Abstract
Garcinia humilis is popularly used to treat digestive, intestinal and inflammatory illness. We investigated the in vivo and in vitro effects of the methanol extract of G. humilis leaves (MEGh) on inflammatory cells behavior (migration and chemical mediators release) and hypersensitivity. Anti-inflammatory activity was investigated using carrageenan-induced inflammation in the subcutaneous tissue of male Swiss mice treated orally with MEGh (0.1–30 mg/kg). Leucocyte migration, chemical mediators secretion (TNF, IL-1β, IL-6 and CXCL1) and protein exudation were quantified in the exudate. The adhesion molecules expression (CD62L and CD18), chemical mediators and chemotaxis was evaluated using neutrophils or macrophages RAW.264.7 previously treated with the extract (1–100 µg/mL) and activated with LPS. The anti-inflammatory activity of the isolated compounds friedelin, canophyllol, amentoflavone and 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone (10 μM) was evaluated in macrophages nitric oxide (NO) and TNF release. MEGh, given orally (30 mg/kg), significantly reduced neutrophil migration and decreased TNF, IL-1β and CXCL1 levels, without interfering with protein exudation and IL-6. In vitro, the extract significantly reduced IL-1β and IL-6 levels but did not alter TNF and CXCL1. The MEGh also reduced the expression of CD62L and CD18 and consequently neutrophil chemotaxis. The compounds friedelin, amentoflavone and 3-demethyl-2-geranyl-4-prenylbellidypholine xanthone decreased the secretion of NO and TNF by RAW264.7. The MEGh effects were extended to the pain-like behaviour induced by carrageenan in the mice hindpaw. MEGh presented important anti-inflammatory effects probably due to its activity on neutrophil migration and on important chemical mediator release, scientifically reinforcing its use as medicinal plant.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Inflammation is a pathophysiological response caused by infection or tissue damage, which consists of cellular and molecular events to eliminate the causative stimulus and return to homeostasis. In response to injury, leukocytes, primarily neutrophils, are recruited to the inflammatory focus. These cells release pro-inflammatory mediators, such as cytokines and chemokines, which attract other inflammatory cells and assemble effector functions to destroy pathogens (Savill et al. 2002; Pantarelli and Welch 2018).
Extravasation of leukocytes to the damaged tissue requires adhesion of these cells to the capillary endothelium. This process is mediated by the sequential expression of glycoprotein receptors, termed adhesion molecules, which are expressed in leukocytes and in endothelial cells. The first class of adhesion molecules involved in this process belongs to the selectins family (P, E and L-selectin), responsible for the leukocytes rolling on the surface of the endothelium. Endothelial cells express two types of selectins, called P-selectin (CD62P) and E-selectin (CD62E). A third selectin, termed L-selectin (CD62L), is expressed on the surface of leukocytes. This glycoprotein serves for the binding of leukocytes to endothelial cells that are activated by inflammatory mediators during inflammation. After activation, L-selectin is cleaved into inactive fragments by membrane metalloproteases, such as ADAM17 (Uchimura and Rosen 2006; Geng et al. 1990; Smith 2007; Ley et al. 2007; Freire and Van Dyke 2013).
Leukocyte arrest is mediated by the activation of G protein-coupled receptors (GPCRs) by inflammatory chemokine’s, which mediate the activation of integrins, another class of adhesion molecules. The most important subfamily in neutrophils is the β2-integrin family. β2-integrin is expressed on the surface of leukocytes and binds to glycoproteins expressed on the endothelial surface, including ICAM-1 or VCAM-1. This binding allows the reduction of the leukocyte rolling speed, firm adhesion of the leukocytes to the endothelium, as well as alteration of the cells compliance, and then transmigration to the focus of the lesion (Ghandour et al. 2007; Williams et al. 2011; Herter and Zarbock 2013).
During the inflammatory process, the presence of pain is also common, which is resulted basically from the interaction between damaged tissue and peripheral sensory neurons through the activity of inflammatory mediators, such as cytokines and chemokines, which are known for sensitizing the nociceptive response (Patapoutian et al. 2009; Grace et al. 2014).
Garcinia humilis, popularly known as “achachairu” comes from the region of Santa Cruz-Bolivia, but very well adapted in the southern region of Brazil. In Brazil, the G. humilis is little known and sometimes confused by the lay public with fruits of other species, such as bacuripari and bacurizinho. In folk medicine, fruits and leaves are used as healing, digestive and laxative and in treatment of rheumatism, gastric ulcer and inflammation (Dal Molin et al. 2012; Marques et al. 2012).
Data from the literature demonstrate the great potential of the Garcinia genus, highlighting the anti-inflammatory activity (Martins et al. 2008), antiproliferative (Cao et al. 2007) and antimalarial (Konziase 2015). Despite the popular use, the species G. humilis has few studies, the methanolic extract of the seeds of G. achachairu (now called G. humilis) from which a new phenolic compound called Guttiferona A was extracted, was active against pain in models induced by formalin, capsaicin, glutamate and carrageenan (Dal Molin et al. 2012). In addition, it presented gastroprotective effects against some ulcer models in mice (Niero et al. 2012). The methanolic extract of the seeds also has xanthones with a gastroprotective effect (Mariano et al. 2016a, b) and antiproliferative effect against breast, prostate and kidney cancer cell lines (Mariano et al. 2016a, b).
Acute inflammation is associated with several pathologies and if not properly treated can progress to chronic state leading to loss of function. Although extracts of G. humilis and some of its isolated compounds have been subjected to some pharmacological studies, there is no report in the literature of the effects of the extract of G. humilis’ leaves on acute inflammation and inflammatory pain. Thus, the present work aimed to investigate the anti-inflammatory and antinociceptive activity of the extract obtained from G. humilis’ leaves using in vivo and in vitro methodologies, aiming to confirm the ethnopharmacological use of this herbal product to treat inflammatory and painful processes.
Materials and methods
Drugs and reagents
Indomethacin, Morphine (Dimorf®), Lipopolysaccharide from E. coli (Sigma), Carrageenan-λ (Sigma), Ketamine and xilazyne (Vitrocell, Campinhas, SP, Brazil), Filter (TPP Spritzen-Syringe-Filter 0.22 μm), Phosphate buffered saline (PBS) Sodium Chloride (NaCl), May-Grünwald Dye (New Prov, Brazil), Giemsa Dye (New Prov), Formaldehyde (Quemis), DMEM (Dulbecco's Modified Eagle Medium), Oyster Glycogen (Sigma Aldrich), Bradford), Tetrazolium thiazolyl bromide (Sigma-Aldrich), Agarose (Invitrogen ™), Hank’s Balanced Salt solution (HBSS) (Sigma Aldrich, St. Louis, MO, USA), Anti-CD62L and anti-CD18 monoclonal antibodies (BD Pharmingen™), anti-CD62L-phycoerythrin (PE), anti-CD18-fluorescein-isothiocyanate (FITC), Fibroblast L929 murine NCTC lineage, Mouse antibody TNF, IL-1β, IL-6 and CXCL1/KC (R&D Systems - DuoSet®).
Chromatographic conditions and apparatus
Column silica gel (E. Merck; 70–230 mesh) and flash silica gel (E. Merck; 230–400 mesh) were used for open column chromatography. The purity of the isolated compounds was examined by thin layer chromatography (TLC) using Merck silica gel pre-coated aluminum plates (thickness = 200 µm) on different solvent systems with increasing of polarities. Plates were visualized under UV at 254 and 366 nm and by spraying with anisaldehyde sulphuric or FeCl3 reagents. 1H NMR spectra were recorded on a Bruker AV-300 at 300 MHz, while the 13C NMR were recorded on a 75 MHz spectrometer in CDCl3 or CD3OD. Chemical shifts (δ) and coupling constants were expressed in ppm Hz, respectively. All the reagents and solvents used were of analytical grade.
Plant material
G. humilis leaves was collected in Camboriú City, SC, Brazil (Latitude 27°04′24.7′′S and longitude 48°42′45.1′′W) in August 2010 and identified by Dr. Oscar B. Iza (University of Vale do Itajaí). The material was collected on private which does not require specific permissions and does not involve endangered or protected species. A voucher specimen was deposited at the Barbosa Rodrigues Herbarium (Itajaí-SC) under number HBR 52,637.
Extraction and isolation
The extraction and isolation were carried out as described by Mariano et al. (2016a, b) with minor modifications. Briefly, air-dried powdered leaves (1.5 kg), were extracted with methanol (3L) at room temperature for seven days. The extracted portion was concentrated under reduced pressure in a rotatory evaporator, yield a dark-brown residue (135 g; 8.62%). All the extract was suspended in a methanol:water (65:35) mixture (500 mL) and subjected to liquid–liquid partition using solvents of increasing polarity: dichloromethane and ethyl acetate (300 mL; 4 × each), yielding (25.12 g; 20%) and (22.68 g;18.1%), respectively. Part of the soluble dichloromethane fraction (10 g) was subjected to open column chromatography (OCC) and eluted with dichloromethane:methanol (100:0 0:100) in increasing order of polarity to afford 168 fractions. Ethyl acetate fraction was subjected to a similar chromatographic method as mentioned above, furnishing 132 factions.
HPLC–UV analysis
A Shimadzu LC-10AD LC system (Shimadzu, Tokyo, Japan) consisting of a binary pump and a Shimadzu SPD-M10A photodiode array detector were used. Injections (20 μL) were carried out on a Phenomenex® (Torrance, California, USA) Luna C18, 5 μm (250 × 4.6 mm) column at 30 °C with detection at 300 nm, adapting the methodology previously developed (Dutra et al. 2014). The mobile phase, at 1.0 mL/min, consisted of methanol (A) and pH 3.0 water acidified with phosphoric acid 0.5% (B) according to the following gradient (A:B v/v): 40:60 (0–40 min); 100 (40–45 min); 60:40 (45–50 min keeping until 50 min). Methanol extract was dissolved in 1:1 v/v of methanol:water pH 3.0 with phosphoric acid and a diluted to 2.5 mg mL−1 solution.
Animals
Male Swiss mice (20–30 g) were obtained from the Central Animal House from Universidade do Vale do Itajaí (UNIVALI). The animals were kept in enriched environment with controlled temperature (22 ± 1 °C), humidity (40–60%) in cycles of 12 light/12 h dark, with food and water provide ad libitum. The animals were acclimated for a period of 5 days before the experiments. During experimental procedures, animals were anaesthetized with ketamine (150 mg/kg, i. p.) and xylazine (15 mg/kg i. p.) to avoid any stress condition. All procedures were performed according to the Brazilian Society of Science of Laboratory Animals guidelines for the proper care and use of experimental animals. All experiments were approved by local ethics committee (protocol CEUA #061/17).
In vivo leukocyte migration: air pouch model
The air pouches were produced on the back of the mice according to the procedure described by Sedwick (1986) and Jain and Parmar (2011). Initially, 3 mL of sterile air were inflated into the dorsal subcutaneous tissue of previously anesthetized mice using a filter (TPP Spritzen-Syringe-Filter 0.22 µm). Three days later, further 3 mL of sterile air were inflated. Six days after the first injection, the animals were orally pre-treated with G. humilis extract (0.1, 1, 10 or 30 mg/kg) diluted in PBS, indomethacin (30 mg/kg, positive control) diluted in PBS or phosphate-buffered saline (PBS, negative control). One hour after the treatments, a solution of carrageenan (1%; 3 mL/cavity) was injected directly into the air pouch. Naïve animals received only PBS and anesthesia for collection of exudate washing, without pharmacological treatments. Four hours later, a small incision was made in the air pouch. The inflammatory infiltrate was collected washing the cavity with 2 mL of sterile PBS. The sample was centrifuged at 600g for 10 min at 4 °C. Cells were counted using Neubauer chamber (Fig. 1). Differential cell counting was performed manufacturing slides stained with Giemsa dye following the manufacturer’s recommendation. Hundred cells per slide were counted to obtain the absolute number and then the relative number of migrated polymorphonuclear cells was calculated. Results were expressed as total number of cells/mL. The exudate was submitted to the ELISA protocol for cytokines quantification and the protein concentration was measured in a spectrophotometer at 590 nm using the Bradford reagent and bovine albumin was used as standard (Bradford 1976).
The air pouch lining tissue was collected for histological analysis and preserved in formaldehyde solution until the blades were made. The preparation consisted of insertion of the tissue fragment into a cassette. The sample then underwent a dehydration process for 1 h first in 70% alcohol, followed by 96% alcohol and finally absolute alcohol. Subsequently, the sample was placed in xylol and then in paraffin bath. After this process, the cassette with the sample was placed in the equipment called a microtome where serial cuts were made between 3 or 4 microns and the tape produced in this process was placed in a water bath with a temperature of approximately 56 °C. After stretching the tape in the bath, the required number of fragments were removed and these were placed on the slide. Xylol, hematoxylin and eosin were used for the staining of the slides. The stained samples were covered with a cover slip and the microscopic evaluation was performed under an optical microscope on the 4, 10 and 40 × objectives.
In vitro anti-inflammatory effects
LPS-stimulated neutrophil
Neutrophils were obtained from Swiss male mice 4 h after i. p. injection of 3 mL of PBS solution containing sterile oyster glycogen 1%. After this period, the animals were anesthetized with ketamine (150 mg/kg i. p.) and xylazine (15 mg/kg i. p.), and the cells were collected by rinsing the abdominal cavity with 3 mL of sterile PBS. The number of viable cells (98%) was counted in Neubauer chamber using a light microscope (Olympus) by the Trypan blue exclusion test.
The cells were incubated with DMEM medium in the presence or absence of LPS (5 µg/mL) and treated simultaneously with G. humilis (0.1, 1, 10 or 100 µg/mL) diluted in DMEM medium for 18 h. Control cells were treated only with DMEM. Then, 10 µL of neutrophil suspension from each well were mixed with 10 µL of 0.4% trypan blue solution. The sample (10 µL) was placed in Neubauer chamber, and 200 cells were counted to calculate the percentage of viable cells. The supernatant was then collected and kept frozen (− 80 °C) until analysis of cytokine and NO levels.
In vitro chemotaxis assay
The assay was performed using the methodology described by Nelson et al. (1975) and modified by Heit and Kubes (2003). Neutrophils obtained as previously described were used for the assay. Agarose gel was prepared in the following proportions: agarose (1.2%), HBSS (50%), and DMEM supplemented with 20% FBS (50%). 3 mL of gel per well were placed in 6-well plates. After solidification, wells of 3.5 mm of diameter were made in line in the gel, at 2.4 mm between each well. fMLP was used as chemotactic agent at the concentration of 0.1 µM. Cells were treated with G. humilis extract (0.1, 1, 10 or 100 µg/mL) diluted in DMEM medium and then placed in the wells of both extremities at concentration of 1 × 107 cells/mL. After 4 h into the cell incubator at 37 °C and 5% CO2, the number of cells that migrated towards the central well containing fMLP was counted.
Expression of adhesion molecules
The effect of G. humilis extract on expression of CD62L and CD18 by neutrophils was investigated by flow cytometry. Neutrophils were incubated in the absence or presence of G. humilis (1, 10 or 30 µg/mL) diluted in DMEM medium and simultaneously stimulated with LPS (5 µg/mL; for 1 h). Subsequently, cells were washed with cold PBS and incubated with monoclonal antibody anti-CD62L conjugated with FITC, or anti-CD18 conjugated with PE at 4 °C during 20 min in the dark. Cells were fixed by adding paraphormaldehyde solution (2%). Cells were analyzed on Accuri C6 flow Cytometer (Immunocytometry System, BD Bioscience), and data from 10.000 events were obtained. The results were expressed as mean fluorescence intensity (MFI).
LPS-stimulated macrophages
The murine RAW 264.7 macrophage cell line was purchased from the Bank of Cells from Rio de Janeiro (BCRJ, Rio de Janeiro, RJ, Brazil) and maintained in DMEM supplemented with 10% FBS, 100 μg/L streptomycin and 100 IU/mL penicillin at 37 °C in a 5% CO2 atmosphere. The cell viability was evaluated by MTT method. The mitochondrial-dependent reduction of MTT to formazan was used to measure cell respiration as an indicator of cell viability. Briefly, RAW 264.7 cells were seeded in 96-well plates at the density of 1 × 105 cells/well together with DMEM. After 24 h of incubation, the adhered cells were treated with friedelin, canophyllol, amentoflavone or 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone (10 μM) diluted in DMEM. Twenty-four hours later, after changing the medium, MTT was added to a final concentration of 0.5 mg/mL, and the cells were incubated for 4 h at 37 °C and 5% CO2. The medium was then removed and the precipitate was solubilized in DMSO.
In another set of experiment, the RAW 264.7 cells were seeded at a density of 1 × 105 cells well in 96 well plates and incubated for 24 h at 37 °C and 5% CO2. Then the aged media in each well were aspirated and replaced with fresh FBS-free DMEM media. The cells were treated with the extract G. humilis (10 µg/mL), friedelin, canophyllol, amentoflavone or 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone (10 μM) prepared in FBS-free DMEM. After 1 h treatment, cells were stimulated with 1 μg/mL of LPS for 24 h. After 24 h the supernatant was removed to NO2− and TNF levels evaluation.
Cytokines and NO2 − analysis
Samples of exudate collected from mice submitted to the air-pouch model and the supernatant obtained from the cell cultures were submitted to biochemical analysis (100 μl) to quantify IL-1β, TNF, IL-6 and CLXCL1 by ELISA assay following manufacturer’s instruction (R&D System).
Nitrite level in the culture supernatant was used as the measurement of NO production by neutrophils and macrophages. Samples (100 µL) were incubated for 10 min with 100 µL of Griess reagent (1% sulfanilamide, 0.1% naphthylethylenediamine dihydrochloride in 5% phosphoric acid). The absorbance was determined at 540 nm using a multiwell plate reader (SPECTROstar Nano - BMG Labtech). The amount of nitrite was calculated from sodium nitrite (NaNO2) standard curve and the results were reported as µM of NO2− (Grisham et al. 1996).
Inflammatory hypersensitivity induced by carrageenan
For the induction of inflammatory pain, the mice received an i. pl. injection of 50 µL of carrageenan (300 µg/paw) on the plantar surface of the right hind paw. Initially, the animals were pretreated with G. humilis (1, 10 or 30 mg/kg) or indomethacin (5 mg/kg), 1 h prior to induction of hypersensitivity. The animals were then i.pl. injected with carrageenan and were evaluated for mechanical hypersensitivity using the von Frey monofilament at the intervals of 1, 3. 4, and 6 h after carrageenan injection (Quintão et al. 2005; Cobos et al. 2012). All substances, including carrageenan, extract and indomethacin were diluted in PBS solution.
Locomotor activity assessment
To exclude possible nonspecific effects, mice treated with G. humilis (30 mg/kg, p. o.) or morphine (5 mg/kg, s. c.) were submitted to the open-field test 1 h after the treatments. The open-field apparatus consists of a transparent glass-circular arena, 60 cm in diameter and 50 cm in height, with the floor divided into 12 quadrants by intersecting lines drawn on its surface. The number of squares crosses with all paws (“crossings”) was counted in a 6 min session. The control mice received vehicle (Quintão et al. 2005).
Statistical analysis
The results obtained in the experiments were expressed as mean ± standard error of the mean (S. E. M.), except the DI50 values (doses of the drug that produced the response in 50% in relation to the control group), which are presented as the geometric mean accompanied by confidence limits at the 95% level. Inhibition percentages were quoted as the mean ± standard error of the mean of the difference (in percentage) between the areas under the curves obtained for each individual experiment relative to the corresponding control group. Data analysis was performed using the multiple-comparison variance (ANOVA) and, when necessary, the Tukey–Kramer or Dunett test was used as a post-test. All analyses were performed using the GraphPad PRISM® program (GraphPad Software, San Diego, CA, USA).
Results
Phytochemical profile and HPLC–UV analysis
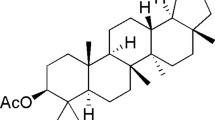
Based on OCC procedures and TLC profiles, is was obtained four compounds: friedelin (1), canophyllol (2) and 3-demethyl-2-geranyl-4-prenylbellidypholine xanthone (3) obtained from dichloromethane soluble fraction and amentoflavone (4) obtained from ethyl acetate soluble fraction. All substances were identified by NMR direct comparison data with those reported previously (Almeida et al. 2011; Nozaki et al. 1986; Ricaldez et al. 2000; Torrico et al. 2008; Mariano et al. 2016a, b) (Fig. 2).
The benzophenone Guttiferone A was previously isolated from the G. humilis’ seed and, besides its biological effects, such as gastroprotection (Niero et al. 2012) and anti-hypersensitivity (Dal Molin et al. 2012), it also caused important genotoxic effects in several tissues (Terrazas et al. 2013). As observed in Fig. 3b, the extract obtained by G. humilis’ leaves does not present Gutiferone A (Fig. 3a), represented by the peak appearing in the retention time 47 min (Fig. 3a).
G. humilis inhibits in vivo neutrophil migration
The effects of different doses of G. humilis on neutrophil migration (in vivo) were evaluated in the dorsal subcutaneous tissue of Swiss mice during the action of carrageenan. The data presented in Fig. 4a demonstrates that G. humilis, at doses of 1, 10 or 30 mg/kg was able to reduce the migration of leukocytes to the air pouch when compared to the control group, with inhibition percentage of 93.10 ± 1.78% in the highest used dose (30 mg/kg). The ID50 value was 9.07 (6.19–13.29) mg/kg. Indomethacin, a drug used as positive control, also reduced cell migration, with inhibition of 89.8 ± 5.02%. Differential analysis of cells migrated to the air pouch demonstrated that the treatment with G. humilis at doses of ten or 30 mg/kg, as well as indomethacin, reduced the migration of leukocytes, specifically polymorphonuclear cells to the focus of the inflammation (Fig. 4b).
Air pouches were induced in the dorsal subcutaneous tissue of Swiss mice. The animals were treated orally one hour before the injection of 2 mL of carrageenan (1%). The lavage of the inflammatory infiltrate was collected 4 h after the injection of carrageenan into the air pouch. The determination of the (a) total number of exudate cells was performed on a Neubauer chamber and the (b) differential value was performed on smears stained by the May-Grunwald Giemsa method. (c) The protein concentration was measured in a spectrophotometer at 590 nm using the Bradford reagent and bovine albumin was used as standard. Values express the mean ± S.E.M. of tests performed with the inflammatory exudate obtained from six animals per group. *p < 0.05 and ***p < 0.001 vs control group significantly different from naïve group #p < 0.001 (one-way ANOVA followed by Tukey’s post hoc test)
In order to verify the concentration of proteins in the extravascular space the concentration of total proteins in the lavage infiltrate was measured by Bradford method. The data obtained show that G. humilis did not alter protein concentration in the exudate of the air pouch (Fig. 4c).
The histological analysis (Fig. 5) of air pouch lining tissue demonstrated reduction of leukocytes to the inflammatory tissue, as well as reduction of edema when compared to the control group.
The air pouch liner was fixed in 10% formaldehyde for histological evaluation. The microscopic evaluation was performed under an optical microscope on the 4, 10 and 40 ×. Dotted black arrows indicate the thickness of the connective tissue. Red arrows indicate the presence of inflammatory infiltrate. Keys indicate adema in the connective tissue (color figure online)
In vivo G. humilis reduces TNF, IL-1β and CXCL1 levels, but not IL-6
During the inflammatory process cytokines and inflammatory chemokines such as TNF, IL-1β, IL-6 and CXCL1 contribute for the extension of the inflammatory process. In order to verify the secretion of these inflammatory mediators, the cytokine dosage was performed by the ELISA immunoassay. Treatment of animals with G. humilis (30 mg/kg) was able to decrease the levels of TNF, IL-1β and CXCL1 (Fig. 6a, b, d) when compared to the control group, but without interference in IL-6 level (Fig. 6c).
Air pouch were induced in the dorsal subcutaneous tissue of Swiss mice. The animals were treated orally one hour before the injection of 2 mL of carrageenan. The lavage of the inflammatory infiltrate was collected 4 h after the injection of carrageenan into the air pouch. The levels of a TNF b IL-1β c IL-6 and d CXCL1 were determined by the ELISA method according to the manufacture’s standards. Values express the mean ± S.E.M. of tests performed with the inflammatory exudate obtained from 6 animals per group. *p < 0.05, **p < 0.01 and ****p < 0.0001, vs control group significantly different from naïve group #p < 0.001 (one way ANOVA fallowed by Tukey’s post hoc test)
G. humilis acts on the neutrophil secretion of cytokines and NO
To exclude possible cytotoxic effects of G. humilis extract, cell viability assays by the Trypan Blue technique and tetrazolium salt reduction (MTT) were performed. The extract of G. humilis did not produce cytotoxic effects on neutrophils and fibroblasts (data not shown). The interaction of neutrophils with resident cells, local inflammatory mediators and/or extracellular matrix lead to the production of several other mediators, including cytokines, degrading enzymes and reactive oxygen species, which can amplify the inflammatory response and damage the surrounding tissue. The treatment of neutrophils with G. humilis was able to decrease the levels of IL-1β (Fig. 7b), IL-6 (Fig. 7c) and NO2− (Fig. 7e), but did not interfere with the TNF (Fig. 7a) or CXCL1 (Fig. 7d) levels.
Neutrophils were obtained from the peritoneal cavity of Swiss mice (1% oyster glycogen). Neutrophils (1 × 106) were incubated in the presence or absence of LPS (5 µg/mL) along with G. humilis (0.1, 1, 10 or 100 µg/mL). The levels of a TNF, b IL-1β, c IL-6 and d CLXL1 were determined by the ELISA method according to the manufacture’s standards. The quantification of nitrite was performed by Griess reaction (e). The values express the mean ± S.E.M. of tests performed with cell obtained from 8 animals in per group. *p < 0.05 **p < 0.01 and ***p < 0.001 vs LPS group significantly different from basal group. #p < 0.001 (one way ANOVA followed by Tukey’s post hoc test)
In vitro G. humilis reduce neutrophil chemotaxis and adhesion molecule expression
The assays for the evaluation of the anti-inflammatory activity in vivo evidenced the decrease in the migration of inflammatory cells. Considering that the process of neutrophil migration from the blood vessel to the damaged tissue requires the action of adhesion molecules, we evaluated the expression of L-selectin (CD62L) and β2-integrin (CD18) on the surface of neutrophils stimulated with LPS.
The obtained results showed that treatment with G. humiluis increased the percentage of neutrophils positive for L-selectin (CD62L) (Fig. 8b). However, they reduced the expression of β2-integrin (CD18) in neutrophils (Fig. 8c), demonstrating that the treatment with G. humilis was able to affect the expression of adhesion molecules, especially β2-integrin in neutrophils.
For the chemotaxis assay (a), neutrophil cell suspension (1 × 107) was incubated with different concentrations of G. humilis (1, 10 or 100 µg/mL) for 15 min and placed in front of the fMLP (1 µM) on an agarose plate. Cells were counted using inverted microscopy at 20 × magnification. Quantification was made from the margin of the peripheral perforations towards the chemotactic agent (central perforation). For evaluation of the neutrophil adhesion molecules were obtained from the peritoneal cavity of Swiss mice (1% oyster glycogen). Neutrophils (1 × 106) were incubated in the presence or absence of LPS (5 µg/mL) along with G. humilis (0.1, 1, 10 or 100 µg/mL). Quantification of adhesion molecule expression was assessed by labeling with (b) CD62L and (c) CD18 detection antibodies by the flow cytometry method. Values express the mean ± S.E.M of tests performed with cells obtained from eight animals per group. ***p < 0.001 and ****p < 0.0001 vs LPS group significantly different from basal group #p < 0.001 (one way ANOVA followed by Turkey’s post hoc test)
In addition to the action of the adhesion molecules for cell migration, the presence of a chemotactic agent is necessary to attract the displacement of the neutrophil. To evaluate the direct action of G. humilis on the neutrophil locomotion properties, an in vitro chemotaxis induced by fMLP was performed.
Figure 8 demonstrated that the neutrophils significantly responded to chemotactic agent, moving towards the fMLP. On the other hand, the neutrophils treated with G. humilis presented significant reduction in chemotaxis against fMLP, reducing cell migration (Fig. 8a).
G. humilis and compounds isolated acts on the macrophages secretion of NO
To investigate the anti-inflammatory effects of G. humilis (10 µg/mL) and compounds friedelin, canophyllol, amentoflavone and 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone (10 μM), NO production was determined in the presence of the compounds in LPS-induced RAW 264.7 cells. When LPS was incubated with macrophages, the nitrite generation increased from the basal level after 24 h incubation (Fig. 9a and b). LPS-induced nitrite generation was significantly attenuated by G. humilis (10 µg/mL), friedelin (C1), amentoflavone (C3) and 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone (C4) at 10 μM (Fig. 9a and b). Regarding TNF secretion, only the 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone compound decreased levels of this cytokine (Fig. 9d). Is important to emphasize that G. humilis and all compounds did not cause cell cytotoxicity at 10 µg/mL and 10 uM (data not shown).
The murine macrophages strain RAW.264.7 was obtained from the Cell Bank of Rio de Janeiro (BCRJ, Rio de Janeiro, RJ, Brazil) and incubated in the absence of LPS (1 μg/mL) together with G. humilis (10 μg/mL) or friedelin (C1), canophyllol (C2), 3-desmethyl-2-geranyl-4-prenylbellidifoline xanthone (C3) and amentoflavone (C4) (10 µM) (a to d). Nitrite quantification was performed by the Griess reaction (a and b). Quantification of TNF was determined by the ELISA method according to the manufacturer's standards (c and d). Values express the mean ± S.E.M. of assays performed with 50.000 cells/well. *p < 0.1, **p < 0.01 and ****p < 0.0001, against the LPS group significantly different from baseline group #p < 0.001 (one-way ANOVA followed by Tukey's post hoc test)
G. humilis reduced inflammatory hypersensitivity induced by carrageenan
Other important inflammation cardinal sign is the hypersensitivity (pain). To establish the most effective dose of G. humilis and its time of action, the model of mechanical hypersensitivity induced by carrageenan was performed. The results presented in Fig. 10 demonstrate that all tested doses of G. humilis were capable of reducing the mechanical hypersensitivity when compared to the control group, frequency of response (Fig. 10a, b) had an inhibition percentage of 54.62 ± 5.5% and the mechanical threshold is an inhibition of 35.10 ± 3.87% in the highest used dose (30 mg/kg) Indomethacin, a drug used as a positive control, also reduced the carrageenan-induced sensitization, with inhibition of 74.78 ± 5.95% on the frequency response and 56.3 ± 8.23% on the mechanical threshold.
Mechanical hypersensitivity of mice previously treated with G. humilis (1, 10 or 30 mg/kg, p.o.), indomethacin (5 mg/kg, p.o.) or vehicle and injected 1 h later with carrageenan (300 µg/paw, i.pl.). The results are presented qualitatively by the (a and b) response frequency obtained with the von Frey filament of 0.6 g and quantitatively by the (c and d) mechanical withdrawal threshold (g). b and d represent the area under curve (AUC) of the respective line graph. Values express the mean ± S.E.M. of tests performed with six animals in each group. Significantly different from Control group, where **p < 0.01, ***p < 0.001 and ****p < 0.0001, # differs significantly from basal threshold
G. humilis does not alter the locomotor activity of the animals
In order to verify a possible sedative or muscle relaxant effect of G. humilis, the open field test was performed. The results showed that the highest dose of G. humilis (30 mg/kg) was not able to affect the locomotor activity of the animals (125.5 ± 17.6 number of crossings) when compared with the control group (138.4 ± 10.8). On the other hand, the mice treated with morphine presented significant increase in the locomotor activity (187.8 ± 28.6).
Discussion
The results obtained in this study demonstrate that the in vivo and in vitro treatment with G. humilis leaf extract exerts effects on the neutrophil function, diminishing the migration of cells to the inflammatory focus, followed by the reduction of inflammatory mediators.
It is important to emphasize that although previous studies indicate that the compound Guttiferone A obtained from the seeds of G. humilis, when administered orally, produces genotoxic effects in leukocytes, liver, bone marrow and testicular and cerebral cells. The extract obtained from the leaves of G. humilis used in this study does not have this compound, assuring its use (Terrazas et al. 2013).
The effects of G. humilis on acute inflammation were evaluated through the induction of inflammatory process by carrageenan injection using the air pouch model. The results obtained showed that the treatment with the extract of G. humilis reduced, in a dose-dependent manner, the number of migrated leukocytes to the air pouch. These results corroborate with studies already published for others species of Garcinia genus. Among them, G. kola (Olaleye et al. 2000), G. hambury (Pathong et al. 2007), G. mangostana (Chen et al. 2008), G. brasiliensis (Martins et al. 2008) and G. morella (Choudhury et al. 2018) demonstrated a pronounced anti-inflammatory effect in the carrageenan-induced paw edema model.
Based on the total leukocyte analysis, the differential analysis of the air pouch-migrated cells showed that the reduction of leukocytes is mainly marked by the decrease of polymorphonuclear cells (mainly neutrophils,) migration. From the air pouch lining tissue, histological analyses were performed to verify the reduction of cell migration, as well as the decrease of the connective tissue edema in comparison to the control group. It is known that edema formation is mediated mainly by increased vascular permeability, leukocyte migration and protein production/exudation (Gordon et al. 2005). Although the extract of G. humilis decreased cell migration, the extract was not able to block the exudation of proteins, because the process of vasodilation still happens, allowing the extravasation of proteins.
The inflammatory process is characterized by the high production and release of inflammatory cytokines and chemokines, among which TNF, IL-1β, IL-6 and CXCL1 play a prominent role. The results obtained show that the animals treated with extract of G. humilis had significant decrease of TNF, IL-1β and CXCL1, but not IL-6. TNF and IL-1β induce the production/secretion of other cytokines and chemokines, expression of adhesion molecules, angiogenesis and mediation of systemic inflammatory effects, such as fever, hypotension, and promotion of neutrophilia. CXCL1 is an important chemokine to attract polymorphonuclear cells to the inflammatory site. The reduction of TNF, IL-1β and CXCL1 directly implies in the progression of the inflammatory process, resulting in decrease of cell migration and production/secretion of inflammatory cytokines, contributing to the recovery of homeostasis (Tincani et al. 2007; Silva et al. 2017). IL-6 is released, in addition to neutrophils, by keratinocytes, fibroblasts and endothelial cells. This wide release by several cells justifies their increased levels in the in vivo model since in addition to inflammatory cells, other resident cells also release IL-6 (Sproston and Ashworth 2018). Although IL-6 is considered a pro-inflammatory cytokine, evidence also indicates that, under certain conditions, IL-6 has anti-inflammatory properties (Barton 1997; Xing et al. 1998).
To complete the in vivo findings, in vitro assays were also performed to evaluate the anti-inflammatory activity of the extract. Then, tests were performed using neutrophils recruited from mice and macrophages. For the in vitro assays, LPS was used to stimulate the inflammatory process. LPS is capable of triggering important inflammatory response from the activation of toll-like receptors (TLRs) that are expressed on the neutrophil cell membrane. After specific activation of the TRL4 by LPS, activation of inflammatory pathways, especially that of NF-κB, occurs, triggering the gene expression of iNOS, TNF, IL-1β, IL-6 and other inflammatory mediators. In addition, LPS is capable of inducing adhesion molecules expression, which are responsible for cell migration (Tucureanu et al. 2017; Naegelen et al. 2015).
In this context, the expression of the major adhesion molecules involved in cell migration, L-selectin and β2-integrin, was analyzed. Data obtained by flow cytometry showed that treatment with the extract of G. humilis alters the expression of CD62L. CD62L is exclusively expressed on the leukocyte membrane and mediates the rolling behavior of neutrophils in the vessel wall. This adhesion molecule becomes highly expressed in the cell membrane when hemodynamic changes occur by actions of inflammatory mediators. After the activation process, CD62L is cleaved by membrane metalloproteases, allowing the activation of CD18 (Uchimura and Rosen 2006). The obtained data demonstrate that the extract of G. humilis reduced the percentage of neutrophils positive for CD18. CD18 binds to different components of the cell membrane and adhesion molecules of the immunoglobulin superfamily (ICAM1 and 2, VCAM-1 and PECAM-1) present in the endothelium. The expression of this molecule in the quiescent endothelium is low, however, during an inflammatory process it increases, providing the firm adhesion of the leukocytes in the vascular endothelium for its subsequent migration to the affected site (Herter and Zarbock 2013). Thus, it is possible to infer that the decrease in cell migration may, in part, could be related to the observed changes in the expression of the adhesion molecules. The hypothesis is that the extract of G. humilis keeps the leukocytes in rolling behavior on the vessel wall, but it prevents the firm adhesion of these leukocytes in the vascular endothelium for later transmigration to the inflamed tissue.
During the transmigration process, leukocytes have become skilled in responding to chemotactic substances, such as fMLP. The fMLP interacts with the FPR1 receptor in neutrophils, promoting the recruitment of these cells, a process known as chemotaxis (Mcdonald et al. 2010; Zhang et al. 2010). The evaluation of chemotaxis was done by agarose migration technique, where it was observed that the cells treated with the leaves extract of G. humilis decreased the cellular migration towards the fMLP. The decrease in response to this agent hinders cell migration and prevents inflammatory cells from accumulating at the site of the lesion (Mcdonald et al. 2010).
To verify if the cytokines and chemokines reduction observed in in vivo protocol were due to a decrease in neutrophils migration, neutrophils collected directly from mice’s peritoneum were treated with the extract and then challenged with LPS. The results obtained in vitro show that the G. humilis extract decreases IL-1β and IL-6, without interfering with the secretion of TNF and CXCL1. The decrease in IL-1β levels in vitro corroborates with the data obtained in vivo. IL-1β is one of the most important markers of inflammation, able to induce inflammatory response associated with acute infection. Decreased levels of this cytokine are important for the decreased cell migration and impairment of inflammatory-cytokine release. IL-6 is secreted by several cells, including neutrophils. The obtained results evidenced that the extract of G. humilis acts in the reduction of neutrophils, which corroborates with the decreased levels of IL-6 secreted by the neutrophils treated with the extract. TNF is a ubiquitous cell signaling protein produced primarily by macrophages, as well as other cells, such as leukocytes, dendritic cells, smooth muscle cells, fibroblasts, endothelial and epithelial cells (Bartekova et al. 2018). Knowing that TNF secretion by neutrophils was not inhibited by treatment with the extract of G. humilis, it is likely that the extract, in addition to acting on neutrophils, also acts on macrophages, and therefore, TNF levels presented decrease in vivo, but not in vitro. The same can be proposed for CXCL1 levels. It is known that resident macrophages are responsible for the production of CXCL1. The results obtained show that the in vivo treatment with extract of G. humilis decreases the levels of CXCL1, but the same is not observed in the in vitro tests, where the treatment of neutrophils with G. humilis does not decrease the levels of CXCL1, once again, the action of the extract also on macrophages. During the inflammatory process, there is an excessive stimulation of NADPH oxidase activity by cytokines and other agents, leading to iNOS expression. INOS synthesizes NO that has a multifaceted role in inflammatory reactions, ranging from increased vasodilation and edema formation through the regulation of leukocyte activity to the modulation of sensory nerve endings and tissue cytotoxicity (Tripathi et al. 2007). Treatment of neutrophils with G. humilis extract was able to reduce NO levels. This data corroborates with results obtained in the study by Chen et al. (2008), which demonstrates the performance of G. mangostana in the reduction of NO through the reduction of iNOS expression.
To verify the activity of G. humilis on macrophages and to analyze which specific compounds are related to the anti-inflammatory activity, the compounds friedelin, canophyillol, amentoflavone and 3-desmethyl-2-geranyl-4-prenylbellidifoline xanthone were evaluated in macrophages stimulated with LPS. All compounds except canophyllol were able to decrease NO secretion. A study by Li et al. (2019) used LPS-stimulated RAW 264.7 macrophages and treated with amentoflavone (50 μM). In this study, amentoflavone was able to decrease NO secretion and the EtOAc fraction containing amentoflavone was able to reduce the secretion of IL-6 and PGE2 but did not decrease TNF levels. Collaborating with these data, Oh et al. (2013) also describes amentoflavone (100 and 200 μM) as an important inhibitor of NO and PGE2 secretion, as well as decreasing the mRNA expression of pro-inflammatory genes such as iNOS, TNF, COX-2 and IL-1β. Both studies corroborate, in part, the results obtained in the present study, suggesting amentoflavone as an important compound for the anti-inflammatory potential of G. humilis. In contrast to the friedelin compound, although the levels of nitric oxide have decreased, the literature only demonstrates its anti-inflammatory, analgesic and antipyretic potential in vivo models (Paulrayer et al. 2011).
The compound that showed better anti-inflammatory profile was 3-desmethyl-2-geranyl-4-prenylbellidypholine xanthone, once it was able to decrease NO and TNF levels. In fact, literature data demonstrate that this class of compounds presents important biological activities, among them the anti-inflammatory effect, mainly reducing the COX-2 and NF-κB activity (Tian-Yong et al. 2019; Ren et al. 2019).
During the inflammatory process, the symptom that causes the most discomfort and disability to individuals is pain. Knowing this, the effect of the extract of G. humilis on the mechanical threshold of paw withdrawal in the model of hypersensitivity induced by carrageenan in mice was evaluated. Treatment with G. humilis reduced the frequency of response and increased the mechanical threshold of carrageenan-stimulated animals. The genus Garcinia, besides possessing anti-inflammatory activity, also has important antinociceptive activity, G. gardneriana (Cechinel Filho et al. 2000), G. hanburyi (Panthong et al. 2007), G. kola (Olaleye et al. 2000) and G. mangostana (Chen et al. 2008), have been described as potent antinociceptive effect.
The pronociceptive action of neutrophils has already been studied by several authors, Levine et al. (1984) have shown that the hypersensitization induced by LTB4, C5a and fMLP depended on the migration of neutrophils and Cunha et al. (2008) also demonstrated that neutrophils are involved in the genesis of inflammatory hypersensitivity, especially carrageenan-induced hypersensitivity. Considering that activated neutrophils produce and release inflammatory cytokines, such as TNF, IL-1β and IL-6, and these can act on receptors present in the peripheral nociceptors and lead to the triggering of the painful process, the reduction of TNF, IL-1β and CXCL1 shown in air pouch model, exhibited by in vivo treatment with extract of G. humilis, contributes to the reduction of the nociceptive sensitivity triggered by mechanical stimuli.
Taken together, the data herein obtained allow us to conclude that the treatment with the extract of G. humilis inhibits some neutrophilic functions, blocking the secretion and migration pathways of neutrophils, besides exerting antinociceptive effects in relation to inflammatory pain, suggesting its therapeutic application in acute inflammatory reactions.
References
Almeida MFO, De Melo ACR, Pinheiro MLB, Silva JRA, De Souza ADL (2011) Constituintes químicos e atividade leishmanicida de gustavia elliptica (lecythidaceae). Quim Nova 34:1182–1187
Bartekova M, Rodosinska J, Jelemensky M, Dhalla NS (2018) Role of cytokines and inflammation in heart function during health and disease. Heart Fail Rev 23:733–758
Barton BE (1997) IL-6 insights into novel biological activities. J Clin Immunol Immunopathol Res 85:16–20
Bradford MMA (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cao S, Brodie PJ, Miller JS, Ratovoson F, Birkinshaw C, Randrianasolo S, Rakotobe E, Rasamison VE, Kingston DG (2007) Guttiferones K and L, antiproliferative compounds of Rheedia calcicola from the Madagascar rain forest. J Nat Prod 70:686–688
Cechinel Filho V, Da Silva KL, De Souza MM, Yunes RA, Guimarães CL, Verdi LG, Simionatto EL, Delle Monache F (2000) I3-naringenin-II8-4'OMe-eriodictyol: a new potential analgesic agent isolated from Rheedia gardneriana leaves. J Nature R 55:820–823
Chen LG, Yang LL, Wang CC (2008) Anti-inflammatory activity of mangostins from Garcinia mangostana. Food Chem Toxicol 46:688–693
Choudhury B, Raghuram K, Elancheran R, Bharali R, Kotoky J (2018) Garcinia morella fruit, a promising source of antioxidant and anti-inflammatory agents induces breast cancer cell death via triggering apoptotic pathway. Biomed Pharmacother 103:562–573
Cobos EJ, Ghasemlou N, Araldi D, Segal D, Duong K, Woolf C (2012) Inflammation-induced decrease in voluntary wheel running in mice: a non-reflexive test for evaluating inflammatory pain and analgesia. Pain 153:876–884
Cunha TM Jr, Verri WA, Schivo IR, Napomoga MH, Parada CA, Poole S, Teixeira MM, Ferreira SH, Cunha F (2008) Crucial role of neutrophils in the development of mechanical inflammatory hypernociception. J Leukoc Biol 83:824–832
Dal Molin MM, Silva S, Alves DR, Quintão NLM, Monache FD, Cechinel Filho V, Niero R (2012) Phytochemical Analysis and Antinociceptive Properties of the Seeds of Garcinia achachairu. Arch Pharmacal Res 35:623–631
Dutra DM, Barth CS, Block LC, Quintão NLM, Couto AG, Cechinel Filho V, Bresolin TMB (2014) Simultaneous determination of four phenolic compounds in extracts of aerial parts of Ipomoea pes-caprae (L.) R. Br. (Convolvulaceae) by HPLC-UV. Quim Nova 37:1510–1514
Freire MO, Van Dyke T (2013) Natural resolution of inflammation Periodontol 2000(63):149–164
Geng JG, Bevilacqua MP, Moore KL, Mcintyre TM, Prescott SM, Kim JM, Bliss GA, Zimmerman GA, Mcever RP (1990) Rapid neutrophil adhesion to activated endothelium mediated by GMP-140. Nature 343:757–760
Ghandour HX, Cullere A, Alvarez FW, Luscinskas T, Mayadas N (2007) Essential role for Rap1 GTPase and its guanine Exchange factor CalDAG-GEFI in LFA-1 but not VLA-4 integrin mediated human T-cell adhesion. Blood 110:3682–3690
Gordon JR, Li F, Zhang X, Wang W, Zhao X, Nayyar A (2005) The combined CXCR1/CXCR2 antagonist CXCL8 (3–74) K11R/G31P blocks neutrophil infiltration, pyrexia and pulmonary vascular pathology in endotoxemic animals. J Leukoc Biol 78:1265–1272
Grace PM, Hutchinson MR, Maier SF, Watkins LR (2014) Pathological pain and the neuroimmune interface. Nat Rev Immunol 14:217–231
Grisham MB, Johnson GG (1996) quantitation of nitrate and nitrite in extracellular fluids. Methods Enzymol 268:237–246
Heit B, Kubes P (2003) Measuring chemotaxis and chemokinesis: the under-agarose cell migration assay. Sci Signal 170:15
Herter J, Zarbock A (2013) Integrin regulation during leucocyte recruitment. J Immunol 190:4451–4457
Jain M, Parmar HS (2011) Evaluation of antioxidative and anti-inflammatory potential of hesperidin and naringin on the rat air pouch model of inflammation. Inflamm Res 60:483–491
Konziase B (2015) Protective activity of biflavanones from Garcinia kola against Plasmodium infection. J Ethnopharmacol 172:214–218
Levine JD, Lau W (1984) Leukotriene B4 produces hyperalgesia that is dependent on polymorphonuclear leukocytes. Science 225:743–745
Ley K, Laudanna C, Cybulsky MI, Nourshargh S (2007) Getting to the site of inflammation: the leukocyte adhesion cascade update. Nat Rev Immunol 7:678–689
Li M, Li B, Hou Y, Tian Y, Chen L, Liu S, Zhang N (2019) Anti-inflammatory effects of chemical components from Ginkgo biloba L. male flowers on lipopolysaccharide-stimulated RAW264.7 macrophages. Phytother Res 33:989–997
Mariano LNB, Silva LM, Souza P, Boeing T, Somensi LB, Bonomini TJ, Monache FD, Cechinel Filho V, Andrade SF, Niero R (2016a) Gastroprotective xanthones isolated from Garcinia achachairu: Study on mucosal defensive factors and H+, K+-ATPase activity. Chem-Biol Interact 258:30–39
Mariano LNB, Vendramini-Costa DB, Ruiz ALTG, De Carvalho JE, Corrêa R, Cechinel Filho V, Monache FD, Niero R (2016b) In vitro antiproliferative activity of uncommon xanthones from branches of Garcinia achachairu. Pharm Biol 54:1697–1704
Marques ES, Silva S, Niero R, Andrade SF, Rosa PSP, Perazzo FF, Maistro EL (2012) Genotoxy assessment of Gacinia achachairu (Clusiaceae) extract in mammalian cells in vivo. J Ethnopharmacol 142:362–366
Martins FT, Doriguetto AC, Souza TC, Souza KR, Dos Santos MH, Moreira ME, Barbosa LC (2008) Composition, and anti-inflammatory and antioxidant activities of the volatile oil from the fruit peel of Garcinia brasiliensis. Chem Biodivers 2:251–258
Mcdonald B, Pittman K, Menezes GB, Hirota SA, Slaba I, Waterhouse CC, Beck PL, Muruve DA, Kubes P (2010) Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science 330:362–366
Naegelen I, Beaume N, Plançon S, Schenten V, Tschirhart EJ, Bréchard S (2015) Regulation of neutrophil degranulation and cytokine secretion: a novel model approach based on linear fitting. J Immunol Res. https://doi.org/10.1155/2015/817038
Nelson RD, Quie PG, Simmons RL (1975) Chemotaxis under agarose: A new and simple method for measuring chemotaxis and spontaneous migration of human polymorphonuclear leukocytes and monocytes. J Immunol 115:1650–1656
Niero R, Dal Molin MM, Silva S, Damian NS, Maia LO, Monache FD, Chechinel Filho V, Andrade SF (2012) Gastroprotective effects of extracts and guttiferone A isolated from Garcinia achachairu Rusby (Clusiaceae) against experimentally induced gastric lesions in mice. Naunyn-Schmiedeberg’s Arch Pharmacol 385:1103–1109
Nozaki H, Suzuki H, Hirayama T, Kasai R, Wu RY, Lee KH (1986) Antitumour triterpenes of Maytenus diversifolia. Phytochemistry 25:479–485
Oh J, Rho HS, Yang Y, Yoon JY, Lee J, Hong YD, Kim HC, Choi SS, Kim TW, Shin SS, Cho JY (2013) Extracellular signal-regulated kinase is a direct target of the anti-inflammatory compound Amentoflavone derived from Torreya nucifera. Mediat Inflamm 2013:761506
Olaleye SB, Farombi EA, Adewoye BV, Owoyele SA, Onasanwo RA (2000) Analgesic and anti-inflammatory effects of kaviiron (a Garcinia kola seed extract). Afr J Biomed Res 3:171–174
Pantarelli C, Welch HCE (2018) Rac-GTPases and Rac-GEFs in neutrophil adhesion, migration. Eur J Clin Investig 48:e12939
Panthong A, Norkaew P, Kanjanapothi D, Taesotikul Anantachoke N, Reutrakul V (2007) Anti-inflammatory, analgesic and antipyretic activities of the extract of gamboge from Garcinia hanburyi Hook. J Ethnopharmacol 111:335–340
Patapoutian A, Tate S, Woolf CJ (2009) Transient receptor potential channels: targeting pain at the source. Nat Rev 8:55–68
Paulrayer A, Veeramuthu D, Savarimuthu I (2011) Anti-inflammatory, analgesic and antipyretic effects of friedelin isolated from Azima tetracantha Lam. in mouse and rat models. J Pharm Pharmacol 63:1070–1077
Quintão LM, Rocha AC, Medeiros R, Campos MM, Fernades ES, Calixto JB (2005) Effects of diacerhein on mechanical allodynia in inflammatory and neuropathic pain. Anesth Analg 101:1763–1769
Ren J, Li L, Wang Y, Zhai J, Chen G, Hu K (2019) Gambogic acid induces heme oxygenase-1 through Nrf2 signaling pathway and inhibits NF-κB and MAPK activation to reduce inflammation in LPS-activated RAW264.7 cells. Biomed Pharmacother 109:555–562
Ricaldez FT, Vega G, Almanza R (2000) Phytochemical study of Rheedia gardneriana. Rev Bol Quim 17:9–14
Savill J, Dransfield I, Gregory C, Haslett C (2002) A blast from the past: clearance of apoptotic cells regulates immune responses. Nat Rev Immunol 2:965–975
Sedgwick AD, Lees P (1986) Studies of eicosanoid production in the air pouch model of synovial inflammation. Agents Actions 18:429–438
Silva RL, Lopes AH, Guimarães RM, Cunha TM (2017) CXCL1/CXCR2 signaling in pathological pain: role in peripheral and central sensitization. Neurobiol Dis 105:109–116
Smith CW (2007) Adhesion molecules and receptors. J Allergy Clin Immunol 121:375–379
Sproston NR, Ashworth JJ (2018) Role of C-reactive protein at sites of inflammation and infection. Front Immunol 9:754
Terrazas PM, Marques ES, Mariano LNB, Cechinel-Filho V, Niero R, Andrade SF, Maistro ED (2013) Benzophenone guttiferone A from Garcinia achachairu Rusby (Clusiaceae) presents Genotoxic Effects in Different Cells of mice. PLoS ONE 11:e76485
Tian-Yong H, Jian-Ming J, Li-Hua M, Li M, Wen-Hui H, Rong-Rong Y, Xue-Qing C, Yan-Yan C, Zhi-Qiang L, Shu-Qi Q, Jun-Ting F, Bao-Hui C (2019) Anti-inflammation action of xanthones from Swertia chirayita by regulating COX-2/NF-κB/MAPks/Akt signaling pathways in RAW 264.7 macrophages cells. Phytomedicine 55:214–221
Tincani A, Adreoli L, Bazzani C, Bosisio D, Sozzani S (2007) Inflammatory molecules: a target for treatment of systemic autoimmune diseases. Autoimmun Rev 7:1–7
Torrico F, Velasco P, Giménez A et al (2008) Xantonas preniladas de Rheedia gardneriana y R. acuminata. In: Feliciano AS, Pérez AL, Del Olmo E. Manual de determinación estructural de compuestos naturales. Programa Iberoamericano de Ciencia y Tecnologia. https://iacobus.usc.es/record=b2169943~S1*gag. Accessed: 23 Mai 2019.
Tripathi P, Tripathi P, Kashyap L, Singh V (2007) The role of nitric oxide in inflammatory reactions. FEMS Immunol Med Microbiol 51:443–452
Tucureanu MM, Rebleanu D, Constantinescu CA, Deleanu M, Voicu G, Butoi E, Calin M, Manduteanu I (2017) Lipopolysaccharide-induced inflammation in monocytes/macrophages is blocked by liposomal delivery of Gi-protein inhibitor. Int J Nanomed 13:63–76
Uchimura K, Rosen SD (2006) Sulfateted I-selectin ligands as a therapeutic target in chronic inflammation. Trends Immunol 38:781–791
Williams MR, Azcutia V, Newton G, Alcaide P, Luscinskas FW (2011) Emerging mechanisms of neutrophil recruitment across endothelium. Cell press 32:461–469
Xing Z, Gauldie J, Cox G, Baumann H, Jordana M, Lei XF, Achong MK (1998) IL-6 is an anti-inflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest 101:311–312
Zhang Q, Raoof M, Chen Y, Sumi Y, Sursal T, Junger W, Brohi K, Itagaki K, Hauser CJ (2010) Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 464:104–107
Acknowledgements
This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, 429505/2018–3), the Coordenação de Aperfeiçoamento Pessoal de Nível Superior (CAPES) and the Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (FAPESC), R.N., L.M.B.N. and M.F.B. were MSc students in Pharmaceutical Science and are recipients of CAPES grants (Financial code 001) and R.D.F. was a graduate student in Biomedicine recipient of CNPq (PIBIC) grant (Financial code 001). N.L.M.Q., R.N. and V.C.F. are researchers granted by CNPq (Financial code 001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nunes, R., Broering, M.F., De Faveri, R. et al. Effect of the metanolic extract from the leaves of Garcinia humilis Vahl (Clusiaceae) on acute inflammation. Inflammopharmacol 29, 423–438 (2021). https://doi.org/10.1007/s10787-019-00645-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-019-00645-x