Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder characterized by intracellular neurofibrillary tangles and extracellular Aβ deposition. Growing experimental evidence indicate diverse biological effects of vitamin D3 including antioxidant, neuroprotective, anti-inflammatory and cardiovascular benefits. However, the underlying neuroprotective mechanism of vitamin D3 is still largely elusive. Therefore, the present study was aimed to investigate the neuroprotective effects of vitamin D3 on ICV-STZ induced sporadic AD. Our study demonstrated that vitamin D3 pretreatment significantly improved spatial learning and memory functions and effectively mitigated ICV-STZ mediated neuronal oxidative stress, mitochondrial aberrations and improved cholinergic functions. Moreover, vitamin D3 attenuated hippocampal neuroinflammatory response and reduced neuronal death in cortex and hippocampus. Our findings indicated that prophylactic vitamin D3 supplementation ameliorated ICV-STZ mediated neurobehavioral alterations, oxidative stress and neuroinflammation thereby improving cholinergic functions and reversed degenerative changes in brain. Thus, our study further provides evidence for its therapeutic supplementation for various neurodegenerative disorders including AD.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease is a multifactorial, age related progressive neurodegenerative disorder (Anand et al. 2014) typically characterized by pathological accumulation of tau protein, extracellular Aβ deposits, loss of neuronal synapses and pyramidal neurons (Kraska et al. 2012). Despite major advances in scientific research, the etiology and underlying mechanisms of Alzheimer’s disease remains yet to be elucidated.
Intracerebroventricular injection of sub-diabetogenic dose of streptozotocin (STZ) induces memory impairment, defects in brain insulin signaling and glucose metabolism, cholinergic deficits, oxidative stress and neuroinflammation culminating in pathological features of AD (Labak et al. 2010). Vitamin D is a fat-soluble vitamin that plays an important role in bone metabolism with distinct anti-inflammatory, anti-oxidant and immune-modulating properties. Vitamin D3 has recently gained attention for its widespread presence in various brain regions exerting neuroprotective effects and its deficiency is associated with various neurological disorders (Buell and Dawson-Hughes 2008; Keeney and Butterfield 2015), indicating an intimate relationship between vitamin D deficiency and impaired cognitive function (Becker et al. 2005; Shen and Ji 2015).
Currently, cholinomimetics as rivastigmine, galantamine and donepezil are the clinically used FDA approved drugs for the symptomatic treatment of AD, however, clinically effective therapeutic drugs for AD are still lacking. Nutritional factors play a central role in promoting health and clinical evidence establishes direct correlation between nutritional deficiencies and cognitive dysfunction (Buell and Dawson-Hughes 2008). These beneficial functions of vitamin D3 in cognitive dysfunction have led us to explore the possible neurotherapeutic potential of vitamin D3 in experimental ICV-STZ induced sporadic Alzheimer’s disease. However, the effect of vitamin D3 on ICV-STZ mediated neurobehavioral, oxidative stress, mitochondrial functions, histological alterations, neuroinflammation and cholinergic functions has not been explored yet. Therefore, with this background, the present study was planned to investigate the neuroprotective potential of Vitamin D3 in ICV-STZ model of sporadic Alzheimer’s disease.
Materials and methods
Experimental animals
Two months old adult male wistar rats (200–230 g) bred in central animal house facility of Punjab University were used. All animals were housed under standard laboratory conditions, maintained on a 12 h light/dark cycle and had free access to food (Ashirwad Industries, Mohali, India) and water. All experimental animals were acclimatized to laboratory conditions prior to neurobehavioural tests. All experiments were carried out between 09.00 and 17.00 h. The experimental protocols were approved by the Institutional Animal Ethics Committee (PU/IAEC/S/15/37) of Punjab University, Chandigarh, India and performed in accordance with the guidelines of Committee for Control and Supervision of Experimentation on Animals (CPCSEA), Government of India, on animal experimentation.
Drugs and treatment
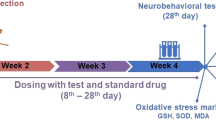
Vitamin D3 was procured from Hi-media, India. Streptozotocin was purchased from Sigma Aldrich, St. Louis, MO, USA. Rivastigmine was procured from Sun Pharmaceuticals Industries Ltd. Tumour Necrosis Factor-α (TNF-α) ELISA kit was purchased from Ray Biotech, USA, while NF-κB p65 ELISA kit was procured from QAYEE-BIO for Life Science, Shanghai. All other chemicals used were of analytical grade. Vitamin D3 solution was freshly prepared immediately prior to oral administration in tween 80 and diluted with distilled water. Streptozotocin was dissolved in artificial cerebrospinal fluid (aCSF) (2.9 mM KCl, 147 mM NaCl, 1.7 mM CaCl2, 1.6 mM MgCl2, and 2.2 mM d-glucose). In pre-treatment groups, vitamin D3 (42 IU) was administered 7 days before ICV-STZ administration and continued till end of the study, Post-treatment vitamin D3 (42 IU) and Rivastigmine (2 mg/kg) were administered by oral gavage once daily 24 h after ICV-STZ administration for 21 days (Fig. 1). The dose of vitamin D3 (Alrefaie and Alhayani 2015; Briones and Darwish 2014) and rivastigmine (Sachdeva and Chopra 2015) were selected based on previously published literature.
Surgical induction of sporadic Alzheimer’s disease using ICV-STZ injection
Intracerebroventricular streptozotocin injection procedure was made according to Sonkusare et al. (2005). Rats were anesthetized with chloral hydrate (TCI, India, 350 mg/kg, i.p.). Briefly, the rat scalp was shaved, and positioned in a stereotaxic apparatus and a midline sagittal incision was made in the scalp. Burr holes were drilled in the skull on both sides over the lateral ventricles using the following coordinates: 0.8 mm posterior to bregma; 1.5 mm lateral to sagittal suture and 3.6 mm beneath the cortical surface of the brain (Paxinos et al. 1980). STZ (3 mg/kg) was prepared freshly by dissolving in aCSF and injected bilaterally in two divided doses on first and third day 1.5 mg/kg (3 μl) each day. The wound was sutured followed by topical application of antiseptic powder (Neosporin®). Control animals received ICV injection of the same volume of aCSF on the first and third day. Postoperatively, the rats were fed with glucose water and normal pellet diet for 4 days, followed by normal pellet diet alone.
Neurobehavioural evaluation
Morris water maze (MWM)
Morris water maze (MWM) was used to assess the learning and memory functions in rat according to Sachdeva and Chopra (2015). Briefly, the water-maze apparatus consisted of a circular pool of 151 cm diameter and 60 cm high, which was divided factitiously into four equal quadrants (1st, 2nd, 3rd and 4th). The pool was filled with water (25 ± 1 °C). A platform (12.5 cm in diameter and 38 cm high) was placed at a constant position in the middle of one quadrant, 2 cm below the surface of water, equidistant from the centre and edge of the pool. The pool was placed in an illuminated room with some external cues, which remained in the same location throughout the training and testing period. On day 16, each animal was subjected to four training trials per day for a period of 5 days with an inter-trial interval of 15 min. In each acquisition trial session, rats were individually placed in the pool facing the wall at one point randomly selected from four designated starting points. The central platform was placed in 4th quadrant for all session, during trial session. Mean escape latency time to locate the hidden platform in water maze was recorded as an index of acquisition or learning. During training sessions, rats were allowed to swim until they found the hidden platform and if they could not find the platform within 90 s, they were gently guided to the platform by the experimenter. Rats were allowed to remain on the platform for 15 s before being removed. Twenty four hours after the fifth day trial, animals were subjected to probe trial for 90 s by removing the platform from the pool. During 5 day training trials, mean escape latency (MEL) and path length to reach the platform were recorded. The average time spent in the target quadrant (TSTQ) and numbers of entries in the target quadrant were recorded during probe trial.
Elevated plus maze task (EPM)
Memory acquisition and retention were tested using elevated plus maze test on day 20 and 21 according to Misra et al. (2011). The elevated plus maze consisted of two opposite open arms (50 × 10 cm), crossed with two closed arms of the same dimensions with 40 cm high walls. The arms were connected with a central square of dimensions 10 × 10 cm; the entire maze was elevated to a height of 50 cm from the floor. Each rat was placed on the end of the open arm, facing outwards. The time taken by the rat to enter the closed arm in the first trial (acquisition trial) on day 20 was noted and was called as initial transfer latency (ITL). Cutoff time was fixed at 90 s, and in case a rat could not find the closed arm within this period, it was gently pushed into one of the closed arms and allowed to explore the maze for 30 s. Second trial (retention trial) was performed 24 h after the acquisition trial (day 21), and retention transfer latency (RTL) was noted.
Biochemical estimations
Brain homogenate preparation
After behavioral experiments, the rats were sacrificed, hippocampus and cortex were isolated, weighed and immediately homogenized in homogenization buffer (1% IGEPAL CA 630, 10 mM Tris and 150 mM MgCl2, 1 mM EDTA, 1 mM EGTA) containing complete protease inhibitor cocktail (Sigma, USA). The homogenate was centrifuged at 4000g for 20 min at 4 °C. The supernatant thus obtained, was used for estimation of acetylcholinesterase activity, lipid peroxidation, reduced glutathione, nitrite, catalase and superoxide dismutase activity.
Determination of lipid peroxidation
Malondialdehyde (MDA) levels were measured to assess lipid peroxidation in hippocampal and cortex homogenates as according to Wills (1966). Briefly, 0.5 ml of tissue homogenate and 0.5 ml of Tris–HCl were incubated at 37 °C for 2 h, after incubation, 1 ml of 10% trichloroacetic acid was added and centrifuged at 1000g for 10 min. To 1 ml of supernatant, 1 ml of 0.67% thiobarbituric acid was added, and the tubes were kept in boiling water for 10 min. After cooling, 1 ml double-distilled water was added, and absorbance was measured at 532 nm using UV–Vis spectrophotometer (Parkin Elmer, USA). The results were calculated using the molar extinction coefficient as 1.56 × 105 M−1 cm−1 and expressed as n moles per mg protein of MDA.
Determination of reduced glutathione
Reduced glutathione (GSH) was estimated according to Jollow et al. (1974). Briefly, 1.0 ml of tissue homogenate was precipitated with 1.0 ml of sulfosalicylic acid (4%). The mixture were kept at 4 °C for at least 1 h and then centrifuged at 1200g for 15 min at 4 °C. To 0.1 ml of supernatant, 2.7 ml of PBS (0.1 mmol/l, pH 8.0), and 0.2 ml of 0.01 M (5,5′ dithio-bis (2-nitro benzoic acid), DTNB) were added. The yellow colour thus developed was measured with UV–Vis spectrophotometer at 412 nm. Results were calculated using the molar extinction coefficient of chromophore as 1.36 × 104 M−1 cm−1 and expressed as µ moles of GSH per mg protein.
Determination of acetylcholinesterase activity
Acetylcholinesterase is the enzyme which catalyzes the breakdown of acetylcholine, marker of cholinergic neurons in the brain. It was assessed in hippocampal and cortical regions according to Ellman et al. (1961). The assay mixture contained 50 µl of supernatant, 3 ml of sodium phosphate buffer (pH 8), 0.1 ml of acetylthiocholine iodide, and 0.1 ml of DTNB (Ellman’s reagent). The change in absorbance was measured for 2 min at 30 s interval at 412 nm using a UV–Vis spectrophotometer. Results were calculated using molar extinction coefficient of chromophore as 1.36 × 104 M−1 cm−1 and expressed as μM of acetylthiocholine iodide hydrolyzed per min per mg of protein.
Determination of nitrite levels
The nitric oxide is short-lived so, spontaneously oxidizes to nitrite and nitrate. The plasma nitrite levels were estimated according to Green et al. (1982) and Ray et al. (2014). Equal volumes of supernatant and Greiss reagent (equal amount of 0.1% naphthyl ethylenediamine dihydrochloride and 1% sulphanilamide in 5% phosphoric acid) were mixed, and the mixture was incubated in the dark for 10 min at room temperature. Absorbance was recorded at 540 nm with a UV–Vis spectrophotometer. The nitrite concentration was calculated from a standard curve using sodium nitrite as standard and expressed as μM per ml.
Determination of superoxide dismutase
Superoxide dismutase (SOD) activity was assayed according to Kono (1978). Briefly, the assay procedure consisted of 0.1 mM EDTA, 50 mM sodium carbonate and 96 mM of nitro blue tetrazolium. In the cuvette, 2 ml of above mixture was taken, and to it, 50 µl of homogenate and 50 µl of hydroxylamine hydrochloride (pH 6.0) were added. The auto-oxidation of hydroxylamine was observed by measuring the change in optical density at 560 nm using a UV–Vis spectrophotometer for 2 min at 30 s intervals and expressed as SOD units per mg protein.
Determination of catalase
Catalase activity was assayed according to Claiborne (1985). Briefly, the assay mixture consisted of 1.95 ml phosphate buffer (0.05 M, pH 7.0), 1.0 ml hydrogen peroxide (0.019 M) and 50 µl tissue homogenate (10%) in a final volume of 3.0 ml. Changes in absorbance were recorded at 240 nm with a UV–Vis spectrophotometer. The results were obtained as micromoles of hydrogen peroxides decomposed per mg of protein/min.
Protein estimation
Protein estimation was done by biuret method using bovine serum albumin (BSA) as standard (Gornall et al. 1949).
Mitochondrial estimation
Isolation of mitochondria
Mitochondria were isolated as described by Berman and Hastings (1999). Briefly, cerebral cortex and hippocampus region were dissected from rat brain and homogenized in ice-cold isolation buffer (215 mM mannitol, 75 mM sucrose, 0.1% BSA, 20 mM HEPES, 1 mM EGTA, and pH 7.2) with EGTA [Ethylene glycol-bis (β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid]. Homogenates were centrifuged at 1000g for 5 min at 4 °C. Pellets were resuspended in isolation buffer with EGTA and spun again at 13,000g for 5 min 4 °C. The resulting supernatants were transferred and topped off with isolation buffer with EGTA and again spun at 13,000g for 10 min 4 °C. Pellets were resuspended in isolation buffer with EGTA and spun again at 13,000g for 5 min 4 °C. It was incubated with 1 ml with digitonin for 10 min and again spun at 13,000g for 10 min at 4 °C. Pellets containing pure mitochondria were resuspended in isolation buffer without EGTA.
NADH dehydrogenase activity (complex I)
Nicotinamide adenine dinucleoside (NADH) dehydrogenase activity was assessed in cerebral cortex and hippocampus according to King and Howard (1967). The method involves catalytic oxidation of NADH to NAD+ with subsequent reduction of cytochrome C. Briefly, the reaction mixture contained 0.2 M glycyl glycine buffer, pH 8.5, 6 mM NADH in 2 mM glycyl glycine buffer, and 10.5 mM cytochrome C. The reaction was initiated by the addition of required amount of solubilized mitochondrial sample, and the absorbance change at 550 nm using UV–Vis spectrophotometer was recorded over the period of 3 min at 1 min interval and the results were expressed as n moles of NADH oxidized/min/mg protein.
Succinate dehydrogenase activity (complex II)
Cortical and hippocampal succinate dehydrogenase activity was measured UV–Vis spectrophotometrically according to king (King and Howard 1967). The method involves oxidation of succinate by an artificial electron acceptor, potassium ferricyanide. The reaction mixture consisted of 0.2 M phosphate buffer, pH 7.8, 1% BSA, 0.6 M succinic acid, and 0.03 M potassium ferricyanide. The reaction was initiated by the addition of mitochondrial sample. The change in absorbance was followed at 420 nm for 3 min at 1 min interval and the results were expressed as n moles of substrate/min/mg protein.
Methylthiazolyldiphenyl-tetrazolium bromide (MTT) ability (complex III)
This is an indirect method to measure the activity of the complex-III. MTT, a pale yellow substrate, produces a purple product when incubated with mitochondrial sample, and the number of viable cells/well that is directly proportional to the production of the product, which follows solubilization with dimethyl sulfoxide (DMSO), can be measured according to (Mosmann 1983). In general, each well is incubated in 10 μl of MTT and incubates the plate in a humidified atmosphere (5% CO2 95% air) at 37 °C for 3 h. The medium was then aspirated off, and lysed with 50% DMSO. The absorbance of the resulting medium was measured using ELISA reader at 580 nm wavelength.
Cytochrome oxidase assay (complex IV)
Cytochrome oxidase activity was assayed in both cortical and hippocampal mitochondrial sample according to Sottocasa et al. (1967). The assay mixture contained 0.3 mM reduced cytochrome C in 75 mM phosphate buffer, and the reaction was started by the addition of mitochondrial sample. The change in absorbance was recorded at 550 nm over the period of 3 min at 1 min interval and the results were expressed as µ moles of Cyt C reductase/min/mg protein.
Estimation of TNF-α and NF-κB levels
The levels of TNF-α and NF-κB in hippocampal tissue homogenate were measured by enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions. The quantity of TNF-α and NF-κB present in the hippocampal were determined using their respective standard curve. Values were expressed as mean ± SEM. TNF-α and NF-κB is expressed as pg/mg protein and ng/mg protein, respectively.
Hematoxylin and eosin staining
At the end of experiment, randomly 2 animals were chosen from each group. The brains were rapidly excised and fixed by immersion in 10% formalin. Subsequently, they were embedded in paraffin wax. Both hippocampal and cortical paraffin (thickness 7 μm) were dewaxed and hydrated using different gradient of alcohol for hematoxylin–eosin (H&E) staining. The morphology of the pyramidal neurons in CA1 and CA3 region of hippocampus and cortex was examined under light microscopy.
Statistical analysis
Results were expressed as mean ± SEM. The intergroup variation was expressed by one-way analysis of variance (ANOVA) followed by Tukey’s test in all the parameters to assess the significance. However, two-way ANOVA followed by Bonferroni’s test was employed to analyze the learning performance for 5 days in MWM trial in MEL and pathlength. In all the test, p < 0.05 was taken as criterion for statistical significance. Prism Graphpad 5.0 (Graphpad Software Inc, Ca, USA) was used to analyze statistics.
Results
Neurobehavioural observations
Effect of vitamin D3 on cognitive function of rats with ICV-STZ in Morris water maze task
Mean escape latency
The effects of vitamin D3 on ICV-STZ induced spatial memory functions were studied using MWM task. The mean escape latencies of ICV-STZ induced Alzheimer’s animals were significantly increased on day 21 (p < 0.01) compared to sham animals. We observed a significant decrease in mean escape latency in vitamin D3 pre-treated animals on day 21 (p < 0.01) compared to ICV-STZ administered animals. Although, post-treatment vitamin D3 exhibited a marginal decrease in mean escape latency on day 21, the declined escape latency was unable to produce statistical significance compared to ICV-STZ animals. Furthermore, rivastigmine administration produced progressive decrease in mean escape latency indicating significant amelioration of impaired memory functions compared to ICV-STZ animals on day 21 (p < 0.001) (Fig. 2a).
Effects of vitamin D3 on spatial learning and memory function in Morris water maze and elevated pluz maze. a Mean escape latency, b pathlength, c representative Morris water maze tracking memory performances, d time spent in target quadrant, e number of entries in target quadrant, f % initial transfer latency in elevated pluz maze (n = 7–8). Values were expressed as mean ± SEM. Mean escape latency and pathlength are expressed by two-way ANOVA followed by Bonferroni’s test. TSTQ, number of entries into the target quadrant and % initial transfer latency are expressed by one-way ANOVA followed by Tukey’s test. ### p < 0.001, ## p < 0.01, # p < 0.05—sham versus ICV-STZ; **p < 0.01, *p < 0.05—ICV-STZ versus vitamin D3 pre-treatment; % p < 0.05—vitamin D3 pre-treatment versus vitamin D3 post-treatment; $$$ p < 0.001, $$ p < 0.01, $ p < 0.05—ICV-STZ versus rivastigmine (colour figure online)
Path length
Intracerebroventricular STZ injection displayed significant increase in path length on day 21 (p < 0.001) in MWM compared to sham operated animals. Pre-treatment with vitamin D3 elicited a significant decrease in path length (p < 0.01) compared to ICV-STZ administered animals whereas post-treatment vitamin D3 failed to show any significant improvement in memory functions compared with ICV-STZ animals. Further, rivastigmine treated animals displayed significant decrease in path length of acquisition trial compared to ICV-STZ animals, indicating of improvement in memory performances (p < 0.01) (Fig. 2b).
Time spent in target quadrant (TSTQ)
In probe trial, time spent in the target quadrant is a putative measure of spatial learning and memory performance. ICV-STZ animals spent considerably lesser duration in the target quadrant compared to sham animals (p < 0.001). Besides, vitamin D3 pre-treated animals spent significantly increased time in the target quadrant compared to ICV-STZ administered animals (p < 0.01). Conversely, post-treated vitamin D3 animals failed to exhibit any significant improvement in time spent in target quadrant compared to ICV-STZ administered animals. Treatment with rivastigmine demonstrated significant increase in TSTQ compared to ICV-STZ administered animals (p < 0.05) (Fig. 2d).
Number of entries in target quadrant
During probe trial, number of entries into target quadrant was evaluated as a measure of memory performances. Intracerebroventricular injection of sub-diabetogenic streptozotocin exhibited significantly decreased number of entries into the target quadrant compared to sham operated animals (p < 0.05). Further, vitamin D3 pre-treatment revealed significant increase in the number of entries into the target quadrant compared to ICV-STZ animals (p < 0.01). Moreover, we did not observe any significant change in number of entries into target quadrant in post-treated vitamin D3 animals compared to ICV-STZ animals. Moreover, Rivastigmine treatment exhibited substantial increase in the number of entries compared to ICV-STZ animals implying improved memory performances (p < 0.05) (Fig. 2E).
Effect of vitamin D3 on spatial memory in elevated plus maze test
Elevated plus maze task was performed to ascertain the influence of vitamin D3 on spatial memory performances. In EPM paradigm, the time taken by the rat to move from the open arm to the enclosed arm (transfer latency, TL) was taken as an index of memory retention. Retention transfer latency observed on day 21 indicates sham animals exhibited improved spatial memory than ICV-STZ injected animals (p < 0.01). The pre-treated with vitamin D3 and rivastigmine significantly improved spatial memory functions (p < 0.01) in EPM compared to ICV-STZ animals. Furthermore, the transfer latencies in vitamin D3 post-treatment animals did not show any significant improvement compared to ICV-STZ animals (Fig. 2f).
Biochemical observations
Effect of vitamin D3 on SOD levels in cortex and hippocampus tissue
Superoxide dismutase levels in ICV-STZ animals were significantly decreased in cortex (p < 0.01) and hippocampus (p < 0.001) compared to sham operated animals. Pre-treatment with vitamin D3 significantly restored the ICV-STZ induced decrease in SOD levels in cortex (p < 0.05) and hippocampus (p < 0.01) compared to ICV-STZ animals. Nevertheless, post-treatment failed to rescue the declined SOD level in cortex and hippocampus indicative of increased brain oxidative stress. Additionally, we observed a significant difference in hippocampal SOD levels between vitamin D3 pre-treatment and post-treatment (p < 0.05), but not in cortex. Rivastigmine treatment demonstrated significant restoration of hippocampal and cortical SOD levels compared to ICV-STZ animals rescuing the brain cells from oxidative stress (p < 0.05) (Fig. 3a, b).
Effects of vitamin D3 on SOD, catalase and GSH levels in ICV-STZ rats in cortex and hippocampus. a, b SOD units, c, d catalase activity and e, f GSH levels. Values were expressed as mean ± SEM. ### p < 0.001, ## p < 0.01—sham versus ICV-STZ; **p < 0.01, *p < 0.05—ICV-STZ versus vitamin D3 pre-treatment; % p < 0.05—Vitamin D3 pre-treatment versus vitamin D3 post-treatment; $$ p < 0.01, $ p < 0.05—ICV-STZ versus rivastigmine
Effect of vitamin D3 on catalase levels in cortex and hippocampus tissue
Intracerebroventricular injection of STZ resulted in significantly decreased catalase activity in cortex (p < 0.001) and hippocampus (p < 0.001) compared to sham animals. Pre-treatment with vitamin D3 significantly reversed the declined catalase activity in cortex (p < 0.01) and hippocampus (p < 0.05) compared to ICV-STZ animals. However, vitamin D3 post-treatment failed to restore the catalase activity in cortex and hippocampus. Moreover, rivastigmine treatment effectively restored catalase activity in cortex (p < 0.01) and hippocampus (p < 0.01) indicating improved endogenous anti-oxidant defense (Fig. 3c, d).
Effect of vitamin D3 on GSH levels in cortex and hippocampus
Reduced glutathione levels were found to be significantly decreased in ICV-STZ animals compared to sham animals in both cortex (p < 0.001) and hippocampus (p < 0.01). Pre-treatment with vitamin D3 demonstrate significant restoration of the decreased GSH levels in cortex (p < 0.01) and hippocampus (p < 0.01) indicating its potent anti-oxidant potential. Furthermore, post-treatment with vitamin D3 failed to demonstrate any significant change in GSH levels compared to ICV-STZ animals. Furthermore, treatment with rivastigmine displayed comparable increase in GSH level in cortex (p < 0.01) and hippocampus (p < 0.05) with pre-treatment animals (Fig. 3e, f).
Effect of vitamin D3 on ICV-STZ induced lipid peroxidation in cortex and hippocampus tissue
Elevated levels of malondialdehyde (MDA) in brain tissue measures the extent of lipid peroxidative damage owing to cellular oxidative stress. Malondialdehyde levels were significantly increased in both cortex (p < 0.01) and hippocampus (p < 0.01) in ICV-STZ animals as compared to sham animals. Pre-treatment with vitamin D3 significantly attenuated the elevated MDA level in cortex (p < 0.01) and hippocampus (p < 0.05) compared to ICV-STZ animals. Also, post-treated vitamin D3 animals also exhibited decreased MDA levels in cortex (p < 0.05) but not in hippocampus. Moreover, rivastigmine treatment significantly decreased MDA levels in both cortex (p < 0.01) and hippocampus (p < 0.01) indicating amelioration of cellular oxidative stress (Fig. 4a, b).
Effects of vitamin D3 on MDA levels in a cortex, b hippocampus and c plasma nitrite levels in ICV-STZ rats. Values were expressed as mean ± SEM. ### p < 0.001, ## p < 0.01—sham versus ICV-STZ; **p < 0.01, *p < 0.05 —ICV-STZ versus vitamin D3 pre-treatment; @ p < 0.05—ICV-STZ versus vitamin D3 post-treatment; $$ p < 0.01, $ p < 0.05—ICV-STZ versus rivastigmine
Effect of vitamin D3 on plasma nitric oxide levels
Nitric oxide is a short-lived molecule and there is a marked release of NO in the injured brain leading to nitrative stress. The involvement of NO in ICV-STZ induced AD followed by vitamin D3 administration was studied using Griess assay. In the present study, a significantly elevated plasma nitrite levels were observed in ICV-STZ animals compared to sham operated animals (p < 0.001). Pre-treatment with vitamin D3 significantly reversed the elevated plasma nitrite level (p < 0.01) compared to ICV-STZ animals indicating reduced nitrative stress. Post-treated vitamin D3 animals also demonstrated decreased nitrite levels, but did not achieve statistical significance compared to ICV-STZ animals. Moreover, rivastigmine administration led to significant decrease in plasma nitrite levels compared to ICV-STZ animals (p < 0.01) indicating reduced nitrative stress (Fig. 4c).
Effect of vitamin D3 on cortical and hippocampal acetylcholinesterase activity in ICV-STZ induced rats
The acetylcholine levels in the hippocampal synapse are maintained primarily by acetylcholinesterase activity. Acetylcholinesterase plays an important role in modulating cognitive functions through catalytic breakdown of acetylcholine. Increased AChE activity leads to decrease in functional ACh levels at the synapse and is widely implicated in various neurodegenerative disorders such as AD. Acetylcholinesterase activity was significantly increased in cortex and hippocampus (p < 0.01) of ICV-STZ animals compared to sham operated animals. Further, pre-treatment with vitamin D3 significantly attenuated the elevated acetylcholinesterase activity in cortex (p < 0.01) and hippocampus (p < 0.05) compared to ICV-STZ animals. Additionally, vitamin D3 post-treatment demonstrated marginal decrease in AChE activity in cortex (p < 0.05), but not in hippocampus. Furthermore, rivastigmine treated animals caused significant decrease in acetylcholinesterase levels in cortex (p < 0.01) and hippocampus (p < 0.05) compared to ICV-STZ animals (Fig. 5a, b).
Effects of vitamin D3 on AChE levels in cortex (a) and hippocampus (b) in ICV-STZ rats. Values were expressed as mean ± SEM. ## p < 0.01—sham animals versus ICV-STZ; **p < 0.01, *p < 0.05—ICV-STZ versus vitamin D3 pre-treatment; @ p < 0.05—ICV-STZ versus vitamin D3 post-treatment; $$ p < 0.01, $ p < 0.05—ICV-STZ versus rivastigmine
Mitochondrial estimations
Effect of vitamin D3 on mitochondrial enzyme complex I activity
Mitochondrial complex I activity was found to be drastically reduced in ICV-STZ animals compared to sham animals in both cortex (p < 0.05) and hippocampus (p < 0.01). In the present study, vitamin D3 pre-treatment significantly restored the decreased mitochondrial complex I activity in both hippocampus and cortex (p < 0.01) compared to ICV-STZ animals. Vitamin D3 post-treatment also displayed a marginal increase in mitochondrial complex I activity, however, the increasing trend did not achieve statistical significance compared to ICV-STZ animals. Additionally, we observed a significant difference between post-treatment and pre-treatment vitamin D3 in cortical mitochondrial complex I activity (p < 0.05), but not in hippocampus. Furthermore, rivastigmine treated animals exhibited significant increase in mitochondrial complex I activity, compared to ICV-STZ animals in cortex and hippocampus (p < 0.05) (Fig. 6a, b).
Effects of vitamin D3 on mitochondrial complex in hippocampus and cortex of ICV-STZ rats. Mitochondrial complex I (a, b), complex II (c, d), complex III (e, f), complex IV (g, h). Values were expressed as mean ± SEM. ## p < 0.01, # p < 0.05—sham animals versus ICV-STZ; **p < 0.01, *p < 0.05—ICV-STZ versus pre-treatment vitamin D3; @ p < 0.05—ICV-STZ versus post-treatment vitamin D3; % p < 0.05—pre-treatment vitamin D3 versus post-treatment vitamin D3 animals; $$ p < 0.01, $ p < 0.05—ICV-STZ versus rivastigmine. (one-way ANOVA, post hoc Tukey’s test)
Effect of vitamin D3 on mitochondrial enzyme complex II activity
Mitochondrial complex II levels were significantly decreased in ICV-STZ animals compared to sham animals in both cortex and hippocampus (p < 0.01). In the present study, we observed that vitamin D3 pre-treatment significantly increased mitochondrial complex II activity in both hippocampus and cortex (p < 0.05) compared to ICV-STZ animals. Post-treatment vitamin D3 exhibited an increasing pattern of mitochondrial complex II, but failed to display any significance compared to ICV-STZ animals. Furthermore, rivastigmine treated animals also demonstrated significant increase in mitochondrial complex II levels in both cortex and hippocampus (p < 0.01) (Fig. 6c, d).
Effect of vitamin D3 on mitochondrial enzyme complex III activity
Cortical (p < 0.01) and hippocampal (p < 0.05) mitochondrial complex III activity were significantly decreased in ICV-STZ animals compared to sham operated animals. In the present study, vitamin D3 pre-treatment demonstrated significant improvement in mitochondrial complex III activity in hippocampus (p < 0.05) and cortex (p < 0.01) compared to ICV-STZ animals. Moreover, vitamin D3 post-treatment revealed increase in hippocampal mitochondrial complex III activity (p < 0.01), but without changes in cortex compared to ICV-STZ animals. Furthermore, rivastigmine treated animals demonstrated significant increase in mitochondrial complex III activity compared to ICV-STZ animals in both cortex (p < 0.05) and hippocampus (p < 0.05) (Fig. 6e, f).
Effect of vitamin D3 on mitochondrial enzyme complex IV activity
Mitochondrial complex IV activity was decreased in ICV-STZ animals as compared to sham animals in both cortex (p < 0.1) and hippocampus (p < 0.05). Pre-treatment with vitamin D3 showed significantly increased mitochondria complex IV activity in hippocampus (p < 0.05) and cortex (p < 0.01) compared with ICV-STZ animals. Post-treatment vitamin D3 marginally increased the mitochondrial complex IV levels, but did not show significant increase compared to ICV-STZ animals. There was a significant difference between post-treatment vitamin D3 and pre-treatment vitamin D3 (p < 0.05) in both hippocampus and cortex. Similarly, rivastigmine treatment also demonstrated significant increase in mitochondrial complex IV activity in both cortex (p < 0.01) and hippocampus (p < 0.05) compared to ICV-STZ animals (Fig. 6g, h).
Neuroinflammation studies
Effect of vitamin D3 on NF-κB in hippocampus
Hippocampal NF-κB levels were significantly increased in ICV-STZ administered animals compared to sham animals (p < 0.001). Pre-treatment with vitamin D3 significantly attenuated the elevated NF-κB p65 subunit expression in the nuclear fraction compared to ICV-STZ animals (p < 0.01). Post-treatment with vitamin D3 animals failed to attenuate the increased levels of NF-κB in hippocampus. Rivastigmine treatment also showed significantly decreased NF-κB levels compared to ICV-STZ levels (p < 0.001) (Fig. 7a).
Effects of vitamin D3 on NFκβ (a) and TNF-α (b) levels in hippocampus of ICV-STZ rats. Values were expressed as mean ± SEM. ### p < 0.001—sham versus ICV-STZ; ***p < 0.001, **p < 0.01—ICV-STZ versus pre-treatment vitamin D3; @ p < 0.05—ICV-STZ versus post-treatment vitamin D3; %%% p < 0.001, % p < 0.05—pre-treatment vitamin D3 versus post-treatment vitamin D3 animals; $$$ p < 0.001—ICV-STZ versus rivastigmine. (one-way ANOVA, post hoc Tukey’s test)
Effect of vitamin D3 on TNF-α in hippocampus
TNF-α levels were significantly increased in hippocampus homogenate of ICV-STZ administered rats compared to sham animals (p < 0.001) suggesting involvement of neuroinflammatory component in ICV-STZ induced AD. Pre-treatment with vitamin D3 significantly attenuated the rise in TNF-α levels compared to ICV-STZ rats (p < 0.001). Post-treatment with vitamin D3 showed slight decrease in TNF-α levels compared to ICV-STZ rats (p < 0.05). Additionally, rivastigmine significantly reduced the TNF-α levels compared to ICV-STZ rats (p < 0.001). Furthermore, we observed significant difference between pre-treatment and post-treatment with vitamin D3 (p < 0.001) (Fig. 7b).
Effects of vitamin D3 on histopathological analysis
Histopathological analysis of hippocampii of sham animals showed more number of optimum sized, undamaged neuronal cells and cell layers. However, neuroinflammation and increased number of apoptotic cells were observed in the CA1 and CA3 region of the hippocampus in ICV-STZ-injected rats, indicating significant neurodegeneration. Pre-treatment with vitamin D3 caused significant attenuation of histological and morphological changes along with significantly lesser number of apoptotic bodies in hippocampal CA1 and CA3 region compared to ICV-STZ administered group. Post-treatment with vitamin D3 treatment did not result in significant improvement changes in morphological and histological structures demonstrating comparable histopathological characteristics as ICV-STZ administered rats. Rivastigmine significantly attenuated the ICV-STZ induced cell loss and pyknotic cells, but few degenerating cells with changes of morphology were observed.
The neurons in the sham group were large, conical shaped cells with well-delineated amphophilic cytoplasm and round vesicular nuclei with prominent nucleoli in CA1 hippocampal region. However, in ICV-STZ treated animals, the CA1 layer neurons showed pronounced shrinkage of the neuronal bodies with the nuclei losing their regular outlines and becoming hyperchromatic. The shrinkage of the cells also caused a clear space (vacuoles) to form around each neuron. Vitamin D3 pretreated animals exhibited partial to full recovery comparable to the ICV-STZ animals.
Moreover, neuronal cells in cerebral cortex have prominent nucleolus typical to large neurons in sham group, smaller nuclei may be those of smaller neurons or of associated glial cells in sham animals. ICV-STZ animals demonstrate severe neuronal death and change in cell shape as been observed which was significantly reversed in pre-treatment vitamin D3 and rivastigmine administered group. However, vitamin D3 post-treatment showed neuronal death in cortical region, resembling ICV-STZ animals (Fig. 8a–c).
Discussion
In the present study, we evaluated the neuroprotective potential of vitamin D3 supplementation in rat model of ICV-STZ induced neurobehavioral, mitochondrial, histopathological alterations and cholinergic functions. Besides alteration in cerebral glucose and energy metabolism, intracerebroventricular injection of STZ, a commonly used experimental tool to study sporadic AD, induces neuroinflammation reproducing classical features of AD (Sharma et al. 2012). Therefore, we evaluated the effect of vitamin D3 (42 IU/day) using two treatment modalities, i.e., pre-treatment and post-treatment compared with standard drug rivastigmine.
Intracerebroventricular administration of STZ significantly impaired learning and memory performance and exhibited a higher score of errors and took considerably longer duration of time to reach the target platform during training, indicating impaired spatial learning. The progressive increase in mean escape latency and swim path length to find the hidden platform during training trials along with decreased number of entries and time spent in the target quadrant corroborates the impaired retention memory elicited by ICV-STZ rats during spatial navigating task in MWM. Vitamin D3 pre-treatment (42 IU/day), significantly decreased the mean escape latency and found the hidden platform sooner than vitamin D3 post-treatment animals and ICV-STZ animals. Our results along with previous reports revealed that vitamin D3 treatment improved spatial learning, and memory functions in MWM (Latimer et al. 2014; Mohamed et al. 2015). Further, vitamin D3 pre-treatment also significantly shortened the transfer latency into the enclosed arm, eliciting improved retention memory compared to ICV-STZ animals and vitamin D3 post-treatment in elevated plus maze. Although, both pre-treatment and post-treatment strategies elicited improved spatial learning, vitamin D3 pre-treatment evidenced prominent superior efficacy in attenuating ICV-STZ induced neuronal damage and behavioral dysfunction.
Brain is essentially one of the most metabolically active organs and owing to its limited availability of endogenous defense systems, it is extremely prone to free radical induced oxidative tissue damage (Butterfield 2006; Markesbery 1997; Wang and Michaelis 2010). Brain oxidative stress may be effectively countered through endogenous antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) (Gandhi and Abramov 2012). An increased neuronal oxidative stress trigger neuronal damage affecting major cellular biomolecules (lipids, proteins, DNA and RNA) and plays a crucial role in the development and progression of neurodegenerative disorders such as AD (Javed et al. 2011). In the present study, vitamin D3 pretreatment significantly rescued the ICV-STZ induced oxidative trigger and defective endogenous anti-oxidant defense system back to near basal levels. Thus, our results are concordant with an earlier report, which demonstrate restoration of depleted endogenous anti-oxidant defense system, depicting the anti-oxidant potential of vitamin D3 in attenuating oxidative damage (Kuzmenko et al. 1997).
Increased neuronal oxidative stress worsens peroxidative damage of membrane lipids through MDA generation, which in turn amplifies neuronal damage severely affecting key cellular biomolecules and is commonly used as biomarkers of oxidative damage. In the present study, overproduction of free radical triggered MDA mediated hippocampal and cortical oxidative damage. Our findings corroborate with earlier experimental evidences suggesting defective anti-oxidant defense system potentiating oxidative damage (Javed et al. 2012; Rai et al. 2014; Sharma and Gupta 2001).
Furthermore, ICV-STZ administration elevated plasma total nitric oxide levels fuelling oxidative-nitrative damage, which provoked peroxynitrite arbitrated neuronal damage and compromised normal brain functions. Moreover, vitamin D3 administration mitigated iNOS expression, limiting NO mediated AD pathogenesis (Dulla et al. 2016; Garcion et al. 1997). Besides, NO and MDA generation, intracerebroventricular STZ displayed mitochondrial aberrations, perturbing cortical and hippocampal mitochondrial complex (I, II, III and IV) activities triggering neuronal apoptosis (Correia et al. 2013; Lin and Beal 2006; Paidi et al. 2015; Witte et al. 2010). Further, vitamin D3 pre-treatment effectively ameliorated ICV-STZ mediated oxidative–nitrative damage and restored mitochondrial complex activities, further substantiating the neuroprotective potential of vitamin D3 (Longoni et al. 2016). Therefore, it is imperative that the anti-oxidant properties of vitamin D3 are critical for attenuating ICV-STZ induced mitochondrial aberrations, neurotoxicity and cognitive impairment.
Hippocampal and cortical neurons play a crucial role in modulating AD pathology, often characterized by marked neuronal damage to these brain structures (Tiwari et al. 2009). Histopathological evidence suggests that ICV-STZ promote structural and functional damage to cortical and hippocampal structures along with β-amyloid peptide-like aggregates in brain capillaries (Salkovic-Petrisic et al. 2006; Shoham et al. 2003). In the present study, we observed marked morphological changes in the hippocampal CA1 and CA3 region and cortex of ICV-STZ induced AD animals. Pre-treatment with vitamin D3 attenuated ICV-STZ induced morphological alterations in the hippocampal CA1, CA3 and cortical regions displaying functional neurotherapeutic benefits further substantiating neurobehavioral and biochemical results thereby improving cognitive functions.
Growing experimental evidence suggests that chronic neuroinflammatory conditions modulate disease progression in AD pathology. Neuroinflammation is driven by activated glial cells especially microglia and astrocytes, along with peripheral leucocytes, which in turn release copious amounts of proinflammatory cytokine such as TNF-α, in response to various neuropathological sequalae (Akiyama et al. 2000; Frankola et al. 2011; Rai et al. 2014). Therefore, targeting anti-inflammatory mechanisms might prove beneficial in delaying the AD progression (Wyss-Coray and Rogers 2012). Clinicopathological evidences revealed elevated levels of TNF-α have been negatively correlated with neurodegenerative diseases such as AD (Buchhave et al. 2010; Kim et al. 2017). Further, NF-κB plays complex roles in regulating inflammatory disease progression during CNS stress, which may potentially modulate neuronal survival through transcription of chemokines, proinflammatory cytokines and transcription factors. Recent experimental evidences demonstrate anti-inflammatory properties of vitamin D3 (Dulla et al. 2016; Jiang et al. 2015; Nissou et al. 2014). In the present study, we found that vitamin D3 supplementation effectively mitigated ICV-STZ induced inflammatory damage in cortex and hippocampus, which can be correlated well with reversal of neuronal injury indicative of increased cell survival.
The central cholinergic system, through its neurotransmitter, acetylcholine, regulates neurocognitive functions and is implicated in various neurodegenerative diseases including AD, vascular dementia, Huntington’s disease, stroke, cardiac arrest, Sepsis (Everitt and Robbins 1997), etc. Cholinergic innervation is high in cortex and hippocampal region and any neuronal loss in these brain regions can be linked to altered cognitive status (Arendt et al. 1995; Bobinski et al. 1998). Acetylcholinesterases hydrolyses acetylcholine into choline and acetate and elevated AChE levels is associated with deregulated cholinergic system, a measure of severity of cognitive impairment during various neurodegenerative conditions including AD (Sarter and Parikh 2005). Cholinergic degeneration is closely linked to AD progression in patients and experimental animal models and clinically, AChE inhibitors have been widely used to symptomatically treat AD induced cognitive impairment (Sonkusare et al. 2005). However, the role of vitamin D3 in regulating the cholinergic neurotransmission and memory functions has not been fully explored.
ICV-STZ administration demonstrated a significant increase in cortical and hippocampal AChE activity as a result of oxidative-nitrative stress and inflammatory degeneration, which mimics AD pathology (Kaizer et al. 2005; Sharma et al. 2010; Sonkusare et al. 2005). Furthermore, increased AChE activity can possibly modulate cytokine levels, exacerbating neuronal injury and memory dysfunction (Pavlov et al. 2003; Ray et al. 2014). The results clearly indicate that pre-treatment with vitamin D3 markedly inhibited AChE activity and alleviated the cholinergic insufficiency thereby increasing the synaptic levels of available ACh improving neurotransmission. Consistent with previous reports, evidently, our results indicate that vitamin D3 supplementation improved cholinergic functions (Kumar et al. 2011; Peeyush et al. 2010) and improved memory functions. Also recent studies reported that vitamin D3 enhanced Aβ clearance (Guo et al. 2016; Ito et al. 2011), decreased amyloid burden (Briones and Darwish 2012; Durk et al. 2014; Yu et al. 2011) and attenuated neuroinflammatory responses (Briones and Darwish 2012) and prevented axonal degeneration (Annweiler et al. 2014) thereby eliciting neuroprotective effect against experimental AD.
Conclusion
In conclusion, our results revealed that vitamin D3 pre-treatment attenuated ICV-STZ induced neurobehavioral, biochemical and histological abnormalities and memory deficits whereas post-treatment with vitamin D3 failed to exert any considerable beneficial effects on neurobehavioral, histopathological and molecular alterations. The neuroprotective effect of vitamin D3 could be attributed to its possible involvement in modulating inflammatory brain damage, decreasing neuronal oxidative stress, improving mitochondrial functions and enhanced cholinergic neurotransmission. Therefore, our study underscores the functional importance of prophylactic maintenance of adequate vitamin D3 levels, might offer novel therapeutic strategy for the treatment of neurodegenerative diseases such as AD.
References
Akiyama H et al (2000) Inflammation and Alzheimer’s disease. Neurobiol Aging 21:383–421
Alrefaie Z, Alhayani A (2015) vitamin D(3) improves decline in cognitive function and cholinergic transmission in prefrontal cortex of streptozotocin-induced diabetic rats. Behav Brain Res 287:156–162. doi:10.1016/j.bbr.2015.03.050
Anand R, Gill KD, Mahdi AA (2014) Therapeutics of Alzheimer’s disease: past, present and future. Neuropharmacol 76:27–50
Annweiler C, Brugg B, Peyrin JM, Bartha R, Beauchet O (2014) Combination of memantine and vitamin D prevents axon degeneration induced by amyloid-beta and glutamate. Neurobiol Aging 35:331–335. doi:10.1016/j.neurobiolaging.2013.07.029
Arendt T, Bruckner MK, Bigl V, Marcova L (1995) Dendritic reorganisation in the basal forebrain under degenerative conditions and its defects in Alzheimer’s disease. III. The basal forebrain compared with other subcortical areas. J Comp Neurol 351:223–246. doi:10.1002/cne.903510204
Becker A, Eyles DW, McGrath JJ, Grecksch G (2005) Transient prenatal vitamin D deficiency is associated with subtle alterations in learning and memory functions in adult rats. Behav Brain Res 161:306–312. doi:10.1016/j.bbr.2005.02.015
Berman SB, Hastings TG (1999) Dopamine oxidation alters mitochondrial respiration and induces permeability transition in brain mitochondria. J Neurochem 73:1127–1137
Bobinski M, de Leon MJ, Tarnawski M, Wegiel J, Reisberg B, Miller DC, Wisniewski HM (1998) Neuronal and volume loss in CA1 of the hippocampal formation uniquely predicts duration and severity of Alzheimer disease. Brain Res 805:267–269
Briones TL, Darwish H (2012) Vitamin D mitigates age-related cognitive decline through the modulation of pro-inflammatory state and decrease in amyloid burden. J Neuroinflamm 9:244. doi:10.1186/1742-2094-9-244
Briones TL, Darwish H (2014) Decrease in age-related tau hyperphosphorylation and cognitive improvement following vitamin D supplementation are associated with modulation of brain energy metabolism and redox state. Neuroscience 262:143–155. doi:10.1016/j.neuroscience.2013.12.064
Buchhave P, Zetterberg H, Blennow K, Minthon L, Janciauskiene S, Hansson O (2010) Soluble TNF receptors are associated with Abeta metabolism and conversion to dementia in subjects with mild cognitive impairment. Neurobiol Aging 31:1877–1884. doi:10.1016/j.neurobiolaging.2008.10.012
Buell JS, Dawson-Hughes B (2008) Vitamin D and neurocognitive dysfunction: preventing “D”ecline? Mol Aspects Med 29:415–422. doi:10.1016/j.mam.2008.05.001
Butterfield DA (2006) Oxidative stress in neurodegenerative disorders. Antioxid Redox Signal 8:1971–1973. doi:10.1089/ars.2006.8.1971
Claiborne A (1985) Catalase activity. In: GreenWald RA (ed) CRC handbook of methods for oxygen radical research, vol 1. CRC Press, Boca Raton, pp 283–284
Correia SC et al (2013) Mitochondrial abnormalities in a streptozotocin-induced rat model of sporadic Alzheimer’s disease. Curr Alzheimer Res 10:406–419
Dulla YA, Kurauchi Y, Hisatsune A, Seki T, Shudo K, Katsuki H (2016) Regulatory mechanisms of vitamin D3 on production of nitric oxide and pro-inflammatory cytokines in microglial BV-2 cells neurochemical research. Neurochem Res 41(11):2848–2858. doi:10.1007/s11064-016-2000-3
Durk MR, Han K, Chow EC, Ahrens R, Henderson JT, Fraser PE, Pang KS (2014) 1alpha,25-Dihydroxyvitamin D3 reduces cerebral amyloid-beta accumulation and improves cognition in mouse models of Alzheimer’s disease. J Neurosci 34:7091–7101. doi:10.1523/JNEUROSCI.2711-13.2014
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Everitt BJ, Robbins TW (1997) Central cholinergic systems and cognition. Annu Rev Psychol 48:649–684. doi:10.1146/annurev.psych.48.1.649
Frankola KA, Greig NH, Luo W, Tweedie D (2011) Targeting TNF-alpha to elucidate and ameliorate neuroinflammation in neurodegenerative diseases. CNS Neurol Disord Drug Targets 10:391–403
Gandhi S, Abramov AY (2012) Mechanism of oxidative stress in neurodegeneration. Oxidative Med Cell Longev 2012:428010. doi:10.1155/2012/428010
Garcion E, Nataf S, Berod A, Darcy F, Brachet P (1997) 1,25-Dihydroxyvitamin D3 inhibits the expression of inducible nitric oxide synthase in rat central nervous system during experimental allergic encephalomyelitis Brain research. Mol Brain Res 45:255–267
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177:751–766
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126:131–138
Guo YX, He LY, Zhang M, Wang F, Liu F, Peng WX (2016) 1,25-Dihydroxyvitamin D3 regulates expression of LRP1 and RAGE in vitro and in vivo, enhancing Abeta1-40 brain-to-blood efflux and peripheral uptake transport. Neuroscience 322:28–38. doi:10.1016/j.neuroscience.2016.01.041
Ito S, Ohtsuki S, Nezu Y, Koitabashi Y, Murata S, Terasaki T (2011) 1alpha,25-Dihydroxyvitamin D3 enhances cerebral clearance of human amyloid-beta peptide(1-40) from mouse brain across the blood-brain barrier. Fluids Barriers CNS 8:20. doi:10.1186/2045-8118-8-20
Javed H et al (2011) S-allyl cysteine attenuates oxidative stress associated cognitive impairment and neurodegeneration in mouse model of streptozotocin-induced experimental dementia of Alzheimer’s type. Brain Res 1389:133–142. doi:10.1016/j.brainres.2011.02.072
Javed H et al (2012) Rutin prevents cognitive impairments by ameliorating oxidative stress and neuroinflammation in rat model of sporadic dementia of Alzheimer type. Neuroscience 210:340–352. doi:10.1016/j.neuroscience.2012.02.046
Jiang J et al (2015) Vitamin D inhibits lipopolysaccharide-induced inflammatory response potentially through the toll-like receptor 4 signalling pathway in the intestine and enterocytes of juvenile Jian carp (Cyprinus carpio var. Jian). Br J Nutr 114:1560–1568. doi:10.1017/S0007114515003256
Jollow D, Mitchell J, Na Zampaglione, Gillette J (1974) Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3, 4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 11:151–169
Kaizer RR, Correa MC, Spanevello RM, Morsch VM, Mazzanti CM, Goncalves JF, Schetinger MR (2005) Acetylcholinesterase activation and enhanced lipid peroxidation after long-term exposure to low levels of aluminum on different mouse brain regions. J Inorg Biochem 99:1865–1870. doi:10.1016/j.jinorgbio.2005.06.015
Keeney JT, Butterfield DA (2015) Vitamin D deficiency and Alzheimer disease: common links. Neurobiol Dis 84:84–98. doi:10.1016/j.nbd.2015.06.020
Kim YS, Lee KJ, Kim H (2017) Serum tumour necrosis factor-alpha and interleukin-6 levels in Alzheimer’s disease and mild cognitive impairment. Psychogeriatrics. doi:10.1111/psyg.12218
King TE, Howard RL (1967) [52] Preparations and properties of soluble NADH dehydrogenases from cardiac muscle. Methods Enzymol 10:275–294
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Kraska A et al (2012) In vivo cross-sectional characterization of cerebral alterations induced by intracerebroventricular administration of streptozotocin. PloS ONE 7:e46196. doi:10.1371/journal.pone.0046196
Kumar PT, Antony S, Nandhu MS, Sadanandan J, Naijil G, Paulose CS (2011) Vitamin D3 restores altered cholinergic and insulin receptor expression in the cerebral cortex and muscarinic M3 receptor expression in pancreatic islets of streptozotocin induced diabetic rats. J Nutr Biochem 22:418–425. doi:10.1016/j.jnutbio.2010.03.010
Kuzmenko AI, Morozova RP, Nikolenko IA, Korniets GV, Kholodova YuD (1997) Effects of vitamin D3 and ecdysterone on free-radical lipid peroxidation. Biochem Biokhimiia 62:609–612
Labak M, Foniok T, Kirk D, Rushforth D, Tomanek B, Jasiński A, Grieb P (2010) Metabolic changes in rat brain following intracerebroventricular injections of streptozotocin: a model of sporadic Alzheimer’s disease. Acta Neurochir Suppl 106:177–181. doi:10.1007/978-3-211-98811-4_32
Latimer CS et al (2014) Vitamin D prevents cognitive decline and enhances hippocampal synaptic function in aging rats. Proc Natl Acad Sci USA 111:E4359–E4366. doi:10.1073/pnas.1404477111
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795
Longoni A et al (2016) 1,25-Dihydroxyvitamin D3 exerts neuroprotective effects in an ex vivo model of mild hyperhomocysteinemia. Int J Dev Neurosci 48:71–79. doi:10.1016/j.ijdevneu.2015.11.005
Markesbery WR (1997) Oxidative stress hypothesis in Alzheimer’s disease. Free Radic Biol Med 23:134–147
Misra S, Tiwari V, Kuhad A, Chopra K (2011) Modulation of nitrergic pathway by sesamol prevents cognitive deficits and associated biochemical alterations in intracerebroventricular streptozotocin administered rats. Eur J Pharmacol 659:177–186
Mohamed AR, Soliman GY, Ismail CA, Mannaa HF (2015) Neuroprotective role of vitamin D3 in colchicine-induced Alzheimer’s disease in rats. Alex J Med 51:127–136
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Nissou MF, Guttin A, Zenga C, Berger F, Issartel JP, Wion D (2014) Additional clues for a protective role of vitamin D in neurodegenerative diseases: 1,25-dihydroxyvitamin D3 triggers an anti-inflammatory response in brain pericytes. J Alzheimer’s Dis 42:789–799. doi:10.3233/JAD-140411
Paidi RK, Nthenge-Ngumbau DN, Singh R, Kankanala T, Mehta H, Mohanakumar KP (2015) Mitochondrial deficits accompany cognitive decline following single bilateral intracerebroventricular streptozotocin. Curr Alzheimer Res 12:785–795
Pavlov VA, Wang H, Czura CJ, Friedman SG, Tracey KJ (2003) The cholinergic anti-inflammatory pathway: a missing link in neuroimmunomodulation. Mol Med 9:125–134
Paxinos G, Watson CR, Emson PC (1980) AChE-stained horizontal sections of the rat brain in stereotaxic coordinates. J Neurosci Methods 3:129–149
Peeyush KT, Savitha B, Sherin A, Anju TR, Jes P, Paulose CS (2010) Cholinergic, dopaminergic and insulin receptors gene expression in the cerebellum of streptozotocin-induced diabetic rats: functional regulation with vitamin D3 supplementation. Pharmacol Biochem Behav 95:216–222. doi:10.1016/j.pbb.2010.01.008
Rai S, Kamat PK, Nath C, Shukla R (2014) Glial activation and post-synaptic neurotoxicity: the key events in streptozotocin (ICV) induced memory impairment in rats. Pharmacol Biochem Behav 117:104–117. doi:10.1016/j.pbb.2013.11.035
Ray RS, Rai S, Katyal A (2014) Cholinergic receptor blockade by scopolamine and mecamylamine exacerbates global cerebral ischemia induced memory dysfunction in C57BL/6J mice. Nitric Oxide 43:62–73. doi:10.1016/j.niox.2014.08.009
Sachdeva AK, Chopra K (2015) Naringin mitigate okadaic acid-induced cognitive impairment in an experimental paradigm of Alzheimer’s disease. J Funct Foods 19:110–125
Salkovic-Petrisic M, Tribl F, Schmidt M, Hoyer S, Riederer P (2006) Alzheimer-like changes in protein kinase B and glycogen synthase kinase-3 in rat frontal cortex and hippocampus after damage to the insulin signalling pathway. J Neurochem 96:1005–1015. doi:10.1111/j.1471-4159.2005.03637.x
Sarter M, Parikh V (2005) Choline transporters, cholinergic transmission and cognition. Nat Rev Neurosci 6:48–56. doi:10.1038/nrn1588
Sharma M, Gupta YK (2001) Intracerebroventricular injection of streptozotocin in rats produces both oxidative stress in the brain and cognitive impairment. Life Sci 68:1021–1029
Sharma N, Deshmukh R, Bedi KL (2010) SP600125, a competitive inhibitor of JNK attenuates streptozotocin induced neurocognitive deficit and oxidative stress in rats. Pharmacol Biochem Behav 96:386–394. doi:10.1016/j.pbb.2010.06.010
Sharma V, Bala A, Deshmukh R, Bedi KL, Sharma PL (2012) Neuroprotective effect of RO-20-1724-a phosphodiesterase4 inhibitor against intracerebroventricular streptozotocin induced cognitive deficit and oxidative stress in rats. Pharmacol Biochem Behav 101:239–245. doi:10.1016/j.pbb.2012.01.004
Shen L, Ji HF (2015) Vitamin D deficiency is associated with increased risk of Alzheimer’s disease and dementia: evidence from meta-analysis. Nutr J 14:76. doi:10.1186/s12937-015-0063-7
Shoham S, Bejar C, Kovalev E, Weinstock M (2003) Intracerebroventricular injection of streptozotocin causes neurotoxicity to myelin that contributes to spatial memory deficits in rats. Exp Neurol 184:1043–1052. doi:10.1016/j.expneurol.2003.08.015
Sonkusare S, Srinivasan K, Kaul C, Ramarao P (2005) Effect of donepezil and lercanidipine on memory impairment induced by intracerebroventricular streptozotocin in rats. Life Sci 77:1–14. doi:10.1016/j.lfs.2004.10.036
Sottocasa GL, Kuylenstierna B, Ernster L, Bergstrand A (1967) An electron-transport system associated with the outer membrane of liver mitochondria a biochemical and morphological study. J Cell Biol 32:415–438
Tiwari V, Kuhad A, Bishnoi M, Chopra K (2009) Chronic treatment with tocotrienol, an isoform of vitamin E, prevents intracerebroventricular streptozotocin-induced cognitive impairment and oxidative-nitrosative stress in rats. Pharmacol Biochem Behav 93:183–189. doi:10.1016/j.pbb.2009.05.009
Wang X, Michaelis EK (2010) Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci 2:12. doi:10.3389/fnagi.2010.00012
Wills E (1966) Mechanisms of lipid peroxide formation in animal tissues. Biochem J 99:667
Witte ME, Geurts JJ, de Vries HE, van der Valk P, van Horssen J (2010) Mitochondrial dysfunction: a potential link between neuroinflammation and neurodegeneration? Mitochondrion 10:411–418. doi:10.1016/j.mito.2010.05.014
Wyss-Coray T, Rogers J (2012) Inflammation in Alzheimer disease-a brief review of the basic science and clinical literature. Cold Spring Harb Perspect Med 2:a006346. doi:10.1101/cshperspect.a006346
Yu J, Gattoni-Celli M, Zhu H, Bhat NR, Sambamurti K, Gattoni-Celli S, Kindy MS (2011) Vitamin D3-enriched diet correlates with a decrease of amyloid plaques in the brain of AbetaPP transgenic mice. J Alzheimer’s Dis 25:295–307. doi:10.3233/JAD-2011-101986
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Yamini, P., Ray, R.S. & Chopra, K. Vitamin D3 attenuates cognitive deficits and neuroinflammatory responses in ICV-STZ induced sporadic Alzheimer’s disease. Inflammopharmacol 26, 39–55 (2018). https://doi.org/10.1007/s10787-017-0372-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-017-0372-x