Abstract
5-Lipoxygenase inhibitor zileuton has been demonstrated to attenuate ischemic brain damage in rats of permanent focal cerebral ischemia in previous work. To further investigate the mechanism underlying zileuton's neuroprotection, adult male Sprague-Dawley rats underwent permanent middle cerebral artery occlusion (MCAO), then received treatment with zileuton or vehicle after the onset of ischemia. Neurological deficit, cerebral infarction, and morphological characteristic were measured 6 and 24 h after MCAO. The enzymatic activity of myeloperoxidase (MPO) was assessed 6 and 24 h after MCAO and the lipid peroxidation levels were evaluated by malondialdehyde assay. Expression of nuclear factor-kappa B (NF-κB) p65 in rat brain was detected by immunohistochemistry and Western blot. Expression of inducible nitric oxide synthase (iNOS) in rat brain was determined by RT-PCR and Western blot. Nitric oxide production in rat brain was also measured 24 h after MCAO. The concentration of TNF-α and IL-1β in serum were detected by ELISA. Zileuton significantly reduced neurological deficit scores, cerebral infarct volume, MPO activity, and the lipid peroxidation levels. It also inhibited the expression of NF-κB and decreased the expression and activity of iNOS in rat brain. In addition, zileuton attenuated the release of TNF-α and IL-1β in serum. Our results suggest that zileuton reduces inflammatory reaction and brain damage in a rat model of permanent focal cerebral ischemia. The neuroprotective effect of zileuton in cerebral ischemia might be associated with the inhibition of inflammatory reaction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stroke is one of the leading causes of death and disability. Brain damage following focal cerebral ischemia develops from a complex series of pathophysiological mechanisms including ion homeostasis imbalance, excitotoxicity, oxidative stress, blood–brain barrier disruption, inflammation, and apoptosis [1, 2]. In animal models of ischemic stroke, potential neuroprotective agents targeted at different pathophysiological mechanisms in ischemic brain damage have been explored [3].
Several reports demonstrated that post-ischemic inflammation contributes to the development of neuronal injury and cerebral infarction [4, 5]. 5-Lipoxygenase (5-LOX) is one of the major enzymes in leukotrienes (LTs) biosynthesis and is widely recognized as an important role in cerebral ischemia [6]. Zhou et al. [7] demonstrated that cerebral ischemia leads to enhanced expression of 5-LOX, which might result in the expansion of ischemic brain damage. Selective 5-LOX inhibitors AA-861 and BW-B 70C have been demonstrated to protect brain against ischemic damage in an animal model of focal cerebral ischemia [6, 8]. 5-LOX mediates the generation of reactive oxygen species, which is implicated in the activation of nuclear factor-kappa B (NF-κB) [9]. NF-κB is a key transcription factor involved the inducible expression of inflammatory mediators such as inducible nitric oxide synthase (iNOS), tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β) that are the major mechanisms by which post-inflammation contributes to ischemic brain damage [10–12].
Recent work from our laboratory demonstrated that zileuton attenuates ischemic brain damage in a rat model of permanent focal cerebral ischemia [13]. Zileuton reduced brain edema and infarct volume 24 h after permanent middle cerebral artery occlusion (MCAO) in rats; it also decreased the expression of 5-LOX and matrix metalloproteinase-9 in rat brain [13]. To further explore the potential neuroprotective mechanism of zileuton in cerebral ischemia, we, for the first time, explored the effect of zileuton on inflammatory reaction and inflammatory mediators in rats of permanent focal cerebral ischemia.
Materials and Methods
Experimental Animals and Permanent MCAO Model
All animal experiments were conducted according to the National Institute of Health Guide for the Care and Use of Laboratory Animals. Adult male Sprague-Dawley rats weighing 250–300 g were obtained from Shanghai Laboratory Animal Center, Chinese Academy of Sciences. Animals were housed in a colony room under controlled temperature (22°C) and a 12:12 light-dark cycle, with food and water available. Rats were anesthetized with an intraperitoneal injection of chloral hydrate (300 mg/kg) and subjected to permanent MCAO as described previously [13]. In brief, the right common carotid artery, external carotid artery (ECA), and internal carotid artery (ICA) were carefully exposed. A nylon monofilament suture (0.24 mm in diameter) with a distal cylinder (0.32 mm in diameter) was inserted from the ECA into the ICA and gently advanced to occlude the origin of the right MCA. Sham-operated animals were manipulated in the same way, but the MCA was not occluded. Rectal temperature was maintained at 37.0°C with a heating pad and warm light during the surgical procedure.
Drug Administration and Experimental Groups
Zileuton (Comens Chemical Co. Ltd, Beijing, China) was dissolved in absolute alcohol and diluted to 0.5% alcohol concentration with normal saline and then administered to rats by intragastric gavage after the onset of ischemia. Rats were divided into three groups as follows: (1) sham control group, which underwent sham operation and received vehicle; (2) MCAO group, which was subjected to permanent MCAO and received vehicle; (3) Zileuton group, which was subjected to permanent MCAO and treated with zileuton 50 mg/kg. The dosage of zileuton was determined according to our previous report [13], in which showed that the zileuton at 50 mg/kg could produce the best protective effect on the brain of rats subjected to permanent focal cerebral ischemia. Rats were treated with normal saline as the vehicle control at the same volume and time point as zileuton.
Assessment of Neurological Deficit
Neurological deficit was assessed at 6 and 24 h after permanent MCAO according to the previous method described by Bederson et al. [14], as follows: 0, no observable deficit; 1, contralateral forelimb flexion; 2, decreased resistance to lateral push without circling; 3, circling to the contralateral side.
Assessment of Cerebral Infarction
Rats were sacrificed under deep anesthesia, brains were rapidly removed and coronally sliced into 2.0-mm-thick sections. Brain slices were incubated in 2% 2,3,5-triphenyltetrazolium chloride (TTC, Amresco) for 20 min. The infarcted brain tissue appeared white, whereas the non-infarcted region appeared red. Then, all slices were fixed by immersion in 4% formaldehyde solution overnight. The sections were digitized, and the infarct areas were measured using Photoshop software by tracing around the white area in each brain section. Infarct volume was calculated according to the following formula: V = t × (A 1 + A 2 + . . .An). V is the infarct volume, t is the thickness of slice, and A is the infarct area. The degree of cerebral infarction was expressed as a percent volume of the contralateral hemisphere. Correction for edema of infarct area was performed as described by Lin et al. [15].
Biochemical Analysis
The infiltration of neutrophils was evaluated by measuring the enzymatic activity of myeloperoxidase (MPO). MPO activity in rat brain was measured according to the manufacturer's instructions from the assay kit (Nanjing Jiancheng Bioengineering Institute, China). The results were expressed as unit per gram tissue.
The lipid peroxidation was estimated by measuring the formation of malondialdehyde (MDA). The content of MDA in rat brain was detected according to the manufacturer's instructions from the assay kit (Nanjing Jiancheng Bioengineering Institute, China). The results were expressed as micromole per gram protein.
Nitric oxide (NO), the reaction product of iNOS, is usually used as an indicator of the enzymatic activity of iNOS. The content of NO in rat brain was determined according to the manufacturer's instructions from the assay kit (Nanjing Jiancheng Bioengineering Institute, China). The results were expressed as micromole per gram protein.
Histological Examination
Rats were deeply anesthetized and perfused with 4% paraformaldehyde in phosphate-buffered saline (PBS, pH 7.4). Then the brains were removed, fixed, and embedded in paraffin. The paraffin-embedded sections (4 μm thick) were deparaffinized with xylene and rehydrated with graded alcohol for hematoxylin and eosin (HE) staining and immunohistochemical analysis. For HE staining, coronal sections were counterstained with hematoxylin and eosin. The sections were visualized with a microscope (Olympus, Japan).
Immunohistochemical Analysis
For immunohistochemical analysis, the paraffin-embedded sections (4 μm thick) were deparaffinized with xylene and rehydrated with graded alcohol. Then, brain sections were incubated in 3% H2O2 for 15 min to block endogenous peroxidase activity. After washing in PBS, sections were incubated overnight at 4°C with a mouse monoclonal antibody against NF-κB p65 (1:50, Santa Cruz, USA). Biotinylated goat anti-mouse IgG (Zhongshan Biotechnology CO. LTD, Beijing, China) was used as a secondary antibody. Diaminobenzidine was used as a color substrate. PBS was applied instead of the primary antibody as a negative control. The sections were visualized with a microscope (Olympus, Japan).
Western Blot
Samples from ischemic brain tissue were used for experiments and total protein was extracted using protein extraction kit (Beyotime Biotech. CO., China) according to the manufacturer's instructions. Protein samples (100 μg) were separated on 8% SDS polyacrylamide gels, then transferred to nitrocellulose (NC) membranes and blocked in 5% nonfat dry milk buffer. The membranes were incubated overnight at 4°C with a mouse monoclonal antibody against NF-κB p65 (1:200, Santa Cruz, USA), or a rabbit polyclonal antibody against iNOS (1:500, Santa Cruz, USA). After extensive rinsing with Tris-buffered saline with Tween-20 buffer, the NC membranes were incubated with horseradish-peroxidase conjugated secondary antibodies (1:2,000, KPL Inc, USA) for 2 h at 25°C and developed with the use of an enhanced chemiluminescence detection system (KPL Inc, USA). GAPDH was used as a loading control. The optical densities of protein bands were analyzed by the Quantity one software (Bio-Rad).
Reverse Transcription-Polymerase Chain Reaction
Total RNA was extracted from brain sample using Trizol reagents (Invitrogen, USA) and reverse-transcribed to obtain single-strand cDNA using a Reverse Transcription System (Promega, USA) according to the manufacturer's instructions. Single-strand cDNA was amplified by PCR in a 100-μl reaction mixture containing 50 mM KCl, 10 mM Tris-HCl (pH 9.0), 2 mM MgCl2, 200 μM dNTPs, 0.5 μM sense and antisense primers, and 2.5 units Taq DNA polymerase (Promega, USA). The primer sequences were as follows: iNOS (primers, sense 5′-ATC CCG AAA CGC TAC ACT T-3′; antisense 5′-TCT GGC GAA GAA CAA TCC-3′, product size 314 bp), β-actin as an internal standard (primers, sense 5′-CAC CCT GTG CTG CTC ACC GAG GCC-3′; antisense 5′-CCA CAC AGA TGA CTT GCG CTC AGG-3′, product size 720 bp). The reactions were initially heated at 94°C for 4 min, then at 94°C for 40 s, 55°C for 40 s, and 72°C for 50 s, totally 40 cycles, finally stopped at 72°C for 7 min. PCR products were electrophoresed through 2% agarose gels containing ethidium bromide (0.5 µg/ml). The optical densities of DNA bands were analyzed using the UVP gel analysis system (Quantity one, Bio-Rad).
Serum Content of TNF-α and IL-1β
Twenty-four hours after the induction of MCAO, blood samples (1 ml) were drawn from femoral vein of rats. After centrifugation at 3,000 r/min for 15 min, the supernatant was collected and stored at –80°C in refrigerator. Serum contents of inflammatory cytokines (TNF-α and IL-1β) were measured using a rat TNF-α or IL-1β immunoassay enzyme-linked immunosorbent assay kits (R&D Systems) according to the manufacturer's instructions.
Statistical Analysis
Experimental data were presented as mean ± SEM. Statistical analysis was performed using ANOVA followed by Bonferroni test for individual comparisons between group means. A value P < 0.05 was considered statistically significant.
Results
Effect of Zileuton on Neurological Deficit and Cerebral Infarction
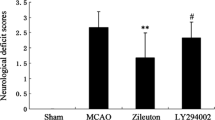
MCAO caused an obvious increase of neurological deficit scores and cerebral infarct volume. Zileuton administration significantly reduced the neurological deficit scores 6 and 24 h after MCAO (p < 0.05 and p < 0.01, Fig. 1b), it also significantly reduced cerebral infarct volume 6 and 24 h after MCAO (p < 0.01, Fig. 1a, c). Sham-operated rats did not have any neurological deficit and cerebral infarction.
Effect of zileuton on neurological deficit and cerebral infarction after MCAO. Representative TTC stained sections were presented where rats were subjected to permanent MCAO (6 and 24 h) and treated with vehicle or zileuton 50 mg/kg (a), and the results were summarized in (c). Neurological deficit scores (b) were significantly reduced by zileuton 50 mg/kg at both 6 and 24 h after MCAO (p < 0.01). Bars represent mean ± SEM (n = 6).
Morphological Changes
HE staining showed the morphological characteristic of neurons from sham-operated, vehicle-treated, and zileuton-treated rats. In sham-operated rats, no neuronal injury was observed in brain tissue (Fig. 2a, d). About 6 and 24 h after the induction of MCAO, most neurons in the ischemic brain tissue appeared shrunken with triangulated pyknotic nuclei and cavitation, necrotic neurons lacked normal cellular architecture (Fig. 2b, e). Zileuton reduced the necrotic or ischemic injured neurons at both 6 and 24 h after MCAO (Fig. 2c, f).
Morphological changes of cortical neurons following permanent MCAO was determined by HE staining. Representative microphotographs of neuronal injury from sham-operated, vehicle-treated, and zileuton-treated rats at 6 h (a–c) and 24 h (d–f) after MCAO were shown. Necrotic and ischemic injured neurons were obviously reduced by zileuton administration. Scale bar = 50 µm.
Effect of Zileuton on Neutrophil Infiltration
MPO activity was measured for quantitative indication of neutrophil infiltration and inflammatory reaction in ischemic brain. Experimental results showed that MPO activities were increased at both 6 and 24 h after MCAO compared with sham control (p < 0.01), both of which were significantly inhibited by zileuton administration (p < 0.05 and p < 0.01, Fig. 3a).
Effect of zileuton on MPO activity and MDA content in rat brain after MCAO. a Assay of MPO activities in rat brain at 6 and 24 h after MCAO. Zileuton administration significantly inhibited the enzymatic activities of MPO at 6 h (p < 0.05) and 24 h (p < 0.01) after MCAO. b Assay of MDA content in rat brain at 6 and 24 h after MCAO. Zileuton administration significantly decreased the MDA content at 6 h (p < 0.01) and 24 h (p < 0.05) after MCAO. n = 6; number sign, p < 0.01 compared with sham control.
Effect of Zileuton on Lipid Peroxidation
The content of MDA in rat brain, an index of lipid peroxidation, was increased at both 6 and 24 h after MCAO compared with sham control (p < 0.01), both of which were significantly decreased by zileuton administration (p < 0.01 and p < 0.05, Fig. 3b).
Effect of Zileuton on the NF-κB p65 Expression
Immunohistochemical results showed that NF-κB p65 immunoreactivity was little in sham-operated rat (Fig. 4a), but dramatically increased in ischemic brain 24 h after MCAO (Fig. 4b). Zileuton administration obviously reduced the NF-κB p65 immuno-positive cells (Fig. 4c). Western blot analysis showed an increase of NF-κB p65 in ischemic brain of vehicle-treated MCAO rats compared with sham control (p < 0.01), which was significantly inhibited by zileuton administration (p < 0.01, Fig. 4d, e).
Effect of zileuton on the NF-κB p65 expression in rat brain after MCAO. a–c Representative microphotographs of NF-κB p65 immuno-positive cells in ipsilateral hemisphere from sham-operated, vehicle-treated, and zileuton-treated rats. Scale bar = 50 µm. Representative protein bands of NF-κB p65 expression in sham-operated, vehicle-treated, and zileuton-treated groups were detected by Western blot (d), and the data were summarized in (e). n = 6; number sign, p < 0.01 compared with sham control, **p < 0.01 compared with vehicle-treated group.
Effect of Zileuton on the Expression and Enzymatic Activity of iNOS
MCAO caused an up-regulated expression of both iNOS mRNA and iNOS protein in rat brain at 24 h when compared with sham control (p < 0.01, Fig. 5a–d). NO, the reaction product of iNOS, was also increased in rat brain 24 h after MCAO (p < 0.01, Fig. 5e). Zileuton administration significantly down-regulated both the mRNA and protein expression of iNOS (p < 0.01 and p < 0.05, Fig. 5a–d), it also decreased the NO production in ischemic brain following permanent MCAO (p < 0.05, Fig. 5e).
Effect of zileuton on the expression and enzymatic activity of iNOS in rat brain after MCAO. Representative bands of iNOS mRNA expression in sham-operated, vehicle-treated and zileuton-treated groups were detected by RT-PCR (a), and the data were summarized in (c). Representative bands of iNOS protein expression in sham-operated, vehicle-treated, and zileuton-treated groups were determined by Western blot (b), and the data were summarized in (d). MCAO caused an elevation of NO product in rat brain, which was significantly decreased by zileuton administration (e). n = 6; number sign, p < 0.01 compared with sham control, *p < 0.05 and **p < 0.01 compared with vehicle-treated group.
Effect of Zileuton on the Content of TNF-α and IL-1β in Serum
Serum content of TNF-α and IL-1β were increased 24 h after MCAO compared with sham control, both of which were significantly attenuated by zileuton administration (p < 0.05 and p < 0.01, Fig. 6).
Effect of zileuton on the serum content of TNF-α and IL-1β in rats of permanent MCAO. MCAO caused an increased content of TNF-α and IL-1β in serum, both of which were significantly attenuated by zileuton administration. n = 6, number sign, p < 0.01 compared with sham control, *p < 0.05 and **p < 0.01 compared with vehicle-treated group.
Discussion
Accumulating evidence showed that cerebral ischemia is accompanied by a marked inflammatory reaction, which is initiated by ischemia-induced expression of inflammatory enzymes, cytokines, and other inflammatory mediators [4, 16]. 5-LOX, a key enzyme in LT biosynthesis, is implicated in several inflammation-associated diseases [17], including acute ischemic stroke [7]. Zileuton, a selective 5-LOX inhibitor, possesses the anti-inflammatory property and exerts an inhibitory effect on the inflammatory diseases [18, 19]. Our previous study showed that zileuton reduces cerebral infarct volume and brain edema 24 h after the onset of ischemia in rats [13]. In this study, we first demonstrated that the administration of zileuton significantly reduces neurological deficit scores, cerebral infarct volume, and neuronal injury at both 6 and 24 h after permanent MCAO. Then, we further investigated the mechanisms underlying the neuroprotective effect of zileuton on permanent focal cerebral ischemia. This is the first paper, to our best knowledge, indicating that zileuton could inhibit inflammatory reaction and down-regulate the expression of inflammatory mediators in a rat model of permanent MCAO.
In the present study, we found that zileuton inhibits the enzymatic activity of MPO in rats of permanent MCAO. MPO, an enzyme exclusive to neutrophils, was usually examined for quantitative indication of the presence of inflammatory reaction in the ischemic brain [20]. The hallmark of the inflammatory reaction in cerebral ischemia is the initial infiltration of neutrophils into the ischemic area of brain parenchyma [21]. Several studies showed that the infiltration of neutrophils into the brain parenchyma following an ischemic stroke leads to an expansion of cerebral infarction, and the depletion of neutrophil infiltration has been demonstrated to reduce infarct volume [22]. Recent study demonstrated that zileuton exerts a protective effect on mouse experimental spinal cord injury via inhibiting neutrophil infiltration. Accordingly, the inhibitory effect of zileuton on neutrophil infiltration in rat experimental ischemic stroke in our experiments may partially explain zileuton's neuroprotection in cerebral ischemia [23]. In addition, MDA content, an indicator of lipid peroxidation, was elevated in ischemic brain after permanent MCAO in this study, which verified the oxidative damage in ischemic tissue. Our results demonstrated that zileuton reduced MDA content in rats of permanent MCAO, therefore, the underlying neuroprotection of zileuton might be in part associated with the inhibitory effect of zileuton on the oxidative damage.
We demonstrated that selective 5-LOX inhibitor zileuton inhibited the activation of NF-κB and reduced the expression and activation of iNOS in rats of permanent MCAO. NF-κB is an inflammation responsive transcription factor known to be activated in response to ischemic stroke [10]. Inhibition of NF-κB activation results in a significant reduction of brain damage in both transient and permanent cerebral ischemia models, suggesting that NF-κB plays a detrimental role in ischemic stroke [24, 25]. In addition, NF-κB has been shown to regulate the expression of iNOS and other inflammatory mediators that contributes to the evolution of ischemic brain damage. Attenuation of iNOS expression and NO production has been demonstrated to protect brain against ischemic damage [26, 27]. There are some differences in pathophysiological processes between the transient MCAO and permanent MCAO models, however, our results concerning the neuroprotective mechanism of zileuton in permanent MCAO was in part consistent with the previous study [8] which demonstrated that another selective 5-LOX inhibitor BW-B 70C down-regulated the expression of iNOS via inhibiting the activation of NF-κB in transient MCAO.
In cerebral ischemia, NF-κB also regulates the expression of inflammatory cytokines TNF-α and IL-1β that propagate the inflammatory cascade reaction, eventually resulting in the expansion of brain damage. Brain concentrations of TNF-α and IL-1β were increased following transient cerebral ischemia in gerbils [12]. Previous research showed that activated neutrophils infiltrated in brain parenchyma could release TNF-α and IL-1β [28]. Inhibition of TNF-α and IL-1β has been shown to reduce ischemic brain damage [29, 30]. Recent report demonstrated that zileuton protects mouse experimental spinal cord injury by attenuating the expression of TNF-α [23]. Our experimental results demonstrated that the content of TNF-α and IL-1β were elevated in blood following permanent MCAO, both of which were attenuated by zileuton administration.
In summary, selective 5-lipoxygenase inhibitor zileuton is discovered to reduce brain damage both at 6 and 24 h after permanent cerebral ischemia. To our knowledge, this is the first demonstration of zileuton's effect on inflammatory reaction and inflammatory mediators in the setting of permanent focal cerebral ischemia in rats. The neuroprotective effect of zileuton in cerebral ischemia may be associated with the attenuation of inflammatory reaction and the inhibition of inflammatory mediators.
References
Dirnagl, U., C. Iadecola, and M.A. Moskowitz. 1999. Pathobiology of ischaemic stroke: an integrated view. Trends in Neurosciences 22: 391–397.
Durukan, A., and T. Tatlisumak. 2007. Acute ischemic stroke: overview of major experimental rodent models, pathophysiology, and therapy of focal cerebral ischemia. Pharmacology Biochemistry and Behavior 87: 179–197.
Lapchak, P.A., and D.M. Araujo. 2007. Advances in ischemic stroke treatment: neuroprotective and combination therapies. Expert Opinion Emerging Drugs 12: 97–112.
Huang, J., U.M. Upadhyay, and R.J. Tamargo. 2006. Inflammation in stroke and focal cerebral ischemia. Surgical Neurology 66: 232–245.
Mabuchi, T., K. Kitagawa, T. Ohtsuki, K. Kuwabara, Y. Yagita, T. Yanagihara, M. Hori, and M. Matsumoto. 2000. Contribution of microglia/macrophages to expansion of infarction and response of oligodendrocytes after focal cerebral ischemia in rats. Stroke 31: 1735–1743.
Baskaya, M.K., Y. Hu, D. Donaldson, M. Maley, A.M. Rao, M.R. Prasad, and R.J. Dempsey. 1996. Protective effect of the 5-lipoxygenase inhibitor AA-861 on cerebral edema after transient ischemia. Journal of Neurosurgery 85: 112–116.
Zhou, Y., E.Q. Wei, S.H. Fang, L.S. Chu, M.L. Wang, W.P. Zhang, G.L. Yu, Y.L. Ye, S.C. Lin, and Z. Chen. 2006. Spatio-temporal properties of 5-lipoxygenase expression and activation in the brain after focal cerebral ischemia in rats. Life Sciences 79: 1645–1656.
Jatana, M., S. Giri, M.A. Ansari, C. Elango, A.K. Singh, I. Singh, and M. Khan. 2006. Inhibition of NF-kappaB activation by 5-lipoxygenase inhibitors protects brain against injury in a rat model of focal cerebral ischemia. Journal de Neuroinflammation 3: 12.
Bonizzi, G., J. Piette, S. Schoonbroodt, R. Greimers, L. Havard, M.P. Merville, and V. Bours. 1999. Reactive oxygen intermediate-dependent NF-kappaB activation by interleukin-1beta requires 5-lipoxygenase or NADPH oxidase activity. Molecular and Cellular Biology 19: 1950–1960.
Ridder, D.A., and M. Schwaninger. 2009. NF-kappaB signaling in cerebral ischemia. Neuroscience 158: 995–1006.
Iadecola, C., F. Zhang, S. Xu, R. Casey, and M.E. Ross. 1995. Inducible nitric oxide synthase gene expression in brain following cerebral ischemia. Journal of Cerebral Blood Flow and Metabolism 15: 378–384.
Saito, K., K. Suyama, K. Nishida, Y. Sei, and A.S. Basile. 1996. Early increases in TNF-alpha, IL-6 and IL-1 beta levels following transient cerebral ischemia in gerbil brain. Neuroscience Letters 206: 149–152.
Tu, X.K., W.Z. Yang, S.S. Shi, C.M. Chen, and C.H. Wang. 2009. 5-Lipoxygenase inhibitor zileuton attenuates ischemic brain damage: involvement of matrix metalloproteinase 9. Neurological Research 31: 848–852.
Bederson, J.B., L.H. Pitts, M. Tsuji, M.C. Nishimura, R.L. Davis, and H. Bartkowski. 1986. Rat middle cerebral artery occlusion: evaluation of the model and development of a neurologic examination. Stroke 17: 472–476.
Lin, T.N., Y.Y. He, G. Wu, M. Khan, and C.Y. Hsu. 1993. Effect of brain edema on infarct volume in a focal cerebral ischemia model in rats. Stroke 24: 117–121.
Iadecola, C., and M. Alexander. 2001. Cerebral ischemia and inflammation. Current Opinion in Neurology 14: 89–94.
Martel-Pelletier, J., D. Lajeunesse, P. Reboul, and J.P. Pelletier. 2003. Therapeutic role of dual inhibitors of 5-LOX and COX, selective and non-selective non-steroidal anti-inflammatory drugs. Annals of the Rheumatic Diseases 62: 501–509.
Bell, R.L., P.R. Young, D. Albert, C. Lanni, J.B. Summers, D.W. Brooks, P. Rubin, and G.W. Carter. 1992. The discovery and development of zileuton: an orally active 5-lipoxygenase inhibitor. International Journal of Immunopharmacology 14: 505–510.
Patel, N.S., S. Cuzzocrea, P.K. Chatterjee, R. Di Paola, L. Sautebin, D. Britti, and C. Thiemermann. 2004. Reduction of renal ischemia-reperfusion injury in 5-lipoxygenase knockout mice and by the 5-lipoxygenase inhibitor zileuton. Molecular Pharmacology 66: 220–227.
Lerouet, D., V. Beray-Berthat, B. Palmier, M. Plotkine, and I. Margaill. 2002. Changes in oxidative stress, iNOS activity and neutrophil infiltration in severe transient focal cerebral ischemia in rats. Brain Research 958: 166–175.
Weston, R.M., N.M. Jones, B. Jarrott, and J.K. Callaway. 2007. Inflammatory cell infiltration after endothelin-1-induced cerebral ischemia: histochemical and myeloperoxidase correlation with temporal changes in brain injury. Journal of Cerebral Blood Flow and Metabolism 27: 100–114.
Beray-Berthat, V., N. Croci, M. Plotkine, and I. Margaill. 2003. Polymorphonuclear neutrophils contribute to infarction and oxidative stress in the cortex but not in the striatum after ischemia-reperfusion in rats. Brain Research 987: 32–38.
Genovese, T., A. Rossi, E. Mazzon, R.D. Paola, C. Muia, R. Caminiti, P. Bramanti, L. Sautebin, and S. Cuzzocrea. 2008. Effects of zileuton and montelukast in mouse experimental spinal cord injury. British Journal of Pharmacology 153: 568–582.
Nurmi, A., P.J. Lindsberg, M. Koistinaho, W. Zhang, E. Juettler, M.L. Karjalainen-Lindsberg, F. Weih, N. Frank, M. Schwaninger, and J. Koistinaho. 2004. Nuclear factor-kappaB contributes to infarction after permanent focal ischemia. Stroke 35: 987–991.
Schneider, A., A. Martin-Villalba, F. Weih, J. Vogel, T. Wirth, and M. Schwaninger. 1999. NF-kappaB is activated and promotes cell death in focal cerebral ischemia. Natural Medicines 5: 554–559.
Tu, X.K., W.Z. Yang, S.S. Shi, C.H. Wang, and C.M. Chen. 2009. Neuroprotective effect of baicalin in a rat model of permanent focal cerebral ischemia. Neurochemical Research 34: 1626–1634.
Yang, Y., X. Xia, Y. Zhang, Q. Wang, L. Li, G. Luo, and Y. Xia. 2009. Delta–opioid receptor activation attenuates oxidative injury in the ischemic rat brain. BMC Biology 7: 55.
Cassatella, M.A. 1995. The production of cytokines by polymorphonuclear neutrophils. Immunology Today 16: 21–26.
Barone, F.C., B. Arvin, R.F. White, A. Miller, C.L. Webb, R.N. Willette, P.G. Lysko, and G.Z. Feuerstein. 1997. Tumor necrosis factor-alpha. A mediator of focal ischemic brain injury. Stroke 28: 1233–1244.
Boutin, H., R.A. LeFeuvre, R. Horai, M. Asano, Y. Iwakura, and N.J. Rothwell. 2001. Role of IL-1alpha and IL-1beta in ischemic brain damage. Journal of Neuroscience 21: 5528–5534.
Acknowledgements
The key laboratory (Neurosurgical Department) funding from the Affiliated Union Hospital of Fujian Medical University, and the Professor Academic Development Fund of Fujian Medical University (JS0610), supported this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tu, Xk., Yang, Wz., Wang, Ch. et al. Zileuton Reduces Inflammatory Reaction and Brain Damage Following Permanent Cerebral Ischemia in Rats. Inflammation 33, 344–352 (2010). https://doi.org/10.1007/s10753-010-9191-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-010-9191-6