Abstract
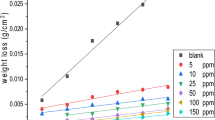
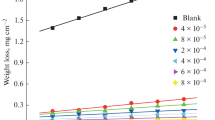
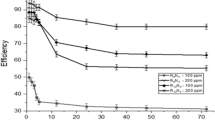
Four novel non-ionic ethoxylated fatty alkyl amine surfactants (I–IV) were synthesised and investigated as corrosion inhibitors of carbon steel in 1 M hydrochloric acid solution using gravimetric, open circuit potential and potentiostatic polarisation techniques. The percentage inhibition efficiency (η%) for each inhibitor increased with increasing concentration until the critical micelle concentration (cmc) was reached. The maximum inhibition efficiency approached 95.1% in the presence of 400 ppm of the inhibitor (IV). It was found that the adsorption of the surfactants on carbon steel followed the Langmuir adsorption isotherm. Potentiostatic polarisation data indicated that these surfactants act as mixed type inhibitors. The values of activation energy (E a*) of carbon steel dissolution in 1 M HCl were calculated in the absence and presence of 400 ppm of each inhibitor. Finally, scanning electron microscopy (SEM) was used to examine the surface morphology of polished carbon steel surfaces and those immersed in 1 M HCl in the absence and presence of 400 ppm of inhibitor (IV).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
L. Garverick, Corrosion in the Petrochemical Industry (ASM, New York, 1994)
A. Popova, M. Christor, S. Raicheva and E. Sokolova, Corros. Sci. 46 (2004) 1333
M.A. Migahed, A.A. El-Safei, A.S. Fouda and M.A. Morsi, Egypt. J. Chem. 45 (2002) 571
M.A. Migahed, H.M. Mohamed and A.M. Al-Sabagh, Mat. Chem .Phys. 80 (2003) 169
M.A. Migahed, E.M.S. Azzam and A.M. Al-Sabagh, Mat.Chem.Phys. 85 (2004) 273
M.A. Migahed, R.O. Aly and A.M. Al-Sabagh, Corros. Sci. 46 (2004) 253
M.M. Osman, A.M. Omar and A.M. Al-Sabagh, Mat. Chem. Phys. 50 (1997) 271
M.M. Osman, R.A. El-Ghazawy and A.M. Al-Sabagh, Mat. Chem. Phys. 80 (2003) 55
Z. Abdel-Hamid, T.Y. Soror, H.A. El-Dahan and A.M. Omar, Anti-Corros. Meth. Mat. 45 (1998) 306
G. Latha and S. Rajeswari, Anti-Corros. Meth. Mat. 43 (1996) 19
N. Pebere, M. Duprat, F. Dabosi and A. Lattes, J. Appl. Electrochem. 18 (1988) 225
M.L. Free, Corros. Sci. 44 (2002) 2865
D.P. Weinsberg and V. Ashworth, Corros. Sci. 28 (1988) 539
W. Hreczuch, A. Kozlek, Tens. Surf. Det. 38 (1996) 621
N.D. Greene (ed.), Experimental Electrode Kinetics (Rensselaer Polytechnic Institute, New York, 1965)
K.F. Bonhoffer and K.E. Heascer, Z. Phys. Chem. N.F. 8 (1956) 930
R. Narayan, An introduction to Metallic Corrosion and its Prevention (IBH publishing Co., Oxford, 1983)
M. Stern and A.L. Geary, J. Electrochem. Soc. 104 (1957) 56
J.M. Abd El- Kader, A.A. El-Warraky and A.M. Abd El-Aziz, Br. Corros. J. 33 (1998) 139
A.A. El-Shafei, S.A. Abd El-Maksoud, A.S. Fouda, Corros. Sci. 46 (2003) 579
K.K. Al-Neami, A.K. Mohamed, I.M. Kenawy and A.S. Fouda, Montach. Chem. 126 (1995) 369
Z.I. Osipow, Surface Chemistry (Reinhold publisher Co., New York, 1960)
H. Luo, Y.C. Guan and K.N. Han, Corrosion 54 (1998) 619
ASTME 45-87, Annual Book of ASTM Standard, Vol. 11 (ASTM, Philadelphia, PA, 1980), p. 125
I.L. Rozenfeld, Corrosion Inhibitors (New York, 1981)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Migahed, M., Abd-El-Raouf, M., Al-Sabagh, A. et al. Corrosion inhibition of carbon steel in acid chloride solution using ethoxylated fatty alkyl amine surfactants. J Appl Electrochem 36, 395–402 (2006). https://doi.org/10.1007/s10800-005-9094-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-005-9094-7