Abstract
The study of variation of species composition among sites is key to understanding community ecology, but few studies have assessed beta diversity patterns in highly dynamic stream networks in the Neotropical region. We assessed aquatic insect patterns of local contribution to beta diversity (LCBD) and species contribution to beta diversity (SCDB) in a Neotropical drainage network composed of both perennial and intermittent streams in a dry period. We evaluated if environmental and/or spatial predictors drive patterns of LCBD. We sampled aquatic insects in 12 intermittent headwater streams and 34 perennial streams. The intermittent compared to perennial streams had higher LCBDs and lower richness. The pure environmental component significantly explained 19% of the variation of LCBD, while the pure spatial components were not significant. Forty-six taxa contributed to beta diversity above the mean of the 199 taxa. We detected the association of oxygen tolerant and good dispersal ability taxa to intermittent streams and species riffle-adapted taxa as indicators of perennial streams. We showed a disproportional contribution of intermittent streams to the regional species pool. In summary, we demonstrated that when streams dry out, compositional uniqueness may increase during the dry period making them critical to conservation planning of dynamic stream networks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The study of spatial variation in species composition (i.e., beta diversity) is a cornerstone of community ecology and it is useful for defining conservation actions (Socolar et al., 2016). Recently, Legendre & De Cáceres (2013) developed a single-number estimate of beta diversity through the calculation of the total variance of the species composition. This method allows the assessment of two other measures: species contributions to beta diversity (SCDB) and local contributions to beta diversity (LCBD) (Legendre & De Cáceres, 2013). The latter is an estimate of the uniqueness of sampling sites in terms of species composition (i.e., unusual species combinations), which can be used to understand drivers of beta diversity (e.g., Landeiro et al., 2018) and may indicate site’s conservation value (Legendre & De Cáceres, 2013). This conservation value may be especially interesting when high LCBD values coincide with low speciose sites, a common pattern in stream communities (Legendre & De Cáceres, 2013; Heino & Grönroos, 2017; Landeiro et al., 2018). This pattern would indicate sites that disproportionally contribute to regional species pool relative to species richness and may be particularly useful to identify keystone sites (Legendre & De Cáceres, 2013; Ruhí et al., 2017).
Correlative methods between LCBD patterns and environmental variables can be used to better understand LCBD drivers. Because LCBD represents an estimate of the uniqueness of the sampling sites in terms of species composition, higher values are expected in sites with specific environmental conditions. In addition, spatially distributed environmental variables, dispersal-related processes, and stochastic events can create strong spatial patterns in LCBD, leading to a correlation of composition uniqueness with spatial variables. Disentangling these two possible drivers can shed light on mechanisms behind compositional uniqueness patterns, a fundamental topic in metacommunity ecology (Leibold et al., 2004; Leibold & Chase, 2018). In this context, Landeiro et al. (2018) explored 14 biological dataset groups in Amazonia and reported that LCBD in plants was mainly explained by environmental conditions—soil clay content, slope, and distance to the nearest stream—and in animals by both environmental conditions—distance to the nearest stream and soil phosphorus content—and large-scale spatial variables. In lotic systems, macroinvertebrate compositional uniqueness are better explained by environmental variables than spatial predictors (Tonkin et al., 2016; Sor et al., 2018; Tolonen et al., 2018), despite some studies reported low predictability in beta diversity patterns and also that these patterns may be explained by stochastic forces (Heino et al., 2015; Leibold & Chase, 2018; Valente-Neto et al., 2018a).
River networks are naturally complex hierarchical landscapes connected by unidirectional flow, in which headwaters coalesce to form large rivers (Altermatt, 2013). In addition to this complexity, some drainage networks are composed of both perennial and intermittent streams in a given period of time (hereafter called highly dynamic stream networks), and shifts can occur seasonally among these two states along the year (Datry et al., 2016a). Flow intermittence can be caused by transmission loss, cessation of spring discharge or groundwater discharge (Datry et al., 2017a). Recently, intermittent streams have been included more explicitly in the freshwater ecology agenda (Datry et al., 2014, 2016a) and studies demonstrated that they comprise nearly 50% of global river length, and provide significant ecosystem services, such as water provision and purification, carbon storage, and nutrient cycling (Datry et al., 2017a).
Highly dynamic stream networks may create conditions for the species co-occurrence at regional scale, of both lotic (e.g., riffle-adapted organisms) and lentic adapted fauna (e.g., oxygen tolerant and good dispersal ability organisms) during the non-flowing cycle of intermittent streams (i.e., dry season—all intermittent streams without flow, characterized by dry sections and isolated pools). This lack of flow would typically increase total beta diversity compared to flowing season (i.e., wet season—all streams with running waters). The presence of both intermittent and perennial streams in such networks strengthens the environmental gradient and may increase the expectation of environmental selection structuring aquatic insect metacommunities (Datry et al., 2016a). Local factors, including hydrological parameters, and dispersal distances accounted for most of the dissimilarity among metacommunities of freshwater invertebrates in stream network composed of intermittent and perennial streams in south-eastern Arizona (Cañedo-Argüelles et al., 2015). Despite the importance of both perennial and intermittent streams contributing to the regional species pool of stream networks, ecologists usually study the biodiversity patterns in either perennial or intermittent streams (e.g., Datry et al., 2014; but see Cañedo-Argüelles et al., 2015). Furthermore, few studies have assessed beta diversity patterns in highly dynamic stream networks in the Neotropical region (e.g., Datry et al., 2016b). For example, intermittence (measured as environmental harshness) has been shown to mediate the role of dispersal and environmental selection on community similarity in headwater streams from Bolivia (Datry et al., 2016b). However, in general, the contribution of intermittent streams to regional biodiversity of highly dynamic stream network is poorly known.
We investigated aquatic insect compositional uniqueness (LCBD) in a highly dynamic stream network (composed by both perennial and intermittent streams) from the Neotropical region in the dry season and its correlation with taxa richness. We also tested whether LCBD was explained by environmental and spatial predictors. We predicted higher LCBD values for intermittent streams than for perennial ones because the environmental selection due to changes in the flow of water (Boulton, 2003) would filter out species in intermittent streams relative to perennial ones, increasing LCBD of intermittent streams. The presence of both perennial and intermittent streams in the stream network can create strong environmental variability. Consequently, we expected environmental selection would better explain LCBD patterns of aquatic insects. We also assessed species contributions to beta diversity (SCBD) and investigated which species are indicators of permanent and intermittent streams. We predicted that oxygen-tolerant taxa, and insects with good dispersal ability would have higher SCBD values and greater probability to be indicator species of intermittent streams. We also predicted that riffle-adapted species would have higher SCBD and greater probability to occur and to be abundant in perennial streams.
Materials and methods
Study area and sampling
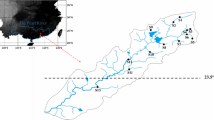
We selected 46 sites along the Betione river network, located in the Bodoquena Plateau, southwest Mato Grosso do Sul, Brazil. The region is transitional between two Brazilian biodiversity hotspots, Cerrado (Brazilian savanna) and Atlantic Forest (Myers et al., 2000). Streams and rivers from Bodoquena Plateau drain into Pantanal floodplain, one of the largest wetlands in the world (Tomas et al., 2019). The region is marked by a dry (April to September) and wet (October to March) season. The river network flows through karstic geology and contains areas of sub-surface flow and fluctuating groundwater discharge, common causes of stream intermittence in karstic regions (Datry et al., 2017a).
Stream intermittence can be established by different criteria (Datry et al., 2017a). Here we used two. First, for each stream, we delimited a minimum of 200 m reach to assess visually areas of flow cessation, characterized by dried riffles and a series of isolated sections and pools. Second, because all sites studied were located on farm lands, we interviewed landowners and asked them if the selected streams often undergo annual drying events. We only considered intermittent streams as those streams that were known to have flow cessation at the moment of sampling and frequently flow-cessation periods (informed by landowners). In this way, we selected 12 intermittent headwater streams, all of them located in the middle and final portion of the stream network (Fig. 1). The remaining sites (34) were perennial (flowing year-round) streams, of which seven sites were third-order channels.
Aquatic insect sampling was carried out in September 2013, during the non-flowing phase of intermittent streams, and at the end of dry season and the at beginning of regional wet season. The period of sampling was marked by only 27 mm of precipitation during the month of September (INMET, 2018), distributed in 6 days (ranging from a maximum of 9 mm and a minimum of 1 mm). Sites in intermittent streams were close to headwater areas, with some spring or groundwater discharges that could maintain isolated pool areas. In the field, we observed that downstream areas of the intermittent streams sampled were dried. Each sample included the sum of 20 kick net (0.5 mm mesh size) sub-samples per stream, proportionally distributed between all major habitats available (Barbour et al., 1996). We visually estimated the major habitats in three reaches of 10 m, including rock outcrops, rock cobble, gravel, sand, mud silt, organic matter, wood, aquatic vegetation, leaf litter, and roots. Then, we averaged the proportion of habitats from the three 10 m reaches and proportionally distributed the sampling units among habitats. Each sub-sample consisted of 1 m length using a kick net (covering 0.3 m2), totaling a sampling effort of 6 m2 of stream-bottom habitats. The 20 sub-samples were pooled, preserved in formaldehyde in the field, and transported to the laboratory for sorting and identification. At the laboratory, samples were processed and insects were sorted on transilluminated trays and identified to the lowest viable taxonomic level (mostly to the genus level). Due to restricted taxonomic knowledge, some Diptera (Ceratopogonidae, Culicidae, Tipulidae, Empididae, Ephydridae, Dixidae, and Stratiomyidae) and Coleoptera (Curculionidae, Lampyridae, and Scirtidae) were identified at family level. These dipterans and coleopterans comprised less than 5% of the total abundance found in our study.
Environmental variables
In each site, we measured several environmental variables, including water parameters (pH, dissolved oxygen, temperature, conductivity, turbidity, velocity), physical features (stream width, depth, altitude, shading), landscape measures (vegetation cover), and intermittence of flow (dummy variable). We took three measures of water parameters to estimate the mean for each 30 m reach. Stream widths and depths were measured every 6 m along the 30 m reach and the five measures were averaged. We took three digital photographs of the canopy to estimate shading by riparian cover (using the software ImageJ). The vegetation cover was estimated from a 200-m riparian buffer and a vegetation cover image (30 m resolution) provided by Environmental Institute of the state of Mato Grosso do Sul (IMASUL, 2014). We used only environmental variables that had correlation values less than 0.8 (Dormann et al., 2013). Considering this criterion, we excluded turbidity because its correlation with conductivity exceeded 0.8. All these variables were included in the environmental matrix.
Spatial variables
Geographical distances are the base information to model spatial structures. We calculated watercourse distance between each pair of two sampling sites following the river network using three sources of information: (i) Betione network flowline vector provided by Brazilian Institute of Geography and Statistics (IBGE) (scale 1:250,000); (ii) high-resolution Google Earth images (1 m); and (iii) geographic position of sites. We used (ii) and (iii) to correct distortions present in (i) due to the low spatial resolution. We used the ArcGis (version 10.1 ESRI, Redlands, California, USA) (Network Analysis Toolbox/OD Cost Matrix Analysis Tool) to calculate the watercourse distances between sites.
Distance-based Moran’s eigenvector maps (dbMEM) were applied on watercourse distances to model spatial structures of sampling sites (Borcard & Legendre, 2002; Dray et al., 2006). For each matrix, the minimum spanning tree distance that keeps all sites connected was used as truncation threshold to construct the truncated matrix. The truncated matrix was submitted to a Principal Coordinate Analysis (PCoA), and the eigenvectors with significant patterns of spatial autocorrelation, i.e., with significant (P < 0.05) and positive Moran’s I (Sokal & Oden, 1978) were selected. Eigenvectors represent distinct spatial structures of relationships among the sampling sites, from broad-scale to fine-scale patterns (Dray et al., 2006; Griffith & Peres-Neto, 2006). We used the selected eigenvectors (MEMs) as spatial explanatory variables in data analyses.
We recognized that geographical distances between sampling sites can be calculated in different ways, especially in stream ecology where the dendritic structure can matter for species dispersal (Schmera et al., 2018; Tonkin et al., 2018). Considering that aquatic insects can use different dispersal routes (Tonkin et al., 2018), we also tested overland (straight line distance between two sampling sites) and directional (asymmetric eigenvector maps) distances. However, because they did not change the pattern we detected, the results were not reported here (but see Supplementary material—section Other spatial structures).
Data analysis
We calculated total beta diversity (BDtotal), the local contribution to beta diversity (LCBD), and species contribution to beta diversity (SCBD) (Legendre & De Cáceres, 2013). We first applied Hellinger transformation to community composition and, then, estimated BDtotal as the unbiased total sum of square of the species composition data, which can be used to compare beta diversity between studies. We did not partition beta diversity into nestedness and turnover components because they both equally contributed to beta diversity (see Supplementary material—section Partitioning beta diversity). Computing beta diversity as the total variation in the species composition data allowed us to assess LCBD, which is the relative contribution of each sampling unit to beta diversity (Legendre & De Cáceres, 2013). This measure was calculated by the division of sum of squares corresponding to each sampling unit by the total sum of square of the species composition data. We used Pearson correlation to test the relationship between LCBD and taxa richness.
To determine which taxa mostly contributed to beta diversity patterns of intermittent and perennial streams, we used two approaches. First, we retained those taxa with SCBD values greater than the mean of all taxa (i.e., species that has a disproportional contribution to beta diversity patterns) (Legendre & De Cáceres, 2013). SCBD represents the degree of variation of individual species in the study area (Legendre & De Cáceres, 2013). This subset of taxa was used in species indicator analysis (IndVal) (Dufrêne & Legendre, 1997) to identify indicator species characterizing intermittent and perennial streams. To calculate indicator species, IndVal uses within-species abundance (specificity) and occurrence (fidelity) comparisons between groups. A random reallocation of sites among groups (1000 permutations) was used to test the significance of indicator values.
Commonly, variation partitioning is used in linear models to verify if the variation in a response variable is affected by environmental and/or spatial variables. However, variation partitioning is only required when both biological and environmental matrices are spatially structured, which would be necessary to filter out the effects of spatial correlation of environmental predictors (Legendre et al., 2002; Peres-Neto & Legendre, 2010). In this way, we separately ran environmental and spatial global models (carried out a priori and including all variables) to estimate significance (in our case P < 0.05) and adjusted R2 (global \(R_{\text{adj}}^{2}\)). Then, if a given model was significant, we used forward selection as implemented by Blanchet et al. (2008) that requires two fixed criteria to select variables from an explanatory matrix: the significance (P < 0.05) and \(R_{\text{adj}}^{2}\) have to be below the global \(R_{\text{adj}}^{2}\) (Blanchet et al., 2008). Forward selection was not used for non-significant global models and, if this condition was true, we just reported individual multiple regression model after forward selection. If both global models were significant, we applied variation partitioning to decompose the LCBD variation into four components: pure environmental component (a), the amount of variation shared by environmental component and spatial component (b), pure specific spatial component (c), and non-explained variation (residual) (d). The significance of fractions (a) and (c) were tested via permutation-based tests of partial multiple regressions models.
We used R language to perform all analyses, using vegan (Oksanen et al., 2009), packfor (Dray et al., 2011), labdsv (Roberts, 2016), and adespatial packages (Dray et al., 2017).
Results
We collected 17,560 insects belonging to 199 taxa. The mean abundance at each site was 381 individuals. Richness ranged from 9 to 74 taxa. The total beta diversity was 0.62. The mean local contribution to beta diversity was 0.02 (ranging from 0.011 to 0.043) (Fig. 2). Sites with the highest values (LCBD ≥ 0.030) had significant LCBDs (seven sites, six intermittent), while sites with values lower than 0.030 had not significative LCBDs. LCBD was negatively correlated with taxa richness (Pearson correlation = − 0.42, P = 0.003; Fig. 3a).
Relationship between local contribution to beta diversity (LCBD) of aquatic insects and a richness, b intermittence, and c conductivity. Aquatic insects were sampled in 46 sites from Betione river network. Dark blue and light blue dots in a denote intermittent and perennial streams, respectively. 0 and 1 in c denote perennial and intermittent streams, respectively. Shadings represent the confidence interval (0.95) for the linear model fitted
Forty-six taxa contributed to beta diversity above the mean of the 199 taxa (Table S1). Indicator value analysis showed that intermittent streams had nine indicator species, most of them Coleoptera (Curculionidae, Derallus, Scirtidae, Tropisternus, and Enochrus), and the remaining were Culicidae (Diptera) and Acanthagrion (Odonata) (Table S1). Perennial sites had four indicator species, three chironomids (Endotribleos, Stenochironomus, and Tanytarsus) and one riffle beetle (Hexacylloepus) (Table S1).
The environmental global model was significant (P = 0.01; adjusted R2 = 0.29) and the variables selected were conductivity and intermittence. Unlike, spatial global model were not significant (watercourse: P = 0.31; adjusted R2 = 0.03) and, consequently, we did not proceed with variation partitioning and just reported synthetic environmental model. Environmental component composed by conductivity and intermittence significantly explained variance in LCBD values (adjusted R2 = 0.19; P = 0.004; F = 6.40). The relationships between LCBD and environmental variables showed that LCBD increase with intermittence (i.e., intermittent streams had higher values of LCBDs compared to perennial ones) and decrease with conductivity (Fig. 3b, c).
Discussion
We found that intermittent streams had higher values of LCBDs compared to perennial streams in a highly dynamic stream network. The variation in the LCBDs was explained by local environmental variables, mainly intermittence and conductivity. This latter result demonstrated that environmental selection better explained aquatic insects community pattern compared to dispersal-related, agreeing with previous studies (Warfe et al., 2013; Cañedo-Argüelles et al., 2015; Sor et al., 2018; Tolonen et al., 2018; Valente-Neto et al., 2018b). The disproportional contribution of intermittent streams to the stream network can be interpreted as keystone sites in a metacommunity context (Mouquet et al., 2013; Ruhí et al., 2017). This result makes them critical to conservation strategies of dynamic aquatic systems.
Intermittent streams are extremely important worldwide, provide many ecosystem services, such as water provision, carbon and nutrient cycling, and habitat for aquatic biodiversity (Acuña et al., 2014; Datry et al., 2014). Our results showed that these systems contain a unique and small set of organisms (low richness) of aquatic insects compared to perennial streams (see also Soria et al., 2017). This pattern indicated that intermittent streams disproportionally contribute to beta diversity of a highly dynamic stream network. The negative relationship between LCBD and richness showed here is frequently reported in aquatic and terrestrial systems (Legendre & De Cáceres, 2013; Heino & Grönroos, 2017; Landeiro et al., 2018). This negative relationship can be the result of the loss of connectivity among intermittent streams that eliminates the downstream drift of aquatic insect from upstream habitats combined with the harshness of local conditions for many lotic species and stronger biotic interactions, such as predator aggregation (Datry, 2012; Datry et al., 2016a). Similarly, in a river network from eastern France, most intermittent sites contained few species of macroinvertebrates, but they had large contribution to beta diversity (Ruhí et al., 2017).
Our results also showed that environmental selection was an important driver of LCBD variation, as demonstrated by other studies in intermittent systems (Warfe et al., 2013; Cañedo-Argüelles et al., 2015). This result suggests that in the stream network studied here, aquatic insect dispersal would not be limiting nor in excess (i.e., mass effects) and species could reach suitable sites and could track environmental variation in the stream network. The absence of relationship between LCBD values and spatial pure components (see also Supplementary material—section Other spatial structures) reinforces these inferences as well as the role of environmental selection. For example, both perennial and intermittent communities of fish, macroinvertebrate and vegetation were best explained by environmental variables in Australian streams, specifically flow regime and channel width (Warfe et al., 2013). In our case, the variable selection procedure demonstrates that intermittence and conductivity were able to explain 19% of LCBD variation. These variables contributed to the overall heterogeneity of sites in the stream network and created habitat differences where species may track suitable sites. Similarly, stream invertebrates beta diversity from New Zealand were mainly driven by local habitat heterogeneity and was not affected by regional and landscape-scale variables (Astorga et al., 2014).
The effects of flow regime on stream biodiversity have a long-established history in stream ecology (Fisher et al., 1982; Poff et al., 1997; Allan & Castillo, 2007) and intermittence can profoundly change stream communities (Datry et al., 2016a, 2017b), from lotic to terrestrial fauna. We detected the association of oxygen-tolerant taxa to intermittent streams (i.e., genera of Hydrophilidae, Scirtidae, Culicidae), including some insects with good dispersal ability (e.g., Tropisternus and Acanthagrion), as also found by Bogan & Boersma (2012). Overall, most shredders were found in low abundance (e.g., Phylloicus) or they were not found in intermittent streams (e.g., some chironomids and caddisflies). The reduction in density and richness of shredders is a common pattern in intermittent streams (Martínez et al., 2015) and it can decrease organic matter processing rates compared to permanent streams (Datry et al., 2011). For example, dissolved oxygen levels are also often reduced as a result of both flow cessation and the concentration of individuals and organic matter during the initial phases of drying (Datry et al., 2016a). Whereas flow cessation is a strong environmental selection pressure to many lotic organisms, the concentration of individuals could lead to strong biotic interactions among predator and prey species. These forces combined lead to the simplification of stream communities to those with lentic tolerant species (Datry et al., 2016a), as we found. On the other hand, the water flow of perennial streams selects those species adapted to lotic systems, including many genera of riffle beetles, mayflies, and caddisflies. In this way, future studies should assess the compositional uniqueness of perennial and intermittent streams in terms of taxa functional traits.
Although we did not measure intermittence variables such as flow permanence (i.e., percentage of time with surface flow), flow regimes (frequency of maximum and minimum discharge), and the duration and frequency of drying (e.g., Datry, 2012) due to equipment and time restrictions, the categorical variable “intermittence” was based on our field observation and landowner knowledge as a good proxy of processes related to intermittence in the study area. In the future, it would be useful to incorporate the long-term temporal dynamics of Neotropical intermittent streams and evaluate whether the pattern we showed here is maintained in the different phases of intermittence (flowing, non-flowing, and drying) (Ruhí et al., 2017). Temporal dynamics may change the strength of environmental selection and dispersal-related processes on species distribution and, consequently, on LCBD patterns (Datry et al., 2016a). Ruhí et al. (2017) demonstrated that the contribution of richness and replacement components to total beta diversity of aquatic insects from France changed through time (beginning of the dry phase and beginning of flowing phase over 4 years), altering LCBD patterns. They concluded that analyzing spatial beta diversity over time improved their understanding of the variation in community composition present in highly dynamic systems and the identification of keystone sites.
Conductivity was also influential for LCBD variation and can reflect a natural variation of water characteristics due to local geology (Griffith, 2014) and concentration of solutes during drying process (Boulton, 2003). Many streams in the study area are located in karst landscapes (Silva et al., 2017) and have high conductivity because of high levels of dissolved calcium and bicarbonate. Karstic streams often have travertine deposits that cover the stream substrates, delaying organic substrates decomposition and affecting benthic aquatic insects (Casas & Gessner, 1999). In addition to the effect on decomposition, high conductivity levels may affect metabolism of aquatic insects and increase their downstream drift, mainly among mayflies (Clements & Kotalik, 2016). Some streams in the study area are not influenced by karstic zones and they typically had low and acidic waters, enhancing decomposition of organic matter.
Although our results indicated that environmental selection was more important than dispersal-related processes to explain LCBD variation, a large proportion of variation remained still to be explained (71% using all environmental variables and 81% using selected environmental variables). The low explanation of biodiversity patterns is a common pattern in lotic systems (Heino et al., 2015; Schmera et al., 2018) and the main explanation is usually stochasticity caused by flow variability (Heino et al., 2015). Flow variability includes a series of components, such as flash floods that can remove most organisms from a suitable patch (Flecker & Feifarek, 1994), leading to unexpected absences and, in the other extreme, the absence of flow (intermittence). The dynamics of intermittent stream ecosystems are largely dependent on precipitation but the flow interruption is unpredictable to most stream species although some consequences of stream drying on aquatic insects may be niche-determined (Datry et al., 2016a). Because our study was carried in the non-flowing phase of the intermittent streams and we did not have an estimative of flow interruption, it is possible that some organisms present in recent disconnected intermittent streams would not persist, creating unexpected effects (i.e., extinction debt). In a simulation modeling study parameterized by empirical data, Valente-Neto et al. (2018a) showed that a niche–stochastic assembly scenario improved predictions for river network communities in Neotropical streams. In this way, we believe the inclusion of flow variability can improve model explanation. Also, the large proportion of unexplained variation may be caused by ecological interactions and unmeasured variables.
Although the idea of identifying keystone species has a long history in ecology and conservation (Mills et al., 1993), only recently this idea was extended to communities. Mouquet et al. (2013) defined a keystone community as communities with a disproportional positive impact relative to their weight in the metacommunity, which can better inform decision-making in conservation. However, currently there is a lack of methods to operationalize this concept. Compositional uniqueness is a measure of the relative site impact in the metacommunity and, thus, a potential candidate to be part of the tool box of metrics to identify keystone communities. In this way, LCBD (a measure of contribution of local sites to the metacommunity) in combination with species richness (a measure of size of local communities) are both good candidates to estimate keystone communities (Ruhí et al., 2017). Using this approach, a keystone community would have high LCBD (impact) and low species richness (weight). Our results suggest that intermittent streams are good candidates to be keystone communities because they have a disproportional contribution to regional species pool relative to their local species number.
Intermittent streams are frequently undervalued by landowners due to their irregular flow (Armstrong et al., 2012). Many intermittent streams and rivers have been degraded due to their use as rubbish and sewage disposal and land use conversion and fragmentation (Leigh et al., 2016) or even buried (Acuña et al., 2014), affecting the biodiversity in these important habitats. Brazilian biomes are highly threatened by land conversion for agro-business expansion, especially in the Cerrado biome, where our study region is located (Strassburg et al., 2017). The lack of concern of landowners for intermittent streams combined with agro-business expansion in our study region threaten these systems. Thus, some conservation strategies are needed to maintain the disproportional contribution of intermittent streams to the regional species pool of the stream network, including (i) the detection and mapping of such streams (as we did here); (ii) the definition of targets to conserve intermittent streams (Acuña et al., 2017) using cost–benefit conservation tools, such as the systematic planning approach; (iii) the payment for ecosystem services and taxa incentives; and (iv) education of landowners and people about the ecosystem services that intermittent streams provide (Acuña et al., 2017; Leigh et al., 2019), including their higher compositional uniqueness compared to perennial streams.
Conclusion
In summary, we demonstrated that when streams dried out, the compositional uniqueness of insect communities increased, (i.e., intermittent streams had more unique communities compared to perennial streams). We also showed that site uniqueness was better explained by environmental variables than dispersal-related processes, and that conductivity and intermittence were important drivers of LCBD values. Our results agreed with recent studies emphasizing the need for protection of intermittent streams worldwide (Acuña et al., 2014; Datry et al., 2017a), due their high contribution to regional species pool and the increasing effects of climate change on stream flows (Acuña et al., 2017).
References
Acuña, V., T. Datry, J. Marshall, D. Barcelo, C. N. Dahm, A. Ginebreda, G. McGregor, S. Sabater, K. Tockner & M. A. Palmer, 2014. Why should we care about temporary waterways? Science 343: 1080–1081.
Acuña, V., M. Hunter & A. Ruhí, 2017. Managing temporary streams and rivers as unique rather than second-class ecosystems. Biological Conservation 211: 12–19.
Allan, J. D. & M. M. Castillo, 2007. Stream ecology: structure and function of running waters. Springer, Netherlands.
Altermatt, F., 2013. Diversity in riverine metacommunities: a network perspective. Aquatic Ecology 47: 365–377.
Armstrong, A., R. C. Stedman, J. A. Bishop & P. J. Sullivan, 2012. What’s a stream without water? Disproportionality in headwater regions impacting water quality. Environmental Management 50: 849–860.
Astorga, A., R. Death, F. Death, R. Paavola, M. Chakraborty & T. Muotka, 2014. Habitat heterogeneity drives the geographical distribution of beta diversity: the case of New Zealand stream invertebrates. Ecology and Evolution 4: 2693–2702.
Barbour, M., J. Gerritsen, G. Griffith, R. Frydenborg, E. McCarron, J. White, & M. Bastian, 1996. A framework for biological criteria for Florida streams using benthic macroinvertebrates. Journal of the North American Benthological Society 15: 185–211.
Blanchet, G., P. Legendre & D. Borcard, 2008. Forward selection of spatial explanatory variables. Ecology 89: 2623–2632.
Bogan, M. T. & K. S. Boersma, 2012. Aerial dispersal of aquatic invertebrates along and away from arid-land streams. Freshwater Science 31: 1131–1144.
Borcard, D. & P. Legendre, 2002. All scale spatial analysis of ecological data by means of principal coordinates of neighbor matrices. Ecological Modeling 153: 51–68.
Boulton, A. J., 2003. Parallels and contrasts in the effects of drought on stream macroinvertebrate assemblages. Freshwater Biology 48: 1173–1185.
Cañedo-Argüelles, M., K. S. Boersma, M. T. Bogan, J. D. Olden, I. Phillipsen, T. A. Schriever & D. A. Lytle, 2015. Dispersal strength determines meta-community structure in a dendritic riverine network. Journal of Biogeography 42: 778–790.
Casas, J. J. & M. O. Gessner, 1999. Leaf litter breakdown in a Mediterranean stream characterised by travertine precipitation. Freshwater Biology 41: 781–793.
Clements, W. H. & C. Kotalik, 2016. Effects of major ions on natural benthic communities: an experimental assessment of the US Environmental Protection Agency aquatic life benchmark for conductivity. Freshwater Science 35: 126–138.
Datry, T., 2012. Benthic and hyporheic invertebrate assemblages along a flow intermittence gradient: effects of duration of dry events. Freshwater Biology 57: 563–574.
Datry, T., R. Corti, C. Claret & M. Philippe, 2011. Flow intermittence controls leaf litter breakdown in a French temporary alluvial river: the “drying memory”. Aquatic Sciences 73: 471–483.
Datry, T., S. T. Larned & K. Tockner, 2014. Intermittent rivers: a challenge for freshwater ecology. BioScience 64: 229–235.
Datry, T., N. Bonada & J. Heino, 2016a. Towards understanding the organisation of metacommunities in highly dynamic ecological systems. Oikos 125: 149–159.
Datry, T., A. S. Melo, N. Moya, J. Zubieta, E. De la Barra & T. Oberdorff, 2016b. Metacommunity patterns across three Neotropical catchments with varying environmental harshness. Freshwater Biology 61: 277–292.
Datry, T., N. Bonada & A. Boulton, 2017a. Intermittent Rivers and Ephemeral Streams. Ecology and Management. Academic Press, New York.
Datry, T., R. Corti, J. Heino, B. Hugueny, R. J. Rolls, & A. Ruhí, 2017b. Habitat fragmentation and metapopulation, metacommunity, and metaecosystem dynamics in intermittent rivers and ephemeral streams. Intermittent Rivers and Ephemeral Streams: Ecology and Management. Academic Press
Dormann, C. F., J. Elith, S. Bacher, C. Buchmann, G. Carl, G. Carré, J. R. G. Marquéz, B. Gruber, B. Lafourcade, P. J. Leitão, T. Münkemüller, C. McClean, P. E. Osborne, B. Reineking, B. Schröder, A. K. Skidmore, D. Zurell & S. Lautenbach, 2013. Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36: 27–46.
Dray, A. S., G. Blanchet, D. Borcard, S. Clappe, G. Guenard, T. Jombart, G. Larocque, P. Legendre, N. Madi, & H. H. Wagner, 2017. Package ‘adespatial.’
Dray, S., P. Legendre & P. R. Peres-Neto, 2006. Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecological Modelling 196: 483–493.
Dray, S., P. Legendre, & G. Blanchet, 2011. Packfor: forward selection with permutation.
Dufrêne, M. & P. Legendre, 1997. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecological Monographs 67: 345–366.
Fisher, S. G., L. J. Gray, N. B. Grimm & D. E. Busch, 1982. Temporal succession in a desert stream ecosystem following flash flooding. Ecological Monographs 52: 93–110.
Flecker, A. S. & B. Feifarek, 1994. Disturbance and the temporal variability of invertebrate assemblages in two Andean streams. Freshwater Biology 31: 131–142.
Griffith, M. B., 2014. Natural variation and current reference for specific conductivity and major ions in wadeable streams of the conterminous USA. Freshwater Science 33: 1–17.
Griffith, D. A. & P. R. Peres-Neto, 2006. Spatial modeling in ecology: the flexibility of eigenfunction spatial analyses. Ecology 87: 2603–2613.
Heino, J. & M. Grönroos, 2017. Exploring species and site contributions to beta diversity in stream insect assemblages. Oecologia 183: 151–160.
Heino, J., A. S. Melo, L. M. Bini, F. Altermatt, S. A. Al-Shami, D. G. Angeler, N. Bonada, C. Brand, M. Callisto, K. Cottenie, O. Dangles, D. Dudgeon, A. Encalada, E. Göthe, M. Grönroos, N. Hamada, D. Jacobsen, V. L. Landeiro, R. Ligeiro, R. T. Martins, M. L. Miserendino, C. S. Md Rawi, M. E. Rodrigues, F. O. de Roque, L. Sandin, D. Schmera, L. F. Sgarbi, J. P. Simaika, T. Siqueira, R. M. Thompson & C. R. Townsend, 2015. A comparative analysis reveals weak relationships between ecological factors and beta diversity of stream insect metacommunities at two spatial levels. Ecology and Evolution 5: 1235–1248.
IMASUL, 2014. Sistema de Suporte ao Licenciamento Ambiental., http://sisla.imasul.ms.gov.br/sisla.
INMET, 2018. Instituto Nacional de Metereologia - National Institute of Meteorology. Data available at: http://www.inmet.gov.br/portal/index.php?r=home/page&page=rede_estacoes_auto_graf.
Landeiro, V. L., B. Franz, J. Heino, T. Siqueira & L. M. Bini, 2018. Species-poor and low-lying sites are more ecologically unique in a hyperdiverse Amazon region: evidence from multiple taxonomic groups. Diversity and Distributions 24: 966–977.
Legendre, P. & M. De Cáceres, 2013. Beta diversity as the variance of community data: dissimilarity coefficients and partitioning. Ecology Letters 16: 951–963.
Legendre, P., M. R. T. Dale, M.-J. Fortin, J. Gurevitch, M. Hohn & D. Myers, 2002. The consequences of spatial structure for the design and analysis of ecological field surveys. Ecography 25: 601–615.
Leibold, M. A. & J. M. Chase, 2018. Metacommunity Ecology. Princeton University Press, Princeton.
Leibold, M. A., M. Holyoak, N. Mouquet, P. Amarasekare, J. M. Chase, M. F. Hoopes, R. D. Holt, J. B. Shurin, R. Law, D. Tilman, M. Loreau & A. Gonzalez, 2004. The metacommunity concept: a framework for multi-scale community ecology. Ecology Letters 7: 601–613.
Leigh, C., A. J. Boulton, J. L. Courtwright, K. Fritz, C. L. May, R. H. Walker & T. Datry, 2016. Ecological research and management of intermittent rivers: an historical review and future directions. Freshwater Biology 61: 1181–1199.
Leigh, C., K. S. Boersma, M. L. Galatowitsch, V. S. Milner & R. Stubbington, 2019. Are all rivers equal? The role of education in attitudes towards temporary and perennial rivers. People and Nature. https://doi.org/10.1002/pan3.22.
Martínez, A., J. Pérez, J. Molinero, M. Sagarduy & J. Pozo, 2015. Effects of flow scarcity on leaf-litter processing under oceanic climate conditions in calcareous streams. Science of The Total Environment 503–504: 251–257.
Mills, L. S., M. E. Soule & D. F. Doak, 1993. The keystone-species concept in ecology and conservation. BioScience 43: 219–224.
Mouquet, N., D. Gravel, F. Massol & V. Calcagno, 2013. Extending the concept of keystone species to communities and ecosystems. Supporting Information. Ecology Letters 16: 1–8.
Myers, N., R. A. Mittermeier, C. G. Mittermeier, G. A. B. Da Fonseca & J. Kent, 2000. Biodiversity hotspots for conservation priorities. Nature 403: 853–858.
Oksanen, J., R. Kindt, P. Legendre, B. O’Hara, G. L. Simpson, P. Solymos, H. Stevens, & H. Wagner, 2009. The Vegan Package: Communiity Ecology Package.
Peres-Neto, P. R. & P. Legendre, 2010. Estimating and controlling for spatial structure in the study of ecological communities. Global Ecology and Biogeography 19: 174–184.
Poff, N. L., J. D. Allan, M. B. Bain, J. R. Karr, K. L. Prestegaard, B. D. Richter, R. E. Sparks & J. C. Stromberg, 1997. The natural flow regime. BioScience 47: 769–784.
Roberts, D. W., 2016. Labdsv: Ordination and multivariate analysis in ecology.
Ruhí, A., T. Datry & J. L. Sabo, 2017. Interpreting beta-diversity components over time to conserve metacommunities in highly dynamic ecosystems. Conservation Biology 31: 1459–1468.
Schmera, D., D. Árva, P. Boda, E. Bódis, Á. Bolgovics, G. Borics, A. Csercsa, C. Deák, E. Á. Krasznai, B. A. Lukács, P. Mauchart, A. Móra, P. Sály, A. Specziár, K. Süveges, I. Szivák, P. Takács, M. Tóth, G. Várbíró, A. E. Vojtkó & T. Erős, 2018. Does isolation influence the relative role of environmental and dispersal-related processes in stream networks? An empirical test of the network position hypothesis using multiple taxa. Freshwater Biology 63: 74–85.
Silva, M. B., L. H. C. dos Anjos, M. G. Pereira, J. A. Schiavo, M. Cooper & R. S. de Cavassani, 2017. Soils in the karst landscape of Bodoquena plateau in cerrado region of Brazil. CATENA 154: 107–117.
Socolar, J. B., J. J. Gilroy, W. E. Kunin & D. P. Edwards, 2016. How should beta-diversity inform biodiversity conservation? Trends in Ecology & Evolution 31: 67–80.
Sokal, R. R. & N. L. Oden, 1978. Spatial autocorrelation i n biology 1. Methodology. Biological Journal of the Linnean Society 10: 199–228.
Sor, R., P. Legendre & S. Lek, 2018. Uniqueness of sampling site contributions to the total variance of macroinvertebrate communities in the Lower Mekong Basin. Ecological Indicators 84: 425–432.
Soria, M., C. Leigh, T. Datry, L. M. Bini & N. Bonada, 2017. Biodiversity in perennial and intermittent rivers: a meta-analysis. Oikos 126: 1078–1089.
Strassburg, B. B. N., T. Brooks, R. Feltran-Barbieri, A. Iribarrem, R. Crouzeilles, R. Loyola, A. E. Latawiec, F. J. B. Oliveira Filho, C. A. M. De Scaramuzza, F. R. Scarano, B. Soares-Filho & A. Balmford, 2017. Moment of truth for the Cerrado hotspot. Nature Ecology and Evolution 1: 1–3.
Tolonen, K. E., K. Leinonen, J. Erkinaro & J. Heino, 2018. Ecological uniqueness of macroinvertebrate communities in high-latitude streams is a consequence of deterministic environmental filtering processes. Aquatic Ecology 52: 17–33.
Tomas, W. M., F. de Oliveira Roque, R. G. Morato, P. E. Medici, R. M. Chiaravalloti, F. R. Tortato, J. M. F. Penha, T. J. Izzo, L. C. Garcia, R. F. F. Lourival, P. Girard, N. R. Albuquerque, M. Almeida-Gomes, M. H. da S. Andrade, F. A. S. Araujo, A. C. Araujo, E. C. de Arruda, V. A. Assunção, L. D. Battirola, M. Benites, F. P. Bolzan, J. C. Boock, I. M. Bortolotto, M. da S. Brasil, A. R. Camilo, Z. Campos, M. A. Carniello, A. C. Catella, C. C. Cheida, P. G. Crawshaw, S. M. A. Crispim, G. A. D. Junior, A. L. J. Desbiez, F. A. Dias, D. P. Eaton, G. P. Faggioni, M. A. Farinaccio, J. F. A. Fernandes, V. L. Ferreira, E. A. Fischer, C. E. Fragoso, G. O. Freitas, F. Galvani, A. S. Garcia, C. M. Garcia, G. Graciolli, R. D. Guariento, N. M. R. Guedes, A. Guerra, H. M. Herrera, R. Hoogesteijn, S. C. Ikeda, R. S. Juliano, D. L. Z. K. Kantek, A. Keuroghlian, A. C. R. Lacerda, A. L. R. Lacerda, V. L. Landeiro, R. R. Laps, V. Layme, P. Leimgruber, F. L. Rocha, S. Mamede, D. K. S. Marques, M. I. Marques, L. A. F. Mateus, R. N. Moraes, T. A. Moreira, G. M. Mourão, R. D. Nicola, D. G. Nogueira, A. P. Nunes, C. da Nunes da Cunha, M. D. Oliveira, M. R. Oliveira, G. M. Paggi, A. O. Pellegrin, G. M. F. Pereira, I. A. H. F. S. Peres, J. B. Pinho, J. O. P. Pinto, A. Pott, D. B. Provete, V. D. A. dos Reis, L. K. dos Reis, P.-C. Renaud, D. B. Ribeiro, O. C. Rossetto, J. Sabino, D. Rumiz, S. M. Salis, D. J. Santana, S. A. Santos, Â. L. Sartori, M. Sato, K.-L. Schuchmann, E. Scremin-Dias, G. H. F. Seixas, F. Severo-Neto, M. R. Sigrist, A. Silva, C. J. Silva, A. L. Siqueira, B. M. A. Soriano, L. M. Sousa, F. L. Souza, C. Strussmann, L. S. M. Sugai, N. Tocantins, C. Urbanetz, F. Valente-Neto, D. P. Viana, A. Yanosky, & W. J. Junk, 2019. Sustainability Agenda for the Pantanal Wetland: Perspectives on a Collaborative Interface for Science, Policy, and Decision-Making. Tropical Conservation Science 12: 1–30
Tonkin, J. D., J. Heino, A. Sundermann, P. Haase & S. C. Jähnig, 2016. Context dependency in biodiversity patterns of central German stream metacommunities. Freshwater Biology 61: 607–620.
Tonkin, J. D., F. Altermatt, D. Finn, J. Heino, J. D. Olden, S. U. Pauls, D. A. Lytle, D. S. Finn, J. Heino, J. D. Olden, S. U. Pauls & D. A. Lytle, 2018. The role of dispersal in river network metacommunities: patterns, processes, and pathways. Freshwater Biology 63: 141–163.
Valente-Neto, F., L. Durães, T. Siqueira & F. O. Roque, 2018a. Metacommunity detectives: confronting models based on niche and stochastic assembly scenarios with empirical data from a tropical stream network. Freshwater Biology 63: 86–99.
Valente-Neto, F., M. E. Rodrigues & F. O. de Roque, 2018b. Selecting indicators based on biodiversity surrogacy and environmental response in a riverine network: bringing operationality to biomonitoring. Ecological Indicators 94: 198–206.
Warfe, D. M., N. E. Pettit, R. H. Magierowski, B. J. Pusey, P. M. Davies, M. M. Douglas & S. E. Bunn, 2013. Hydrological connectivity structures concordant plant and animal assemblages according to niche rather than dispersal processes. Freshwater Biology 58: 292–305.
Acknowledgements
We are grateful to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul (FUNDECT) by grants that allowed the fieldwork. We are also thankful to two anonymous reviewers and to the associate editor Luis Mauricio Bini for their valuable contributions that improved this manuscript. The research was partially supported by the Long Term Ecological Research “Planalto da Bodoquena: redes de interações em longo Prazo” (CNPq-Fundect). Specifically, FVN was supported by grant number 88882.317337/2019-01, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and FOR was supported by CNPq grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Luis Mauricio Bini
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Valente-Neto, F., da Silva, F.H., Covich, A.P. et al. Streams dry and ecological uniqueness rise: environmental selection drives aquatic insect patterns in a stream network prone to intermittence. Hydrobiologia 847, 617–628 (2020). https://doi.org/10.1007/s10750-019-04125-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-04125-9