Abstract
New Chlorphenoxamine (Ch) ion selective plastic membrane electrodes for both conventional and coated wire types based on the ion associate of Ch-HCl with sodium tetraphenylborate (NaTPB) and phosphotungestic acid (PTA) were prepared. The conventional type electrode was fully characterized in terms of membrane composition, plasticizer, filling solution, life span, pH, ionic strength, temperature. The membranes of optimum compositions were used for the preparation of both graphite and cupper coated wire electrodes. The prepared electrodes were used for the potentiometric determination of the investigated drug in raw material and pharmaceutical preparations under batch and flow injection analysis conditions. The selectivity of the electrodes towards a large number of excipients like inorganic cations, sugars, amino acids and other antihistaminic drugs was tested. Determination of Ch-HCl, the solubility product of the ion associate and the formation constant of the precipitation reaction leading to the ion associate formation of Ch-HCl with NaTPB and PTA were carried out using conductimetric measurements.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Chlorphenoxamine Hydrochloride is a fast acting anti-allargic agent with antipruritic proprties and pleasantly cooling and sedative effect. Chemically, it is 2-[1-(4-chlorophenyl)-1-phenylethoxyl]-N,N-dimethylethanamine hydrochloride. (Molecular formula C18H22ClNO·HCl and molecular weight 340.29) [1].
Several methods have been reported in the literature for the determination of Ch-HCl including spectrophotometric methods [2–8], potentiometric method using NIO Metrhom electrode [9], thin layer chromatography [10, 11], plastic membrane ion selective electrode based on Tetrakis (4-chlorophenyl)borate [12], indirectly via its complexation with metal ions using Atomic Absorption Spectrometry [13], polarography [14] and differential pulse voltammetry [15].
In the present work, new selective plastic membranes electrodes for Chlorphenoxamine ion (Ch) of both conventional and coated wire type have been constructed; they are based on the incorporation of Ch-TPB or Ch-PTA ion exchangers in polyvinyl chloride (PVC) membrane plasticized with a proper plasticizer (dioctyl phthalate (DOP) or dibutyl phthalate (DBP)). The electrodes were fully characterized under batch conditions, and used for the determination of the drugs in pure solutions and in their pharmaceutical preparations by applying the standard additions and direct potentiometric titration methods in batch conditions and peak heights comparison in FIA conditions.
The conductimetric measurements were also applied to throw more light on the solubility and molar ratio of the used ion-exchangers and for the drugs determination in their pure state or pharmaceutical formulations. 
2 Experimental
2.1 Reagent and solutions
All reagents used were chemically pure grade. Bi-distilled water was used throughout all experiments. Pure grade Ch-HCl and its pharmaceutical preparations (Allergex and Allergex caffeine 20 mg/capsule) were provided by EPICO, Egypt. Poly (vinyl chloride) (PVC) of high molecular weight (Aldrich, England), dioctyl phthalate (DOP), dibutyl phthalate (DBP), nitrobenzene (NB), Paraffin oil (Fluka, Germany), tetrahydrofuran (THF) (Aldrich, England), sodium tetraphenylborate (NaTPB) and phosphotungestic acid (PTA) (Fluka, Germany) were used. During the FIA measurements the carrier and reagent solutions were de-gassed by mean of vacuum suction. Stock solutions 5.0 × 10−2 mol L−1 of Ch-HCl, TPB and PTA were prepared by dissolving the accurately weighed amounts in water and completed to the mark in a 50 mL volumetric flask. For tablet solution, 10 tablets of the pharmaceutical preparation were weighed, finely grounded and amount equivalent to the calculated weight of pharmaceutical preparations to produce a 1.0 × 10−2 mol L−1 solution was weighed and dissolved in bi-distilled water then filtered in 50 mL measuring flask and completed to the mark. Lower concentration solutions were prepared by appropriate dilutions.
2.2 Apparatuses
The potentiometric measurements in the batch mode were carried out with multimeter (Sanwa, Japan). A Techne circulator thermostat Model C-100 (UK) was used to control the temperature of the test solution. Saturated calomel electrode (SCE) was used as external reference electrode.
The flow injection set-up was composed of four channel peristaltic pump (ISM 827) (Ismatec, Zurich, Switzerland) and an injection valve (model 5020) with exchangeable sample loop from Rheodyne (Cotati, CA, USA). The electrode was connected to a WTW pMX 2000 microprocessor pH/ion meter and interfaced to a Model BD111 strip chart recorder from Kipp and Zonn (Delft, The Netherlands). The flow injection measurements were carried out in a two line system [16, 17] where the sample was injected into a carrier stream, which then merged with another stream; the same tubing size was used in both lines to offer the same flow rate. A wall-jet cell, providing a low dead volume, fast response, good wash conditions and compatibility with electrodes of different shape and size, was used in flow measurements, where a Perspex cup with axially positioned inlet polypropylene tubing was mounted at the sensing surface of the electrode body. The optimized distance between the nozzle and the sensing surface of the electrode was 3 mm; this provides the minimum thickness of the diffusion layer and consequently a fast response [18]. The ion selective electrode (ISE) with flow cup, reference electrode (SCE) and the outlet tubes were placed in a beaker, where the level of solution was kept 1 cm above the electrode surface.
For conductimetric determination a Jenway conductivity meter model 4510 was used for conductance measurements, the bridge is connected with a temperature sensor for temperature measurements and the cell constant k cell = 1.0.
2.3 Preparation of the ion exchanger
Chlorphenoxaminum tetraphenylborate (Ch-TPB) ion exchanger was prepared by mixing 100 mL (1.0 × 10−2 mol L−1) of NaTPB with 100 mL (1.0 × 10−2 mol L−1) of Ch-HCl while the chlorphenoxaminum phosphotungestate (Ch-PTA) ion exchanger was prepared by mixing 100 mL (1.0 × 10−2 mol L−1) of phosphotungestic acid with 300 mL (1.0 × 10−2 mol L−1) of Ch-HCl.
The resulting white precipitates of both Ch-TPB and Ch-PTA were left in contact with their mother liquors overnight to assure complete digestion. The precipitates were then filtered, washed by double distilled water until chloride ion free (as tested by acidic solution of AgNO3) and dried at room temperature for at least 48 h. The product was subjected to elemental analysis for C, H, and N in the Micro analytical Center of Cairo University-Egypt. The results showed a good agreement with the calculated values for a 1:1 and 1:3 ion exchangers for both Ch-TPB and Ch-PTA, respectively.
2.4 Preparation of conventional and coated type electrodes
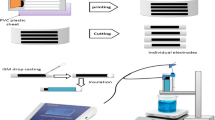
Membranes of different composition were prepared by dissolving the required amounts of PVC, plasticizer (DOP, DBP, Nitrobenzen or paraffin oil) and ion exchanger(s) of total weight 0.4 g in about 10 mL tetrahydrofuran, the solution mixture was poured into a 7.5 cm Petri-dish and left to dry in air (not less than 24 h), The conventional electrode was constructed as previously described [19]. A 12 mm diameter disk was cut out from the membrane and glued to the polished end of a PVC plastic cap attached to a glass tube. The electrode body was filled with a solution that is 1.0 × 10−1 mol L−1 in NaCl and 1.0 × 10−3, 1.0 × 10−4 or 5 × 10−4 mol L−1 in Ch-HCl. A Ag/AgCl wire was immersed in the internal solution of the electrode to act as internal reference.
The coated graphite or cupper type electrodes were prepared using graphite or cupper rods that are 5 cm length and 4 mm diameter. One of the two ends of the rod was used for connection while the other, about 1 cm length, was dipped in a solution of the same optimum membrane composition used for the conventional type and left to dry in air. The process was repeated several times till a layer of the proper thickness was formed covering the terminal of the rod. Soaking for half an hour in Ch-HCl solution of the same concentration of the filling solution was required to precondition the conventional type electrodes, while the coated graphite and cupper types can be used without preconditioning.
2.5 Construction of the calibration graphs
For batch measurements, suitable increments of standard drug solutions were added to 50 mL bi-distilled water to cover the concentration range 1.0 × 10−6–5.0 × 10−2 mol L−1. The sensor and the reference electrodes were immersed in the solution and the e.m.f. value was recorded at 25 ± 1 °C, after each addition. The mV readings were plotted versus the negative logarithmic values of the drug concentration (pC).
For FIA measurements, a series of freshly prepared solutions of the drug covering the same concentration range used in batch measurements was injected to the flow stream and the corresponding peak heights were recorded and used to draw the calibration graphs.
2.6 Potentiometric determination of Ch-HCl
Ch-HCl was determined potentiometrically using the investigated electrodes under batch conditions by applying the standard addition [20] and potentiometric titration [21] methods. In the standard addition method known volumes of standard drug solution were added to 50 mL water containing different amounts of pure compound or the pharmaceutical preparations (allergex and allergex caffeine tablets). The change in mV reading was recorded for each increment and used to calculate the concentration of the drug in sample solution using the following equation:
where Cx and Vx are the concentration and the volume of the unknown solution respectively, Cs and Vs are the concentration and the volume of the standard solution, respectively, S is the slope of the calibration graph and ΔE is the change in millivolt due to the addition of the standard solution.
In potentiometric titration different weights of the investigated drug were transferred into 100 mL cell and diluted to 50 mL by distilled water. The resulting solutions were then titrated against 1 × 10−2 mol L−1 NaTPB or 5 × 10−3 mol L−1 PTA according to the corresponding ion-exchanger used in constructing the electrode. The relation between the volume added and the mV reading were graphically constructed and the end point was determined by the conventional S-shaped curves. The differential graph of the titration curves have also been constructed whenever it needed to obtain well defined and sharp end points using the computer program Origin lab.
Under FIA conditions, a series of solutions of different concentrations was prepared from allergex and allergex caffeine (20 mg/tablet) or unknown pure solution and the peak heights were measured using flow rate 9.7 mL min−1 and then compared with those obtained by injecting the standard series solutions under the same conditions and the recovery could be calculated as the ratio between the peak height of a sample and that of its corresponding standard concentration [21].
2.7 Conductimetric determination of the investigated drug and the solubility products of the ion-exchangers
Chlorphenoxamine Hydrochloride has been determined conductimetrically by taking different volumes of 5.0 × 10−3 mol L−1 stock solution of pure drug (Ch-HCl) or its pharmaceutical preparations, the taken volumes were transferred to a 50 mL volumetric flask and made to the mark with bi-distilled water. The contents of the volumetric flask were transferred to a beaker and the conductivity cell was immersed, then 5.0 × 10−3 mol L−1 NaTPB or PTA solution was added from micro burette and the conductance was measured subsequent to each addition of the reagent solution after thorough stirring. The conductance reading, taken after 1–2 min, after each addition was corrected for dilution [22] by mean of the following equation, assuming that conductivity is a linear function of dilution:
where Ω is the electrolytic conductivity, V1 is the initial volume and V2 is the volume of the added reagent (corr., corrected; obs., observed). A graph of corrected conductivity versus the volume of titrant added was constructed and the end point was determined graphically at the intersection of two lines.
To determine the drug–titrant ratio, 2 or 6 mL of 5 × 10−3 mol L−1 Ch-HCl completed to the required volume with bi-distilled water was titrated with 5 × 10−3 mol L−1 of NaTPB and PTA, respectively and the conductance was measured after each addition with thorough stirring.
For determining the solubility product a series of solutions of different concentrations (C = 1.0 × 10−5 to 5.0 × 10−3 mol L−1) was prepared for each of Ch-HCl, PTA and Na-TPB. The measured conductivities of these solutions at 25 °C were used to calculate the specific conductivities (K), corrected for the effects of the solvent and dilution, then the equivalent conductivities (λ) of the solutions, (λ = 1000 K/C) were obtained. Straight-line plots of λ versus (C1/2) were constructed and the equivalent conductance at infinite dilution (λ0) values was determined for Ch-HCl (λ0 Ch-HCl), PTA (λoPTA) and Na-TPB (λ0 Na-TPB) from the intercept of respective lines with the λ-axis. The activity coefficients of the ions employed were taken as unity because all solutions were sufficiently dilute and λ0Ch-TPB and λ0Ch-PTA were calculated from Kohlrausch,s law of independent migration of ions [23]. The solubility (S) and solubility product (Ksp) of the ion pair were obtained applying the following equations:
and
where Ks is the specific conductivity of a saturated solution of the ion exchanger determined at 25 °C and corrected for the effect of the solvent. This was done by stirring suspensions of the solid precipitates in distilled water for 3 h and measuring the conductivities.
3 Results and discussions
3.1 Optimization of the ISEs response in batch conditions
3.1.1 Effect of membrane composition and plasticizer
Several membrane compositions were investigated in which the content of the ion exchanger ranged from 3 to 10% of Ch-PTA and Ch-TPB. For each composition, the electrodes were repeatedly prepared four times. The preparation process was highly reproducible, as revealed from the low standard deviation SD values of the obtained slope for calibration graphs of the prepared membranes (SD average 1.30). The best performances were obtained by using membrane compositions containing 3.0% Ch-TPB, 48.5% plasticizer and 48.5% PVC for Ch-TPB electrodes and 7.0% Ch-PTA, 46.5% plasticizer and 46.5% PVC for Ch-PTA electrodes. Plasticizer plays a vital role in the sensor performance, as it is responsible for the ion-pair solvation and distribution in the membrane, thus controlling the detection limit [24], affecting the selectivity and sensitivity [25, 26] and give the membrane the proper elasticity and strength. Therefore the effect of four different plasticizers (DOP, DBP, nitrobenzene and Paraffin oil) on the performance characteristics of the prepared electrodes has been investigated. It was found that the membranes prepared using paraffin oil were too rigid to be attached to the plastic cape of the electrodes and those prepared using nitrobenzene as plasticizer have weak response to the concentration change where the slope of their calibration graphs never exceed 38 mV/conc. decade. It seems that DOP or DBP, as a low polarity solvent mediator, provides more appropriate conditions for incorporation of the highly lipophilic Ch+ ions into the membrane, therefore DOP or DBP has been used throughout the present investigation.
It was found that DOP is the proper plasticizer for Ch-PTA membrane, where the slope of its calibration graph was 55.80 mV/conc. decade within the concentration range 2.0 × 10−6 to 5.0 × 10−2 mol L−1 and DBP is the proper one for Ch-TPB membrane where the slope of its calibration graph was 55.65 mV/conc. decade within the concentration range 4.0 × 10−6 to 1.0 × 10−2 mol L−1. The optimum membrane compositions were also used for the preparation of coated wire electrodes where the slope of their calibration graphs was 53.00, 54.10 and 62.50 mV/conc. decade within the concentration ranges 4.0 × 10−6–5.0 × 10−2, 2.0 × 10−6–1.0 × 10−2 and 1.2 × 10−5–1.0 × 10−2 mol L−1 for Ch-TPB graphite, Ch-TPB cupper and Ch-PTA graphite electrodes, respectively. Ch-PTA copper electrode could not be constructed since during its drying a green color is formed on the copper wire and imparts the membrane a green color and the potential reading of the constructed electrode was fluctuated and did not show Nernestian behavior.
3.1.2 Effect of filling solution
Varying the composition of internal reference solution could considerably improve the linear range and limit of detection [27, 28], therefore the response of the electrode in relation to the variation of internal solution was investigated using the optimum membrane composition. Three different concentrations of Ch-HCl (1.0 × 10−3, 5.0 × 10−4 and 1.0 × 10−4 mol L−1) with 1 × 10−1 mol L−1 NaCl in filling solution were investigated. It was found that the Ch-TPB and Ch-PTA electrodes containing 1.0 × 10−4 mol L−1 of Ch-HCl were nearly Nernestian with stable reading and smaller response time (15 s) within the concentration ranges 4.0 × 10−6–1.0 × 10−2 and 2.0 × 10−6–5.0 × 10−2 mol L−1 for Ch-TPB and Ch-PTA electrodes, respectively.
3.1.3 Effect of soaking
For these study the constructed electrodes were soaked in a solution (1.0 × 10−4 mol L−1) of the respective drug at different intervals and the effect of soaking on the slope of their calibration graph, usable concentration range were studied for each electrode independently. The soaking process firstly activates the surface of the freshly prepared electrode to form an infinitesimal thin gel layer at the inter-junction boundary separating the membrane from the test solution; however the continuous soaking affects negatively the response of the electrode due to the leaching of the active ingredients (ion-exchanger and plasticizer) to the bathing solution [29]. It was found that the Ch-TPB conventional type electrodes worked in nearly Nernestian behavior in the first 12 days then the slope decreased gradually to reach 50 mV/conc. decade after about 22 days. The Ch-PTA electrode showed longer life time where the electrode worked in nearly Nernestian behavior for about 22 then the slope decreased gradually to reach 51.7 mV/conc. decades after 37 days. It was noted that, the electrodes which had been kept dry in closed vessel and stored in a refrigerator showed nearly constant slope values and response properties for several months if they were preconditioned before using by soaking in 1.0 × 10−4 mol L−1 Ch-HCl for 1 h. It was found that the life spans of the coated type electrodes are less than those of conventional type which may be attributed to the poor mechanical adhesion of the PVC-based sensitive layer to the conductive bed surface and insufficient electrochemical stability [30]. However, Some of the studied electrodes showed reasonable life span which indicated by their near-Nernestian behavior and wide concentration linearity range for 5 days or more, Ch-TPB/graphite (6 days), Ch-PTA/graphite (40 days), Ch-TPB/cupper (10 days), if they were kept in dry vessel at room temperature and presoaked for half hour before use.
3.2 Optimization of the ISEs response in FIA conditions
To evaluate the working characteristics of the constructed electrodes, and to make the developed FIA system suitable for the determination of the studied drugs in different samples, several parameters that controlling the system should be optimized, which include, dispersion coefficient, injection volume, flow rate and carrier composition.
3.2.1 Effect of injection volume
Different sample loops of different volumes range from 20.0 to 500.0 μL were connected to the flow injection system to select the most appropriate one for the measurements. In general the higher the sample volume, the higher are the peak heights but with a longer time to reach the steady state, base line and greater consumption of sample. A sample volume of 150 μL and 75 μL was used through out this work for Ch-PTA and Ch-TPB electrodes, respectively, giving about 96% of the maximum peak height obtained by a 500 μL loop but with a shorter time to reach the base line and less reagent consumption [31].
3.2.2 Effect of flow rate
The dependence of the peak height and time to reach the base line on the flow rate were studied. The response of the electrode to a solution that is 5 × 10−3 mol L−1 was studied at different flow rates (4.15, 5.35, 7.50, 9.70, 12.50, 17.85, 23.25, 25.00, 27.00 and 30.00 mL min−1). It was found that with a constant injection volume, the residence time of the sample was inversely proportional to the flow rate and the recovery time increased linearly with residence time of the sample at the active membrane surface [32]. It was found also that as the flow rate increased, the peaks become sharper until a flow rate of 7.5 and 9.5 mL min−1 was reached for Ch-TPB and Ch-PTA electrodes, respectively, above this values the peak heights were nearly the same over that a noise was observed in the base line at higher flow rates, for these reasons flow rates of 7.5 and 9.70 mL min−1 were chosen for further investigations for Ch-TPB and Ch-PTA electrodes, respectively, where about 99% of the maximum peak height is achieved with stable base line, short time to reach the base line and less consumption of the carrier.
3.2.3 Carrier composition
The base line attainment for the studied electrodes takes a very low short time by using bi-distilled water as a carrier stream. The use of other carrier solutions such as buffer or ionic strength adjuster such as potassium chloride in the second channel of the flow system did not affect the stabilization time, i.e. the time required to attain the base line, but only led to a decrease in the peak heights and higher consumption of reagents, therefore bi-distilled water was used as a carrier in both channels in the system through out the present work.
Under the aforementioned optimum conditions of injection volumes and flow rates the dispersion coefficients were calculated as the ratio between the peak height obtained at steady state conditions (where the sample act as carrier stream) and at the state of maximum peak height, maximum dispersion (where the sample is injected in the carrier stream) [33]. The dispersion coefficients were found to be 1.24 and 1.27 for Ch-TPB and Ch-PTA electrodes, respectively, these values reflect the limited dispersion of the sample that provide optimum sensitivity and fast response of the electrodes [34].
3.2.4 Electrode response under optimum FIA conditions
In potentiometric measurements using FIA, the electrode potential depends on the activity of the main ion sensed and this dependence is semi-logarithmic over a wide analyte activity range which is the main advantage of the device; however, the main unfavorable feature of this method is the slow response of electrode potential to concentration change and that is the reason for the super-Nernstian sensitivities obtained in FIA measurements using the investigated electrodes at different flow rates [35].
An increase in the slope of the calibration plots was observed compared with batch measurements, where potential is measured under conditions very close to the equilibrium at the membrane-solution interface [36]. It was found that the slope of the calibration graphs under FIA conditions ranged from 60.00 to 71.14 compared with 55.80 mV/conc. decade under batch condition for Ch-PTA electrode and from 56.0 to 67.10 under FIA conditions compared with 55.65 mV/conc. decade under batch condition for Ch-TPB electrode within the usable concentration range 1.0 × 10−5–5.0 × 10−2 mol L−1 for Ch-TPB and Ch-PTA electrodes. Figure 1a and b represent the recordings obtained by the studied electrodes at optimum FIA conditions.
3.3 Effect of the temperature of the test solution on the electrodes response
It is very important to study the effect of temperature on the performance characteristics of the plastic membrane electrode because many potentiometric measurements concerning biological media and fluids are carried out at different temperatures. For this purpose the calibration graphs of optimum composition were constructed at different test solution temperatures covering the range 25–70 °C and the isothermal coefficient (dE°/dt) was calculated as the slope of the straight line obtained by plotting \( E_{\rm elec}^{o} \) versus (t − 25), according to Antropov’s equation [37]:
where \( E_{\rm elec}^{o} \) is the standard electrode potential with respect to the normal hydrogen electrode at different temperatures which obtained from the calibration graphs as the intercepts at pCh = 0 (after subtracting the values of the standard electrode potential of the calomel electrode at this temperatures), t is the temperature of the test solution in °C and E 0(25) is the standard electrode potential at 25 °C.
It was found that, the slope of the calibration graphs of the constructed electrodes is stable up to 70 °C with isothermal coefficient 1.80 and 1.48 mV/°C for Ch-TPB and Ch-PTA electrodes, respectively. These low isothermal temperature coefficients values reveal the successful applicability of the electrode over a wide temperature range. Figure 2 represents the variation of the slope of the calibration graph with the temperature of the test solution.
3.4 Effect of pH of the test solution on the electrodes response
The variation of potential with changes of pH of the test solution (5 × 10−2, 1 × 10−3 and 5 × 10−4 mol L−1) was recorded. The pH of the solution was varied over the range of 1–8.3. The mV readings were plotted against the pH values for the different drug concentration corresponding to each electrode, Fig. 3.
In FIA, a series of 5.0 × 10−3 mol L−1 Ch-HCl solutions of different pH values ranging from 1.0 to 8.3 was injected in the flow stream and the peak heights, representing variation of potential response with pH were measured, solutions of higher pH values can’t be prepared due to the precipitation of chlorphenoxamine as base. It was found that Ch-TPB and Ch-PTA electrodes do not respond to pH changes in the range 2.5–7.8 for Ch-TPB and Ch-PTA electrodes under batch and FIA conditions. Ch-PTA electrode shows a relatively wider rang under batch conditions where it does not respond to the pH change in the range 1.8–7.8, Fig. 3. At pH values lower than previously mentioned ranges the potential and peak height decreased gradually which, may be attributed to either the interference of the hydronium ion or the penetration of chloride ions into the membrane network at the test solution-membrane interface while the decrease in potential and peak height at pH higher than 8.0 may be attributed to deprotonation of the investigated drug, which lead to a consequent decrease in its concentration [38].
3.5 Effect of the ionic strength of the test solution on the electrode response
Using ionic strength adjustment solution helps not only in the adjustment of the ionic strength but also, the activity coefficient, and the activity of the electroactive species present in the test solution, this provide accurate determination of the concentration of a given ion potentiometrically, regardless of its original environment. For this purpose, the performance characteristics for Ch-TPB and Ch-PTA electrodes were studied as a function of variation of potassium chloride concentration as ionic strength adjustor. It was found that the slope of the calibration graph for Ch-PTA electrode decreases with the increase of the KCl concentration, while the slope of Ch-TPB electrodes slightly increases by increasing the concentration of KCl till reach its maximum value of 56.71 mV/conc. decade in 1.0 × 10−3 mol L−1 of KCl then decreased in higher concentrations of KCl reaching 55.2 mV/concentration decade in 1.0 × 10−2 mol L−1. In all cases the decrease in the slope of the calibration graphs depends on the degree of interference of the K+ with the investigated drugs.
3.6 Selectivity of the electrodes
Selectivity is the ability of the electrode to respond primarily to only one species in the presence of others. The selectivity coefficient \( K_{{{\text{drug}},j}}^{\text{pot}} \) is the prime source of information concerning the effect of interferences on the electrode response. The response of the electrodes towards different substances and ionic species such as inorganic cations, amino acids, sugars and other antihistamine drugs (phenylpropanolamine hydrochloride and diphenylpyraline hydrochloride) that might be co-existing with the analyte as excipients, was checked under both of batch and FIA conditions using separate and mixed methods [39, 40] and the values of selectivity coefficients, shown in Table 1, were used to evaluate their degree of interference. The apparent selectivity coefficient \( {\text{K}}_{{{\text{Ch}},j^{ + } }}^{\text{pot}} \)measured under transient flow injection conditions might differ significantly from that measured under batch conditions [41, 42]. This is interpreted by the difference in the time of interaction of interferent with the membrane surface in comparison with the main sensed ion, and also the interference process is highly dependent on the rate of diffusion and the exchange reaction of the interfering ion [42]. The determined selectivity coefficients of the Ch-TPB and Ch-PTA electrodes, Table 1, reflect a very high selectivity of the investigated electrodes for chlorphenoxamine cations under both FIA and batch conditions. Also the results obtained in case of amino acids and sugars under FIA are in a good agreement with those under batch conditions, where it was found that the Ch-PTA electrode could be used safely without interference in presence of glucose, maltose, sucrose, lactose, fructose, urea and glycine up to 950, 850, 3600, 1200, 1050, 330 and 2040 folds of chlorphenoxamine, respectively. On the other hand the Ch-PTA electrode could be used safely without interference in the presence of glucose, maltose, sucrose, lactose, fructose, urea and glycine up to 1120, 335, 1820, 1450, 1260, 185, 421 folds of chlorphenoxamine, respectively. Also, the electrodes can be used safely in the presence of two antihistaminic drugs, phenylpropanolamine hydrochloride and diphenylpyraline hydrochloride, as shown in Table 1.
3.7 Response time
The response time [43] of each electrode was tested by measuring the time required to achieve a steady state potential (within ± 1 mV) after successive immersion of the electrode in a series of its respective drug solution, each having a 10-fold increase in concentration from 2.0 × 10−6 to 1.0 × 10−2 mol L−1. The electrodes yielded steady potentials within 10–25 s. The potential reading stay constant, to within ± 1 mV, for at least 3 min. The typical potential plot for the response characteristics of Ch-PTA conventional electrode is shown in Fig. 4.
3.8 Solubility product of Ch-PTA and Ch-TPB ion exchangers
The determination of the solubility product of the ion-pair is important since its reciprocal is approximately equal to the formation constant of the ion-pair, which is tightly related to the degree of its hydrophobicity. So, as the hydrophobicity of the ion exchangers increases, the leaching process of them to the aqueous bathing solution decreases and this is the main determining factor in the lifetime of the electrodes membranes. The solubility product of the Ch-TPB and Ch-PTA was determined conductimetrically as described in the experimental part and was found to be 1.53 × 10−8 and 2.10 × 10−14 for Ch-TPB and Ch-PTA ion exchangers, respectively, indicating very low solubility of the ion exchangers (1.24 × 10−4 and 9.20 × 10−6 mol L−1 for Ch-TPB and Ch-PTA ion exchanger, respectively). Consequently, the formation constant of the following reactions, Ch + TPB ⇔ Ch-TPB and 3Ch + PTA ⇔ (Ch)3-PTA are 6.50 × 107 and 4.75 × 1013, revealing that the degree of completeness of the reactions is more than 99.9%. In the above equilibria, the solubility of the undissociated ion exchangers in water (i.e., the intrinsic solubility) was omitted as it provides too small contribution to the total solubility [19].
3.9 Analytical application
3.9.1 Determination of Ch-HCl using potentiometric titration
The potentiometric titration of Ch-HCl is based on the decrease of the drug cation concentration by precipitation with PTA and NaTPB, the feasibility of such titration depends on the degree of completeness of the reaction, since the equilibrium constant (K) of precipitation titration is inversely proportional to the solubility product; it is natural that the smaller the solubility product of the formed ion associate the sharper is the end point. The potentiometric titration was used successfully in the determination of Ch-HCl in pure solution and its pharmaceutical preparations (allergex and allergex caffeine tablets).
The mean recoveries obtained for pure solution based on Ch-TPB ion pair, Table 2, were 102.63, 105.83 and 103.60% with SD 0.48 and 0.53 and 0.47 for conventional, graphite and copper coated wire electrode, respectively, and those based on Ch-PTA, Table 3, ion associate were 102.02, 102.46% with SD 0.34 and 0.36 for conventional and graphite coated wire electrodes, respectively.
The mean recoveries obtained for the determination of Ch-HCl in allergex tablets, Table 2, based on Ch-TPB ion pair were 102.79, 96.80 and 99.86% with SD 0.26, 0.49 and 0.53 for conventional, graphite and copper coated wire electrodes, respectively, and those based on Ch-PTA, Table 3, ion associate were 101.28 and 100.47% with SD 0.35 and 0.47 for conventional and graphite coated wire electrodes, respectively.
Also, Ch-HCl was determined successfully in Allergex caffeine tablets using potentiometric titration, where the mean recoveries obtained based on Ch-TPB ion pair, Table 2, were 105.13, 98.95 and 101.33% with SD 0.34, 0.33 and 0.36 for conventional, graphite and copper coated wire electrodes, respectively, and those based on Ch-PTA ion associate, Table 3, were 98.86, 100.54% with SD 0.34 and 0.43 for conventional, graphite coated wire electrodes, respectively. Representative Potentiometric curves are shown in Figs. 5 and 6.
3.9.2 Determination of Ch-HCl using standard addition method
The standard addition method described in the experimental part, was proved to be successful for the determination of the investigated drugs in their pure solution and in pharmaceutical preparations in batch conditions, using the respective electrodes as sensors.
Different weights of Ch-HCl in pure solution (5.00–20.00 mg) were determined by applying standard addition method using Ch-TPB and Ch-PTA constructed electrodes and the mean recoveries obtained were 101.21, 100.16 and 100.24% with SD 0.47, 0.25 and 0.20 using Ch-TPB conventional, graphite and copper coated wire electrodes, respectively and those obtained using Ch-PTA electrodes were 99.08 and 102.43% with SD 0.35 and 0.27 using conventional and graphite coated wire electrodes, respectively, Tables 4 and 5.
Solutions of allergex and allergex caffeine contain the same weight range (5.00–20.00 mg) were also prepared and determined using the constructed electrodes by applying the standard addition method, where the mean recoveries obtained using Ch-TPB electrodes for the determination of Ch-HCl in allergex tablets were 99.28, 99.56 and 99.17% with SD 0.59, 0.20 and 0.42 for conventional, graphite and copper coated wire electrodes, respectively, and those obtained using Ch-PTA electrode were 99.40 and 103.33% with SD 0.33 and 0.24 for conventional and graphite coated wire electrodes, respectively, Tables 4 and 5.
The mean recoveries obtained using Ch-TPB electrodes for the determination of Ch-HCl in allergex caffeine tablets were 100.63, 100.20 and 100.57% with SD 0.30, 0.21 and 0.28 for conventional, graphite and copper coated wire electrodes, respectively and those obtained using Ch-PTA were 99.61 and 101.30% with SD 0.42 and 0.35 for conventional and graphite coated wire electrodes, respectively, Tables 4 and 5.
3.9.3 Determination of Ch-HCl using peak height comparison in FIA conditions
In FIA measurements the peak height comparison was used for the determination of the studied drugs in their pharmaceutical tablets where the peak heights corresponding to their solutions were measured and compared by those obtained by series of standard drug solutions under the same conditions as shown in Fig. 7. The percentage recovery is obtained as the ratio of the peak heights and thus the concentration could be calculated. The mean recoveries obtained using Ch-TPB electrode were 101.32 and 97.70% with SD 0.25 and 0.32 for Allergex and Allergex caffeine tablet, respectively, and those obtained using Ch-PTA electrode were 101.19 and 97.93% with SD 0.41 and 0.34 for Allergex and Allergex caffeine tablet, respectively, Table 6.
3.9.4 Conductimetric determination of Ch-HCl
The conductance measurements are used successfully in quantitative titration systems in which the conductance of the solution varies before and after the equivalence point. The sudden change in the slope after the end point may be related to the formation of RNHx+ and OH− by hydrolysis. On adding NaTPB and PTA the ion pair formed replaces RNH +x ions by mobile Na + and H+ and the conductivity increases [43]. After the end point, more reagent is added and the conductivity increase more rapidly.
Titrations in different media were attempted to obtain the best results, where it was found that aqueous medium is more suitable for obtaining a stable conductimetric reading with less consumption of reagents for the determination of Ch-HCl in raw material and pharmaceutical preparations, where the mean recoveries obtained using NaTPB were 102.22, 97.38 and 100.52 with SD 0.52, 0.46 and 0.61 for raw material, allergex and allergex caffeine, respectively and those obtained using PTA were 104.27, 98.75 and 98.02 with SD 0.47, 0.40 and 0.32 for raw material, Allergex and Allergex caffeine respectively, Table 7.
Figure 8 shows the corrected conductimetric titration curves of the drug versus PTA from which the end points were determined
Finally, the results were subjected to linear regression analysis (found values versus taken), using the computer program sigma plot-2 in order to establish whether the investigated electrodes exhibit any fixed or proportional bias. The slopes and intercepts of the regression lines did not differ significantly from the ideal values, revealing the absence of a systematic error during the measurements within the investigated concentration range.
The F- and t-tests [45] were performed to compare the average and SD of the results of the proposed methods with those obtained using spectrophotometric method [4]. The results shown in Table 8 indicate that the calculated F and t values were lower than those tabulated which indicate there is no significant difference or constant error between the two methods at the indicated significant level.
4 Conclusion
In the present work five electrodes of both conventional and coated wire types have been constructed for the determination of chlorphenoxamine hydrochloride in raw material and pharmaceutical preparations with high degree of precision, selectivity and accuracy in comparison with the reference method, within wide concentration, temperature and pH ranges that have been confirmed by recoveries,standard deviations, F- and t-values. Over that, the coated wire types are simple in construction and easy in store where no filling or soaking solutions are needed. In addition, applying flow injection technique shortened the time needed for the determination when compared with batch technique or previously published methods. Also, the conductimetric titration was used successfully for the determination of the investigated drug which is considered a simple way of application and of low expense. In general the proposed methods are valid for the determination of the investigated drug and can be used as a shelf stability test in drug stores and in quality control and quality assurance during the manufacturing of pharmaceutical preparations.
References
The United States pharmacopoeia (2006) The national formulary, USP 29/NF24, Rockville, MD
Kelani KM (2005) J AOAC Int 88(4):1126
Dinc E, Palabiyik IM, Ustundag O, Yurtsever F, Onur F (2002) J Pharm Biomed Anal 28(3–4):591
Ayad M, Saleh H, El–Bolkiny M, El–Henawee M (1993) Anal Lett 26(5):913
Barary MH, Wahbi AM (1991) Drug Dev Ind Pharm 17(3):457
El-Henawee MM, Mohamed HS, El-Bolkiny N, Ayad MM (1990) Spectrosc Lett 23(3):273
Amine AS, El-Henawee MM (1995) Mikrochim Acta 118(3–4):177
Hassan WS, El-Henawi MM, Gouda AA (2008) Spectrochim Acta Part A Mol Biomol Spectrosc 69:245
Metrohm Appliction Bulletin (2001) Technical report 263/1e: 11
Bebawy LI, El-Kousy MN (1999) J Pharm Biomed Anal 20(4):663
Goeneches S, Ruecker G, Brzezinka H, Hoffmann G, Neugebauer M, Pyzik T(1987) Arzneim Forsch 37(II)(7):854
Zareh MM, Teleb SM, Ahmed ES (2002) Anal Lett 35:17
Isaa YM, Amin AS (1996) Analusis 24:139
Temizer A, Ozaltin N (1986) J AOAC Int 69(2):192
Abdel-Ghani NT, Abu-Elenien GM, Hussein SH (2010) J Appl Electrochem 40(3):499
Abdel-Ghani NT, Shoukry AF, Hussein SH (2002) J Pharm Biomed Anal 30:601
Segundo MA, Maglhaes LM (2006) Anal Sci 22(1):3
Abdel-Ghani NT, Hussein SH (2010) Anal Lett 43(4):582
Abdel-Ghani NT, Shoukry AF, EL-Nashar RM (2001) Analyst 126:79
Shoukry AF, Abdel-Ghani NT, Issa YM, Ahmed HM (1999) Electroanalysis 11:443
Abdel-Ghani NT, Hussein SH (2002) IL FARMACO 58:581
Lingan JJ (1985) Electroanalytical Chemistry, 2nd edn. Interscience, New York
West DM, Holler FJ, Grouch SR (2004) Fundamental of analytical chemistry, 8thed. Thomson Brooks/Cole, USA
Armstrong RD, Harai G (1990) Electrochim Acta 35:1
Cobben PLHM, Egberink RJM, Bomer JG, Bergveld P, Reinhoudt DN (1994) J Electroanal Chem 368:193
Morf WE (1981) The principle of ion selective electrodes and of membrane transport. Elsevier, New York
Sokalski T, Ceresa A, Zwicki T, Bakker A, Pretch E (1999) Anal Chim 71:1210
Abbas MN, Abdel Fattah AA, Zahran E (2004) Anal Sci 20:1137
Linder E, Toth K, Pungor E (1988) FL. CRC Press, Boca Raton
Al-Momani IF, Awni I, Kilalil HS, Esmadi F (1999) Anal Lett 32:2977
Frenzel W, Bratter P (1986) Anal Chim Acta 185:127
Morf WE, Lindner E, Simon W (1975) Anal Chem 47:1596
Ruzicka J, Hansen EH (1980) Anal Chim Acta 114:19
Trojanowicz M, Matuszewski W (1983) Anal Chim Acta 151:77
Rizk MS, Abdel-Ghani NT, EL-Nashar RM (2001) Microchem J 70:93
Ilcheva L, Trojanowicz M, Vel Krawczyk TK (1987) Fresenius Z Anal Chem 328:27
Antropov LL (1977) Theoretical Electrochemistry. MIR publisher, Moscow
El-Nashar RM, Abdel-Ghani NT, Bioumy AA (2004) Microchem J 78:107
Cosofret VV, Buck RP (1984) Analyst 109:1321
Pungor E, Toth K (1969) Anal Chim Acta 47:291
Trojanowicz M, Matuszewski W (1982) Anal Chim Acta 138:71
Davey DE, Mulcahy DE, Oconnel GR, Smart RSC (1995) Electroanal 7:461
Buck P, Linder E (1994) Pure Appl Chem 66:2527
Bahbouh MS, Salem AA, Issa YM (1998) Mikrochemic. Acta 128:57
Christian GD (1994) Analytical chemistr. John Wiley & Sons, USA
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Ghani, N.T., Hussein, S.H. Flow injection potentiometric determination of Chlorphenoxamine Hydrochloride. J Appl Electrochem 40, 2077–2090 (2010). https://doi.org/10.1007/s10800-010-0189-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-010-0189-4