Abstract
Disturbances caused by rainfall are common in streams with the impact on stream inhabitants determined by the frequency, intensity, and predictability of the event. Here, we examine the response of winter-emerging Chironomidae (Diptera) to extreme flooding disturbance. In August of 2007, a severe flood impacted southeastern Minnesota, imparting stress on aquatic communities. Chironomid pupal exuviae collections were obtained biweekly from 18 southeast Minnesota streams during the following winter to assess resistance and resilience of winter-active chironomids to flooding. Streams examined were divided into moderate (2.5–10 cm), heavy (10–20 cm), or extreme (20+ cm) rainfall categories with rainfall amounts in each category representing total precipitation during the 3-day storm. Post-flood samples were compared to samples obtained from the same localities during prior winters. Our findings contradict studies of responses by Chironomidae to flooding during warmer-water conditions and show that winter-emerging Chironomidae are resistant to stress imposed by summer spates. Significantly more taxa emerged during winter after flooding as compared to historic collections, and the number of species emerging in winter was positively correlated with rainfall severity, indicating that 15 species responded opportunistically to disturbance. This indicates that winter-active Chironomidae may be resistant to increased severity of summer spates associated with climate change predictions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In environments that are constantly in a state of flux, such as stream systems, disturbances are best defined as events whose frequency and intensity falls outside of an otherwise predictable range (Resh et al., 1988). Disturbances caused by severe rainfall are common in streams and can have severe short-term consequences on the organisms living within these systems. The resulting impact on stream inhabitants is determined by the frequency, intensity, extent, and predictability of a particular disturbance event (Sousa, 1984; Resh et al., 1988; Grimm & Fisher, 1989; Townsend et al., 1997).
Global climate warming is accelerating evaporation rates worldwide; this extra water vapor that is added to the atmosphere is among the key drivers necessary to fuel intense storm events (Madsen & Willcox, 2012). Across the contiguous United States, extreme precipitation events that are among the largest experienced at a specific location are occurring 30% more often now as compared to 1948 (Madsen & Willcox, 2012). Other recent climate change predictions suggest that Central North America will undergo a 3% increase in mean annual precipitation (Christensen et al., 2007) and, perhaps more importantly, an increase in the intensity of individual precipitation events (Trenberth et al., 2007, Madsen & Willcox, 2012). Specifically, over the last 65 years, precipitation intensity has increased by 10% nationwide (Madsen & Willcox, 2012). Consequently, we can expect more precipitation-related disturbances to impact stream communities. This type of disturbance directly modifies flow regimes of rivers and streams and can cause increased amounts of sedimentation and streambed scouring with many studies indicating significant changes in species density, richness, and composition (e.g., Fisher et al., 1982; Miller & Golladay, 1996; Matthaei et al., 2000; McCabe & Gotelli, 2000; Suren & Jowett, 2006). Furthermore, disturbances experienced during one season may impact the recovery, diversity, and abundance of organisms active in subsequent seasons.
The consequences of disturbance to streams are often measured by assessing the resistance and resilience of stream biota, which are interpreted in the context of the ecological stability of the disturbed system (Grimm & Fisher, 1989; Boulton et al., 1992). Organisms that are resistant to a particular disturbance have the ability to absorb or deflect the effects of the disturbance with very minimal changes in density (Fig. 1a). Resilient organisms, by contrast, initially exhibit dramatic declines in density; however, are able to quickly recover to pre-disturbance levels (Fig. 1b). Some species may have both low resistance and resilience (Fig. 1c) and are either slow or unable to recover. In addition to resistant or resilient response trajectories, we propose that some species of Chironomidae (Insecta: Diptera), represent an opportunistic response to high levels of disturbance associated with flooding. By our definition, opportunistic taxa quickly become abundant following a disturbance due to the new environmental conditions (Fig. 1d), thereby influencing the short-term seasonal trajectory of recovery. These taxa may later exhibit declines after competitively superior, but less resilient, taxa are able to recolonize.
Potential responses to disturbance. (a) Community exhibiting high resistance following a disturbance. (b) Community exhibiting low resistance and high resilience. (c) Community exhibiting low resistance and low resilience. (d) Opportunistic community that is able to become abundant following disturbance
Several studies have examined the resistance and resilience of the aquatic community to severe floods during spring, summer, and fall (e.g. Fisher et al., 1982; Mackay, 1992; Miller & Golladay, 1996; Hax & Golladay, 1998; Gendron & Laville, 2000; Melo et al., 2003; Dodds et al., 2004; Fritz & Dodds, 2004). While initially imparting severe reductions in taxa abundance and richness following devastating floods, stream communities typically exhibit high levels of resilience, with populations often rebounding within days or weeks of the disturbance (Reice, 1985). For example, a study examining the effects of flooding on prairie streams found low resistance (90% decreases in total density) of the benthic community, but a high degree of resilience, especially by Chironomidae, over subsequent seasons (Miller & Golladay, 1996). This short recovery period may be facilitated by the high fecundity exhibited by most macroinvertebrates (Reice, 1985), rapid recolonization by species living in suboptimal areas such as the hyperheos (Reice, 1985; Gendron & Laville, 2000), or recolonization by individuals that were able to take advantage of sheltered refuges such as backwaters (Reice, 1985), downstream drift (Fisher et al., 1982; Gendron & Laville, 2000), or aerial dispersal (Fisher et al., 1982). While this strong research base documents effects of disturbance on the macroinvertebrate community during warmer seasons, there is little documentation of the effects of seasonal disturbances during spring or summer on winter active aquatic invertebrate populations, such as certain species of Chironomidae, which characterize streams of higher latitudes.
The Chironomidae play a particularly important role in stream dynamics. They typically comprise over 50% of aquatic insects within a stream system (Coffman & Ferrington, 1996), are among the most species-rich of aquatic insect families (Ferrington, 2008), and are a major protein source and thereby promote the growth of stream fish (Armitage, 1995b). Chironomidae are also unique in that they are among the few aquatic insect families in which several species emerge from streams and are active as adults during the cold winter months that characterize the Midwestern United States (e.g., Ferrington, 2000; Ferrington, 2007; Bouchard & Ferrington, 2009; Anderson et al., 2011). Species within the subfamilies Orthocladiinae and Diamesinae, as well as a limited number of species in the Chironominae tribe Tanytarsini, are the primary constituents of the winter chironomid community in temperate streams and rivers (Ferrington, 2000; Ferrington, 2007; Bouchard & Ferrington, 2008; Eggermont & Heiri, 2012). Some species show very specialized adaptations to cold, such as adults of Diamesa mendotae (Muttkowski), which have the ability to survive air temperature less than −20°C via freezing point depression (Carrillo et al., 2004; Bouchard et al., 2006). Over 50 Chironomidae species have been shown to emerge within different water temperature ranges from Kansas streams during winter months (Ferrington, 2000) and in this study we show that 39 species emerge from groundwater dominated trout streams in southeastern Minnesota during winter (Table 1) when ambient water temperatures do not exceed 8°C.
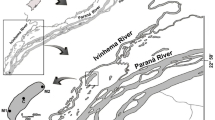
In August of 2007, a large area of southeastern Minnesota experienced extreme flooding (Fig. 2), with some areas receiving over 38 cm of rainfall in a 24-h period, far surpassing the state’s previous 24-h record rainfall total of 27 cm (Minnesota State Climatology Office, 2008). This late-summer spate imposed varying degrees of severe stress (e.g. stream bed scouring, siltation, increased runoff) on aquatic communities inhabiting the numerous economically important trout streams characterizing the area. High-quality historic data, dating back to 2002, of the winter-emerging Chironomidae in southeast Minnesota trout streams provided us with the opportunity to evaluate the effects of this late summer flood on the resistance, resilience, and response trajectory of winter-emerging Chironomidae. Specifically, we test the null hypothesis that there is no difference in chironomid species density pre- and post-flood, nor any difference in chironomid richness between streams impacted at different levels.
Southeastern Minnesota Trout streams sampled for Chironomidae. Streams sampled were divided into three rainfall categories. Actual rainfall totals for the area are depicted in the map inset on the top right (Rainfall totals inset modified from Minnesota State Climatology Office, 2008)
Materials and methods
Study area
Southeast Minnesota, USA is surrounded, but virtually untouched, by the effects of the last glaciation (Waters, 1977). This lack of glacial activity accounts for the unique landscape that distinguishes the area. The region is characterized by steep limestone bluffs, sandstone valleys, and an array of 188 spring-fed trout streams that comprise a total of 788 stream miles (Dieterman & Merten, 2003). Winter water temperatures typically range from 2 to 8°C with most streams exhibiting a temperature around 4°C.
Eighteen trout streams in southeastern Minnesota with long-term chironomid winter-emergence data were selected for study. Streams were chosen to include multiple representatives of three rainfall categories: extreme (20+ cm) (N = 6 streams), heavy (10–20 cm) (N = 5 streams), and moderate (2.5–10 cm) (N = 7 streams) amounts of rainfall (Fig. 2); each rainfall category represents total precipitation received during the 3-day storm (18–20 August, 2007). Rainfall data were obtained from the Minnesota State Climatology Office.
Field and lab methodology
Samples of chironomid pupal exuviae, the surface-floating cast-skins left behind by recently emerged adults, were collected biweekly from each stream from December 2007 to March 2008, following methods of Ferrington et al. (1991), yielding a total of 108 samples from the 18 streams over the 3-month period. Briefly, pupal exuviae were collected from multiple areas of known accumulation (e.g., areas of the stream containing foam or debris buildup) by scooping exuviae, debris, and water into a white enamel pan and pouring all contents through a 125-μm sieve. This procedure was repeated for 10 min, starting at the downstream end of each sample site and working upstream for a total distance of approximately 100 m. All scoops from an individual stream and sample date were pooled into a single sample. An important benefit of using pupal exuviae is that this technique provides estimates of individuals that were able to successfully survive all immature stages and emerge as adults. In addition, because collections are made from “natural” areas of accumulation within a stream reach, this method allows evaluation of individuals originating from a wide variety of larval habitats that might be missed with other sampling methods.
All pupal exuviae were preserved in the field using 70% ethanol and later sorted in the lab under 12× magnification to insure accurate quantification of small to large species. When available, three representatives of every taxon represented in each sample were slide mounted in Euparal® for species-level identification. Genus-level identification was achieved using Wiederholm (1986) or Ferrington et al. (2008) and species level identification was accomplished with reference to the most recent revision available for each genus. Slide-mounted voucher specimens are located in the chironomid collection at the University of Minnesota Insect Collection in St. Paul, MN (UMSP).
Historic collections
Between one and six collections of pupal exuviae were made during winter by the second author in each of the eighteen study streams between 2002 and 2007 with a cumulative total of 40 samples collected historically across the eighteen streams. All sampling techniques and sampling localities were identical to those of the post-flood collections. Species identification information from the historic collections is restricted to presence-absence data, as abundance information is not available for all historic samples.
Data analysis
A variety of numerical analysis methods were used to assess the response of winter-emerging Chironomidae to this severe flood event. In our analyses, we test the null hypothesis that there is no difference in chironomid species density pre- and post-flood, nor any difference in chironomid richness between streams impacted at different levels. Statistical programs used were Number Cruncher Statistical Software (NCSS) (Hintze, 2001), JMP Pro 9.0.2, and R (version 2.14.2). First, considering that many of the sites in each rainfall category are clustered in space (Fig. 2), we tested for any possible effects of geographic location for the historic samples. Sample localities were lumped into four categories based on latitude and analyzed by one-way analysis of variance (ANOVA) with the total number of chironomid taxa as the response variable. One-way ANOVA was also used to verify that historic samples were not significantly different from each other when grouping samples by rainfall category, again, using total number of taxa as the response. In order to evaluate whether post-flood taxonomic sample density of winter-emerging Chironomidae differed from historic sample density values for streams in each of the three rainfall categories, we chose to analyze the data by a 2 × 3 level two-way ANOVA (α = 0.05); sample density for each historic and post-flood sample was assessed and included in this analysis. The numbers of taxa emerging per sample date were not normally distributed, but closely approximated a Poisson distribution. Thus, data were transformed by adding 0.5 to the count and subsequently taking the square root. Means were then back-calculated to yield values reported here. Analysis of Variance was followed with a Tukey–Kramer Multiple Comparison Test.
Linear regression was used to analyze the relationship between actual rainfall amount incurred during the 3-day flood and the average number of taxa collected per stream in historic and post-flood samples. This analysis included data from the 40 post-flood sampling events that matched closest temporally to the 40 historic sampling occasions. Degree-day data were not available for comparison of pre- and post-flood sample sites, consequently we chose to match our samples by the nearest calendar date for the temporal comparisons; while we recognize that there are limitations with this method, we believe it is the best option given the data available.
Non-metric multidimensional scaling (NMDS) was used to assess similarity among sites based on pre- and post-flood data for temporally comparable sample dates. Post-flood patterns were also evaluated separately based on rainfall intensity and included data for all 108 post-flood samples. For both analyses, the NCSS NMDS software was set to create similarities based on presence/absence data for all taxa. Initial configuration was set to use a random starting configuration with 50 iterations and the default values for minimum stress, minimum stress change, and minimum gradient sum. Considering that we achieved stress values less than 0.10, which corresponds to a good ordination with little risk of drawing false interpretations (Clarke, 1993), we constrained our analysis to two dimensions. NMDS maps were saved as figures.
Individual-based rarefaction curves were generated by means of the vegan package (Oksanen et al., 2012) in R version 2.14.2 to assess differences in expected generic richness for a given number of randomly drawn individuals from the Moderate, Heavy, and Extreme rainfall categories. Since abundance data were not available for historic collections, only post-flood data are presented here. Data from all samples and streams within a particular rainfall category were pooled to develop seasonal richness estimates specific for each category. Considering that only a subset of all species collected in a sample could be consistently and accurately identified to species level, we chose to constrain the resolution of our abundance data to the generic level for this richness assessment; all other analyses were conducted with species level data.
Results
A total of 20 species in 14 genera were collected in the 40 historic samples, while 29 taxa in 14 genera were collected in the 40 post-flood collections that were temporally closest to the day and month of each historic sample. A cumulative total of 39 taxa within 20 genera were collected across all 108 post-flood samples (Table 1). Of the 39 taxa collected post-flood, 26 were within the subfamily Orthocladiinae (Table 1). Only two taxa, Diplocladius cultriger and Tvetenia sp. 2, were unique to historic collections; conversely, 15 taxa were unique to post-flood collections. The most common and abundant taxon in both historic and post-flood collections was Diamesa sp. Species within the genera Orthocladius and Micropsectra were also quite common.
One-way ANOVA indicated no significant difference in the number of taxa detected when comparing historic samples grouped into either latitudinal (F 3,39 = 2.38, P = 0.086) or rainfall categories (F 2,39 = 2.16, P = 0.129). Two-way ANOVA found a significant effect when examining the number of taxa collected in historic versus post-flood collections with historic samples (N = 40) significantly different than that in post-flood collections (N = 108) (F 1, 147 = 371.92, P = 0.0023). Post-flood collections exhibited an average of 2.05 more taxa per sample than historic collections. Similarly, this test found a significant effect when examining differences between rainfall categories (F 2, 147 = 9.66, P = 0.0027). A Tukey–Kramer Multiple Comparison Test indicated that all rainfall categories were significantly different from one another with the Extreme rainfall category having 2.6 more taxa than the Moderate category, and 0.9 more taxa than the Heavy category, and the Heavy rainfall category having 1.6 more taxa than the Moderate rainfall category. No interaction was detected between the collection period and rainfall category (F 2,147 = 0.01, P = 0.985).
Linear regression models indicated that in post-flood collections, the average number of taxa collected per sample per stream as a function of total rainfall amount indicated a trend of increasing number of taxa with increasing rainfall (R 2 = 0.259, P = 0.031). Historic collections, conversely, indicated no significant differences in taxa collected among the same streams during non-flood years (R 2 = 0.040, P = 0.425) (Fig. 3). The trend of increasing taxa with increasing rainfall in post-flood collections is also quite apparent in Table 1 when examining the comprehensive list of taxa collected in each rainfall category, with 20 taxa found in streams receiving moderate rainfall, 25 taxa in those receiving heavy rainfall, and a total of 31 taxa collected in streams receiving extreme amounts of rainfall (Table 1). Table 2 shows a similar trend when comparing average taxa collected per sample post-flood as compared to pre-flood.
Linear regression of average number of taxa collected per stream by rainfall amount. Points were jittered horizontally and vertically to overcome complete overlap. Linear regression analysis indicated a significant response in post-flood collections, indicating that the average number of taxa collected/stream/sample is positively correlated with rainfall (R 2 = 0.259; P = 0.031). No trend was apparent in historic collections (R 2 = 0.040; P = 0.425)
Differences among streams based on historic data were primarily captured on the Dimension 1 axis of the NMDS (Fig. 4a, stress = 0.075), where we plot samples in species space, reflecting the date-to-date differences in emergence of the smaller number of taxa pre-flood. By contrast, post-flood samples exhibited not only the date-to-date separation on Dimension 1 but also exhibited greater separation of streams along Dimension 2 with the streams that had a higher incidence of opportunistic taxa (e.g., Brillia retifinis, Corynoneura sp., Eukiefferiella spp., Dicrotendipes fumidus, Polypedilum sp., Micropsectra spp., Rheotanytarsus distinctissimus, Tanytarsus nearcticus, Zavrelimyia sinuosa) being much more strongly differentiated in this dimension. The influence of opportunistic taxa is also illustrated in Fig. 4b (stress = 0.086), where we plot streams in species space, coded by rainfall categories. Most streams with moderate amounts of rainfall occur near the middle of the NMDS plot and streams with heavy and extreme rainfall more strongly separated by both axes.
Non-metric multidimensional scaling maps of similarity among historic and post-flood data. (a) Comparison of historic (open circles) and post-flood (filled circles) data (stress = 0.075). (b) Comparison of rainfall categories using data from all post-flood samples. Open circles represent streams from the “moderate” category, gray circles represent the “heavy” category, and black circles represent streams from the “extreme” rainfall category (stress = 0.086)
Rarefaction curves of chironomid generic richness also indicated clear separation between rainfall categories with the Extreme category exhibiting the highest taxa richness estimates with increasing sample size. Streams within the Heavy category exhibited intermediate richness values, while generic richness was lowest in the Moderate rainfall category (Fig. 5). For example, based on a subsample of 1,000 individuals, sites within the Extreme category have an expected generic richness of 14.0 (SE = 1.2), while those in the Heavy or Moderate categories have expected values of 11.0 (SE = .98) and 9.6 (SE = 1.1), respectively.
Discussion
Our results indicate that winter-emerging Chironomidae exhibited high resistance to late summer flooding and were not negatively impacted by severe flooding during the preceding summer season as demonstrated by the higher number of taxa in post-flood samples. As mentioned, abundance data were not available for historic samples, which restricted the use of taxa richness estimates to post-flood data. This constrained all comparisons made between historic and post-flood data to comparisons of species density. We recognize that there are shortcomings with this type of comparison, especially considering the tendency for more abundant samples to have a higher density of species (McCabe & Gotelli, 2000; Gotelli & Colwell, 2011). However, we believe that the consistency found in the results of all analyses used to compare historic and post-flood sites, combined with the pattern of richness results in the post-flood dataset, provide a strong case in support of the conclusion that we have detected a true increase in the number of winter-emerging taxa in the post-flood samples that were most severely impacted by the flood event.
This post-flood increase in Chironomidae taxa is likely triggered by the interplay of a suite of factors. Substrate characteristics, for example, may play a major role in the reduction or persistence of aquatic insect species following severe disturbance. Studies following a single habitat sampling protocol typically sample only in erosional zones because invertebrate density and diversity are generally highest in these areas (Barbour et al., 1999). Research has shown, however, that these habitats, characterized by high amounts of loose cobble and gravel, typically have greatly reduced density and richness following spates as compared to habitats characterized by stable, embedded substrate (Matthaei et al., 2000).
Some aquatic invertebrates may be adapted to seek out areas of refugia during periods of disturbance (Sedell et al., 1990; Dole-Olivier et al., 1997). Matthaei et al. (2000), for example, found that “stable stone” substrates are used as refugia by certain aquatic insects, including Chironomidae. Analogous to the results of our study, chironomid density and richness were found to be significantly higher following the repeated disturbance in post-disturbance samples as compared to pre-disturbance, suggesting that these species may identify and seek these “stable” sites as refugia during periods of high flow (Matthaei et al., 2000).
Several of the winter-emerging species found in our research do not emerge during summer months when water temperatures are warmer, and larvae of Diamesa and five Orthocladius species from this study are not found actively growing on surface substrates during mid-summer. Although further research is needed to characterize the summer habitat and life stages of several of these winter-emerging Chironomidae species, we presume they are diapausing deep within the hyporheos, using this region as a stable refugium during the summer months before shifting to the stream bed in late autumn and winter. Benthic organisms (Füreder, 1999; Malard et al., 2002; Dole-Olivier, 2011), including several species of chironomids (Ferrington, 1984; Ferrington, 1987; Dole-Olivier et al., 1997; Donley et al., 1998), are known to use the hyporheic zone as a habitat or a refuge from environmental disturbance including floods and warm summer temperatures. For example, chironomid larvae have been found invading the hyporheos at depths of up to 2 m following high discharge events (Dole-Olivier et al., 1997). In addition, Dole-Olivier (2011) emphasizes the importance of the hyporheic zone as a protected “nursery” for eggs, early instar larva, and quiescent stages of benthic invertebrates. It is quite plausible that winter-emerging taxa found in our study followed a similar strategy by aestivating in the hyporheos as eggs or early instar larvae, thus avoiding mortality associated with stream bed redistribution due to severe flooding, and helping to explain the persistence and resistance of these taxa.
Studies of rainfall disturbance often emphasize the devastating impacts of floods on the aquatic invertebrate community, highlighting low resistance of the community (e.g., Fisher et al., 1982; Miller & Golladay, 1996; Gendron & Laville, 2000). For example, density reductions in excess of 90% are not uncommon following spates (e.g., Fisher et al., 1982; Miller & Golladay, 1996). Contrastingly, we predict that some species of Chironomidae actually benefit from extreme flood events. Of the 39 winter-emerging species collected after the late summer spate, 15 were unique to the post-flood collections and 10 of the 15 were restricted to streams receiving either heavy or extreme amounts of rainfall (Table 1). We believe that these taxa exhibited an opportunistic response, and thus were able to flourish in the altered stream conditions by quickly taking over void niches left by late-summer developing taxa that are not resistant to severe flooding, thus resulting in reduced competition levels. A study of chironomids in the River Aude also concluded that chironomid diversity was higher following flooding disturbance, and included the appearance of cold stenothermic species (Gendron & Laville, 2000). Conversely, though, while overall species density was higher the year following the flood, Gendron & Laville (2000) also reported that a number of species seemed to be expelled by flooding, and conclude that overall, chironomid resistance to flooding was low, with many populations not rebounding for several months.
In an experimental spate disturbance study, McCabe & Gotelli (2000) also reported higher invertebrate species richness following disturbance. They concluded that their results followed predictions of Huston’s (1979) “dynamic equilibrium model” for explaining ecological disturbance (McCabe & Gotelli, 2000). Under Huston’s model (Huston, 1979), which has been deemed applicable to stream systems (e.g. Resh et al., 1988), if the recurrence interval of a disturbance is less than the time required for competitive exclusion to occur, then species that are not strong competitors can thrive in the system, thereby increasing species richness. In addition, and perhaps more importantly for this research, competitive exclusion of species can be counteracted by population reductions caused by disturbance (Huston, 1979). In the case of our study, we propose that since the flood occurred at the end of the summer season and that returning to a state of semi-equilibrium within the most severely affected streams likely did not occur until the onset of cooler weather, it is possible that some competitively superior aquatic invertebrates did not have time or were otherwise unable to securely re-establishing their position before winter. It is also plausible that some species went into an early seasonal diapause to avoid consequences of the flood; behavioral responses such as suggested here are not uncommon in response to disturbance (Resh et al., 1988).
Thus, without the predominance of these “superior” species, competition within the impacted streams may have been reduced, allowing certain chironomids to take advantage of the new conditions and emerge earlier than expected by more typical phenology patterns. If, as proposed earlier, certain chironomids are able to take advantage of refugia, such as the deep hyporheos, this could certainly allow them to quickly take advantage of vacant niches. Thus, these species show an alternate, opportunistic type of behavioral response by shortening the time required for development and perhaps also altering patterns of development and voltinism. Future research testing these ideas, as well as incorporating abundance of other groups of aquatic insects along with chironomids, is crucial for evaluating these hypotheses.
Another mechanism that might stimulate these opportunistic species is related to change in the quality and quantity of food available to the aquatic insect community following the disturbance. Increased flows are likely to remove leaves and other types of coarse particulate organic matter (CPOM), causing decreases in this type of food resource (Sweeney et al., 1992). This may be especially critical in headwaters of low order streams, such as those examined in this study, shifting the dominant feeding group from shredders, who rely heavily on CPOM, to collectors, which are able to filter the fine particulate organic matter (FPOM) ingested with sediments (e.g., Lenat, 1984; Genito et al., 2002). Importantly, many chironomids, particularly some of the dominant winter-emerging species in our study, are collectors. Twelve of the unique species that were only found in post-flood collections (Table 1) are classified as collectors, thus it is quite plausible that these species are able to opportunistically use these additional food resources, such as increased FPOM resources, resulting in higher population densities or faster growth rates.
When considered at the community level, the opportunistic species introduce more heterogeneity in emergence across all of the study streams during winter. Both the historic data and the results for streams with only moderate rainfall, indicate that a small number of taxa form the nucleus of winter emergence. The more intense disturbance increased the stream-to-stream variability during the first winter following the flood. We are not sure how long this effect could persist. However, based on our interpretation that these opportunistic taxa are not efficient competitors, we expect that during one or more subsequent winters without intensive flooding these taxa will disappear or emerge later into the spring and emergence through winter will shift back to a community pattern that is more homogeneous and dominated by the nucleus of winter emerging taxa that predominated in the streams with moderate rainfall.
Work by Bouchard (2007) provides a strong basis for additional comparisons of our post-disturbance winter-emergence data. Bouchard (2007) compiled a database with estimates of the mean and ranges of water temperatures for emergence of 69 Chironomidae taxa in groundwater dominated streams. We used this database when comparing mean emergence temperature of the chironomids collected in this study (Online Supplemental Resource) and also to compare dates of first emergence for the more warm-water adapted species, defined as species with a mean emergence temperature greater than 13°C. Our data show that 9 warm-water and one colder-water adapted species are emerging earlier in our post-flood collections than recorded by Bouchard (2007). For example, the earliest emergence date for Dicrotendipes fumidus recorded by Bouchard (2007) was 21-April. Similarly, the earliest emergence for Zavrelimyia sinuosa was 10-April (Bouchard, 2007). In our collections, we found D. fumidus as early as 23-February and Z. sinuosa as early as 6-January. While we acknowledge that these results could be due to differences in sampling intensity or the individual conducting the actual sampling event, we believe this is unlikely since identical field collection protocols were used in our study, the historic collection, as well as the work done by Bouchard (2007). We believe it is more plausible that changes of in-stream condition associated with the flooding are the primary drivers accounting for earlier detection of these species.
This idea of earlier emergence is further strengthened by examining results for several additional Orthocladiinae taxa. Species within the subfamily Orthocladiinae are recognized as containing very cold-adapted taxa to less cold-adapted species, but with emergence recorded at lower temperatures than taxa within the subfamilies or tribes Tanypodinae, Chironomini, and Tanytarsini (Oliver, 1971; Coffman, 1973; Berg & Hellenthal, 1992; Ferrington et al., 1993; Armitage, 1995a; Bouchard & Ferrington, 2008). Bouchard (2007) found very few Orthocladiinae emerging during the early-winter season; the only taxon that predictably emerged throughout the early-winter was the cold-hardy Diamesa sp. Most Orthocladiinae were detected in later-winter or spring collections (Bouchard, 2007). Conversely, our results showed that many Orthocladiinae species were found throughout the early and late winter season, and as with the warm-adapted species, these species with varying degrees of cold-adaptedness were much more prevalent in the Heavy and Extreme rainfall categories. Interestingly, the most cold-adapted species found in our collections, Diamesa sp. were not found more predictably during early versus late season collections or in streams incurring more extreme rainfall. This seems to further suggest that trends of earlier emergence post-flooding are most important for warm-adapted species and moderately cold-tolerant Orthocladiinae.
Considering current climate change predictions, our results are quite significant, indicating that a strong base of winter-developing Chironomidae species will be present during the winter months as a food resource for stream fish and other higher vertebrates to feed upon. This may be particularly important for fish residing in streams recovering from devastating spring or summer floods when many alternative species of aquatic insects are temporarily reduced or displaced. Mechanisms used by the winter-emerging species described in our study consist of high resistance especially among the most cold-adapted species, life cycle flexibility in moderately cold-adapted species, and earlier emergence combined with potential increases in voltinism in species that are less cold-adapted and constrained to emergence at higher temperatures in streams with low levels of disturbance.
References
Anderson, A. M., R. W. Bouchard & L. C. Ferrington, 2011. Hibernal emergence of Chironomidae (Diptera) in relation to stream size in Kansas, USA. In Wang, X. & W. Liu (eds), Contemporary Chironomid Studies—Proceedings of the 17th International Symposium on Chironomidae. Nankai University Press, Tianjin, China: 247–255.
Armitage, P. D., 1995a. Behaviour and ecology of adults. In Armitage, P. D., P. S. Cranston & L. C. V. Pinder (eds), The Chironomidae: The Biology and Ecology of Non-biting Midges. Chapman and Hall, London: 194–219.
Armitage, P. D., 1995b. Chironomidae as food. In Armitage, P. D., P. S. Cranston & L. C. V. Pinder (eds), The Chironomidae: Biology and Ecology of Nonbiting Midges. Chapman & Hall, London: 423–435.
Barbour, M. T., J. Gerritsen, B. D. Snyder & J. B. Stribling, 1999. Rapid bioassessment protocols for use in streams and wadeable rivers: Periphyton, benthic macroinvertebrates and fish, 2nd ed. EPA/841/B/99/002. Office of Water, U.S. Environmental Protection Agency, Washington, DC.
Berg, M. B. & R. A. Hellenthal, 1992. The role of Chironomidae in energy flow of a lotic ecosystem. Netherlands Journal of Aquatic Ecology 26: 471–476.
Bouchard, R. W., 2007. Phenology and taxonomic composition of lotic Chironomidae (Diptera) communities in contrasting thermal regimes. M.S/PhD Thesis(?) University of Minnesota.
Bouchard, R. W. & L. C. Ferrington, 2008. Determination of Chironomidae thermal preferences and thermal partitioning among closely related taxa in a Minnesota stream using surface floating pupal exuviae. Boletim do Museu Municipal do Funchal Supplement 13: 191–198.
Bouchard, R. W. & L. C. Ferrington, 2009. Winter growth, development, and emergence of Diamesa mendotae (Diptera: Chironomidae) in Minnesota streams. Environmental Entomology 38: 250–259.
Bouchard, R. W., M. A. Carrillo & L. C. Ferrington, 2006. Lower lethal temperature for adult male Diamesa mendotae Muttkowski (Diptera: Chironomidae), a winter-emerging aquatic insect. Aquatic Insects 28: 57–66.
Boulton, A. J., C. G. Peterson, N. B. Grimm & S. G. Fisher, 1992. Stability of an aquatic macroinvertebrate community in a multiyear hydrologic disturbance regime. Ecology 73: 2192–2207.
Carrillo, M. A., C. A. Cannon & L. C. Ferrington Jr., 2004. Effect of sex and age on the supercooling point of the winter-active Diamesa mendotae Muttkowski (Diptera: Chironomidae). Aquatic Insects 26: 243–251.
Christensen, J. H., B. Hewitson, A. Busuioc, A. Chen, X. Gao, I. Held, R. Jones, R. K. Kolli, W. T. Kwon, R. Laprise, V. Magana Rueda, L. Mearns, C. G. Menedez, J. Raeisaenen, A. Rinke, A. Sarr & P. Whetton, 2007. Regional climate projections. In Solomon, S., et al. (eds), Climate Change 2007: The Physical Science Basis Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change.
Clarke, K. R., 1993. Nonparametric multivariate analyses of changes in community structure. Australian Journal of Ecology 18: 117–143.
Coffman, W. P., 1973. Energy flow in a woodland stream ecosystem: II. The taxonomic composition of the Chironomidae as determined by the collection of pupal exuviae. Archiv fuer Hydrobiologie 71: 281–322.
Coffman, W. P. & L. C. Ferrington Jr., 1996. Chironomidae. In Meritt, R. W. & K. W. Cummins (eds), An Introduction to the Aquatic Insects of North America, 3rd ed. Kendall/Hunt Publishing Company, Dubuque, IA: 635–754.
Dieterman, D. J. & M. Merten, 2003. History of trout management in southeast Minnesota, 1874–2003. Staff Report 61. Division of Fisheries, Minnesota Department of Natural Resources, St. Paul, MN.
Dodds, W. K., K. Gido, M. R. Whiles, K. M. Fritz & W. J. Matthews, 2004. Life on the edge: the ecology of great plains prairie streams. BioScience 54: 205–216.
Dole-Olivier, M. J., 2011. The hyporheic refuge hypothesis reconsidered: a review of hydrological aspects. Marine and Freshwater Research 62: 1281–1302.
Dole-Olivier, M. J., P. Marmonier & J. L. Beffy, 1997. Response of invertebrates to lotic disturbance: is the hyporheic zone a patchy refugium? Freshwater Biology 37: 257–276.
Donley, S., L. C. Ferrington & D. Strayer, 1998. The habitat of Paraboreochlus larvae (Diptera: Chironomidae). Journal of the Kansas Entomological Society 71: 501–504.
Eggermont, H. & O. Heiri, 2012. The chironomid-temperature relationship: expression in nature and palaeoenvironmental implications. Biological Reviews 87: 430–456.
Ferrington, L. C., 1984. Evidence for the hyporheic zone as a microhabitat of Krenosmittia spp. larvae (Diptera, Chironomidae). Journal of Freshwater Ecology 2: 353–358.
Ferrington, L. C., 1987. Microhabitat preferences of larvae of three Orthocladiinae species (Diptera, Chironomidae) in Big Springs, a sandbottom spring in the High-Plains of western Kansas. Entomologica Scandinavica 29: 361–368.
Ferrington, L. C., 2000. Hibernal emergence patterns of Chironomidae in lotic habitats of Kansas versus ambient air and water temperatures. In Hoffrichter, O. (ed.), Late 20th Century Research on Chironomidae: an Anthology from the 13th International Symposium on Chironomidae. Shaker Verlag, Aachen: 375–382.
Ferrington, L. C., 2007. Hibernal emergence patterns of Chironomidae in lotic habitats of Kansas versus substrate composition. In Anderson, T. (ed.), Contributions to the Systematics and Ecology of Aquatic Diptera—A Tribute to Ole A Saether. The Caddis Press: 99–105.
Ferrington, L. C., 2008. Global diversity of non-biting midges (Chironomidae; Insecta-Diptera) in freshwater. Hydrobiologia 595: 447–455.
Ferrington, L. C., M. A. Blackwood, C. A. Wright, N. H. Crisp, J. L. Kavanaugh & F. J. Schmidt, 1991. A protocol for using surface-floating pupal exuviae of Chironomidae for rapid bioassessment of changing water-quality. IAHS Publication 203: 181–190.
Ferrington, L. C., K. M. Buzby & E. C. Masteller, 1993. Composition and temporal abundance of Chironomidae emergence from a tropical rain-forest stream at El-Verde, Puerto-Rico. Journal of the Kansas Entomological Society 66: 167–180.
Ferrington, L. C., M. B. Berg & W. P. Coffman, 2008. Chironomidae. In Merritt, R. W., K. W. Cummins & M. B. Berg (eds), An Introduction to the Aquatic Insects of North America, Vol. 4th, 4th ed., Kendall/Hunt Publishing Company Dubuque, IA: 847–989.
Fisher, S. G., L. J. Gray, N. B. Grimm & D. E. Busch, 1982. Temporal succession in a desert stream ecosystem following flash flooding. Ecological Monographs 52: 93–110.
Fritz, K. M. & W. K. Dodds, 2004. Resistance and resilience of macroinvertebrate assemblages to drying and flood in a tallgrass prairie stream system. Hydrobiologia 527: 99–112.
Füreder, L., 1999. High alpine streams: cold habitats for insect larvae. In Margesin, R. & F. Schinner (eds), Cold Adapted Organisms: Ecology Physiology, Enzymology, and Molecular Biology. Springer-Verlag, Berlin, Germany: 181–196.
Gendron, J. M. & H. Laville, 2000. Impact of a catastrophic flood on the chironomid populations (Diptera: Chironomidae) of the hyporhithral (4th order) zone of the river Aude (Eastern Pyrenees). In Hoffrichter, O. (ed.), Late 20th Century Research on Chironomidae: An Anthology from the 13th International Symposium on Chironomidae. Shaker Verlag, Aachen: 383–391.
Genito, D., W. J. Gburek & A. N. Sharpley, 2002. Response of stream macroinvertebrates to agricultural land cover in a small watershed. Journal of Freshwater Ecology 17: 109–119.
Gotelli, N. J. & R. K. Colwell, 2011. Estimating species richness. In Magurran, A. E. & B. J. McGill (eds), Biological Diversity. Oxford University Press, New York.
Grimm, N. B. & S. G. Fisher, 1989. Stability of periphyton and macroinvertebrates to disturbance by flash floods in a desert stream. Journal of the North American Benthological Society 8: 293–307.
Hax, C. L. & S. W. Golladay, 1998. Flow disturbance of macroinvertebrates inhabiting sediments and woody debris in a prairie stream. American Midland Naturalist 139: 210–223.
Hintze, J., 2001. NCSS and Pass. Number Cruncher Statistical Systems, Kaysville, Utah.
Huston, M., 1979. A general hypothesis of species diversity. American Naturalist 113: 81–101.
Lenat, D. R., 1984. Agriculture and stream water-quality—a biological evaluation of erosion control practices. Environmental Management 8: 333–343.
Mackay, R. J., 1992. Colonization by lotic macroinvertebrates: a review of processes and patterns. Canadian Journal of Fisheries and Aquatic Sciences 49: 617–628.
Madsen, T. & N. Willcox, 2012. When it Rains it Pours. Environment America Research and Policy Center, Boston, MA.
Malard, F., K. Tockner, M. J. Dole-Olivier & J. V. Ward, 2002. A landscape perspective of surface-subsurface hydrological exchanges in river corridors. Freshwater Biology 47: 621–640.
Matthaei, C. D., C. J. Arbuckle & C. R. Townsend, 2000. Stable surface stones as refugia for invertebrates during disturbance in a New Zealand stream. Journal of the North American Benthological Society 19: 82–93.
McCabe, D. J. & N. J. Gotelli, 2000. Effects of disturbance frequency, intensity, and area on assemblages of stream macroinvertebrates. Oecologia 124: 270–279.
Melo, A. S., D. K. Niyogi, C. D. Matthaei & C. R. Townsend, 2003. Resistance, resilience, and patchiness of invertebrate assemblages in native tussock and pasture streams in New Zealand after a hydrological disturbance. Canadian Journal of Fisheries and Aquatic Sciences 60: 731–739.
Miller, A. M. & S. W. Golladay, 1996. Effects of spates and drying on macroinvertebrate assemblages of an intermittent and a perennial prairie stream. Journal of the North American Benthological Society 15: 670–689.
Minnesota State Climatology Office, 2008. Heavy rain falls on southeastern Minnesota: August 18-20, 2007. http://climate.umn.edu/doc/journal/flash_floods/ff070820.htm Accessed Web Page 2009.
Oksanen, J., F. G. Blanchet, R. Kindt, P. Legendre, P. R. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens, & H. Wagner. 2012. vegan: Community ecology package. R package version 2.0-4. http://CRAN.R-project.org/package=vegan.
Oliver, D. R., 1971. Life history of the Chironomidae. Annual Review of Entomology 16: 211–230.
Reice, S. R., 1985. Experimental disturbance and the maintenance of species diversity in a stream community. Oecologia 67: 90–97.
Resh, V. H., A. V. Brown, A. P. Covich, M. E. Gurtz, H. W. Li, G. W. Minshall, S. R. Reice, A. L. Sheldon, J. B. Wallace & R. C. Wissmar, 1988. The Role of Disturbance in Stream Ecology. Journal of the North American Benthological Society 7: 433–455.
Sedell, J. R., G. H. Reeves, F. R. Hauer, J. A. Stanford & C. P. Hawkins, 1990. Role of refugia in recovery from disturbances: modern fragmented and disconnected river systems. Environmental Management 14: 711–724.
Sousa, W. P., 1984. The role of disturbance in natural communities. Annual Review of Ecologyical Systems 15: 353–391.
Suren, A. M. & I. G. Jowett, 2006. Effecs of floods versus low flows on invertebrates in a New Zealand gravel-bed river. Freshwater Biology 51: 2207–2227.
Sweeney, B. W., J. K. Jackson, J. D. Newbold & D. H. Funk, 1992. Climate change and the life histories and biogeography of aquatic insects in eastern North America. In Firth, P. & S. G. Fisher (eds), Global Climate Change and Freshwater Ecosystems. Springer-Verlag, New York: 143–176.
Townsend, C. R., M. R. Scarsbrook & S. Doledec, 1997. The intermediate disturbance hypothesis, refugia, and biodiversity in streams. Limnology and Oceanography 42: 938–949.
Trenberth, K. E., R. D. Jones, P. Ambenje, R. Bojariu, D. Easterling, A. Klein Tank, D. Parker, F. Rahimzadeh, J. A. Renwick, M. Rusticucci, B. Soden & P. Zhai, 2007. Observations: surface and atmospheric climate change. In Solomon, S., et al. (eds), Climate Change 2007: The Physical Science Basis Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA.
Waters, T. F., 1977. The Streams and Rivers of Minnesota. University of Minnesota Press, Minneapolis, MN.
Wiederholm, T., 1986. Chironomidae of the Holarctic region—keys and diagnoses, Part 2, Pupae. Entomologica Scandinavica, Supplement 28: 1–471.
Acknowledgments
The authors wish to thank J. Anderson, S. Anderson, D. Ferrington, and three anonymous reviewers for their critical reading of this manuscript and W. Bouchard, E. Hannesdottir, and B. Scheutz for help with conceptualization of ideas. Thanks also to J. Anderson, S. Anderson, C. Jefferson, and J. Jarvis for field work assistance. This work was supported by the Minnesota Agricultural Experiment Station Project, the University of Minnesota College Of Food, Agriculture, and Natural Resource Sciences Global and Environmental Change Graduate Fellowship, and the University of Minnesota Department of Entomology Sping and Ying-ngoh T. Ling Graduate Fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Katya E. Kovalenko
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Anderson, A.M., Ferrington, L.C. Resistance and resilience of winter-emerging Chironomidae (Diptera) to a flood event: implications for Minnesota trout streams. Hydrobiologia 707, 59–71 (2013). https://doi.org/10.1007/s10750-012-1406-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-012-1406-4