Abstract
While the correlation and chronology of appearance of diabetic nephropathy and retinopathy is well known in diabetes mellitus (DM) type 1 patients, in DM type 2 this correlation is less clear. A retrospective study including 917 patients with type 2 diabetes. Diabetic retinopathy (DR) was diagnosed based on fundus photographs taken with a non-mydriatic camera. Diabetic nephropathy (DN) was diagnosed based on urinary albumin concentration in a morning urine sample. Statistical analysis was performed with a seemingly unrelated regression (SUR) model. Our SUR model is statistically significant: the test for “model versus saturated” is 2.20 and its significance level is 0.8205. The model revealed that creatinine and glomerular filtration rate (GFR) have strong influence on albuminuria, while body mass index (BMI) and HbA1c have less significant impact. DR is affected positively by diabetes duration, insulin treatment, glucose levels, and HbA1c, and it is affected negatively by GFR, triglyceride levels, and BMI. The association between DR and DN was statistically significant and had a unidirectional correlation, which can be explained by chronological order; that is, DN precedes DR. The present study indicates that the level of renal impairment is proportional to the level of damage to the eye. Furthermore, such an association has a chronological aspect; the renal injury precedes retinal damage.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes is the modern world’s most prevalent disease, and its morbidity is increasing constantly with no signs of slowing[1, 2]. Out of the many microvascular complications caused by diabetes, nephropathy and retinopathy are unquestionably the major ones. Indeed, in the U.S., among the working population, diabetic retinopathy is one of the leading causes of blindness; in Israel, diabetic retinopathy is responsible for 10.1 % of all blindness cases, and this figure is constantly rising [3].
As to nephropathy caused by type 2 diabetes, this is the most frequent cause of end-stage renal disease in the United States, Europe, and Japan. In the United States, the incidence of diabetic nephropathy has increased by 150 % in the past 10 years [4].
The first sign of renal involvement in patients with type 2 diabetes is most often microalbuminuria. Microalbuminuria affects 20–40 % of patients 10–15 years after the onset of diabetes. Progression to macroalbuminuria occurs in 20–40 % of patients over a period of 15–20 years after the onset of diabetes. Once macroalbuminuria is present, creatinine clearance declines by an average of 10–12 ml/minute each year in untreated patients [4].
To reduce the risk or slow the progression of microvascular complications of diabetes, current recommendations for patients with type 2 disease are to perform an initial eye examination by an ophthalmologist shortly after diagnosis for retinopathy, and test urinary albumin excretion for nephropathy. Subsequent examinations should be repeated annually [5]. Understanding the association between different complications of diabetes could lead to a better understanding of the disease processes and subsequently better screening and management techniques.
The correlation between diabetic nephropathy and retinopathy is well known in DM type 1 patients. In DM type 2 this correlation is less clear. The chronology of appearance of microvascular complications in DM type 1 is retinopathy prior to nephropathy, while in DM type 2 the chronology of complications is not clear [6].
Very few studies demonstrated an association between diabetic nephropathy and diabetic retinopathy in DM type 2 patients. Most agree that there is a correlation;[7–10] however, not all agree [11, 12]. Some of these studies observed small communities with unique lifestyles. All those studies divided retinopathy to only two severity levels (proliferative and non-proliferative). No similar studies were conducted in Israel.
The purpose of this study is to evaluate the possible correlation between the severity of diabetic nephropathy and diabetic retinopathy in patients with type 2 diabetes mellitus.
Methods
Study population
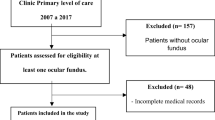
The research includes patients with diabetes mellitus type 2, whose fundus photographs were taken by a non-mydriatic camera, between January 1, 2009 and June 30, 2010. All the patients are clients of Clalit Health Fund and live in various cities and towns (Beer-Sheva, Rahat, Dimona, Sderot, Ashkelon, Kiryat Gat, Netivot, Arad, and Ofakim) in southern Israel.
Included in this research were patients with diabetes type 2, older than 18 years of age.
There were 1,004 subjects who had digital fundus photographs taken during the study period, of them 87 were excluded based on our exclusion criteria (most had additional eye pathology). Therefore 917 subjects were included in research; of them 56 % were males and 44 % females, and 12 % belonged to the Arab community. In this area of Israel, most of the patients affiliated with the Arab community are Bedouins. As 95 % of the non-Arab population in Israel is Jewish [13], the other 88 % of our subjects are mostly Jews.
All the fundus photographs were taken by a skilled technician with the same non-mydriatic camera (Topcon TRC NW6S). The interpretation was performed by a single fellowship-trained retina specialist. After the interpretation, only records that met the above-mentioned criteria were included in the research. Excluded from research were patients without the required blood and urinary exams, and patients who had other eye pathologies (e.g., cataract, drusen).
Since all pictures were reviewed by the same ophthalmologist, we also calculated the intra-observer agreement. Three hundred random pictures were evaluated in a masked fashion at least 8 months after the first examination. The overall proportion of agreement was of 97.7 % with an “almost perfect” kappa coefficient of 0.92. Table 1 shows the characteristics of the study population.
Study design
This retrospective study was approved by the local medical ethics committee in Soroka Medical Center, and was conducted in accordance with the guidelines of the Helsinki declaration.
All the patients data were extracted from their medical records. The collected data were: age, sex, affiliation with Arab community (yes/no), body mass index (BMI), smoking, diabetes duration, cholesterol, LDL, HDL, triglycerides, HbA1c, hypertension, insulin treatment (yes/no), blood creatinine, blood glucose, urine microalbumin, and diagnosis with digital photography. All the values included were measured through year 2010. When several measured values for the same variable were present, the average value was used. Thus, for most of the blood- and urine-related variables averages were used.
Insulin treatment was checked based on pharmacy records during the research period rather than just by a doctor’s prescription.
Hypertension was graded based on JNC7 guidelines as normal/pre-hypertension/HTN-stage-1/HTN-stage-2 [14].
Analytical methods
The glomerular filtration rate (GFR) was calculated using an MDRD formula for adults (186 × Creatinine(−1.154) × age(−0.203) and for females multiplied by 0.742)[15].
Diabetic nephropathy assessment is made by measuring albumin excretion in urine [5]. The gold standard for measuring microalbuminuria is measuring albumin in a 24-hour urine collection. Two less cumbersome and well-accepted alternatives to this test are albumin/creatinine ratio and albumin concentration measured in a spot-morning urine sample. We used the data of measured albumin concentration in a morning spot urine test, which is satisfactory and comparable to the other two tests [16]. Levels less than 30 were defined as normal; 30–299—microalbuminuria; ≥300—macroalbuminuria [5]. Most of the patients had more than one measurement; therefore an average value was used.
Diabetic retinopathy (DR) was diagnosed by a single fellowship-trained retina specialist, based on fundus photographs made by a non-mydriatic Topcon TRC NW6S. This is a well-accepted method for evaluation of DR comparable to the regular ophthalmologic exam [17–19]. DR was graded to non-proliferative-mild (NPDR-mild), non-proliferative-moderate (NPDR-moderate), or proliferative (PDR).
Statistical analysis
As is well known, in regression models only observed variables are modeled, and only the dependent variable has an error term. The ordinary assumption is that independent variables are modeled without error.
Unlike regression models, but like structural equation models (SEM), in seemingly unrelated regression (SUR) models independent variables can be both causes and effects of other variables. That is, SUR models appear to be joint estimates from several regression models, each with its error term. These regressions are related because the errors associated with the dependent variables may be correlated [20].
Generally, SUR may lead to more efficient estimates than running regression models separately as well as allowing joint tests. Moreover, using SEM for SUR models (instead of traditional regression procedures) has the benefit that measures of model fit, modification indices, and other aspects of SEM output become available. Another reason one might want to use SEM is that SEM will provide robust standard errors whereas conventional SUR does not [21].
In SUR implemented via structural equation modeling (SEM) the quality of the regression model can be evaluated with a goodness-of-fit Chi square (χ2GoF). However, unlike a conventional Chi square (χ2), which tests the difference between the observed data and the expected findings (where the expected values are those we would see under the null hypothesis of no association between the variables), with a χ2GoF we are testing the difference between the observed data and our hypothesized model. Because we hope that our proposed model does fit the data, we do not want to see too much difference; in fact, the ideal would be to have no difference between the two. So, the smaller χ2GoF is (i.e., the further up the p value from 0.05 threshold), the better.
We exploited Stata 12 (Data Analysis and Statistical Software; version 12; StataCorp LP, Lakeway Drive, Texas, USA) software for SUR modeling using Stata’s SEM module.
Results
The results of the SUR model are summarized in Table 2. Partial coefficients were calculated using only the independent variables in a direct path to the endogenous variables.
Figure 1 depicts the results graphically. In the figure, the arrows from all independent variables end at the dependent variables, and only significant correlations appear in the figure (marked by arrows). All the coefficients in the figure are standardized.
Altogether, 917 patients were included. Our SUR analysis is statistically significant: P(χ 2) = 0.8205.
There are variables that were collected but do not appear in the model: sex, smoking history, total cholesterol, HDL, LDL, and hypertension. No significant correlation was found between these variables and either DR or diabetic nephropathy.
The analysis revealed that blood creatinine and GFR associate strongly with albuminuria, whereas the link of BMI and HbA1c with albuminuria is less significant. Age links to albuminuria through its association with GFR. Analogously, duration of diabetes relates to albuminuria through its association with HbA1c. Affiliation with the Arabic community also influences albuminuria through its association with HbA1c.
Diabetic retinopathy is affected by more factors. It is positively affected (more severe retinopathy) by diabetes duration, insulin treatment, glucose levels, HbA1c, and affiliation with the Arabic community. It is negatively affected by GFR, triglyceride levels, and BMI. We can say that creatinine and age influence retinopathy through their association with GFR. The negative association between creatinine and GFR is unsurprising because higher creatinine means lower GFR. The same is true for the association between age and GFR. The negative impact of GFR on retinopathy is also expected. Lowering GFR means worsening kidney function, which is associated with more severe retinopathy.
Among other associations found significant are glucose levels and HbA1c, glucose levels and insulin treatment, HbA1c and insulin treatment, diabetes duration and glucose levels, and diabetes duration and insulin treatment.
Estimated model
The model equations are
UA (urine concentration of albumin), DR (diabetic retinopathy), ar (affiliation with the Arabic cmmunity), bmi (BMI score), t (duration of diabetes), tg (triglycerides), hba (HbA1c level), int (insulin treatment), bg (blood glucose), bc (blood creatinine), and gfr (GFR).
Significant association between urine albumin indicating diabetic nephropathy and DR was established at the significance p < 0.001.
Moreover, the association between UA and DR has been established only in one direction—only albuminuria has a statistically significant effect on retinopathy. Such a link can be explained as a chronological effect, namely microalbuminuria precedes the emergence of retinopathy, not the other way around.
Discussion
The correlation between diabetic nephropathy and retinopathy is well known in DM type 1 patients. The chronology of appearance of microvascular complications in DM type 1 is retinopathy prior to nephropathy. In DM type 2 this correlation is less clear [6].
Our study indicates that there is a strong correlation between diabetic retinopathy and diabetic nephropathy. The severity of renal impairment is associated with the severity of the damage to the eye. Moreover, this correlation has a chronological aspect—the renal injury (as measured by albumin in urine) precedes retinal damage.
Based on this finding, future studies may give us a better insight into the mechanisms leading to microvascular damage in diabetes, and better ways to screen, treat, and control the disease.
The prevalence of diabetes is expected to grow throughout the world [1, 2]. Diabetes currently imposes a considerable economic burden on the healthcare services [22, 23]. Currently, the screening for both nephropathy and retinopathy is recommended starting soon after the diagnosis of diabetes type 2 [5]. The screening for nephropathy is easy as it is done by a simple annual urine exam. The screening for retinopathy is more complicated as it requires ophthalmologic examination once a year [5]. This leads to a lower compliance for the screening of retinopathy and a greater burden on the healthcare system.
The HbA1c monitoring is the best way to assess glycemic control in type 2 diabetic patients [5]. Therefore, it was expected to find a significant correlation between HbA1c and both retinopathy and nephropathy.
We also found that in the Arabic community the levels of HbA1c are higher; therefore, the glycemic control in this community is less satisfying. This finding is supported by two other studies that were performed recently in Israel [24, 25]. As a result, the patients from this community had worse retinopathy and nephropathy. The reason for this difference may stem from the lifestyle of the Arabic community in southern Israel. Most of them belong to the Bedouin Community. Previous studies explained the differences mainly by metabolic changes that occurred during the transition from a traditional Bedouin lifestyle to a more modern one that occurred during the last decades, to lower compliance for medical treatment, and to less access to medical services. There also might be a higher genetic predisposition for complications of diabetes in the Bedouin community [26, 27].
There are several limitations to this study:
We examined only the population that lives in Beer-Sheva and the surrounding cities in southern Israel. Most of this population is of middle and lower socioeconomic status [28]. It is well known that a patient with lower socioeconomic status has higher disease prevalence and more complications [24, 29].
As ethnicity is not recorded in the medical files, affiliation with the Arab community was based on the patient’s location of residence. Our study area is characterized by a separation between the Arab community dwelling and the major population residential areas; therefore, we believe the error in the patients’ affiliation estimation to be minimal.
We did not find any correlation between hypertension and albuminuria, despite the fact that the association between chronic kidney disease and albuminuria is acknowledged, and hypertension is a known cause of renal damage [30]. The reason for this might stem from the manner in which the blood pressure data was recorded—as a degree of severity rather than a numerical value.
In conclusion, our study indicates that there is a strong correlation between the severity of diabetic retinopathy and the severity of diabetic nephropathy. We also found that renal injury precedes retinal damage. This finding should be confirmed by future prospective studies.
Disclosure
The authors have no conflict of interest that can create a risk that professional judgment or actions regarding a primary interest will be unduly influenced by a secondary interest in any of the materials or techniques used in this study.
References
Colagiuri S, Borch-Johnsen K, Glumer C, Vistisen D (2005) There really is an epidemic of type 2 diabetes. Diabetologia 48:1459–1463
Wild S, Roglic G, Green A, Sicree R, King H (2004) Global prevalence of diabetes. Diabetes Care 27:1047–1053
Avisar R, Friling R, Snir M, Avisar I, Weinberger D (2006) Estimation of prevalence and incidence rates and causes of blindness in Israel, 1998–2003. IMAJ 8:880–881
Remuzzi G, Schieppati A, Ruggenenti P (2002) Nephropathy in patients with type 2 diabetes. N Engl J Med 346:1145–1151
American Diabetes Association (2012) Standards of medical care in diabetes-2012. Diabetes Care 35(Suppl 1):S11–S63
Romero-Aroca P, Mendez-Marin I, Baget-Bernaldiz M, Fernandez-Ballart J, Santos-Blanco E (2010) Review of the relationship between renal and retinal microangiopathy in diabetes mellitus patients. Curr Diabetes Rev 6:88–101
Rani PK, Raman R, Gupta A, Pal SS, Kulothungan V, Sharma T (2011) Albuminuria and diabetic retinopathy in Type 2 diabetes mellitus Sankara Nethralaya diabetic retinopathy epidemiology and molecular genetic study. Diabetol Metab Syndr 3:9
Savage S, Estacio RO, Jeffers B, Schrier RW (1996) Urinary albumin excretion as a predictor of diabetic retinopathy, neuropathy, and cardiovascular disease in NIDDM. Diabetes Care 19(11):1243–1248
Looker HC, Krakoff J, Knowler WC, Bennett PH, Klein R, Hanson RL (2003) Longitudinal studies of incidence and progression of diabetic retinopathy assessed by retinal photography in Pima Indians. Diabetes Care 26(2):320–326
Kim HK, Kim CH, Kim SW, Park JY, Hong SK, Yoon YH et al (1998) Development and Progression of diabetic retinopathy in Koreans with NIDDM. Diabetes Care 21:134–138
Entacio RO, McFarling E, Biggerstaff S, Jeffers BW, Johnson D, Schrier RW (1998) Overt albuminuria predicts diabetic retinopathy in Hispanics with NIDDM. Am J Kidney Dis 31(6):947–953
Erasmus RT, Oyeyinka G, Arije A (1992) Microalbuminuria in non-insulin-dependent (type 2) nigerian diabetics: relation to glycaemic control, blood pressure and retinopathy. Postgrad Med J 68:638–642
Israeli Central Bureau of Statistics: population and demography, 64th Independence day report (2012). http://www1.cbs.gov.il/reader/newhodaot/hodaa_template.html?hodaa=201211106
The seventh report of the joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure (JNC 7) of the U.S. Department of Health & Human Services. http://www.nhlbi.nih.gov/guidelines/hypertension/
Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Ann Intern Med 130:461–470
Gansevoort RT, Verhave JC, Hillege HL, Burgerhof JGM, Bakker SJL, Zeeuw D et al (2005) The validity of screening based on spot morning urine samples to detect subjects with microalbuminuria in the general population. Kidney Int Suppl 94:S28–S35
Massin P, Erginay A, Ben Mehidi A, Vicaut E, Quentel G, Victor Z et al (2003) Evaluation of a new non-mydriatic digital camera for detection of diabetic retinopathy. Diabet Med 20:635–641
Murgatroyd H, Ellingford A, Cox A, Binnie M, Ellis JD, MacEwen CJ et al (2004) Effect of mydriasis and different field strategies on digital image screening of diabetic eye disease. Br J Ophthalmol 88:920–924
Aptel F, Denis P, Rouberol F, Thivolet C (2008) Screening of diabetic retinopathy: effect of field number and mydriasis on sensitivity and specificity of digital fundus photography. Diabetes Metab 34:290–293
Zellner A (1962) An efficient method of estimating seemingly unrelated regressions and tests for aggregation bias. J Am Stat Assoc 57:348–368
Cameron AC, Trivedi PK (2010) Microeconometrics using stata. Stata Press, College Station
Dall TM, Zhang Y, Chen YJ, Quick WW, Yang WG, Fogli J (2010) The economic burden of diabetes. Health Affairs (Millwood) 29(2):297–303
Chodick G, Porath A, Alapi H, Sella T, Flash S, Wood F et al (2010) The direct medical cost of cardiovascular diseases, hypertension, diabetes, cancer, pregnancy and female infertility in a large HMO in Israel. Health Policy 95:271–276
Wilf-Miron R, Peled R, Yaari E, Shem-Tov O, Weinner VA, Porath A et al (2010) Disparities in diabetes care: role of the patient’s socio-demographic characteristics. BMC Public Health 10:729
Chorny A, Lifshits T, Kratz A, Levy J, Golfarb D, Zlotnik A et al (2011) Prevalence and risk factors for diabetic retinopathy in type 2 diabetes patients in Jewish and Bedouin populations in southern Israel. Harefuah 150:906–910
Abou-Rbiah Y, Weitzman S (2002) Diabetes among Bedouins in the Negev: the transition from a rare to a highly prevalent condition. IMAJ 4(9):687–689
Tamir O, Peleg R, Dreiher J, Abu-Hammad T et al (2007) Cardiovascular risk factors in the bedouin population: management and compliance. IMAJ 9(9):652–655
Israeli Central Bureau of statistics: characterization and classification of local authorities by the socio-economic level of the population 2006 http://www1.cbs.gov.il/www/publications/local_authorities06/local_authorities_e.htm
Nicklett EJ (2011) Socioeconomic status and race/ethnicity independently predict health decline among older diabetics. BMC Public Health 11:684
Król E, Rutkowski B, Czarniak P, Kraszewska E, Lizakowski S, Szubert R et al (2009) Early detection of chronic kidney disease: results of the Pol Nef Study. Am J Nephrol 29:264–273
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kotlarsky, P., Bolotin, A., Dorfman, K. et al. Link between retinopathy and nephropathy caused by complications of diabetes mellitus type 2. Int Ophthalmol 35, 59–66 (2015). https://doi.org/10.1007/s10792-014-0018-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-014-0018-6