Abstract
Paraquat (PQ), a widely used potent herbicide, generates superoxide anions and other free radicals, leading to severe toxicity and acute lung injury. PQ induces pulmonary fibrosis through epithelial to mesenchymal transition (EMT) characterized by increased number of myofibroblasts. Time-dependent PQ-induced EMT has been evaluated in present investigation where intracellular ROS levels were significantly enhanced after 24 h of PQ intoxication. Anti-inflammatory effects of curcumin have been studied where alveolar epithelial cells (A549 cells) were incubated with curcumin (30 μΜ) for 1 and 3 h before PQ intoxication (700 μM). Western blot and immunocytochemistry studies revealed that pretreatment of A549 cells with curcumin for 3 h before PQ exposure has maintained E-cadherin expression and inhibited PQ induced α-smooth-muscle actin (α-SMA) expression. Transforming growth factor-β (TGF-β) that seems to be involved in PQ-induced EMT was enhanced after PQ intoxication, but curcumin pretreatment has effectively inhibited its expression. Immunostaining studies have shown that curcumin pretreatment has significantly reduced matrix metalloproteinase-9 (MMP-9) expressions, which were elevated after PQ intoxication. These results demonstrate that curcumin can regulate PQ-induced EMT by regulating the expression of TGF-β.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Paraquat (PQ) is widely used as highly toxic weed killer (herbicide), which can cause multiple organ failure in human and animals [1, 2]. Paraquat poisoning is a major medical problem due to lack of effective treatment. The lung is the principle target organ, and respiratory failure (irreversible lung injury) is the most common cause of death from PQ poisoning because its amount concentrate in lung tissue than any other organ [3,4,5]. PQ accumulation in lung occurs through type II pneumocytes via a polyamine uptake system. The respiratory epithelium is the lining of the airways, which is in direct contact with the environment, and acts as physical barrier between external and internal surface, contributing to airway defense by clearing infectious and environmental agents [6, 7]. Airway epithelial cells (AECs) are the most abundant cell type in the lungs, which contribute in transportation of gases to and from the alveoli and therefore plays a central role to protect the lungs from exposure to inflammatory stimuli and antigens [8, 9]. The alveolar epithelium consists of type I and type II epithelial cells; type I pneumocytes are squamous epithelial cells that cover approximately 90% of the alveolar surface and conduct the exchange of gases. Type II alveolar cells are cuboidal secretary cells and have a critical role in the synthesis of surfactant, which reduces the surface tension within the alveoli. Alveolar epithelial type II (AT2) cells also have self-renewal capability and played an important role in the repair of damaged alveolar epithelium after lung injury [10,11,12,13,14]. Injury to the epithelial cells initiates the release of proinflammatory and profibrotic cytokines, thereby generating profibrogenic environment, which is characterized by (myo)fibroblast accumulation, collagen and matrix deposition, and structural remodeling. Pulmonary fibrosis associated with excessive deposition of extracellular matrix is mediated by the recruitment of inflammatory cells and collagen-producing myofibroblast and fibroblasts [15,16,17]. Recent studies revealed that myofibroblasts are the hallmark of lung tissue fibrosis, and the sources of these cells are different and may arise from differentiation of resident lung fibroblasts or through the transdifferentiation of epithelial cells [17, 18]. The transdifferentiation of epithelial cells is a biological process known as epithelial-mesenchymal transition (EMT), whereby polarized epithelial cells undergo various biochemical changes giving rise to motile mesenchymal cells, which are important source of fibroblasts and myofibroblasts. Epithelial and mesenchymal cells differ morphologically and functionally. Epithelial cells are polarized and adherent type of cells, which show strong cell-cell adhesion and have limited migratory potential. Mesenchymal cells are non-polarized, have spindle shape, and show focal cell contact with migratory potential, i.e., motile [19, 20].
Alveolar epithelial cells (AECs) are activated in initial phase of lung injury, and then these AECs create profibrotic phase through the process of EMT, in which activated AECs are converted into collagen producing fibroblasts and myofibroblasts. By initiating the tissue repair through producing ECM proteins, EMT plays a key role in maintaining cellular homeostasis, but excessive production of ECM proteins leads to fibrosis, which may cause organ failure [17, 21, 22]. Other studies also have demonstrated that EMT phenomena are the potential contributor of lung fibrosis [21, 23]. Some recent reports have suggested the involvement of EMT in PQ-induced pulmonary fibrosis through transforming growth factor-β1 signaling pathway in vitro and in vivo [24, 25].
We already have demonstrated that single toxic dose of PQ initiates structural changes and early fibroproliferation in lungs within 48 h, and pretreatment of intranasal curcumin (5 mg/kg) inhibits PQ-induced lung injury and fibroproliferation to some extent [26]. Curcumin is well known for its anti-inflammatory and anti-oxidant properties, used as medicine from ancient times. It has been reported earlier that curcumin suppresses EMT; therefore, in the present study, we have investigated the role of curcumin on PQ-induced EMT through TGF-β-dependent mechanism using A549 human AECs [27, 28].
METHODS AND MATERIAL
Cell Culture
Human alveolar type II-like epithelial A549 cells were purchased from National Centre for Cell Science (NCCS), Pune, India. Cells were maintained in Dulbecco’s modified Eagle medium (DMEM) with 10% heat-inactivated FBS and antibiotics at 37 °C with 5% CO2 in a humidified incubator, and cells were sub-cultured until they were 80–90% confluent. Before every experiment, cells were harvested with TPVG (trypsin phosphate versene glucose) solution, and viability was assessed by trypan blue dye exclusion test. PQ was dissolved in distilled water to make stock solutions, and DMSO was used as solvent for curcumin.
PQ-induced Morphological Changes
Morphological changes in A549 cells were observed after PQ treatment, as 10 × 104 cells were plated in 12-well flat bottom culture plates and incubated overnight. After incubation, fresh media (without FBS and antibiotic solution) were added and treated with different concentrations of PQ for 24 h. Cells were examined under microscope (×10, Nikon inverted microscope).
Cell Viability Assay
The cytotoxicity of PQ on A549 cells was detected by the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay [29]. A549 cells were seeded at density of 3 × 103 cells/well in flat-bottomed 96-well tissue culture plates containing 200 μl media and grown overnight (up to 70–80% confluent). The cells were treated with different concentrations of PQ and curcumin to determine IC50. After 24 h of treatment, MTT (5 mg/ml, 20 μl) was added in each well and incubated for 4 h at 37 °C. After incubation, media was aspirated, and 200-μl DMSO was added to dissolve the formazan (MTT metabolic product). Plates were read at 570 nm in microtiter plate reader. The percentage of cellular viability was assessed relative to the control (only media-treated cells).
Intracellular ROS Detection
The production of intracellular ROS was measured by DCF-DA (2′,7′-dichlorofluorescin diacetate) as an indicator of superoxide production in PQ-treated A549 cells. The cells were cultured in 96-well plate (black) and allowed to 70% confluent. The cells were treated with different concentrations of PQ for 24 h and at the end of treatment; medium was aspirated from all the wells. Cells were stained with 10-μM DCF-DA and incubated for 45 min at 37 °C in dark [30]. Plates were read at 485 (exCitation wavelength) and 535 nm (emission wavelength).
Experimental Design
After determining toxic dose of PQ and safe dose of curcumin, five experimental groups were made where (I) control group, cells received only fresh DMEM media without FBS; (II) PQ group, cells treated with PQ 700 μM for 24 h; (III) curcumin group, cells were pretreated with 30-μΜ curcumin, before 1 and 3 h of 700-μM toxic dose of PQ for 24 h; (IV) vehicle group, DMSO (equal volume to curcumin) given an hour before PQ exposure; (V) curcumin alone group, cells received only (30 μM) curcumin. After 24 h of the PQ incubation, cells were used to prepare lysate for western blotting, and RNA was isolated for RT-PCR (Table 1).
Protein Expressions of E-Cadherin and α-Smooth Muscle Actin
A549 cell lysates were prepared with RIPA buffer containing protease inhibitor cocktail, and protein concentration was determined using BCA kit (bicinchoninic acid assay). Equal amount of protein was separated on 12% SDS-PAGE gels and then transferred to nitrocellulose membranes. The membranes were blocked with blocking buffer (5% BSA in Tris-buffered saline with 0.01% T-Tween 20) for 2 h, followed by incubation with primary antibodies to E-cadherin (1: 1000, CST), α-smooth muscle actin (1:500, Thermo Peirce), and β-actin (1:2000, gene script) antibody overnight at 4 °C. After washing the membranes with TBST for 15 min, the membranes were probed with horseradish peroxidase (HRP) conjugated secondary antibody (1:3000, CST). After three washings with TBST for 15 min, detection of bound antibodies was visualized by ECL reagent (Millipore) with short exposure to X-ray films. β-Actin was analyzed as a loading control, and densitometric values of bands were determined and statistically analyzed using ImageJ software.
Immunocytochemistry
To assess protein expression of MMP-9, E-cadherin, and α-smooth muscle actin in A549 cells, immuocytochemistry was done as described earlier [31]. Sterile coverslips were coated with poly-L-lysine and allowed to dry and sterilize under UV light for 4 h. Cells were grown on cover slip in 6-well plate overnight. After rinsing in phosphate-buffered saline (PBS), cells were pretreated with curcumin followed by PQ for 24 h. After treatment, cells were fixed in ice cold methanol for 10 min at room temperature, permeabilized in 0.1% triton X-100 for 5 min, and washed three times with PBS before blocking (1% BSA for 1 h). Cells were washed three times with PBS and stained with FITC-conjugated secondary antibody for 2 h at room temperature, washed three times with PBS, and nuclear regions were stained with DAPI (4′,6-diamidino-2-phenylindole) for 1 min; stained cells were mounted with glycerol mounting medium on slides and visualized using a confocal microscope.
mRNA Expression of TGF-β by Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
After 24 h of treatment, cells were washed with chilled PBS, and total RNA was extracted from cells using TRIzol reagent [32]. Total RNA concentration was assessed using a Nano Drop at 260 nm, and the quality of the RNA was determined (using ratio at 260/280 nm). cDNA was synthesized using 1 μg RNA and Quanti Tect RT PCR KIT (205311 Qiagen) in a 96-well thermal cycler as per the manufacturer’s instructions. Quantitative real-time (qRT)-PCR was performed using a QuantiTect SYBR PCR kit (204141 Qiagen) and ABI 7500 real-time PCR system. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as house-keeping gene for normalization, and the sequences of the primers were used: TGF-β, 5′-ACATGGTCATGAGCTTTGTGA-3′(forward) and 5′-ACTCTTTGTGGTGTCGCTGA-3′(reverse); GAPDH, 5′-CCCCGGTTTCTATAAATTGAGC-3′ (forward) and 5′-TTTCTCTCCGCCCGTCTT-3′ (reverse). For quantitation, Dct method was applied, and results were presented as expression changes.
STATISTICAL ANALYSIS
All values are presented as mean ± SEM. Differences between groups were determined with one-way ANOVA followed by Tukey’s test. A level of p < 0.05 was considered statistically significant.
RESULTS
Effect of PQ and Curcumin on Cell Viability and Morphological Changes
Morphological changes in PQ and curcumin-treated A549 cells were examined under light microscope, where morphological changes were observed in concentration dependent manner. But after curcumin treatment (incubation up to 50 μM), A549 cells did not undergo such morphological changes, but structural morphology was changed with higher concentration of curcumin. Cell viability was evaluated by MTT assay after 24 h of incubation with different concentrations of PQ (100, 200, 300, 400, 500, 600,750, 1000 μM) and curcumin (10, 25, 50, 100, 250, 500 μM). The survival rate of the cells was significantly decreased after PQ intoxication (300 to 1000 μM) as compared with control group. Survival rate for the PQ concentrations (300, 400, 500, 700, and 1000 μM) was 81, 77, 68, 56, 49, and 36, respectively. No significant change in the cell viability was observed up to 50-μM curcumin, whereas the survival rate decreased up to 25, 39, and 57% with higher curcumin concentrations (100, 250, and 500 μM, respectively) (Figs. 1–2).
A549 cells were exposed to PQ at different concentration, and the cell viability was determined by MTT assay. Survival rate of A549 cells treated with PQ for 24 h was decreased as the concentration increased. Results are expressed as percentage of viability, and the data are shown as means ± SEM (*p < 0.05) compared with control.
Effect of Individual Effects of PQ and Curcumin on Intracellular ROS Level
After 24 h of incubation with different concentrations of PQ (100–1000 μM), intracellular ROS level increased with increase in PQ concentration. Different concentrations of curcumin on ROS production were also studied where almost negligible inductive effect of curcumin on ROS generation was noted up to 50 μM. However, a significant change was observed with higher curcumin concentrations (Figs. 3 and 4).
Curcumin Suppressed PQ-Induced Cytotoxicity
Cell viability was determined by MTT assay to investigate anti-cytotoxic effects of curcumin (30 μM). After PQ (700 μM) treatment, survival rate of A549 cells was reduced up to 48% after 24 h, while the viability of curcumin (30 μM)-treated A549 cells was almost 100%. Survival rate of curcumin pretreated group was 26% higher than only PQ exposed cells (Figs. 5–6).
Curcumin Pretreatment Reduced PQ-Induced ROS Generation in A549 Cells
After treatment with PQ (700 μM) for 24 h, the levels of intracellular ROS (iROS) were increased significantly as compared with those in control and curcumin alone groups. Curcumin concentrations less than 30 μM was not much effective to inhibit PQ-induced ROS production, but 1-h pretreatment with 30- or 50-μM curcumin before PQ intoxication has significantly decreased the intracellular ROS as compared with only PQ-induced group. A549 cells were incubated with different concentrations of PQ and curcumin separately to assess their effects on morphology of A549 cells (Figs. 7–8).
Microscopic observation of growth pattern of A549 cells incubated with different concentrations of PQ after 24 h of treatment: a untreated cells; PQ-treated cells (b–i); b 100 μM, c 200 μM, d 300 μM, e 00 μM, f 500 μM, g 600 μM, h 750 μM, i 1000 μM. All images were taken at ×10 magnification with inverted microscope.
Curcumin Inhibits PQ-Induced Epithelial to Mesenchymal Transition in A549 Cells
Effect on E-Cadherin Protein in A549 Cells
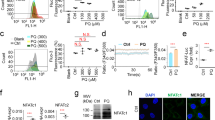
To confirm PQ-induced epithelial to mesenchymal transition in A549 cells, the expression of E-cadherin protein as marker of epithelial cells was measured by western blot and immunofluorescence staining. Our results revealed that marked decrease in the expression of E-cadherin was noted in PQ-induced group as compared with those in the control and curcumin-treated groups, whereas the promotion of E-cadherin protein expression in the curcumin pretreatment group was noted as compared with PQ alone and PQ + DMSO groups. Similar result was found with immunofluorescence staining (Figs. 9–10).
Effect on α-SMA Protein in A549 Cells
To confirm PQ-induced EMT, protein expression of α-SMA as marker of myofibroblast was further studied by western blot analysis and immunofluorescence staining. Protein expression of α-SMA was significantly higher in PQ-induced A549 cells as compared with control. No significant changes were observed in the expression of α-SMA protein after curcumin pretreatment before 1 h of PQ intoxication as compared with only PQ-induced A549 cells. But curcumin treatment (30 μm) before 3 h of PQ intoxication was effective in suppressing PQ induced α-smooth muscle actin expression in A549 cells (Fig. 11).
Effect of Curcumin on TGF-β
TGF-β is a potent inducer of EMT; therefore, its mRNA expression was analyzed by RT-PCR. Our results indicated that the expression of TGF-β was significantly increased after PQ exposure as compared with control and curcumin-treated cells (Fig. 12).
Effect of Curcumin on Matrix Metalloproteinase-9 (MMP-9)
Expression of MMP-9 may play a catalytic role in induction of EMT. Its expression was investigated and determined by immunofluorescence staining of A549 cells, which indicated that PQ-intoxicated cells show more fluorescence for MMP-9, whereas curcumin pretreatment has significantly reduced MMP-9 expression (Fig. 13).
DISCUSSION
PQ, being highly toxic herbicide, is also used in suicidal cases. Lung injury or respiratory failure is the main cause of PQ-related mortality. Oxidative injury to the alveolar epithelium is the main cause of respiratory failure in PQ poisoning. There are numerous in vivo and in vitro studies, which provide the evidence that AEC cells act as precursors, which give rise to fibroblast-like mesenchymal cells [33]. To promote repair in response to injury and severe inflammatory conditions, AECs transdifferentiate into myofibroblast through epithelial to mesenchymal transition (EMT).
EMT is a phenotypic change, which is characterized by loss of epithelial markers, such as E-cadherin and induction of mesenchymal markers like α-smooth muscle actin [34]. There are strong evidences, which suggest that myofibroblasts are the main mediators of pulmonary fibrosis, and increased expression of α-SMA, a marker of myofibroblast after PQ intoxication within 48 h, was also found [26, 33]. Many studies already have revealed the critical role of EMT in the induction of lung fibrosis [21, 23]. In the present study, effects of curcumin have been investigated in PQ-induced EMT in A549 cells.
LD50 (median lethal concentration) for PQ and safe dose of curcumin were determined, and then treatment regimen was decided. Oxidative stress via generation of reactive oxygen species (ROS), inflammation, and hypoxia are main triggers to induce EMT [18]. Our results clearly indicated that the ROS level was significantly less in curcumin-pretreated A549 cells as compared with PQ-induced cells. To determine the effective concentration of curcumin, A549 cells were treated with three different concentrations of curcumin (10, 30, and 50 μM). Surprisingly, curcumin 30 μM has shown the highest activity, whereas curcumin 50 μM was effective but less than 30 μM. Therefore, working concentration of curcumin was selected lower than 50 μM. Pretreatment of curcumin also proved protective against PQ-induced cell death.
TGF-β is a multifunctional cytokine that is regarded as a master cytokine in induced lung fibrosis and many tissues [35]. A number of studies have shown that TGF-β plays a key role in the pathogenesis of PQ-induced fibrosis via induction of EMT [36]. Another study has demonstrated that PQ-induced EMT was abolished by transfection of PQ-treated A549 with TGF-β1-specific small hairpin RNA (shRNA) [37]. Since TGF-β is responsible for the induction of EMT, therefore to determine the role of curcumin on PQ-induced EMT, mRNA expression of TGF-β was investigated in the present study. Pretreatment of A549 cells with curcumin for 1 h before PQ intoxication has significantly suppressed PQ-induced TGF-β level. Increased level of TGF-β causes altered expression of phenotypical markers of epithelial cells. In order to investigate the PQ-induced EMT through TGF-β in A549 cells, protein expression of epithelial and mesenchymal cell markers was determined.
In the present study, potential role of curcumin pretreatment on PQ-induced EMT in AECs was studied by analyzing the expression of phenotypic markers of mesenchymal and epithelial cells. The phenotype of epithelial cells can be examined by expression of many proteins, such as zonula occludens-1 (ZO-1), E-cadherin, and cytokeratins. E-Cadherin is a cell-cell adhesion molecule, expressed in almost all types of epithelial cells except neuronal epithelium [38].
To investigate PQ-induced EMT, protein expression of E-cadherin was studied, which was lost after PQ intoxication, whereas expression of α-smooth muscle actin, which is associated with the differentiation of lung fibroblasts into myofibroblasts, was found significantly higher in PQ-induced A549 cells as compared with those in the control. Decreased expression of E-cadherin and increased expression of a-SMA in A549 cells as transition of epithelial cells to a spindle-shaped morphology indicate successful construction of PQ-induced EMT model in A549 cells. Further, effect of pretreatment of curcumin before 1 h of PQ-induced EMT in A549 cells has significantly increased the expression of epithelial cell marker (E-cadherin) but not much effective in inhibiting the PQ-induced expression of myofibroblast marker, i.e., α-smooth muscle actin, whereas pretreatment of A549 cells with curcumin for 3 h before PQ intoxication has significantly reduced the α-SMA expression as compared with without curcumin pretreatment. The results of immunofluorescence staining of A549 cells also clearly indicated that pretreatment with curcumin for 3 h has better potential to suppress EMT in A549 cells, as compared with 1 h of curcumin pretreatment prior to PQ exposure.
Our results indicated that A549 cells could serve as a model to screen the suitable drug against PQ-induced EMT, and our findings revealed that curcumin not only plays anti-inflammatory and anti-oxidant role in PQ-induced lung injury but also have anti-fibrotic potential by inhibiting epithelial to mesenchymal transition to some extent. Pretreatment of curcumin (30 μm) before 1 h of PQ toxicity in A549 cells was effective to inhibit the PQ-induced cytotoxicity and intracellular ROS production but was not able to inhibit PQ-induced transition of epithelial to mesenchymal cells. But when cells stayed with curcumin for longer time, then PQ-induced EMT was inhibited. On the basis of above results, it can be stated here that expression of TGF-β was regulated by curcumin, and it has some potential to suppress PQ-induced EMT in A549 cells, which may be used to treat PQ-induced toxicity.
References
Eddleston, M. 2000. Patterns and problems of deliberate self-poisoning in the developing world. QJM 93: 715–731.
Sittipunt, C. 2005. Paraquat poisoning. Respiratory Care 50 (3): 383–385.
Bismuth, C., R. Garnier, F.J. Band, J. Muszynski, and C. Keyes. 1990. Paraquat poisoning. An overview of the current status. Drug Safety 5 (4): 243–251.
Dinis-Oliveira, R.J., J.A. Duarte, A. Sanchez-Navarro, F. Remiao, M.L. Bastos, and F. Carvalho. 2008. Paraquat poisonings: Mechanisms of lung toxicity, clinical features and treatment. Critical Reviews in Toxicology 38 (1): 13–71.
Zerin, T., Y.S. Kim, S.Y. Hong, and H.Y. Song. 2012. Protective effect of methylprednisolone on paraquat-induced A549 cell cytotoxicity via induction of efflux transporter, P-glycoprotein expression. Toxicology Letters 208 (2): 101–107.
Knight, D.A., and S.T. Holgate. 2003. The airway epithelium: Structural and functional properties in health and disease. Respirology 8 (4): 432–446.
Chuquimia, O.D., D.H. Petursdottir, N. Periolo, and C. Fernández. 2013. Alveolar epithelial cells are critical in protection of the respiratory tract by secretion of factors able to modulate the activity of pulmonary macrophages and directly control bacterial growth. Infection and Immunity 81 (1): 381–389.
Hiemstra, P.S., P.B. McCray, and R. Bals. 2015. The innate immune function of airway epithelial cells in inflammatory lung disease. European Respiratory Journal 45 (4): 1150–1162.
Bals, R., and P.S. Hiemstra. 2004 Feb. Innate immunity in the lung: How epithelial cells fight against respiratory pathogens. The European Respiratory Journal 23 (2): 327–333.
Parker, D., and A. Prince. 2011 Aug 1. Innate immunity in the respiratory epithelium. American Journal of Respiratory Cell and Molecular Biology 45 (2): 189–201.
Williams, M.C. 2003. Alveolar type I cells: Molecular phenotype and development. Annual Review of Physiology 65 (1): 669–695.
Fereol, S., R. Fodil, G. Pelle, B. Louis, and D. Isabey. 2008. Cell mechanics of alveolar epithelial cells (AECs) and macrophages (AMs). Respiratory Physiology & Neurobiology 163 (1): 3–16.
Castranova, V., J. Rabovsky, J.H. Tucker, and P.R. Miles. 1988. The alveolar type II epithelial cell: A multifunctional pneumocyte. Toxicology and Applied Pharmacology 93 (3): 472–483.
Evans, M.J., L.J. Cabral, R.J. Stephens, and G. Freeman. 1973. Renewal of alveolar epithelium in the rat following exposure to NO2. The American Journal of Pathology 70 (2): 175–198.
Crosby, L.M., and C.M. Waters. 2010. Epithelial repair mechanisms in the lung. American Journal of Physiology. Lung Cellular and Molecular Physiology 298 (6): L715–L731.
Sakai, N., and A.M. Tager. 2013. Fibrosis of two: Epithelial cell-fibroblast interactions in pulmonary fibrosis. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1832 (7): 911–921.
Schissel SL, Layne MD 2006. Telomerase, myofibroblasts, and pulmonary fibrosis. 520–522.
Kage, H., and Z. Borok. 2012. EMT and interstitial lung disease: A mysterious relationship. Current Opinion in Pulmonary Medicine 18 (5): 517–523.
Lamouille, S., J. Xu, and R. Derynck. 2014. Molecular mechanisms of epithelial–mesenchymal transition. Nature Reviews Molecular Cell Biology 15 (3): 178–196.
Kalluri, R., and R.A. Weinberg. 2009. The basics of epithelial-mesenchymal transition. The Journal of Clinical Investigation 119 (6): 1420–1428.
Kalluri, R., and E.G. Neilson. 2003. Epithelial-mesenchymal transition and its implications for fibrosis. Journal of Clinical Investigation 112 (12): 1776–1784.
Kim, K.K., M.C. Kugler, P.J. Wolters, L. Robillard, M.G. Galvez, A.N. Brumwell, D. Sheppard, and H.A. Chapman. 2006. Alveolar epithelial cell mesenchymal transition develops in vivo during pulmonary fibrosis and is regulated by the extracellular matrix. Proceedings of the National Academy of Sciences 103 (35): 13180–13185.
Tanjore, H., X.C. Xu, V.V. Polosukhin, A.L. Degryse, B. Li, W. Han, T.P. Sherrill, D. Plieth, E.G. Neilson, T.S. Blackwell, and W.E. Lawson. 2009. Contribution of epithelial-derived fibroblasts to bleomycin-induced lung fibrosis. American Journal of Respiratory and Critical Care Medicine 180 (7): 657–665.
Yamada, A., T. Aki, K. Unuma, T. Funakoshi, K. Uemura, et al. 2015. Paraquat induces epithelial-mesenchymal transition-like cellular response resulting in fibrogenesis and the prevention of apoptosis in human pulmonary epithelial cells. PLoS One 10 (3): e0120192.
Han, Y.Y., P. Shen, and W.X. Chang. 2015. Involvement of epithelial-to-mesenchymal transition and associated transforming growth factor-β/Smad signaling in paraquat-induced pulmonary fibrosis. Molecular Medicine Reports 12 (6): 7979–7984.
Tyagi, N., D. Dash, and R. Singh. 2016. Curcumin inhibits paraquat induced lung inflammation and fibrosis by extracellular matrix modifications in mouse model. Inflammopharmacology. 24 (6): 335–345.
Xu, S., B. Jiang, H. Wang, C. Shen, H. Chen, and L. Zeng. 2017. Curcumin suppresses intestinal fibrosis by inhibition of pparγ-mediated epithelial-mesenchymal transition. Evidence-based Complementary and Alternative Medicine. https://doi.org/10.1155/2017/7876064.
Wang Q, Qu C, Xie F, Chen L, Liu L, Liang X. Curcumin suppresses epithelial-to-mesenchymal transition and metastasis of pancreatic cancer cells by inhibiting cancer-associated fibroblasts. American Journal of Cancer Research 017;7(1):125.
Mosmann, T. 1983. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Journal of Immunological Methods 65 (1–2): 55–63.
Alaimo, A., R.M. Gorojod, and M.L. Kotler. 2011. The extrinsic and intrinsic apoptotic pathways are involved in manganese toxicity in rat astrocytoma C6 cells. Neurochemistry International 59 (2): 297–308.
Brussel, J.P., G.J. van Steenbrugge, J.C. Romijn, F.H. Schröder, and G.H.J. Mickisch. 1999. Chemosensitivity of prostate cancer cell lines and expression of multidrug resistance-related proteins. European Journal of Cancer 35 (4): 664–671.
Giulietti, A., L. Overbergh, D. Valckx, B. Decallonne, R. Bouillon, and C. Mathieu. 2001. An overview of real-time quantitative PCR: Applications to quantify cytokine gene expression. Methods 25 (4): 386–401.
Willis, B.C., R.M. Dubois, and Z. Borok. 2006. Epithelial origin of myofibroblasts during fibrosis in the lung. Proceedings of the American Thoracic Society 3 (4): 377–382.
Wu, T., J.M. Chen, T.G. Xiao, X.B. Shu, H.C. Xu, L.L. Yang, L.J. Xing, P.Y. Zheng, and G. Ji. 2016. Qinggan Huoxue recipe suppresses epithelial-to-mesenchymal transition in alcoholic liver fibrosis through TGF-β1/Smad signaling pathway. World Journal of Gastroenterology 22 (19): 4695–4706.
Sheppard, D. 2006. Transforming growth factor β: A central modulator of pulmonary and airway inflammation and fibrosis. Proceedings of the American Thoracic Society 3 (5): 413–417.
Kasai, H., J.T. Allen, R.M. Mason, T. Kamimura, and Z. Zhang. 2005. TGF-β1 induces human alveolar epithelial to mesenchymal cell transition (EMT). Respiratory Research 6 (1): 56.
Hay, E.D. 1995. An overview of epithelio-mesenchymal transformation. Cells, Tissues, Organs 154 (1): 8–20.
Willis, B.C., and Z. Borok. 2007. TGF-β-induced EMT: Mechanisms and implications for fibrotic lung disease. American Journal of Physiology Lung Cellular and Molecular Physiology 293 (3): L525–L534.
Funding
The authors are thankful to DST-Science and Engineering Research Board (DST-SERB), New Delhi, India for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tyagi, N., Singh, D.K., Dash, D. et al. Curcumin Modulates Paraquat-Induced Epithelial to Mesenchymal Transition by Regulating Transforming Growth Factor-β (TGF-β) in A549 Cells. Inflammation 42, 1441–1455 (2019). https://doi.org/10.1007/s10753-019-01006-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-019-01006-0