhibited. The absence of significant detrimental effects on in vitro fermentation parameters, in particular the OMdeg, using lower doses (<5 % OM) of Asparagopsis supports the potential for the macroalga to decrease the CH4 production with minimized adverse effects on ruminal fermentation. The use of extracts or purified metabolites from Asparagopsis may further decrease any impact on the production of VFA while maintaining antimethanogenic bioactivity. Consequently, the next challenge in understanding the mechanism of action of Asparagopsis is the quantitative and qualitative analysis of the effects of specific secondary metabolites produced by this alga on the diversity and activity of the rumen microbial biome.
Although Oedogonium was not as effective as Asparagopsis in inhibiting methanogenesis, Oedogonium significantly decreased the production of CH4 as the dose increased. Nevertheless, in this study, Oedogonium was less effective in decreasing the in vitro production of CH4 than reported in previous studies (Dubois et al. 2013; Machado et al. 2014b). Differences in substrate used across these studies may have contributed to the variable antimethanogenic response. In general, substrates with a high protein content generate lower volumes of total gas on a per gram of OM basis (Cone and van Gelder 1999) and methane (Johnson and Johnson 1995) than fibrous low-quality material. The Oedogonium used in this study had higher gross energy and lipid content and 3.4 times higher crude protein content than Rhodes grass hay (Table 1). The inclusion of lipids in ruminant diets has also been demonstrated to decrease methanogenesis in cattle (Beauchemin et al. 2007). The decrease in TGP and production of CH4 may be related to the protein and lipid content of Oedogonium, especially when included at higher doses.
The OMdeg and the production of VFA were significantly decreased for doses of Oedogonium ≥50 % OM. The production of VFA is related primarily to the availability of carbohydrates in the rumen (France and Dijkstra 2005). The increased lipid and protein content associated with the higher doses of Oedogonium decrease the proportion of carbohydrates available for the production of VFA thereby lowering the concentration within the incubations. Nevertheless, Oedogonium has the potential to improve the production of VFA in environments characteristic of northern Australia, where dietary crude protein is a limiting nutrient for the production of rumen microbial crude protein (Poppi and McLennan 1995), particularly during the dry season (Machado et al. 2014a). Oedogonium has the potential to be included in intensive beef cattle production systems as an alternative energy source where algal biomass could be cultured using wastewater (Cole et al. 2015; Hu et al. 2013; Chen et al. 2012). This offers a potentially sustainable and novel feed source to the livestock industry. Additionally, Oedogonium has a high content of polyunsaturated fatty acids (PUFA (Machado et al. 2014b)), with health benefits for ruminants and resulting in improved meat quality (Scollan et al. 2006).
The effectiveness of commonly used feed additives in decreasing the production of CH4 typically varies with the type and quality of substrate (O’Brien et al. 2014). In this study, the addition of 2 % Asparagopsis eliminated the production of CH4 independent of the dose of Oedogonium. The results are consistent with previous halogenated CH4 inhibitors tested in vitro (O'Brien et al. 2014; Lee et al. 2009). Halogenated analogs, such as those found in Asparagopsis, are effective in decreasing the in vitro methanogenesis independent of the type and quality of the basal substrate used here (Rhodes grass only or in combination with Oedogonium). The combination of Asparagopsis and Oedogonium had a cumulative effect on fermentation parameters, indicating that the type and quality of substrates influence the extent of the adverse effects on in vitro fermentation.
In conclusion, this study demonstrated that Asparagopsis is a potent antimethanogen as the optimal dose of 2 % OM decreased the in vitro production of CH4 by over 99 % compared with the control. At this low dose, negative effects on fermentation parameters are minimized. Therefore, Asparagopsis is a potential feed additive for CH4 mitigation in ruminant production systems. Oedogonium was a less potent antimethanogenic agent, but its nutritional value indicates that it could be used as a feed supplement at levels of ≤25 % of the diet (OM).
References
Anderson M, Gorley R, Clarke K (2008) Permanova + for primer: guide to software and statistical methods. Primer-E, Plymouth
Beauchemin K, McGinn S (2006) Methane emissions from beef cattle: effects of fumaric acid, essential oil, and canola oil. J Anim Sci 84:1489–1496
Beauchemin KA, McGinn SM, Petit HV (2007) Methane abatement strategies for cattle: lipid supplementation of diets. Can J Anim Sci 87:431–440
Bodas R, Prieto N, García-González R, Andrés S, Giráldez FJ, López S (2012) Manipulation of rumen fermentation and methane production with plant secondary metabolites. Anim Feed Sci Technol 176:78–93
Chen R, Li R, Deitz L, Liu Y, Stevenson RJ, Liao W (2012) Freshwater algal cultivation with animal waste for nutrient removal and biomass production. Biomass Bioenergy 39:128–138
Cieslak A, Szumacher-Strabel M, Stochmal A, Oleszek W (2013) Plant components with specific activities against rumen methanogens. Animal 7:253–265
Cieslak A, Zmora P, Stochmal A, Pecio L, Oleszek W, Pers-Kamczyc E, Szczechowiak J, Nowak A, Szumacher-Strabel M (2014) Rumen antimethanogenic effect of Saponaria officinalis L. phytochemicals in vitro. J Agri Sci 152:981–993
Clarke KR, Gorley RN (2006) Primer v6: user manual/tutorial. Primer-E Ltd, Plymouth
Cole AJ, de Nys R, Paul NA (2015) Biorecovery of nutrient waste as protein in freshwater macroalgae. Algal Res 7:58–65
Cone JW, van Gelder AH (1999) Influence of protein fermentation on gas production profiles. Anim Feed Sci Technol 76:251–264
Dubois B, Tomkins NW, Kinley RD, Bai M, Seymour S, Paul NA, de Nys R (2013) Effect of tropical algae as additives on rumen in vitro gas production and fermentation characteristics. Am J Plant Sci 4:34–43
France J, Dijkstra J (2005) Volatile fatty acid production. In: Forbers J, France J (eds) Quantitative aspects of ruminant digestion and metabolism. CAB International, Oxon, pp 157–175
Goel G, Makkar HPS (2012) Methane mitigation from ruminants using tannins and saponins. Trop Anim Health Prod 44:729–739
Goel G, Makkar HP, Becker K (2009) Inhibition of methanogens by bromochloromethane: effects on microbial communities and rumen fermentation using batch and continuous fermentations. Br J Nutr 101:1484–1492
Goering H, Van Soest PJ (1970) Forage fiber analyses (apparatus, reagents, procedures, and some applications), vol 379. US Agricultural Research Service, Washington
Hristov A, Oh J, Firkins J, Dijkstra J, Kebreab E, Waghorn G, Makkar H, Adesogan A, Yang W, Lee C (2013) Special topics—Mitigation of methane and nitrous oxide emissions from animal operations: I. A review of enteric methane mitigation options. J Anim Sci 91:5045–5069
Hu B, Zhou W, Min M, Du Z, Chen P, Ma X, Liu Y, Lei H, Shi J, Ruan R (2013) Development of an effective acidogenically digested swine manure-based algal system for improved wastewater treatment and biofuel and feed production. Appl Energy 107:255–263
Johnson KA, Johnson DE (1995) Methane emissions from cattle. J Anim Sci 73:2483–2492
Kinley R, Fredeen A (2014) In vitro evaluation of feeding North Atlantic stormtoss seaweeds on ruminal digestion. J Appl Phycol. doi:10.1007/s10811-014-0487-z
Lawton RJ, de Nys R, Paul NA (2013) Selecting reliable and robust freshwater macroalgae for biomass applications. PLoS ONE 8(5), e64168
Lee S, Yang S, Lee W, Kim H, Shin D, Ha JK (2009) Effect of 2-bromoethanesulfonic acid on in vitro fermentation characteristics and methanogen population. Asian-Australas J Anim Sci 22:42–48
Lourenço SO, Barbarino E, De-Paula JC, Pereira LOS, Marquez UML (2002) Amino acid composition, protein content and calculation of nitrogen‐to‐protein conversion factors for 19 tropical seaweeds. Phycol Res 50:233–241
Machado L, Kinley RD, Magnusson M, de Nys R, Tomkins NW (2014a) The potential of macroalgae for beef production systems in Northern Australia. J Appl Phycol. doi:10.1007/s10811-014-0439-7
Machado L, Magnusson M, Paul NA, de Nys R, Tomkins N (2014b) Effects of marine and freshwater macroalgae on in vitro total gas and methane production. PLoS ONE 9(1), e85289
Martínez-Fernández G, Abecia L, Martín-García A, Ramos-Morales E, Hervás G, Molina-Alcaide E, Yáñez-Ruiz D (2013) In vitro–in vivo study on the effects of plant compounds on rumen fermentation, microbial abundances and methane emissions in goats. Animal 7:1925–1934
Mateos I, Ranilla M, Tejido M, Saro C, Kamel C, Carro MD (2013) The influence of diet type (dairy versus intensive fattening) on the effectiveness of garlic oil and cinnamaldehyde to manipulate in vitro ruminal fermentation and methane production. Anim Prod Sci 53:299–307
Meale S, Chaves A, Baah J, McAllister T (2012) Methane production of different forages in in vitro ruminal fermentation. Asian-Australas J Anim Sci 25:81–91
NHMRC (National Health and Medical Research Council) (2004) Australian code of practice for the care and use of animals for scientific purposes. Canberra, Australia. http://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/ea16_animal_code.pdf
Morgavi D, Forano E, Martin C, Newbold CJ (2010) Microbial ecosystem and methanogenesis in ruminants. Animal 4:1024–1036
Neveux N, Magnusson M, Maschmeyer T, de Nys R, Paul NA (2014) Comparing the potential production and value of high-energy liquid fuels and protein from marine and freshwater macroalgae. GCB Bioenergy. doi:10.1111/gcbb.12171
O'Brien M, Navarro-Villa A, Purcell P, Boland T, O'Kiely P (2014) Reducing in vitro rumen methanogenesis for two contrasting diets using a series of inclusion rates of different additives. Anim Prod Sci 54:141–157
Patra AK (2011) Effects of essential oils on rumen fermentation, microbial ecology and ruminant production. Asian J Anim Vet Adv 6:416–428
Patra AK (2012) Enteric methane mitigation technologies for ruminant livestock: a synthesis of current research and future directions. Environ Monit Assess 184:1929–1952
Paul N, de Nys R, Steinberg P (2006) Chemical defence against bacteria in the red alga Asparagopsis armata: linking structure with function. Mar Ecol Prog Ser 306:87–101
Poppi DP, McLennan SR (1995) Protein and energy-utilization by ruminants at pasture. J Anim Sci 73:278–290
Reay D, Smith P, van Amstel A (2010) Methane sources and the global methane budget. In: Reay D, Smith P, van Amstel A (eds) Methane and climate change. Earthscan Ltd., Washington, pp 1–13
Russell J, O'connor J, Fox D, Van Soest P, Sniffen C (1992) A net carbohydrate and protein system for evaluating cattle diets: I. Ruminal fermentation. J Anim Sci 70:3551–3561
Scollan N, Hocquette J-F, Nuernberg K, Dannenberger D, Richardson I, Moloney A (2006) Innovations in beef production systems that enhance the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci 74:17–33
Tomkins N, Colegate S, Hunter R (2009) A bromochloromethane formulation reduces enteric methanogenesis in cattle fed grain-based diets. Anim Prod Sci 49:1053–1058
Wang Y, Xu Z, Bach S, McAllister T (2008) Effects of phlorotannins from Ascophyllum nodosum (brown seaweed) on in vitro ruminal digestion of mixed forage or barley grain. Anim Feed Sci Technol 145:375–395
Wood J, Kennedy FS, Wolfe R (1968) Reaction of multihalogenated hydrocarbons with free and bound reduced vitamin B12. Biochemistry 7:1707–1713
Woolard FX, Moore RE, Roller PP (1979) Halogenated acetic and acrylic acids from the red alga Asparagopsis taxiformis. Phytochemistry 18:617–620
Zhou Z, Meng Q, Yu Z (2011) Effects of methanogenic inhibitors on methane production and abundances of methanogens and cellulolytic bacteria in in vitro ruminal cultures. Appl Environ Microbiol 77:2634–2639
Acknowledgments
This research is part of the MBD Energy Research and Development program for Biological Carbon Capture and Storage. This project is supported by funding from the Australian Government Department of Agriculture through the National Livestock Methane Production Project, the Australian Government through the Australian Renewable Energy Agency, and the Advanced Manufacturing Cooperative Research Centre (AMCRC), funded through the Australian Government’s Cooperative Research Centre Scheme. We thank Dr. Pedro de Paula Silva for assistance with the experiments and Dr. Matthew Vucko for the advice with the statistical analyses. We also thank Jeffrey Palpratt for handling and maintenance of the donor steers, and Dr. Shane Askew from the Advanced Analytical Centre, JCU, for analytical advice
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Results of full factorial permutational analyses of variance (PERMANOVAs) testing the effects of the fixed factors time (Ti), dose of Oedogonium (Do), and addition of Asparagopsis (Ad) on Gas parameters, VFA profiles, OMdeg, and pH of treatments in the macroalgae combination experiment. Analyses were conducted in Primer v6 (Primer-E Ltd, UK) using Bray-Curtis dissimilarities on fourth root transformed data and 999 unrestricted permutations of raw data. Pseudo F (F) and P values are presented, significant terms shown in bold. (DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Machado, L., Magnusson, M., Paul, N.A. et al. Dose-response effects of Asparagopsis taxiformis and Oedogonium sp. on in vitro fermentation and methane production. J Appl Phycol 28, 1443–1452 (2016). https://doi.org/10.1007/s10811-015-0639-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0639-9