Abstract
To determine if primary selective laser trabeculoplasty (SLT) can be repeated with clinical benefit in patients with primary open-angle glaucoma (POAG). Forty-two eyes of 42 patients with POAG were studied. All patients underwent primary SLT treatment of 40–50 shots to the trabecular meshwork over 360°. The treatment response at the initial post-SLT visit (4 weeks), and second post-SLT visit (mean 4 months), clinical success and duration of clinical success were measured. SLT was repeated in all patients after failure to maintain target intraocular pressure (IOP). The same parameters were measured after repeat SLT. The main outcome measures were success of treatment (as defined by reduction of IOP by at least 20 % and below an individually determined target pressure), duration of treatment success and reduction in IOP. No significant difference between initial and repeat treatments was found for mean reduction in IOP or success rate, or duration of success. Survival analysis found significantly longer benefit for repeat treatment compared to initial treatment (P < 0.01). Repeat SLT treatment in eyes with POAG has similar efficacy to primary SLT treatment with respect to reduction in IOP and success rates, produces a longer duration of treatment success.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary open-angle glaucoma (POAG) is the most common form of glaucoma and by definition occurs in the absence of known ocular or systemic conditions that cause increased aqueous outflow resistance. It therefore represents primary failure of trabecular meshwork function [1].
Laser treatment of the meshwork to reduce intraocular pressure (IOP) has been in use since 1979 and remains a developing field. There are two main procedures currently performed. The first is argon laser trabeculoplasty (ALT) which produces thermal burns in target tissues. The second, more recent therapy is selective laser trabeculoplasty (SLT), and involves using a laser whose action is specifically directed at pigmented cells. The detailed mechanism of IOP-lowering in each method is unknown, though it is thought they act via similar mechanisms [2].
Both SLT and ALT have been shown to have similar efficacy in terms of lowering IOP until a 5-year follow-up [3, 4]. Damji et al.[3, 4] found that SLT was more effective at lowering IOP than ALT in eyes previously treated with ALT . Despite this the success rates were only 32 % for SLT and 21 % for ALT (defined as a reduction in IOP of at least 20 % from baseline and not <3 mmHg, with no additional medications, laser or surgery). This demonstrates the reduction in efficacy with time often seen with laser procedures, resulting in the requirement for further medical or surgical therapy [5, 6].
Furthermore, it has been suggested SLT would be effective as primary treatment for open-angle glaucoma; this is backed up by studies showing similar IOP-lowering efficacy between 360° SLT treatment and latanoprost over a 12-month period, as well as significant reduction of IOP fluctuation in patients with ocular hypertension or POAG [7–9]. SLT has also been shown to be a safe and effective treatment in both phakic and pseudophakic eyes, and in reducing the number of medications required to be used to medically control IOP [9, 10]. The adverse effects of laser therapy are often minor, and most are transient [5, 10]. These effects include an anterior chamber reaction (43−89 %), ocular pain (2−39 %), an initial but temporary IOP increase (2.5−22 %), ocular discomfort (5−39 %), and conjunctival injection (5 %) [6, 8, 11–13]. Other more serious complications include corneal oedema, hyphema, iritis and choroidal effusion which occur in 4.5−11.5 % of patients [7, 8, 11, 12].
SLT uses a 532 nm frequency-doubled, Q-switched neodymium:yttrium–aluminium–garnet (Nd-YAG) laser which delivers <1 % of the energy of ALT treatment. Additionally, the treatment time is significantly shorter than the thermal relaxation time of the trabecular meshwork. Due to these two reasons, there is theoretically minimal damage to the structure of the trabecular meshwork, with no subsequent contractile laser microscars [10, 11, 14, 15]. These important differences suggest that SLT may be repeated after failure of initial treatment, whereas previous attempts to repeat ALT have produced poor results [13, 15, 16].
Materials and methods
This study is a retrospective chart review of patients at a specialist glaucoma practice who underwent SLT as first line IOP-lowering therapy after being given the choice of either topical treatment or SLT. Newly diagnosed POAG patients who had primary SLT treatment failure who subsequently had repeat SLT treatment(s) were included. Patients who had SLT treatment on both eyes had one eye selected (using a random number generator), the fellow eye was excluded. Those with secondary glaucomas including angle-closure glaucoma, pseudoexfoliation syndrome, and pigment dispersion syndrome were excluded.
As part of routine clinical practice, a full ocular examination was performed for each newly referred patient, including best-corrected Snellen visual acuity, slit-lamp evaluation, Goldmann applanation tonometry (this study used two initial IOP measures for each patient to reduce the effect of diurnal variation and improve accuracy), corneal pachymetry, corneal hysteresis measurement, gonioscopy, dilated fundoscopy, visual field testing and imaging as appropriate. SLT was performed using the Q-switched ophthalmic Nd:YAG laser (Laserex Tango Nd:YAG; Ellex Medical, Australia) and Latina contact lens with 40−50 shots to the trabecular meshwork (one spot size apart) over 360° at energy levels titrated to be just below threshold for champagne bubble formation. IOP was recorded at 4 weeks post-treatment and subsequently at three- to four-monthly intervals until treatment failure. Treatment failure was defined as a return to pre-treatment IOP levels or being significantly above target IOP range. At this point repeat SLT treatment with the same settings was offered. For those who elected to undergo repeat SLT treatment the process as above was repeated.
For the purposes of this study, treatment success was defined as a reduction in measured IOP of ≥20 % of initial measured IOP and a post-treatment IOP at or below target IOP. The target IOP was arbitrarily and individually determined at initiation of therapy using the following criteria—normal visual field but early optic disc damage (<20 mmHg); mild to moderate visual field (VF) loss with fixation not threatened (in accordance with Hodapp classification; midteens); advanced VF loss or threatened fixation (low teens) [17].
Basic descriptive statistics were conducted for patient demographics. Comparison of means was performed with the t test. Comparison of success rates was performed with the McNemar test using a binomial distribution. The Kaplan–Meier method was used for survival analysis, while the logrank test was utilized to compare survival curves.
Results
In total, 42 eyes of 42 patients underwent primary and repeat treatment of SLT. Of these, nine eyes underwent a third treatment. The mean duration of follow-up was 10.5 months for first treatment, 15.1 months for second treatment and 9.0 months for third treatment. None of these patients received any other form of IOP-lowering treatment during the study period. The mean age of patients at the first SLT treatment was 61.1 years (standard deviation 10.4). There were 22 male and 21 female eyes that underwent initial and second SLT treatment, and four male and five female eyes that underwent a third SLT treatment.
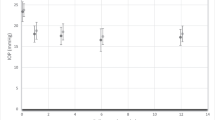
The mean pre-treatment IOP was similar before the primary treatment and second treatment, though slightly lower before third treatment (Table 1). The mean reduction in IOP was 4.76 mmHg (4.4) or 18.9 % for the first visit after the primary SLT treatment, 3.6 mmHg (4.8) or 13.6 % for the second visit after the primary SLT treatment, 19.8 %, 4.7 mmHg (4.5) for the first visit after the second treatment, 4.5 (4.6) for the second visit after the second treatment or 18.1 %, and 3.8 mmHg (2.3) or 18.9 % for the first visit after the third SLT treatment, and 3.6 (2.9) or 17.7 % for the second visit after the third SLT treatment (Table 1). There was no statistically significant difference between the percentage IOP reduction after the primary and second SLT treatments (two-tailed paired t test).
Initial treatment was successful (using the definition described above) in 55 % (23/42 eyes) (Table 2). The second treatment (first repeat treatment) was successful in 67 % (28/42 eyes), and the third successful in 56 % (5/9 eyes) (Table 2). Table 3 summarizes the success rates of the primary and second SLT treatment at the first and second post-SLT visits (mean 1 month and 4 months, respectively). Only 35 % (8/23 eyes) failed to respond to a second SLT treatment after responding to the initial treatment. In addition 68 % (13/19 eyes) that failed to respond to initial SLT treatment subsequently responded to repeat treatment. There was no statistically significant difference in the success rates for the first two visits between the initial and second SLT treatment. The third treatment success was not included in this analysis due to the limited numbers of patients undergoing a third treatment.
The mean duration of successful treatment was 6.9 months (3.4) for the primary treatment, 13.6 months (11.2) for the second treatment, and 9.2 months (4.6) for the third treatment (Table 2). As repeat treatment success durations were limited to the maximum follow-up duration at last visit, we also calculated the mean successful treatment duration for the 28 eyes followed until failure of their second treatment. For these patients, the mean duration of clinical benefit from primary treatment was 6.9 months (3.1), and 13.6 months (11.5) for the second treatment (Table 2). This difference is not statistically significant (two-tailed paired t test).
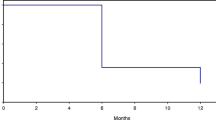
Kaplan–Meier survival analysis found the median survival to be 3 months for the primary treatment (42 eyes), and 11 months for the second treatment (28 eyes) (Fig. 1). The logrank test found this difference to be statistically significant [P < 0.01; hazard ratio 0.43 (95 % confidence interval 0.27−0.69)].
Discussion
This study describes a cohort of POAG patients who underwent repeat SLT after the failure of primary SLT treatment. While the efficacy of SLT as IOP-lowering treatment has been established, limited data currently exist to support the idea that SLT might be repeatable [3, 4]. SLT has also been suggested as a viable alternative to eye drops as an initial treatment of POAG. One of the arguments against initial SLT is the tendency for the treatment effect of SLT to diminish over time, although there is evidence this also occurs with topical treatment [6]. Our results show that for POAG patients, repeat treatment with SLT is of similar effectiveness and of at least comparable duration when compared to the first treatment. Throughout the duration after the primary and repeat SLT treatments when the IOPs were well-controlled without the requirement for any topical medication, patients had the added benefit of avoiding any potential adverse effects from the eyedrops. Our data does not agree with suggestions that because failure of initial SLT occurred in a relatively short time, there was a lesser chance of repeat SLT being effective.
The success rate (using the aforementioned definition of success) was higher for those undergoing a repeat (second) treatment than those undergoing initial treatment although this difference did not achieve statistical significance. In addition, repeat treatment compared favourably in terms of IOP reduction at 4 weeks and 4 months (IOP reductions at 4 weeks of 4.7 mmHg or 19.8 % after the second treatment compared to 4.8 mmHg or 18.9 % after the initial treatment, and reductions of 4.5 (18.1 %) compared to 3.6 (13.6 %) at an average of 4 months, although no statistically significant difference was found. These results suggest that a second SLT treatment may produce an initial clinical benefit similar to that from the first SLT treatment.
In terms of the duration of effect, there was no statistically significant difference in duration of benefit attained with repeat SLT treatment when compared to primary SLT treatment (mean duration 13.1 months for repeat SLT and 6.9 months for primary SLT). Survival analysis, however, found that eyes undergoing repeat SLT treatment had a longer duration of clinical benefit and this was statistically significant. These results suggest that duration of clinical benefit for those undergoing repeat SLT treatment in POAG is at least comparable, and may last significantly longer than initial treatment.
With regard to complication rates, numbers of minor complications were comparable to previous research. Complications in our study included mild discomfort which occurred in 18 % of patients (reported as between 2 and 39 % in the literature). Other complications included headache which occurred in 4 %, and photophobia in 3 % of patients. There were no major complications reported among our subjects [2, 3, 8, 10, 11, 14]. The lower rates of more significant complications could be due to the fewer number of laser spots used in our study compared to other reports.
Despite the promising results shown by this study, there were several limitations that were primarily related to its retrospective design. Firstly, the SLT treatment regime used in our routine clinical practice (40−50 spots over 360°) differed from the 100 spots described in other reports. This was because when we first started performing SLT in our practice, there had not been any published ‘standard’ with regard to the optimum number of SLT spots. Therefore, the results may be related to our reduced SLT treatment regime. However, our SLT treatment regime has been effective in lowering IOP in the great majority of our POAG patients even at the supposedly suboptimal level; so it may be argued that overtreatment may have occurred in the other reports on SLT. There is currently no evidence that has conclusively determined the ideal optimal initial SLT treatment regime. Secondly, there was a group that underwent a third treatment; although the success rates observed were comparable, the number of eyes treated was too small and follow-up too short to perform any meaningful analysis. Thirdly, this study used an observational design and had a relatively small sample. There were only 28 patients followed until failure of the second SLT treatment, which made the duration and survival analysis less reliable and less likely to detect a true difference between treatments.
Hong et al.[13] suggested repeat SLT treatment can be performed as early as 6 months after initial treatment; however, repeat treatment in our study was performed as early as 1 month. It is possible that 1 month may be too soon for any clinical benefit to become apparent, and further study is required to establish what effect such early repeat treatment may have. Our study controlled for this by comparing pressures at both the initial 4-week and second (average 4-month) follow-up visits.
In summary, this study demonstrated that a single repeat treatment with SLT had a similar success rate and a longer duration of clinical benefit as compared to primary treatment in patients with POAG. However, it has to be noted that this cohort of patients had a relatively short duration of SLT clinical benefit which were well below the reported average for response to initial SLT, which may be related to our SLT treatment regime. The benefits of SLT include minimal adverse effects, uncomplicated regimen (single application therapy that can be repeated as necessary) and comparable efficacy to IOP-lowering treatment alternatives. These factors combine to make SLT an excellent management option for those newly diagnosed with POAG, especially if adverse effects from medication and risk factors for poor compliance are taken into account [3, 4, 8, 18, 19]. The use of SLT as a primary treatment option may in turn have wide ranging benefits, from better patient outcomes due to the decrease in eye drop compliance issues, to economic benefits due to a reduction in the number of patients on lifelong eye drops [9, 15].
It will take some years before we can see the results of repeating SLT in eyes where the initial treatment has lasted for several years. Our results suggest that a second treatment might last several more years. If this proves to be the case, there could be a significant decrease in burdens of cost, local side-effects, and daily effort for many patients.
References
American Academy of Ophthalmology (2009) Glaucoma. Singapore: American Academy of Ophthalmology p 3–16 and 85–121
Cvenkel B (2004) One-year follow-up of selective laser trabeculoplasty in open-angle glaucoma. Ophthalmologica 218:14–19
Damji KF, Shah KC, Rock WJ, Bains HS, Hodge WG (1999) Selective laser trabeculoplasty v argon laser trabeculoplasty: a prospective randomised clinical trial. Br J Ophthalmol 83(6):718–722
Damji KF, Bovell AM, Hodge WG, Rock W, Shah K, Buhrmann R, Pan YI (2006) Selective laser trabeculoplasty versus argon laser trabeculoplasty: results from a 1-year randomised clinical trial. Br J Ophthalmol 90:1490–1494
Francis BA, Ianchulev T, Schofield JK, Minckler DS (2005) Selective laser trabeculoplasty as a replacement for medical therapy in open-angle glaucoma. Am J Ophthalmol 140(3):524–525
Juzych MS, Chopra MR, Hughes BA, Kim C, Goulas MT, Shin DH (2004) Comparison of long-term outcomes of selective laser trabeculoplasty versus argon laser trabeculoplasty in open-angle glaucoma. Ophthalmology 111(10):1853–1859
Nagar M, Ogunyomade A, O’Brart D, Howes F, Marshall J (2005) A randomised, prospective study comparing selective laser trabeculoplasty with latanoprost for the control of intraocular pressure in ocular hypertension and open angle glaucoma. Br J Ophthalmol 89(11):1413–1417
Nagar M, Luhishi E, Shah N (2009) Intraocular pressure control and fluctuation: the effect of treatment with selective laser trabeculoplasty. Br J Ophthalmol 93:497–501
Lee R, Hitnik C (2006) Projected cost comparison of selective laser trabeculoplasty versus glaucoma medication in the Ontario health insurance plan. Can J Ophthalmol 41(4):449–456
Werner M, Fran Smith M, Doyle W (2007) Selective laser trabeculoplasty in phakic and pseudophakic eyes. Ophthalmic Surg Lasers imaging 38(3):182–188
Moubayed S, Hamid M, Choremis J, Li G (2009) An unusual finding of corneal edema complicating selective laser trabeculoplasty. Can J Ophthalmol 44(3):337–338
Kim D, Singh A (2008) Severe iritis and choroidal effusion following selective laser trabeculoplasty. Ophthalmic Surg Lasers imaging 39(5):409–411
Hong B, Winer J, Martone J, Wand M, Altman B, Shields B (2009) Repeat selective laser trabeculoplasty. J Glaucoma 18(3):180–183
Nagar M, Ogunyomade A, O’Brart DPS, Howes F, Marshall J (2009) A randomised, prospective study comparing selective laser trabeculoplasty with latanoprost for the control of intraocular pressure in ocular hypertension and open angle glaucoma. Br J Ophthalmol 89:1413–1417
Olthoff C, Schouten J, Van de Borne B, Webbers C (2005) Non compliance with ocular hypotensive treatment in patients with glaucoma or ocular hypertension. Ophthalmology 112:953–961
Pham H, Mansberger S, Brandt JD (2008) Argon laser trabeculoplasty. The gold standard. Surv Ophthalmol 53(6):641–646
Shihadeh WA, Ritch R, Liebmann JM (2006) Hyphema occuring during selective laser trabeculoplasty. Ophthalmic Surg Lasers imaging 37(5):432–433
Hodapp E, Parrish I, Anderson D et al (1993) Clinical decision in glaucoma. CV Mosby, St Louis MO, pp 52–61
Melamed S, Ben Simon G, Levkovitch-Verbin H (2003) Selective laser trabeculoplasty as primary treatment for open-angle glaucoma (a prospective, nonrandomized pilot study). Arch Ophthalmol 121(7):957–960
Acknowledgments
No funding was received for the purposes of this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study has been approved by the appropriate ethics committee.
Rights and permissions
About this article
Cite this article
Avery, N., Ang, G.S., Nicholas, S. et al. Repeatability of primary selective laser trabeculoplasty in patients with primary open-angle glaucoma. Int Ophthalmol 33, 501–506 (2013). https://doi.org/10.1007/s10792-013-9729-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-013-9729-3