Abstract
Reduction of flow constitutes one of the most severe human alterations to rivers, as it affects the key abiotic feature of these ecosystems. While there has been considerable progress in understanding the effects of reduced flow on benthic macroinvertebrates, cascading effects of flow reduction on dissolved oxygen concentrations (DO) have not yet received much attention. We compared the macroinvertebrate composition between reference conditions and a situation after several years of discharge reduction in the Spree River (Brandenburg, Germany). Community composition shifted from rheophilic species to species indifferent to flow conditions. Filter feeders were partially replaced by collector/gatherers, which likely reduces the retention of organic matter, and thus the self-purification capacity of the river section. These shifts were associated with low discharge during summer, cascading into daily DO concentration minima of less than 5 mg l−1 which prevailed 74% of the days in summer. This depletion of DO after flow reduction presumably caused the observed species turnover. Hence, flow reduction in lowland rivers may not only directly impair the ecological functions provided by benthic macroinvertebrates but may also act indirectly by depleting DO concentrations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Reduction of flow affects the key abiotic feature of rivers, and thus may lead to most severe ecological effects (e.g., Castella et al., 1995). Such a “lentification” of river ecosystems (sensu Sabater, 2008) is accompanied by alterations in water depth, turbulence and vertical mixing, and thus results in changes in thermal regime, sedimentation rates, and water chemistry (Dewson et al., 2007).
Alterations of in-stream habitat conditions will result in substantial effects on the benthic macroinvertebrate community and its ecological functions (Dewson et al., 2007). Flow reductions have been reported to directly result in a shift from a lotic community toward a community dominated by lentic species (Bickerton, 1995; Castella et al., 1995; Jowett, 1997; Horrigan & Baird, 2008) and indirectly in increased water temperatures, which may lead to lower macroinvertebrate richness due to loss of cold-stenotherm species (Cazaubon and Giudicelli, 1999). Moreover, at reduced flows, the deposition of organic matter clogs typical river habitats as gravel banks, which alters macroinvertebrate abundance and composition (Wood & Armitage, 1999; Brunke et al., 2001).
In lowland rivers, as in lake outlets, high loads of organic seston often support high densities of macroinvertebrate filter feeders, and a reduction in flow velocity and seston load affects the density of these filter feeders ( Wotton, 1988; Richardson & Mackay, 1991; Poepperl, 1999). This may produce significant cascading effects on other feeding guilds, as the pelagic–benthic coupling performed by filter feeders may support rich populations of benthic collectors/gatherers that feed on their faeces and pseudofaeces (Gergs et al., 2009). However, filter feeder densities may additionally be affected by low dissolved oxygen concentrations (DO) another potential cascading effect of low flow conditions, which may create unfavorable habitat conditions and may especially reduce densities of filter feeding mussels due to their low mobility.
Increased particle sedimentation rate and water temperature during low flow conditions may together result in elevated benthic respiration rate. As a consequence, DO may reach lower daily minima, in extreme cases even leading to anoxic conditions (Parr & Mason, 2004). Especially in eutrophied lowland rivers with high organic matter loads, a daily minimal DO of 3–5 mg l−1 may occur during drought years (Parr & Mason, 2004). Such low DO concentrations are within the range of values reported to severely affect stream macroinvertebrate communities (Nebeker, 1972; Connolly et al., 2004). However, the effect of reduced flow on DO concentration as the governing environmental factor for macroinvertebrate community composition has hardly been studied so far (Miller et al., 2007). Critical DO concentrations have possibly been neglected as a key habitat factor as lowest DO levels most often occur before sunrise and hence are rarely recorded by daytime measurements (Dewson et al., 2007). A study that combines continuous DO measurements with macroinvertebrate community composition measurements may therefore clarify if critical DO concentrations are a key habitat factor controlling macroinvertebrate composition.
In this study, we investigated the cascading effects of low flow on the macroinvertebrate community of a lowland river. We hypothesized that alterations of macroinvertebrate community composition in a lowland river following flow reduction may ultimately be attributed to daily DO minima, and that such alterations will also lead to significant changes in the functioning of the macroinvertebrate community. We studied these cascading effects in the regulated lowland Spree River (Brandenburg, Germany). The studied Krumme Spree section of this river has undergone an anthropogenic reduction of the average discharge by 32% between 1997 and 2007, mainly due to reduced groundwater input into the river linked to declining mining activities in the upstream catchment (Pusch & Hoffmann, 2000). Previous studies conducted in this river section in 1997 and 1998 revealed that the river was inhabited by a lotic macroinvertebrate community typical of a lowland river, with especially high densities of filter feeders downstream a lake outlet (Pusch & Hoffmann, 2000).
Methods
Study site
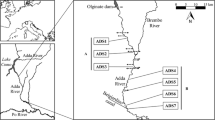
The study was conducted on the 22.4-km long Krumme Spree section of the Spree River (Brandenburg, Germany). There, the river channel has a mean width of 25 m and a mean depth of 1.5 m, after the river channel cross-sectional area has been enlarged in the early twentieth century. The study section is located directly downstream of the eutrophic Lake Neuendorfer See and thus constitutes a typical lake outlet. Natural discharge in this river section amounts to an average of 24 m³ s−1 (measured directly upstream of Lake Neuendorfer See) but which has been artificially increased during the period 1960–1993 to about 33 m³ s−1 due to the increased discharge of groundwater from an upstream mining area (Pusch & Hoffmann, 2000). After 1993 groundwater discharge was substantially reduced, so that annual mean discharge decreased gradually to 14.8 m³ s−1 (2001) and, finally, to 9.5 m³ s−1 (2008) (Fig. 1).
Comparison of the fractions of explained variance (absolute values of Spearman’s ρ) for the regressions between daily minimum DO and water temperature or daily minimum DO and discharge for different ranges of water temperatures (a). b Piecewise regression analysis between daily minimum DO and discharge at water temperatures >16.5°C. Data from March to December in 1997, 1998, 1999, 2007, 2008, and 2010
Macroinvertebrate sampling was performed along the same three cross-sections used by Pusch & Hoffmann (2000). The first cross-section was situated near the lake outlet, the second in the middle of the river section and the third at the downstream end of the river section (Table 1), thereby constituting a longitudinal gradient of decreasing lake influence.
Macroinvertebrates
Macroinvertebrate data from spring 1997 and spring 1998 were taken from Pusch & Hoffmann (2000). In spring 2007, the same methods were applied to take the macroinvertebrate samples. At each of the three cross-sections, samples were taken from eight previously defined mesohabitat categories: Dreissena-bank, unionid-mussel bed, rip rap, alder roots, coarse woody debris (CWD), shifting sand, stable sand, and mud (Pusch & Hoffmann, 2000). Within each cross-section, Dreissena-bank, unionid-mussel bed, rip rap, coarse woody debris (CWD), shifting sand, stable sand, and mud were sampled with three to five replicated samples using a Surber sampler (0.5 mm mesh, 0.09 m² Surber sampler area). The replicated samples covered a total area of 0.27–0.45 m2 (three to five times the area of the Surber sampler) depending on the available area of the mesohabitats in a cross section. Stones and CWD were sampled by a scuba diver using a Surber sampler, subsequently brought ashore, and adhering invertebrates were carefully scraped off with a toothbrush into water-filled boxes. Stones sampled for macroinvertebrates had a polygon shape with distinctively identifiable sides. Thus, the length and height of each stone were measured and its surface area was calculated. CWD surface area was calculated assuming the form of a cylinder. Moreover, with a hand net (0.5 mm mesh), a defined area of alder roots (0.18 m2) was sampled. This area was smaller than that of the other mesohabitats because not enough alder roots could be found to sample larger areas. Invertebrates were preserved in 70% ethanol until further processing in the laboratory.
In the laboratory, macroinvertebrates were sorted using binocular microscopes and identified to species level, except for Diptera (family level), Oligochaeta (class level), and Sphaeriidae (Bivalvia; genus level). Macroinvertebrates were assigned to functional feeding groups and flow preferences according to the AQEM/STAR ecological river classification system (Schmidt-Kloiber et al., 2006). Within the system, the encoding for the functional feeding groups is based on fuzzy coding with probabilities from 0 to 10 for each group and the encoding for the flow preference groups is based on a system with seven classes (limnobiont, limnophilic, limno–rheophilic, rheo–limnophilic, rheophilic, rheobiont, indifferent to flow).
We compared macroinvertebrate community composition between 1997/1998 and 2007 for each of the three cross-sections and used mesohabitats as replicates for each comparison. Invertebrate density for each mesohabitat at each cross-section was calculated as mean abundance per m2. For cross-section-specific comparisons, we used only mesohabitats found in both 1997/1998 and 2007 (Table 1).
An initial analysis of similarity (R = 0.01, P = 0.33, Bray–Curtis similarity, ANOSIM, PRIMER V6, PRIMER-E Ltd., Plymouth, UK) of macroinvertebrate samples from 1997 and 1998 revealed no significant differences in community composition. Thus, macroinvertebrate data from 1997 to 1998 were combined and used as reference data. We then compared macroinvertebrate density at each cross-section between the 1997/1998 and 2007 sampling periods (Paired t test, SPSS Version 16, SPSS Inc., Chicago, Illinois, USA). Prior to analyses, macroinvertebrate densities were ln + 1-transformed to achieve normal distribution.
Compositional differences between 1997/1998 and 2007 were tested using an ANOSIM. A SIMPER analysis (PRIMER V6) was conducted to identify the taxa responsible for any significant differences between 1997/1998 and 2007.
Functional feeding group composition, especially the proportion of filter feeders, may depend on the distance of the cross-sections to the lake outlet (Poepperl, 1999). To account for this, the effects of discharge reduction on functional feeding group composition were compared between 1997/1998 and 2007, using the mesohabitats separately for each of the cross-sections as replicates (n = 14 for the comparison, paired t test, SPSS Version 16, see Table 1 for details on the used mesohabitats). For the comparison of flow preference groups we did not assume a dependence of composition on the distance to the lake outlet and therefore only compared flow preference groups among cross-sections between 1997/1998 and 2007 (paired t test, SPSS Version 16). Prior to analyses, macroinvertebrate community metrics were ln + 1-transformed to achieve normal distribution.
To compare effects of flow reduction on biological and functional diversity, we calculated the Simpson index (diversity function, vegan package in R, Oksanen et al., 2011; R Development Core Team, 2012) and Rao’s quadratic entropy (dbFD function, FD package in R, Botta-Dukát, 2005; Laliberté & Legendre, 2010) for the functional feeding groups. We used the same data as for the analysis of community composition and functional feeding group composition. We compared 1997/1998 and 2007 for each cross-section, using the habitats as replicates (paired t test, t test function in R). The data were normally distributed and thus the paired t test could be applied.
Discharge, water temperature, and DO
Daily discharge data were provided by the regional environmental agency (Landesumweltamt Brandenburg) for the gauging station close to the inflow of the Spree River into Lake Neuendorfer See, which is located 1.9 km upstream of cross-section 1. Since there are no in- or outflows between the gauging station and the studied river section, we assumed that gauging data are also representative for the studied river section. We corrected discharge data for major flooding events and water management measures that were not representative for natural hydrologic conditions, and excluded them. The reason was that water temperature and DO are related to flow conditions and discharge, but rating curves to determine the water temperature/DO–discharge relationships derived from natural hydrologic conditions do not account for major flooding events and water management measures. During major flooding events the river overflows its banks and spreads large quantities of water over its floodplain. Thus, water temperature and DO is altered in the flood plain, which affects measurements and results in the breakdown of water temperature/DO–discharge relationships. During water management measures, large quantities of water may be abstracted from the river instantly, but the water temperature/DO-response to this water abstraction is often delayed in time. Therefore, uncertainty in the rating curve predictions is largely increased by such events. Water management measures were identified as discharge increase peaks that were not caused by precipitation events in the Krumme Spree catchment area in the previous 5 days. The described corrections in discharge for water management measures or flood events were only necessary in the years 2008 and 2010, thus these events did not affect macroinvertebrate assemblages sampled in 1997/1998 or 2007. Precipitation in the catchment was modeled using MONERIS (Venohr et al., 2011) according to Haylock et al. (2008). MONERIS is a semi-empirical, conceptual model which estimates nutrient export, precipitation, and discharge at the catchment scale (Venohr et al., 2011).
Measurements of DO and water temperature were conducted during summer periods (March to December) with varying duration in 1997 (142 days), 1998 (221 days), 1999 (28 days), 2007 (19 days), 2008 (28 days), and 2010 (83 days) every 2 min with either a WTW probe (1997, 1998, 1999; WTW Weilheim, Germany), or every 15 min with an YSI 6600/600 XLM probe (2007, 2008, 2010; YSI Inc., Yellow Springs, Ohio). The probes were installed at cross-section 3 at a distance of 2 m from the shoreline in 1 m water depth. The daily minimum DO value was extracted from every 24-h measurement and related to the discharge value of the same day using non-linear regression analysis (Origin Pro 8, Origin Lab Corporation, Northampton, MA, USA). Daily minimum DO was used as it affects macroinvertebrate community composition more strongly than daily average DO (Dewson et al., 2007; Miltner, 2010).
We used Spearman’s rank correlation (R Development Core Team, 2012) and piece-wise regression (Muggeo, 2003) to predict DO for the periods of time for which no DO measurements were available. We tested both discharge and water temperature as potential predictors. We used non-linear regression to predict water temperature from air temperature (SigmaStat Version 3.5, Systat Software Inc., Chicago, USA) for periods of time for which no measurements of water temperature were available. According to Morrill et al. (2005), we used 3-day averages of water and air temperature (the same day plus the two previous days, see supplement for formula and plot of the regression between air and water temperature). As a threshold for DO depletion situations we used 5 mg l−1, since previous studies have shown that DO concentrations below 5 mg l−1 increase the mortality of benthic macroinvertebrates (Nebeker, 1972; Chen et al., 2001; Connolly et al., 2004) and thus lead to a changes in macroinvertebrate community composition (Canadian Council of Ministers of the Environment, 1999; Miltner, 2010).
Average daily discharge values during summer (June–September) of 1995, 1996, 1997, 2005, and 2006 were compared with a Friedman-Test followed by Dunn’s post hoc (Prism GraphPad version 4, GraphPad Software, La Jolla, CA, USA), because data were not normally distributed and replicates of each year were dependent.
Seston load and quality
In order to account for possible changes in the food quality and quantity available for filter feeders, samples for seston concentration and seston carbon and nitrogen content were taken mostly biweekly from 1999 to 2007 at cross-section 1, except in 2003 where no samples were taken. We chose 1999–2007 for our analysis because seston data were not available for the period before 1999. Seston concentration was measured by filtering 0.5 l of stream water onto GF/F filters (Whatman GmbH, Dassel, Germany) and expressed as mg dry weight per liter. Seston carbon and nitrogen concentrations were determined using a C/N analyzer (Elementar Vario EL, Elementar Analysensysteme GmbH, Hanau, Germany). Subsequently, we converted seston C and N concentrations into molar C:N-ratios. Seston concentration and discharge values from the same day were used to calculate daily seston loads (t d−1) for each sampling date.
We analyzed whether seston concentration, seston load, or seston C:N-ratio changed during the time period of 1999–2007 by calculating the Spearman rank correlations (SPSS Version 16) between each seston parameter and time. Time was given as days since the first measurement in 1999 and the Spearman rank correlation was employed because the data was not normally distributed.
Results
Macroinvertebrates
Mean macroinvertebrate densities in cross-sections 1–3 did not differ significantly between 1997/1998 and 2007 (paired t test; P > 0.05 for all tests).
Biological diversity calculated as Simpson index did not significantly differ between 1997/1998 and 2007 in any of the cross-sections (paired t test, P > 0.05) but a positive trend was apparent: mean Simpson index (±SE) was 0.46 (±0.10) for cross-section 1 in 1997/1998 and 0.55 (±0.08) in 2007. In cross-section 2, mean Simpson index was 0.53 (±0.05) in 1997/1998 and 0.68 (±0.03) in 2007 and in cross-section 3, it was 0.61 (±0.07) in 1997/1998 and 0.75 (±0.04) in 2007.
Functional diversity (mean Rao’s quadratic entropy for the functional feeding group composition) was also not significantly different between years (paired t test, P > 0.05) but tended to be higher in 2007. Functional diversity (±SE) was 2.50 (±0.90) for cross-section 1 in 1997/1998 and 3.16 (±0.65) in 2007. In cross-section 2, functional diversity was 3.91 (±0.90) in 1997/1998 and 5.05 (±0.53) in 2007 and in cross-section 3, it was 4.73 (±0.80) in 1997/1998 and 7.9 (±0.98) in 2007.
Community composition differed significantly between 1997/1998 and 2007 at cross-section 1 (ANOSIM, R = 0.73, P = 0.002, n = 5) and cross-section 3 (R = 0.40, P = 0.005, n = 3), but not at cross-section 2 (R = 0.25, P = 0.14, n = 6). SIMPER-analysis showed that especially Chelicorophium curvispinum Sars (Amphipoda), Chironomidae, Oligochaeta and Dreissena polymorpha Pallas (Bivalvia) contributed to the dissimilarity between 1997/1998 and 2007 (Table 2). Dikerogammarus haemobaphes Eichwald (Amphipoda) was only found in 2007, while average densities of Pisidium sp. Pfeiffer (Bivalvia) and Sphaerium sp. Scopoli (Bivalvia) increased with time (100-fold and fourfold, respectively). Abundance of C. curvispinum tremendously decreased in 2007. Similarly, the average densities of Unio tumidus Philippson (Bivalvia) and Unio pictorum (L.) (Bivalvia) substantially decreased, and the lotic filter-feeding caddis fly Hydropsyche contubernalis McLachlan (Trichoptera) was absent in 2007 (Table 2).
Concerning functional feeding group composition, a comparison across the cross-sections revealed an apparent, but not significant decrease in the density of active filter feeders from 1997/1998 to 2007. Cross-section specific comparisons of the active filter feeder densities revealed significantly lower densities at cross-section 1 in 2007, but no significant temporal change for cross-sections 2 and 3. Conversely, an increase was observed for all other functional feeding groups (Table 3). Densities of collector/gatherer, grazer/scrapers and parasites were significantly higher in 2007 than in 1997/1998 across all cross-sections and in cross-sections 1 and 2. The density of predators increased significantly in cross-section 2.
These changes in community composition were also reflected by flow preference groups. In 1997/1998, the macroinvertebrate community was dominated by rheophilic taxa that decreased on average by 88% until 2007. This difference was, however, not significant due to high variability in the paired differences between mesohabitats of the two campaigns (Table 4). In contrast, the densities of limnophilic, limno–rheophilic taxa, and taxa indifferent to flow increased significantly to more than the double level between 1997/1998 and 2007 (Table 4).
Predictors of DO minima
To predict the occurrence of daily DO minima <5 mg l−1 at high water temperatures also for days for which no measurements of DO were available, the predictive power of temperature and discharge was assessed, respectively. We were especially interested at the prediction of DO when water temperatures were >16.5°C as DO <5 mg l−1 only occurred above this water temperature. To test the predictive power of each water temperature and discharge, daily minimum DO concentrations at several ranges of water temperature (mean daily water temperatures from >5 to >23°C with 0.5°C steps) were correlated to either water temperature or discharge. This analysis revealed that water temperature itself is not a good predictor of daily minimum DO for the summer period as it fails to predict DO for high water temperatures (>16.5°C) typical in summer (absolute value of Spearman’s ρ = 0.40–0.58), whereas at >16.5°C discharge still predicts daily minimum DO at a high level (absolute value of Spearman’s ρ = 0.79–0.85, Fig. 1a). We found that the discharge to daily minimum DO correlation for >16.5°C is best expressed by a piecewise regression, which gives a break point of 5.3 m³ s−1 in the discharge/DO relationship below which discharge strongly affects daily minimum DO (R² = 0.59, P < 0.0001, n = 152, Fig. 1b). This relationship was following the linear equation DO (mg l−1) = 0.75 × Discharge (m3 s−1) + 1.89. Using the linear equation and the equation provided by the air–water temperature relationship for all the days with water temperature >16.5°C and discharge <5.3 m³ s−1, we estimated that DO concentration did not fall below 5 mg l−1 in 1995 and 1996. Conversely, DO was estimated to have fallen below 5 mg l−1 on 13.9% of the days (17 of 122 days in June–September) in 1997, 35.2% of the days (43 days) in 2005 and in 73.8% of the days (90 days) in 2006. Low values of <5 mg l−1 for DO were restricted to the summers when water temperatures were >16.5°C and never occurred in winter or in spring before the sampling dates.
Discharge and seston load
Daily discharge levels did not significantly differ between 1995 and 1996 but were significantly higher than in 1997, 2005, and 2006 (P < 0.05 Friedman-Test with Dunn’s post hoc; Fig. 2). Moreover, daily discharge levels in 1997, 2005, and 2006 were significantly different, with the highest discharges in 1997 and the lowest in 2006 (Fig. 2).
Box plots of daily discharge during summer (June–September) upstream of the studied river section. Letters indicate significant differences (P < 0.05, Friedman-Test with Dunn’s post hoc). The dashed horizontal line represents the calculated discharge threshold to prevent DO concentrations lethal to riverine macroinvertebrates (DO < 5 mg l−1)
Seston concentration (Spearman rank correlation, ρ = 0.25, P = 0.03; Fig. 3) and seston C:N-ratio (ρ = 0.20, P = 0.03) were significantly positively correlated with time, while seston load (ρ = −0.09, P = 0.31) did not change over time.
Discussion
Drivers of DO minima
Dissolved oxygen depletion found during summer probably results from the production of high loads of particulate organic matter flushed out from eutrophic Lake Neuendorfer See located directly upstream of the Krumme Spree section. This organic matter is effectively retained in the studied river section and generates high community respiration rates, especially at high water temperatures in summer (community respiration of >3 mg l−1 d−1 at water temperatures >20°C; Wanner et al., 2002). This relationship best explains the positive correlation which we found between discharge and DO (Fig. 1b), as with lower discharges and longer residence time a higher retention of particulate organic matter will occur. Furthermore, this may explain the fact that above a discharge threshold of 5.3 m³ s−1, the relationship between discharge and DO disappears as, above this threshold, there is probably no substantial change in sedimentation of seston.
Changes in invertebrate composition
Dominant flow preference of the macroinvertebrate community groups changed from lotic in 1997/1998 towards a community dominated by species indifferent to flow velocity. This change could be traced back to the loss of rheophilic species such as C. curvispinum and D. polymorpha and the concomitant increase of Chironomidae. Although individual Chironomidae species differ in their flow preference, the family itself is classified as indifferent to flow (Schmidt-Kloiber et al., 2006). Moreover, recent findings suggest that Chironomidae benefit from low flow conditions due to their short life cycle and small body size (Ledger et al., 2011). Hence, the alteration in composition of the flow preference groups (Table 4) is probably correlated to the 30–40% lower mean annual discharge in 2007 compared to 1997 and 1998 (Fig. 2).
Compared to 1997/1998, the density of filter feeders in cross-section 1 in 2007 was reduced by 80% (Table 3). This cross-section constitutes a typical lake outlet, where large populations of filter feeders usually rely on seston produced in the lake (Malmqvist & Eriksson, 1995; Wotton et al., 1996). Our results suggest that the retention of seston originating from the lake might have been less effective in 2007 due to the lower densities of the filter feeders. Their density was dramatically reduced even though no significant change in seston load occurred between 1999 and 2007. The observed change in the C:N ratio of seston was probably too small (Fig. 3) to affect mussel growth or reproduction. This conjecture is corroborated by a study on the growth efficiency of Crassostrea gigas (Thunberg) that only varies between 0.08 and 0.15 in the range of median C:N ratios (4–8) found in our study (Bayne, 2009). Moreover, invasion success of D. polymorpha in Swedish lakes have been reported to be unrelated to the C:N ratio of seston (Bayne, 2009; Naddafi et al., 2009).
The reduction of the densities of the two filter feeding species C. curvispinum and D. polymorpha explains the positive trend of both biological diversity and functional diversity of the functional feeding groups, since these two species strongly dominated the macroinvertebrate community in 1997/1998, which was not the case in 2007 (Table 2). However, the change in biological or functional diversity was not strong enough to be significant, implying no strong effect of flow reduction on either of them.
Cascading effects of flow reduction on community structure
The observed alteration of the functional feeding group composition could partly be traced back to reduced discharge. Flow preferences of filter-feeding benthic invertebrates in the Krumme Spree section have been specifically analyzed by Brunke et al. (2001) who observed minimum flow requirements of 7.8–9.4 m³ s−1 for the rheophilic and rheo–limnophilic species D. polymorpha, U. tumidus, Neureclipsis bimaculata (L.) (Trichoptera), and H. contubernalis. In our study, D. polymorpha, U. tumidus, and N. bimaculata were found in substantially lower densities in 2007 compared to 1997/1998 (Table 2), and rheophilic H. contubernalis was missing in 2007.
Loss of cold-stenotherm species was not the reason for the found changes in the macroinvertebrate community, since the taxa whose densities declined from 1997/1998 to 2007 (Table 2) are either eurythermic or even slightly thermophile, as was the case for C. curvispinum (Tachet, 2010).
Probably due to the higher percentage of days with DO minima <5 mg l−1 in 2006 (73.8%) than in 1996 (0%) or 1997 (13.9%), species preferring well-oxygenated habitats were less dominant in 2007 than in 1997/1998. The strong decline of the filter-feeding C. curvispinum (Table 2) by more than an order of a magnitude might be related to low DO conditions, because this species actively searches for well-oxygenated habitats, and is sensitive for DO depletion (Harris & Musko, 1999). Similarly, filter-feeding zebra mussel D. polymorpha is a rather weak oxygen regulator and therefore stressed by DO depletion situations, especially at high water temperatures (McMahon, 1996; Alexander & McMahon, 2004). Accordingly, zebra mussel densities were found to be strongly reduced in 2007 (Table 2). Oxygen consumption of unionids has been shown to be stable until a minimum of 2–6 mg l−1, depending on the species (Chen et al., 2001). Below this concentration, their oxygen consumption decreased and the mussels lost their ability to actively regulate oxygen. In our study section, a DO <5 mg l−1 was frequently found in summer which may explain the three-fold density decrease of U. tumidus (Table 2). Densities of other taxa, such as Sphaerium spp. and Pisidium spp. (Sphaeriidae, Bivalvia), were found to be higher in 2007 (Table 2). For Pisidium spp. it was shown that the density is negatively correlated to DO concentration, suggesting better adaptation to situations where DO is depleted (Dussart, 1979). The same was shown for Sphaerium spp. which was only abundant at DO concentrations <4.8 mg l−1 in a study on its environmental preferences (Joyner-Matos et al., 2007).
Macroinvertebrate sampling took place in spring 2007 and low daily minimum DO concentrations were demonstrated for the preceding summer in the river section, with a daily minimum DO below 5 mg l−1 in 73.8% of the days in June–September 2006. Thus, the effect of low DO in the preceding summer on benthic invertebrates was probably followed by slow re-colonization for a part of the macroinvertebrate community. Since the Krumme Spree section is situated below a lake outlet, hardly any benthic species are expected to colonize the study section by drift from upstream. Similarly it has been found in other lake outlets that fast colonization was only observed for aquatic insects with aerial stages and colonization was slower for mussels, snails, and other less mobile species (Malmqvist et al., 1991; Decker et al., 2008). According to those findings, species with low tolerance to low DO and slow colonization rates should be affected most by effects of flow reduction. In our study, this was the case for D. polymorpha, C. curvispinum, and U. tumidus. Therefore, low summer DO concentrations combined with low re-colonization potential are the most probable explanation for the changes in benthic invertebreate communities found between 1997/1998 and 2007.
The anthropogenic modification in terms of channelization, straightening, and incision of the river section may have increased the effects of low flows and low DO. In a large study on English and Danish streams by Dunbar et al. (2010) it was clearly shown that the effects of low flows were stronger in human modified streams than in natural streams, partly probably due to lacking refugia in the modified streams.
Effects of flow reduction on ecosystem functions
The loss of large-bodied, slow growing filter-feeders (as U. tumidus and D. polymorpha) in favor of small-bodied, fast growing collector/gatherers (as Chironomidae) is in accordance to the findings from small stream mesocosms (Ledger et al., 2011, 2012) and implies that also in larger lowland rivers, droughts strongly affect ecosystem functioning as production shifts from large to small species. However, further studies directly assessing production under average and low flow conditions in lowland rivers are needed to confirm these findings.
The low taxonomic resolution of the Chironomidae has affected their functional feeding group affiliation in our study. Chironomidae have been one of the taxa most strongly affected by low flow in our study and due to their high numbers (Table 2), changes in their density may alter the ecosystem functions of the entire macroinvertebrate community. Thus, future studies on low flow effects should identify Chironomidae to the highest taxonomical level possible to better assess the effects of low flow on macroinvertebrate ecosystem functions.
The lower number of filter feeders found in 2007 at the lake outlet (cross-section 1, Table 3) has resulted in a substantially reduced self-purification capacity of the Krumme Spree section, as the filter feeders below the lake retain a substantial portion of the load of particulate organic matter in the Spree (Pusch & Hoffmann, 2000; Lorenz et al., 2012). The original self-purification capacity could only be restored if the number and length of periods of low flow and DO depletion in summer were reduced. The length of these periods is probably increased by channelization and artificially increased channel depth, which leads to a further reduction of flow velocity and hampers vertical mixing of DO (Pusch & Hoffmann, 2000). Thus, a reduction of the effect of low discharges on DO could be reached by restoring a shallower river channel, and by restoration of the natural flow regime. Thus, a given discharge level would be accompanied by higher flow velocities, which would improve vertical mixing of DO even under low flow conditions. Moreover, a further reduction of eutrophication of the upstream lake would decrease loads of particulate organic matter, and thus reduce the probability of low DO levels.
Conclusion
Low flow situations (e.g., Armitage & Petts, 1991; Castella et al., 1995) and “lentification” (sensu Sabater, 2008) are common phenomena in many regulated rivers. Our study shows that in such lowland rivers, DO depletion probably constitutes a major proximate factor leading to significant change in benthic community composition. Due to the replacement of large-bodied filter feeding species (Dreissena polymorpha, Unio sp.) by small-bodied collector/gatherers (Chironomidae), the self-purification capacity of the investigated section of the lowland river was reduced and the macroinvertebrate biomass production may have been altered among potential impairment of other ecological functions. Thus, restoration of the ecological functions of the river system provided by the macroinvertebrate community would require to reduce the number and length of DO depletion periods.
References
Alexander, J. E. & R. F. McMahon, 2004. Respiratory response to temperature and hypoxia in the zebra mussel Dreissena polymorpha. Comparative Biochemistry and Physiology A-Molecular & Integrative Physiology 137: 425–434.
Armitage, P. D. & G. E. Petts, 1991. Biotic score and prediction to assess the effects of water abstractions on river macroinvertebrates for conservation purposes. Aquatic Conservation: Marine and Freshwater Ecosystems 2: 1–17.
Bayne, B. L., 2009. Carbon and nitrogen relationships in the feeding and growth of the Pacific oyster, Crassostrea gigas (Thunberg). Journal of Experimental Marine Biology and Ecology 374: 19–30.
Bickerton, M. A., 1995. Long-term changes of macroinvertebrate communities in relation to flow variations—the River Glen, Lincolnshire, England. Regulated Rivers-Research & Management 10: 81–92.
Botta-Dukát, Z., 2005. Rao’s quadratic entropy as a measure of functional diversity based on multiple traits. Journal of Vegetation Science 16: 533–540.
Brunke, M., A. Hoffmann & M. Pusch, 2001. Use of mesohabitat-specific relationships between flow velocity and river discharge to assess invertebrate minimum flow requirements. Regulated Rivers-Research & Management 17: 667–676.
Canadian Council of Ministers of the Environment, 1999. Canadian water quality guidelines for the protection of aquatic life: dissolved oxygen (freshwater). Canadian environmental quality guidelines.
Castella, E., E. Bickerton, P. D. Armitage & G. E. Petts, 1995. The effects of water abstractions on invertebrate communities in U.K. streams. Hydrobiologia 308: 167–182.
Cazaubon, A. & J. Giudicelli, 1999. Impact of the residual flow on the physical characteristics and benthic community (algae, invertebrates) of a regulated Mediterranean river: The Durance, France. Regulated Rivers-Research & Management 15: 441–461.
Chen, L.-Y., A. G. Heath & R. J. Neves, 2001. Comparison of oxygen consumption in freshwater mussels (Unionidae) from different habitats during declining dissolved oxygen concentration. Hydrobiologia 450: 209–214.
Connolly, N. M., M. R. Crossland & R. G. Pearson, 2004. Effect of low dissolved oxygen on survival, emergence, and drift of tropical stream macroinvertebrates. Journal of the North American Benthological Society 23: 251–270.
Decker, A. S., M. J. Bradford & P. S. Higgins, 2008. Rate of biotic colonization following flow restoration below a diversion dam in the Bridge River, British Columbia. River Research and Applications 24: 876–883.
Dewson, Z. S., A. B. W. James & R. G. Death, 2007. A review of the consequences of decreased flow for instream habitat and macroinvertebrates. Journal of the North American Benthological Society 26: 401–415.
Dunbar, M. J., M. L. Pedersen, D. Cadman, C. Extence, J. Waddingham, R. Chadd & S. E. Larsen, 2010. River discharge and local-scale physical habitat influence macroinvertebrate LIFE scores. Freshwater Biology 55: 226–242.
Dussart, G. B. J., 1979. Sphaerium corneum (L.) and Pisidium spp. Pfeiffer – the ecology of freshwater bivalve molluscs in relation to water chemistry. Journal of Molluscan Studies 45: 19–34.
Gergs, R., K. Rinke & K. O. Rothhaupt, 2009. Zebra mussels mediate benthic-pelagic coupling by biodeposition and changing detrital stoichiometry. Freshwater Biology 54: 1379–1391.
Harris, R. R. & I. B. Musko, 1999. Oxygen consumption, hypoxia, and tube-dwelling in the invasive amphipod Corophium curvispinum. Journal of Crustacean Biology 19: 224–234.
Haylock, M. R., N. Hofstra, A. M. G. Klein Tank, E. J. Klok, P. D. Jones & M. New, 2008. A European daily high-resolution gridded data set of surface temperature and precipitation for 1950–2006. Journal of Geophysical Research 113: D20119.
Horrigan, N. & D. J. Baird, 2008. Trait patterns of aquatic insects across gradients of flow-related factors: a multivariate analysis of Canadian national data. Canadian Journal of Fisheries and Aquatic Sciences 65: 670–680.
Jowett, I., 1997. Environmental effects of extreme flows. In Mosley, M. P. & C. P. Pearson (eds), Floods and Droughts: The New Zealand Experience. Caxton Press, Christchurch, New Zealand: 103–116.
Joyner-Matos, J., L. J. Chapman, C. A. Downs, T. Hofer, C. Leeuwenburgh & D. Julian, 2007. Stress response of a freshwater clam along an abiotic gradient: too much oxygen may limit distribution. Functional Ecology 21: 344–355.
Laliberté, E. & P. Legendre, 2010. A distance-based framework for measuring functional diversity from multiple traits. Ecology 91: 299–305.
Ledger, M. E., F. K. Edwards, L. E. Brown, A. M. Milner & G. Woodward, 2011. Impact of simulated drought on ecosystem biomass production: an experimental test in stream mesocosms. Global Change Biology 17: 2288–2297.
Ledger, M. E., L. E. Brown, F. K. Edwards, A. M. Milner & G. Woodward, 2012. Drought alters the structure and functioning of complex food webs. Nature Climate Change 3: 223–227.
Lorenz, S., F. Gabel, N. Dobra & M. T. Pusch, 2012. Modelling the effects of recreational boating on self-purification activity provided by bivalve mollusks in a lowland river. Freshwater Science 82–93.
Malmqvist, B. & A. Eriksson, 1995. Benthic insects in Swedish lake-outlet streams : patterns in species richness and assemblage structure. Freshwater Biology 34: 285–296.
Malmqvist, B., S. Rundle, C. Brönmark & A. Erlandsson, 1991. Invertebrate colonization of a new, man-made stream in southern Sweden. Freshwater Biology 26: 307–324.
McMahon, R. F., 1996. The physiological ecology of the zebra mussel, Dreissena polymorpha, in North America and Europe. American Zoologist 36: 339–363.
Miller, S. W., D. Wooster & J. Li, 2007. Resistance and resilience of macroinvertebrates to irrigation water withdrawals. Freshwater Biology 52: 2494–2510.
Miltner, R. J., 2010. A method and rationale for deriving nutrient criteria for small rivers and streams in Ohio. Environmental Management 45: 842–855.
Morrill, J., R. Bales & M. Conklin, 2005. Estimating stream temperature from air temperature: implications for future water quality. Journal of Environmental Engineering 131: 139–146.
Muggeo, V. M. R., 2003. Estimating regression models with unknown break-points. Statistics in Medicine 22: 3055–3071.
Naddafi, R., P. Eklov & K. Pettersson, 2009. Stoichiometric constraints do not limit successful invaders: zebra mussels in Swedish lakes. PLoS ONE 4: e5345.
Nebeker, A. V., 1972. Effect of low oxygen concentration on survival and emergence of aquatic insects. Transactions of the American Fisheries Society 101: 675–679.
Oksanen J., F. G. Blanchet, R. Kindt, P. Legendre, P. R. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens & H. Wagner, 2011. Vegan: Community Ecology Package.
Parr, L. B. & C. F. Mason, 2004. Causes of low oxygen in a lowland, regulated eutrophic river in Eastern England. Science of the Total Environment 321: 273–286.
Poepperl, R., 1999. Functional feeding groups of a macroinvertebrate community in a Northern German lake outlet (Lake Belau, Schleswig-Holstein). Limnologica 29: 137–145.
Pusch, M. & A. Hoffmann, 2000. Conservation concept for a river ecosystem (River Spree, Germany) impacted by flow abstraction in a large post-mining area. Landscape and Urban Planning 51: 165–176.
R Development Core Team, 2012. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria.
Richardson, J. S. & R. J. Mackay, 1991. Lake outlets and the distribution of filter feeders – an assessment of hypotheses. Oikos 62: 370–380.
Sabater, S., 2008. Alterations of the global water cycle and their effects on river structure, function and services. Freshwater Reviews 1: 75–88.
Schmidt-Kloiber, A., W. Graf, A. Lorenz & O. Moog, 2006. The AQEM/STAR taxalist – a pan-European macro-invertebrate ecological database and taxa inventory. Hydrobiologia 566: 325–342.
Tachet, H., 2010. Invertébrés d’eau douce: Systématique, biologie, écologie. CNRS, Paris, France.
Venohr, M., U. Hirt, J. Hofmann, D. Opitz, A. Gericke, A. Wetzig, S. Natho, F. Neumann, J. Hürdler, M. Matranga, J. Mahnkopf, M. Gadegast & H. Behrendt, 2011. Modelling of nutrient emissions in river systems – MONERIS – methods and background. International Review of Hydrobiology 96: 435–483.
Wanner, S. C., K. Ockenfeld, M. Brunke, H. Fischer & M. Pusch, 2002. The distribution and turnover of benthic organic matter in a lowland river: influence of hydrology, seston load and impoundment. River Research and Applications 18: 107–122.
Wood, P. J. & P. D. Armitage, 1999. Sediment deposition in a small lowland stream – management implications. Regulated Rivers-Research & Management 15: 199–210.
Wotton, R. S., 1988. Very high secondary production at a lake outlet. Freshwater Biology 2: 341–346.
Wotton, R. S., C. P. Joicey & B. Malmqvist, 1996. Spiralling of particles by suspension feeders in a small lake-outlet stream. Canadian Journal of Zoology-Revue Canadienne de Zoologie 74: 758–761.
Acknowledgments
Thanks to Jürgen Schreiber, Jens Bunzel, Christina Taraschewski and Mark Leszinski for their assistance with the field and laboratory work and to Marianne Graupe, Barbara Meinck, Hannah Winckler and Grit Siegert for the seston measurements. We thank Jens Hürdler for precipitation modeling and the Environmental Agency of Brandenburg (Landesumweltamt Brandenburg) for providing the discharge data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Mariana Meerhoff
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Graeber, D., Pusch, M.T., Lorenz, S. et al. Cascading effects of flow reduction on the benthic invertebrate community in a lowland river. Hydrobiologia 717, 147–159 (2013). https://doi.org/10.1007/s10750-013-1570-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-013-1570-1