Abstract
Ostracods are important members of the benthos and littoral communities of lake ecosystems. Ostracods respond to hydrochemistry (water chemistry) which is influenced by climatic factors such as water balance, temperature, and chemicals in rainfall runoff from the land. Thus, at local scales, environmental preferences of ostracods and characteristics of lakes are used to infer changes in climate, hydrology, and erosion of lake catchments. This study addresses potential drivers of ostracod community structure and biodiversity at multiple spatial scales using NMS, CART®, and multiple regression models. We identified 23 ostracod species from 12 lake sites. Lake area, maximum depth, spring conductivity, chlorophyll a, pH, dissolved oxygen, sedimentary carbonate, and organic matter all influence ostracod community structure based on our NMS. Based on regression analysis, lake depth, chlorophyll a, and total dissolved solids best explained ostracod richness and abundance. Land uses are also important community structuring elements that varied with scale; locally and regionally agriculture, wetlands, and grasslands were important. Nationally, using regression tree analysis of lakes sites in the North American Non-marine ostracod database (NANODe), row-crop agriculture was the most important predictor of biodiversity. Low agriculture corresponded to low species richness but greater landscape heterogeneity produced sites of high ostracod richness.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ostracods have calcium carbonate shells that are well preserved in both marine and lacustrine environments. Ostracods have a long geologic history; they first appeared in the Ordovician Period 485 million years ago (mya) (Williams et al., 2008). The first freshwater ostracods were first found in Devonian (360 mya) deltaic deposits (Swain, 1999) and have since become widespread in all freshwater aquatic environments. High preservation potential and a long geologic history make ostracods particularly useful in paleoecology studies (see Carbonel et al., 1988). There are approximately 300 extant nonmarine ostracod species known from North America and a large number of undescribed species (Martens et al., 2008). Ostracods have become important members of microcrustacean assemblages of lakes (Diner et al., 1986; Dodson et al., 2005).
Previous researchers have focused on single spatial scales to document the occurrence of ostracods and the trace element chemistry (hydrochemistry) of the water in which they are found. There is a large body of research using dated lake cores with the primary goal of extrapolating past climate and environmental conditions based on known species tolerances (Forester, 1986; Smith, 1993; Löffler, 1997; Curry, 1999; Forester et al., 2005a) and temporal changes in shell chemistry (Xia et al., 1997; Saros et al., 2000; Schwalb & Dean, 2002). In contrast to diatoms (Battarbee, 2000), ostracods have been under-utilized in paleoproductivity studies of lacustrine systems. Because ostracod species are indicators of past water chemistry and specific environments under different climate regimes, they are becoming increasingly important in environmental paleoecology studies (Curry, 1999; Battarbee, 2000; Miller et al., 2000; Delorme, 2001; Umbanhower, 2004; Mezquita et al., 2005). At local scales, faunal change has also been related to changes in lakes or their catchments (Alin et al., 1999; Külköylüoglu & Dügel, 2004).
The relationship between species richness and productivity is critical to an understanding of biodiversity in lakes and is a central theme in ecology (Dodson et al., 2000; Mittelbach et al., 2001) and paleoecology (Rosenzweig, 1995; Martens et al., 2008). Despite a great deal of research on richness–productivity relationships, little is known about the changes in ostracod community richness across productivity gradients.
Ostracods have been used as low-cost indicators of human impact on marine and brackish water systems (Frenzel & Boomer, 2005; Ruiz et al., 2005; Keatings et al., 2010), and lakes (Dügel et al., 2008). However, modern relationships between ostracods and the landscape are necessary to document temporal human impacts, for example, pre- and post-European settlement in the Midwest. Land use and land cover change and their effects on water quality and hydrochemistry are well established (Johnson et al., 1997; Jones et al., 2001). Since ostracods respond to hydrochemistry, habitat change and productivity that are affected by human land use, it follows that ostracods and their biodiversity will also be affected by land use. The intensified human use of the landscape (e.g., urban and agriculture) has been correlated with declines in zooplankton richness in small lakes (Dodson et al., 2005). Dodson et al. (2005) found that rare species were eliminated from plankton assemblages in largely agricultural catchments. Similarly, some ostracod studies have shown a relationship between high abundance of cosmopolitan species and poor water quality (Külköylüoglu, 2004; Külköylüoglu & Dügel, 2004).
Although ostracods have been collected from a small percentage of Wisconsin lakes, few in-depth studies have been conducted (Kitchell & Clark, 1979; Smith & Dai, 1993; Smith, 1997; Forester et al., 2005b). Kitchell & Clark (1979) conducted such a study of Lake Mendota, Wisconsin, USA, and contrary to findings for other taxa; they reported that both ostracod species richness and abundance appeared to increase following Euro-American settlement of the region and the introduction of agriculture. This historic change in community structure was accompanied by an increase in the proportion of organic matter versus carbonate in the lake sediment, suggesting a shift in the benthic resource base available to ostracods (Löffler, 1979; Dean & Schwalb, 2002).
The overarching goal of this study was to obtain a better understanding of ostracod ecology and drivers of ostracod biodiversity at multiple spatial scales. Specifically, we had the following objectives in mind:
-
(1)
Identify the chemical and physical characteristics (e.g., salinity, productivity surrogates, and water depth) of the lakes that are important to ostracod species occurrence and biodiversity.
-
(2)
Identify the land attributes that influence ostracod richness, species occurrence and abundance, and the chemical and biological characteristics of lakes to which ostracods respond.
-
(3)
Test for associations between the occurrences of specific macrophytes and ostracods to further document habitat preferences.
-
(4)
Determine the regional ostracod biodiversity in small eutrophic lakes of southeastern Wisconsin, and test the hypothesis that a unimodal ostracod diversity–productivity relationship exists as is true for other microcrustaceans (Dodson et al., 2000).
-
(5)
Assess the relative importance of different land uses on biodiversity of ostracods at broad spatial scales.
Methods
Site selection
Twelve study lakes (Fig. 1) were chosen to be as similar as possible in terms of climate, geography, and morphometry. The purpose of this approach was minimizing the effects of within-lake variation in environmental drivers and increasing the potential for detecting indirect effects of land use on ostracod community structure. The lakes are all located in the formerly glaciated region of southern Wisconsin in the Southeastern Wisconsin Till Plain Ecoregion as defined by Albert (1995), which covers about 14.5 million ha. Other site selection criteria included the following: (1) lakes were dimictic having two periods of stratification and mixing, with surface areas <40 ha, (2) lakes were at least 6.5 m deep, and (3) lakes and regional watersheds were similar in size. In addition, two reference lakes from a reference watershed were chosen to act as controls in a protected area that has experienced minimal impacts from human settlement compared to others in the region.
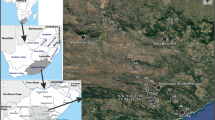
Site locations. Twelve lakes from Southwestern Wisconsin were sampled for this study. All of the lakes fall within the Till Plain Ecoregion as described by Albert (1995)
Lake sampling
The 12 study lakes were sampled four times, once in the spring and once in late summer for each of 2 years (2002 and 2003). Standard lake morphometry and water chemistry parameters were measured (Allen, 2005). The Trophic State Index (TSI) for each lake was calculated from Secchi disk depths (Panuska & Kreider, 2002).
Water samples were analyzed at the Wisconsin State Laboratory of Hygiene for total phosphorus (EPA methods 365.1; colorimetric titration) and chlorophyll a by fluorescence following Welschmeyer (1994). Trace element analyses were conducted at the University of Wisconsin Plant Analysis Laboratory. Major ions (Sr, F, Cl, Br, NO2, NO3, PO4, and SO4) were measured by ion chromatography (DIONEX DX 500 ion chromatography and HPLC system). Elemental analysis (P, K, Ca, Mg, S, Zn, Mn, Bo, Cu, Fe, Na, Al) was completed by inductively coupled plasma mass spectrometry (ICP-MS), except for Cd, Cr, Pb, and Ni, which were analyzed using gas chromatography coupled with mass spectrometry (GCMS). Total organic carbon (TOC) content of water samples was determined by colorimetric titration.
The organic matter and carbonate content of the sediment samples were determined by loss-on-ignition or LOI (Dean, 1974). Insoluble residues, the mineral matter remaining after LOI, were also recorded. Sediment samples were also subjected to several treatments of dilute hydrochloric acid followed by double distilled water rinses and centrifuging to remove carbonate material following a method used by the Paleoecology lab at the University of Wisconsin Climate Research Center (M. Winkler, pers. com., 2004). Samples were then air dried and ground in a pestle and mortar. Pre-treated sediment samples were submitted to the University of Wisconsin Soil and Plant Analysis Laboratory for stable isotope analysis on the organic matter fraction using a high temperature combustion organic carbon mass analyzer (Carlo Erba elemental analyzer coupled with a Europa 20/20 tracemass).
Macrophytes were collected following the procedure outlined in Jessen & Lound (1962). Two rake samples were collected at each littoral ostracod sample site. Species were identified using Fassett (1957) and Borman et al. (1997), and reference collections at the University of Wisconsin herbarium in Madison, WI, and species presence were recorded.
Ostracod sampling and species identification
Standard micropaleontology techniques were applied in the collection and processing of ostracods (Kummel & Raup, 1965) in littoral and profundal sediments. Littoral sediment samples (about 0.2 m depth) were taken from a bed of the most common aquatic macrophyte in each lake, away from inlets or outlets when present. Profundal sediment samples were taken from the deepest point in the lake. The top 2 cm of sediment was skimmed from grab samples collected with an Ekman Dredge and placed in plastic bags. Samples were kept on ice and either refrigerated for immediate analysis or frozen for later analysis. 20 cm3 (2–10 cm3 aliquots of sediment per sample) were sieved using an 80-μm brass sieve. All ostracods from each sample were picked out and placed on standard micropaleontology slides for identification and counting using a binocular microscope.
Ostracod species were identified using a number of standard North American references (Hoff, 1942; Tressler, 1959; Staplin, 1963; Delorme, 1967, 1970a, b, c, d, 1971, 2001; Kitchell & Clark, 1979; Meisch, 2000; Karonovic, 2006). Total ostracod abundance for each lake was calculated by taking the sum of both littoral and profundal counts from each of two 10 cm3 aliquots from each sample and lake region. Ostracod valve abundances from the count data were standardized by dividing the number of valves by the dry sample weights to obtain valve abundance g−1 of dry sediment (Kitchell & Clark, 1979).
Land use analysis
This study relates ostracod ecology to land use at multiple spatial scales: riparian zone, catchment basin, and regional watershed. Additionally, we assessed the effects of land use on biodiversity of ostracods across the country (lower 48 states of the United States; Fig. 2). We defined the riparian zone as the terrestrial environment within 90 m of the lake, using the lake boundaries from Wisconsin Department of Natural Resources (WDNR) hydrography coverage (1999a). Riparian areas were determined by placing a buffer (three grid cells) around each of the study lakes using ArcView 3.0 (Elkie et al., 1999). Catchment basins are land areas that drain from surrounding uplands into a lake. Catchment areas were quantified by processing a 30-m digital elevation model at a 30-m grid cell size using ArcView 3.0 (Elkie et al., 1999) and a 30-m digital elevation model with a 30-m grid cell size, based on data from the University of Wisconsin Forest Ecology and Management laboratory (T. Sickley, per. comm., 1999). We defined the regional watershed as a larger scale surface than the catchment basin; using the WDNR (1999a) identified watersheds. The regional watershed is similar to the level-5 hydrologic cataloging units of the United States Geological Survey (Griffith et al., 1999). There are 328 regional watersheds in the Southeastern Wisconsin Till Plain Ecoregion (WDNR, 1999a), and the regional watershed often includes more than one lake catchment basin. We chose two lakes from each of six regional watersheds that varied in their proportions of human impact. Lakes varied in land use from the least amount of human impact (Nature Conservancy preserves with no agriculture in the riparian zone) to those with approximately 25% agricultural land use in the riparian zone. Land cover proportions for each of the three landscape scales were based on WISCLAND data derived from satellite imagery collected between 1991 and 1993 and processed at a 30-m grid cell size (WDNR, 1999b). The WISCLAND land coverage of 29 land use classes (level III classification) was collapsed to 12 cover classes. Land cover proportions were calculated using Patch Analyst, grid version for ArcView 3.0 (Elkie et al., 1999).
Point coverage of lakes in NANODe. Distribution of lakes sampled for ostracods that are part of the NANODe database (Forester et al., 2005b)
We obtained georeferenced locations of ostracods samples from lakes that form part of the North American Nonmarine ostracod database (NANODe) (B. Curry, IL Geol. Sur., per. comm., 2001) for the continental United States. We intersected the lake sample locations with the hydrologic landscape regions (HLRs) coverage obtained from USGS. HLRs are groups of watersheds that share landform, soil and bedrock permeability, relief, and effective precipitation (mean annual precipitation minus potential evapotranspiration) (Wolock et al., 2004); these attributes are similarly used to establish ecoregions and their subregions. We selected the HLRs corresponding to sampled lakes as a way to minimize climate and concentrate on land use influences. We determined land cover proportions of each HLR based on the 2001 National Land cover data set (NLCD) using Patch Analyst grid in ArcView 3.3.
Statistical analyses
Ordination
We used nonmetric multidimensional scaling (NMS) to produce an ostracod community (species composition) by site ordination using PC-ORD Version 4.17 for Windows (McCune & Mefford, 1999; McCune et al., 2002). Ostracod species presence–absence data were first transformed using Beal’s smoothing, which is appropriate for data sets with large numbers of zeros (>50%). Sørensen dissimilarity distance measures were then used in the ordination, because the Sørensen distance measure is more sensitive to heterogeneous data sets and down-weights outliers better than other techniques (McCune et al., 2002). Lake scores from the species ordination were correlated with untransformed hydrochemical, and land use data, and the macrophyte presence–absence data matrix. A total of 137 physicochemical and biotic variables were assessed for their importance in structuring the ostracod communities.
Regression
StatSoft, Inc. (2000) was used in multiple linear regression analysis to test the significance of the relationship between the response variables species richness and abundance and productivity predictors identified as important from the NMS ordination. We used Mallows Cp values as our criterion for retaining predictors of richness and abundance in multiple linear regression models (StatSoft, Inc., 2007).
In addition, we constructed a Sørensen dissimilarity matrix of common macrophytes (those present in at least two lakes) and performed a Mantel test of the hypothesis that there is no significant relationship between the ostracod and macrophyte dissimilarity (distance) matrices and thus no significant relationship between the occurrence of common ostracods and the occurrence of common macrophytes. The probability of a Type I error, and therefore the significance of the Mantel statistic (r), was determined by randomization by 1000 Monte Carlo permutations (McCune et al., 2002).
CART®
We used the Salford Systems CART Pro 6.0® program for classification and regression tree analysis. CART® is a non-parametric and non-linear method that produces dichotomous decision (regression) trees as predictive models. Both predictors and dependent target variables can be either categorical or continuous. We used 13 land use classes within the HLRs to predict ostracod species richness (our dependent variable) in the 451 lakes from the NANODe database. Land use classes included urban (low, medium, and high intensity), agriculture (pasture and row crops), forest (evergreen, deciduous, and mixed), wetlands (herbaceous and woody), barren areas, grassland, shrubland, and open water. We used cross-validation to validate our models. A series of if–then dichotomous decisions are produced that are based on predictions with the lowest probability of misclassification or the smallest variance.
Results
The ostracod community
Twenty-three ostracod species were identified in the study lakes (Table 1; Table S1). Twenty of these species were found in at least two lakes. Richness varied between 3 and 13 species per lake, with an average of 7.8 species. The overall average abundance of ostracod valves per dry gram of sediment in profundal and littoral zone sediment was 157 ± 87.5 standard deviation valves and 179 ± 125.3 valves, respectively. Differences between duplicate samples (profundal + littoral) of log10(x + 1) transformed data were not significant based on a single factor ANOVA of seven duplicate samples (P = 0.84; F = 0.042).
Ostracod community ordination
An optimal two-dimensional ordination was found for the ostracod communities, using the occurrence of species in the 12 lakes (Fig. 3). This model was determined based on an initial null model that the probability of arriving at a final stress level of randomly generated data sets is less than expected for the observational data set. Monte Carlo simulations based on 200 randomizations were compared to 150 runs with the observational data set. The probability that the solution could be arrived at by chance was less than 5%. The final model produced a stress level of 3.3 and final instability of 0.00028 after 87 iterations. The NMS model accounted for 98.1% of the variability in the ostracod distance matrix; the first axis represented 89.0% and the second axis accounted for 9.1% of the variability.
NMS loadings. Results from the ordination of lakes by the component ostracod species (community composition). The four panels show variables that were highly correlated with the two ordination axes: A ostracod species, B water and sediment chemistry, C common aquatic macrophytes, and D land use variables
The lakes form two clusters along the first axis and are further separated along the gradient of the second axis (Fig. 3). The major division of the ostracod communities and those most correlated with the primary ordination axis is between those containing Potamocypris smaragdina and Physocypria globula to those containing Cypria ophtalmica, cyclocyprids, and Candona (Candona) inopinata (Fig. 3A; Table S2 in supplementary materials contains the results of our ordination). The gradient of the second axis is from communities dominated by abundant large candonids like Candona (Candona) ohioensis and Candona (Candona) crogmaniana and those containing rare specimens of Candona (Candona) acuta.
We identified environmental variables important to the observed distribution of the ostracods by correlating raw physicochemical, land use, and macrophyte presence–absence data with the lake scores obtained from the ostracod species ordination. Subsequent discussions focus on important variables determined in this way.
Lake attributes and ostracod community structure
A complete list of variables used in the NMS analysis and correlated with the ostracod data can be found in Allen (2005). The results presented here include 19 environmental variables and 6 macrophyte species that we found to load heavily on either of the two NMS axes and were therefore interpreted as important variables explaining the distributions of ostracods among the lakes.
Water chemistry
The study lakes included sites ranging from 6.7 to 18.6 m maximum depth and 7.7 to 35.2 ha surface area, respectively. Based on Secchi disk depths (1.0–6.7 m and chlorophyll a (2.0–26.7 μg l−1), the study lakes ranged from mesotrophic to eutrophic. Other chemical variables strongly correlated with the ordination axes include average total phosphorus (20–260 μg l−1), spring pH at the sediment–water interface (7.1–8.8), and spring dissolved oxygen (DO) at the sediment–water interface (0.4–7.0 mg l−1). Daytime average limnetic oxygen concentrations were at or near saturation (8.1–12 mg l−1), but summer oxygen concentrations at the sediment–water interface were more variable (0.4–5.6 mg l−1). Sulfur concentrations ranged from 1.14 to 10.93 mg l−1 and spring conductivity ranged from 280 to 615 mg l−1; these parameters are related to organic matter content (3.5–8.6%) and insoluble residues (2.8–26.4%) of bottom sediment.
Several surrogates of primary productivity were correlated with the first ordination axis (Fig. 3B). The lakes clustered to the left of the graph had higher spring chlorophyll a concentrations and lower oxygen and pH concentrations at the sediment–water interface than those to the right. Factors negatively correlated with the second axis are also positively correlated with total phosphorus, total dissolved solids (TDS) (including the trace elements magnesium and sulfur), spring and summer conductivity, and greater insoluble residue content. The amount of sedimentary organic matter was also positively correlated with this axis.
Relationship between ostracod and macrophyte occurrence
The occurrence of common macrophyte species was correlated with the NMS axes (Fig. 3C). The primary axis loaded negatively on Nymphaea and Nuphar, and positively on the invasive species Potamogeton crispus (curly leafed pondweed). The second axis was negatively correlated with an invasive, Myriophyllum spicatum (Eurasian watermilfoil), and positively correlated with native Myriophyllum heterophyllum.
The significant Mantel statistic (r = 0.43, P = 0.008) indicated a strong positive correlation between ostracod and macrophyte community structure. Both ostracod and macrophyte community structure are based on the most common species that we defined as occurring in two or more study lakes.
Species richness and abundance related to lake depth and productivity
From NMS, we identified TDS, spring chlorophyll a, and average total phosphorus as important productivity surrogates. We also identified lake area, lake depth, and elevation as important lake attributes. Using simple linear regression of these log transformed predictor variables, species richness was found to be dependent on lake depth (R 2 = 0. 39, P = 0.031; Fig. 4A); the highest ostracod species richness, 13, was found in Lulu Lake which is of intermediate water depth. Richness was also dependent on TDS (R 2 = 0.52, P = 0.009; Fig. 4B). On the other hand, although TDS also explains abundance (R 2 = 0.60, P = 0.003), lake depth only marginally explains ostracod abundance (R 2 = 0.31, P = 0.059).
Ostracod richness and abundance related to lake depth and total dissolved solids (TDS). A The relationship between log ostracod species richness or total number of ostracods, and log lake depth. Richness versus depth was significant (R 2 = 0.39; P = 0.03). B The relationship between log ostracod species richness or total number of ostracods and the log of the TDS for the 12 study lakes. Richness versus TDS was significant (R 2 = 0.52; P = 0.009)
Variance inflation factors that measure multi-colinearity of the predictors in the multiple regression models were all less than 10. While the richness model was dependent on TDS and lake depth (R 2 = 0.66; P = 0.008), the best model explaining abundance includes chlorophyll a in addition to TDS (R 2 = 0.71; P = 0.003). We further regressed the residuals from the regression of richness and lake depth on our productivity surrogates, and found that after accounting for lake depth, spring chlorophyll a was also significant (R 2 = 0.33; P = 0.051) (Fig. 5).
Ostracod richness and chlorophyll a after accounting for lake depth. A linear regression of log(10) ostracod richness versus log(10) lake depth was performed. We then regressed the residuals from this regression on important productivity surrogates individually (spring chlorophyll a, average total phosphorus, and TDS) identified in the NMS to test the relationship of species richness to productivity while accounting for the influences of lake depth. The relationship of richness to spring chlorophyll a and accounting for lake depth was significant (R 2 = 0.33; P = 0.051)
Land use and the ostracod community
Land cover types associated with the lakes were determined for riparian areas, lake catchments, and watersheds, and were correlated with the ostracod community ordination axes (Fig. 3D; Table 2, Table S1). The first NMS axis was highly positively correlated with riparian deciduous forests and negatively correlated with regional (watershed scale) total forests and grassland. The second NMS axis was highly correlated with local land cover; it was negatively correlated with row-crop agriculture and coniferous forest land and positively correlated with wetlands in the catchments. Conifers and mixed conifer and deciduous forests are often associated with areas of human impact, whereas higher proportions of grasslands intermixed with forested areas occur in areas receiving less human disturbance.
On a national scale, the Hydrologic landscape regions (HLRs) or mini-ecoregions are each approximately 200,000 ha (2,000 km2). The CART® analysis of the NANODe database produced a decision tree with four terminal nodes. The most important land use predictors of ostracod diversity were cropland (importance value or IV of 100 on a scale of 1 to 100); evergreen forests (IV = 83.56); percent open water on the landscape (IV = 48.15); deciduous forests (IV = 45.72); pastureland (IV = 44.20), mixed forests (IV = 43.45), and herbaceous wetlands (IV = 8.78). Ostracod diversity is largely affected by row-crop agriculture; where row crops were less than 0.3% of the HLRs, the average richness was 2.3; this represented 14.6% of lakes in the database (Fig. 2). Where row crops represented a higher percent of the landscape and where more than 3% of the landscape was open water, the average richness increased to approximately six species and represented 38.4% of the lakes. Terminal node 2 had the greatest number of sites (44.3%) and an average richness of 4.2. This node represents a landscape with >0.3% row crops and <3% open water. The highest average richness values (8.4) were found where row crops were >0.3%, water >3%, deciduous forests were >4.3% and mixed forests and evergreen forest percentages were low but present.
Discussion
The regional ostracod assemblage—richness and productivity
Species richness levels, similar to our results (3–13 species), have been reported for other modern lacustrine ostracod communities in Wisconsin (Kitchell & Clark, 1979; Smith & Dai, 1993; Smith, 1997; Forester et al., 2005a). For Lake Winnebago (56,000 ha), Smith (1997) reported a total of nine ostracod species in the entire sediment record and a maximum of six species in recent surface sediments; total richness for each core sample was not reported. Candona (Candona) ohioensis was the dominant species from 4,800 years ago (4.8 ka) to the present in Lake Winnebago. Miller et al. (2000) and Smith & Dai (1993) reported a total of 13 ostracod species from a core collected from Europe Lake (20 ha); and recent sediment samples that contained 7 species. Kitchell & Clark (1979) reported 19 species from recent sediment of Lake Mendota (3,940 ha) and 10 species from one sediment core. Kitchell & Clark also reported finding Cytherissa lacustris. The NANODe database (Forester et al., 2005b) contains 34 Wisconsin sites; 7 of these sites are in the Till Plain Ecoregion near our study lakes. Forester et al. (2005b) report a total of 22 ostracod species from Till Plain lakes in the database with a species composition similar to lakes in this study. The same species found today are found in Holocene deposits throughout the Midwest. However, species richness cannot be directly compared to lakes that do not meet our selection criteria such as larger lakes, those outside the climate zone, or those that are not the same type (dimictic).
Although temporal change in richness and what drives it are of the utmost interest to Quaternary and environmental paleoecologists, a temporal analysis is beyond the scope of the present study which emphasizes differences in recent ostracod communities over multiple spatial scales. Research on temporal change in richness and indeed differences over large spatial scales must account for differences in regional geomorphic templates and climate, which impose higher level constraints (sensu Allen & Starr, 1982) on ecosystem processes. We attempted to account for climate and geomorphology spatially with an in-depth study of several small lakes from a single ecoregion. We investigated land use drivers only of species richness at a broader national scale and used Hydrologic Landscape Regions or HLRs as our landscape units. Further, it is common practice to substitute data placed in a spatial context for the temporal and one might expect to at least find similarities with past occurrences that represent the circumstances across a broad spatial gradient.
The term Anthropocene was coined to reflect a modern geologic era influenced by recent human activities that have left physical, chemical, and biological evidence that is now part of the sediment record in lakes (Crutzen & Stoermer, 2000). Hoffmann & Dodson (2005) suggested that current human disturbance regimes have modified the richness–productivity relationship among lake taxa. The unimodal model may only hold true when both pristine and developed lakes are considered together as we have done; a linear relation is expected if only pristine or “least impacted” lakes are studied. Testing this hypothesis across the Anthropocene boundary is a fertile research area.
We did not model groundwater–surface water interactions within the lakes. Ostracod distributions may also have been affected by undetected groundwater seeps and springs. Other researchers have found the presence of Darwinula stevensoni, Typhlocypris (Typhlocypris) punctata, and Candona (Candona) crogmaniana to be associated with groundwater seeps (Smith et al., 1997; Smith & Horne, 2002) which may also be suggested here.
The lacustrine ostracods found in the 12 lakes we studied represent three major life modes or guilds: (1) semi-planktonic swimmers, which are commonly found swimming among rooted aquatic plants; (2) epibenthic deposit feeders, which feed on the surface of sediments; and (3) infaunal deposit feeders, which live and feed within sediment interstices. Semi-planktonic ostracods include several species of Cyclocypris, Cypria, Physocypria, Potamocypris, and Cypridopsis vidua. Individuals of this guild have long natatory setae used in swimming. The epibenthic deposit feeders include Candona spp. and Cypricercus spp., while the infaunal deposit feeders include Darwinula spp. and Limnocytherina spp. Along with the epibenthic species, these taxa lack long natatory setae.
In addition to describing the regional ostracod biodiversity and environmental sensitivities, one of our objectives was to test whether ostracods follow a unimodal species richness–pelagic primary productivity relationship. Recent studies of zooplankton (copepod, cladocerans, and rotifers) biodiversity have shown positive monotonic relationships between species richness and lake area (Hoffmann & Dodson 2005), and unimodal relationships between species richness and lake primary productivity (Dodson, 1992, 2000; Mittelbach et al., 2001) using total phosphorus and chlorophyll a as common productivity surrogates. Our study found neither relationship, probably because of the smaller range of lake sizes and productivity measures. However, a significant unimodal relationship between lake depth and ostracod species richness (Fig. 4A) is apparent.
Proxies of pelagic productivity (chlorophyll a, total nitrogen, total phosphorus, Secchi depth, and Trophic State Index) were not directly correlated with species richness. We suspect a lack of direct correlation because both littoral macrophyte production and pelagic primary productivity are both important and influenced by lake depth. However, the concentration of TDS has been used as a productivity proxy (Prepas, 1983). The data for TDS (Fig. 4B) may represent the left side of a unimodal relationship between ostracod richness and productivity. We predict that hypereutrophic lakes with higher TDS levels than those surveyed in our study will have fewer species, thus fitting the general unimodal model. This relationship is presumed because ostracods are dependent on detritus, diatoms, and macroalgae that comprise pelagic, benthic, and littoral (even riparian) production. Hypereutrophic lakes are expected to have fewer sources of food for ostracods than merely eutrophic lakes. For example, a high density of macrophytes or dense algae creates shade that reduces the abundance of benthic algae, an important ostracod food resource (Morris et al., 2002). Similarly, dense macrophyte beds reduce pelagic production, and dense pelagic algae inhibit macrophyte production (Scheffer et al., 2001). Additionally, when lake depth was accounted for, ostracod species richness showed a significant positive correlation with spring chlorophyll a (Fig. 5). However, the best models explaining richness and abundance indicate that although both are influenced by TDS, abundance was dependent on spring chlorophyll a more so than depth, whereas richness was more a function of depth. Increases in TDS may also result from the decomposition of organic matter and erosion of the land; thus, multivariate models of richness and productivity have more explanatory power.
The highest ostracod diversity was found in lakes of medium depths of 8–15 m. We interpret the unimodal relationship between richness and depth as the result of greater habitat complexity in lakes of intermediate depths which provide similar amounts of habitat for the three groups of ostracods with different life habits (guilds), and thus are supportive of the highest diversity. The most diverse lake (Lulu) was of medium depth and similar abundances of ostracods from both littoral and profundal zones. Sedimentary carbonate was also equal to the amount of organic matter in this lake. In shallow lakes dominated by macrophytes, however, the abundance of epibenthic deposit feeders and infaunal deposit feeders is rare to absent; these species occurred only in low alkalinity lakes with inlets or both inlets and outlets suggesting the influence of current flow. Immature candonids were only found in lakes with a maximum depth greater than 8 m.
Regional patterns from ordination—ostracods and lake condition
There were three nearly ubiquitous species in our study lakes: Cypria ophtalmica, Physocypria globula, and Cypridopsis vidua. These species are also widespread in North America (Hoff, 1942; Delorme, 1967) and they are indicative of the general region of north temperate lakes. Several ostracod species were found to be potential indicator species at a finer scale, because their presence–absence distance matrix was significantly correlated with the ordination axes and related to variation in community structure within the ecoregion (Fig. 3A). Potamocypris smaragdina is found in lakes with higher spring DO and pH levels and lower spring chlorophyll a and is strongly correlated with the first ordination axis. This is consistent with previous studies that have found this species in oxygenated, swift running water. Thus, presence of P. smaragdina indicates high oxygen levels and low spring pelagic productivity in the lakes. In our study, Cypria ophtalmica, Cypria turneri, cyclocyprids, and Candona (Candona) inopinata were negatively correlated with the first ordination axis because they were found in wetlands and lakes more productive in the spring. Candona (Candona) ohioensis was negatively correlated with the second axis, while Candona (Candona) acuta was positively correlated with this axis. This suggests that Candona (Candona) ohioensis may have a higher tolerance for human disturbance than Candona (Candona) acuta or that an unmeasured variable is affecting their distributions within the lakes studied. The remaining species were not good indicators of lake condition because they were not strongly correlated with either community ordination axis. These species were distributed at random relative to community composition, they were equally influenced by both gradients (e.g., Darwinula stevensoni) indicated by the two NMS axes, or because they were rare and occurred at a single site.
Regional patterns from ordination—ostracods and land use
The species that indicate spring productivity and greater habitat complexity are also indicative of several landscape conditions that are strongly correlated with the community ordination axes. Potamocypris smaragdina, indicative of low spring pelagic productivity, is also indicative of riparian deciduous forests. This species was also associated with P. crispus, an invasive species (Fig. 3C). The relationship between riparian forests and increased water quality is well established (Peterjohn & Correll, 1984). Lakes in forested areas are generally less productive, have lower TDS, and lower sedimentation rates. Cypria ophtalmica and Candona (Candona) inopinata, indicative of higher spring pelagic productivity, are also indicative of both riparian and catchment-scale wetlands (Fig. 3D). These species are also associated with Nuphar and Nymphaea (water lilies), suggesting that shallow quiet water macrophyte beds in the lake are also important structuring elements. Candona (Candona) ohioensis was associated with M. spicatum, another aquatic invasive plant. The presence of M. spicatum suggests an increase in human disturbance, and the lakes in this study where it was found were associated with higher proportions of agriculture in the riparian zone. Wetlands are also highly productive, exporting large amounts of particulate and dissolved organic material into lakes (Fig. 6).
Regression/decision tree of ostracod species richness predicted by the percentage of 14 different land use classes within their respective hydrologic landscape regions or HLRs across the continental United States (see Wolock et al., 2004 for a full description of the HLRs). N = 451. Of 20 possible HLRs, 19 are represented along an agricultural land use gradient from 0 to 93% agriculture. This data set includes the HLRs representing our 12 study lakes. The resultant regression tree had a relative error of 0.872
At the riparian or catchment scales, agricultural land use is associated with decreased macrophyte diversity and abundance and decreased zooplankton species richness (Dodson et al., 2005). These decreases are probably due to adverse effects of agriculture, such as increased erosion, increased lake temperature from the removal of native vegetation that normally provides shade, and increased input of pesticides. Row crops typically receive high amounts of phosphorus which increases soil phosphorus levels in the region above that which is needed for crop growth (K. Becker, Wisconsin Department of Agriculture, Trade and Consumer Protection, pers. com., 2004). Phosphorus enters the lake adsorbed to eroded soil particles (in runoff, groundwater, or atmospheric deposition) which can stimulate macrophyte and algal growth (e.g., Bennett et al., 1999). Anthropogenic disturbance can promote the spread of invasive aquatic plants such as M. spicatum and P. crispus, which develop high density monospecific stands that displace native plants and decrease habitat complexity. High proportions of agricultural land uses are generally indicative of more productive watersheds, and phosphorus unit-area loads to aquatic resources of small watersheds are highly dependent on watershed size (Corsi et al., 1997). However, macrophyte decomposition can also add substantial amounts of available phosphorus to the water column (Carpenter, 1980; Smith & Adams, 1986). For example, in Hooker Lake, one of the medium depth lakes in our study, M. spicatum, has become a nuisance. The high proportion of riparian agriculture and insoluble residues suggest higher sedimentation in this lake than the other medium-sized lakes in our study. M. spicatum has a high shade tolerance (Davis & Brinson, 1980) and therefore greater tolerance for more turbid water. Our data suggest that where macrophyte community structure is altered by invasive macrophyte species, the ostracod guild structure can also be altered.
The larger longer-lived candonids like Candona (Candona) ohioensis, Candona (Candona) crogmaniana, and to a lesser extent, Candona (Candona) caudata and Eucandona rectangulata may be more tolerant of human impacts by virtue of a greater tolerance to temperature and dissolved oxygen variability, and variability in sedimentation that is suggested by our NMS.
Invasive macrophyte such as M. spicatum though present may not establish a stronghold where there is a steep drop off to the profundal zone and where this zone is much deeper than the average depth of the lake. We found this to be true for Rock Lake. However, where the natural structure of the shallow macrophyte community is compromised by invasive macrophyte species, ostracod guild structure can also be affected directly by chemical management of the invasive macrophytes or indirectly via habitat alteration. In shallow lakes, the depth to which candonids may escape littoral disturbance is, however, limited by potential anoxia in deeper water. Other studies have found that larger candonids, particularly Candona (Candona) ohioensis, are often unable to complete their life cycles where seasonally low DO is significant (Curry & Filippelli, 2010). Contrary to the findings of Kitchell & Clark (1979), Curry & Filippelli (2010) noted post-European settlement decreases in Candona (Candona) ohioensis (Curry & Filippelli, 2010) in Crystal Lake, IL, which they attributed to seasonal anoxia driven by agricultural land use and lake development. Of the 12 lakes we studied, only one had a significant anoxia problem during late summer when we did our sampling (Big Twin Lake, Green Lake County). This lake had steep banks, a high proportion of agriculture land use and only four species of ostracods. Eucandona rectangulata was the only candonid found and like Crystal Lake the ostracod community was dominated by Cypria ophtalmica. Whether or not seasonal anoxia limits the distribution of certain ostracods of course is dependent on lake depth, morphology and the extent of seasonal anoxia which can be driven by changes in land cover (increased agriculture and urban development) and introduction of invasive species along a continuum of human disturbance. Combined spatial and temporal analyses of a larger number of lakes may be required to establish this continuum of human disturbance that single lake studies or spatial analyses alone cannot.
National land use predictors of ostracod biodiversity from regression tree analysis
Nationally, the average lacustrine ostracod richness was 4.6. Richness in our study lakes is basically the same as the National average. Total agriculture ranged from 0 to 92.6% of the HLRs (row crop 0–91.3; pasture 0–47.6). Within the HLRs represented by the Till Plain Ecoregion (humid and subhumid plains with permeable soils and bedrock, subhumid plains with impermeable soils and bedrock, and humid plateaus with impermeable soils and bedrock), agriculture ranged from 0 to 91.3% row crops; from 0 to 47% pasture; and from 0 to 92.9% total agriculture.
Conclusions
Modern lacustrine ostracod communities in southeastern Wisconsin are structured directly by within-lake chemical and physical factors, including: spring conductivity, total phosphorus, lake area and maximum depth, sedimentary carbonate and organic matter, spring pH and DO. Variability in both species richness and abundance in the lakes studied was largely explained by maximum lake depth, TDS, and chlorophyll a. Ostracod biodiversity is also affected by surrounding land uses and these affects are scale dependent. Ostracods do not appear to be macrophyte specific but depend on the overall structure of the macrophyte community. Agriculture practiced in riparian areas and lake catchments is associated with increases in invasive macrophyte growth which may reduce optimal near shore ostracod habitat. In addition to agriculture, forests and wetlands locally and forests and grasslands regionally influence lake chemistry and therefore ostracod community structure. In expanding our analysis of the affect of land use on modern ostracod diversity we found that agriculture was still the most significant land use driver but that the greatest species richness occurred under conditions of landscape heterogeneity that includes a landscape with more than three percent open water.
Results of our multivariate analyses provide new insight into ostracod ecology and improve our understanding of both natural and anthropogenic drivers of biodiversity acting at multiple spatial scales. Our regional sample database includes information on lake productivity and places ostracods in a landscape context. Additional work is underway to expand the national NANODe database to include lake characteristics and water quality data consistent with our regional sampling effort to better understand modern ostracod species biodiversity and better evaluate the relative importance of multiple collinear environmental factors affecting community composition at multiple scales.
References
Albert, D. A., 1995. Regional landscape ecosystems of Michigan, Minnesota and Wisconsin: a working map and classification. U.S. Department of Agriculture, Forest Service, North Central Forest Experiment Station General Technical Report NC-178.
Alin, S. R., A. S. Cohen, R. Bills, M. M. Gashagaza, E. Michel, J. J. Tiercelin, K. Martens, P. Coveliers, S. K. Mboko, K. West, M. Soreghan, S. Kimbadi & G. Ntakimazi, 1999. Effects of landscape disturbance on animal communities in Lake Tanganyika, East Africa. Conservation Biology 13: 1017–1033.
Allen, P. E., 2005. Landscape influences on lake chemistry and ostracod community structure in small southern Wisconsin dimictic lakes. Ph.D. Dissertation, University of Wisconsin, Madison, WI.
Allen, T. F. H. & T. B. Starr, 1982. Hierarchy: Perspectives for Ecological Complexity. University of Chicago Press, Chicago, IL.
Battarbee, R. W., 2000. Palaeolimnological approaches to climate change, with special regard to the biological record. Quaternary Science Reviews 19: 107–124.
Bennett, E. M., T. Reed-Andersen, J. N. Houser, J. R. Gabriel & S. R. Carpenter, 1999. A phosphorus budget for the Lake Mendota watershed. Ecosystems 2: 69–75.
Borman, S., R. Korth, J. Temte & C. Watkins, 1997. Through the Looking Glass: A Field Guide to Aquatic Plants. Wisconsin Lakes Partnership and the University of Wisconsin-Extension, Stevens Point, WI.
Carbonel, P., J.-P. Colin, D. L. Danielopol, H. Löffler & I. Nuestrueva, 1988. Paleoecology of limnic ostracodes: a review of some major topics. Palaeogeography, Palaeoclimatology, Palaeoecology 62: 413–461.
Crutzen, P. J. & E. F. Stoermer, 2000. The ‘Anthropocene’. Global Change Newsletter 41: 17–18.
Carpenter, S. R., 1980. Enrichment of Lake Wingra, Wisconsin by submerged macrophyte decay. Ecology 61: 1145–1155.
Corsi, S. R., D. J. Graczyk, D. W. Owens & R. T. Bannerman, 1997. Unit-area loads of suspended sediment, suspended solids and total phosphorus from small watersheds in Wisconsin. U.S. Geological Survey, Fact Sheet FS-195-97: 4 pp.
Curry, B., 1999. An environmental tolerance index for ostracodes as indicators of physical and chemical factors in aquatic habitats. Palaeogeography, Palaeoclimatology, Palaeoecology 148: 51–63.
Curry, B. B. & G. M. Filippelli, 2010. Episodes of low dissolved oxygen indicated by ostracodes and sediment geochemistry at Crustal Lake, Illinois, USA. Limnology and Oceanography 55: 2403–2423.
Davis, G. J. & M. M. Brinson, 1980. Responses of Submersed Vascular Plant Communities to Environmental Change. United States Fish and Wildlife Service, Kearneysville, WV. 72 pp.
Dean, W. E., 1974. Determination of carbonate and organic matter in calcareous sediments and sedimentary rocks by loss on ignition; comparison with other methods. Journal of Sedimentary Petrology 44: 242–248.
Dean, W. & A. Schwalb, 2002. The lacustrine carbon cycle as illuminated by the waters and sediments of two hydrologically distinct headwater lakes in north-central Minnesota, USA. Journal of Sedimentary Research 72: 416–431.
Delorme, L. D., 1967. Field key and methods of collecting freshwater ostracodes in Canada. Canadian Journal of Zoology 45: 1275–1281.
Delorme, L. D., 1970a. Freshwater ostracodes of Canada. Part I. Family Cypridinae. Canadian Journal of Zoology 48: 153–168.
Delorme, L. D., 1970b. Freshwater ostracodes of Canada. Part II. Subfamily Cypridopsinae and Herpetocypridinae and family Cyclocyprididae. Canadian Journal of Zoology 48: 253–266.
Delorme, L. D., 1970c. Freshwater ostracodes of Canada. Part III. Family Candonidae. Canadian Journal of Zoology 48: 1099–1127.
Delorme, L. D., 1970d. Freshwater ostracodes of Canada. Part IV. Families Ilyocyprididae, Notodromadidae, Darwinulidae, Cytherideidae and Entocytheridae. Canadian Journal of Zoology 48: 1251–1259.
Delorme, L. D., 1971. Freshwater ostracodes of Canada. Part V. Families Limnocytheridae and Loxoconchidae. Canadian Journal of Zoology 49: 43–64.
Delorme, L. D., 2001. Ostracoda. In Thorpe, J. H. & A. P. Covich (eds), Ecology and Classification of North American Freshwater Invertebrates, 2nd ed., Chap. 19. Academic Press, Inc., San Diego, CA: 811–849.
Diner, M. P., E. P. Odum & P. F. Hendrix, 1986. Comparison of the roles of ostracods and cladocerans in regulating community structure and metabolism in freshwater microcosms. Hydrobiologia 133: 59–63.
Dodson, S. I., 1992. Predicting crustacean zooplankton species richness. Limnology and Oceanography 37: 848–856.
Dodson, S. I., S. E. Arnott & K. L. Cottingham, 2000. The relationship in lake communities between primary productivity and species richness. Ecology 8: 2662–2679.
Dodson, S. I., R. Lillie & S. Will-Wolf, 2005. Relationship of land use, aquatic environment, and zooplankton community structure. Ecological Applications 15: 1191–1198.
Dügel, M., O. Külköylüoğlu & M. Kılıç, 2008. Species assemblages and habitat preferences of Ostracoda (Crustacea) in Lake Abant (Bolu, Turkey). Belgian Journal of Zoology 138: 50–59.
Elkie, P. C., R. S. Rempel & A. P. Carr, 1999. Patch analyst user’s manual—a tool for quantifying landscape structure: 12 pp.
Fassett, N. C., 1957. A Manual of Aquatic Plants. University of Wisconsin Press, Madison, WI.
Forester, R. M., 1986. Determination of the dissolved anion composition of ancient lakes from fossil ostracodes. Geology 14: 796–799.
Forester, R. M., T. K. Lowenstein & R. J. Spencer, 2005a. An ostracode based paleolimnologic and paleohydrologic history of Death Valley: 200 to 0 ka. Geological Society of America Bulletin 117: 1379–1386.
Forester, R. M., A. J. Smith, D. F. Palmer & B. B. Curry, 2005b. NANODe: North American Nonmarine Ostracode Database, version 1.0, http://www.kent.edu/NANODe. Kent State University, Kent, OH.
Frenzel, P. & I. Boomer, 2005. The use of ostracods from marginal marine, brackish waters as bioindicators of modern and quaternary environmental change. Palaeogeography, Palaeoclimatology, Palaeoecology 225: 68–92.
Griffith, G. E., J. M. Omernick & A. J. Woods, 1999. Ecoregions, watersheds, basins, and HUCs: how state and federal agencies frame water quality. Journal of Soil and Water Conservation 54: 666–678.
Hoff, C. C., 1942. The Ostracods of Illinois, Their Biology and Taxonomy. The University of Illinois Press, Urbana, IL.
Hoffmann, M. D. & S. I. Dodson, 2005. Land use, primary productivity, and lake area as descriptors of zooplankton diversity. Ecology 86: 255–261.
Jessen, R. & R. Lound, 1962. An evaluation of a survey technique for submerged aquatic plants. Minnesota Department of Conservation, Division of Game and Fish Section on Research and Planning Investigational Report 6: 1–10.
Johnson, L. B., C. Richards, G. E. Host & J. W. Arthurst, 1997. Landscape influences on water chemistry in midwestern stream ecosystems. Freshwater Biology 37: 193–208.
Jones, K. B., A. C. Neale, M. Nash, R. D. Van Remortal, J. D. Wickham, K. H. Riitters & R. V. O’Neil, 2001. Predicting nutrient and sediment loading to streams from landscape metrics: a multiple watershed study from the United States Mid-Atlantic Region. Landscape Ecology 16: 301–312.
Karonovic, I., 2006. Recent Candoninae (Crustacea, Ostracoda) of North America: Western Australian Museum. Records of the Western Australian Museum, Supplement 71, Welshpool DC, Western Australia 6986, 75 pp.
Keatings, K., J. Holmes, R. Flower, D. Horne, J. Whittaker & R. Abu-Zied, 2010. Ostracods and the Holocene palaeolimnology of Lake Qarun, with special reference to past human–environment interactions in the Faiyum (Egypt). Hydrobiologia 654: 155–176.
Kitchell, J. A. & D. L. Clark, 1979. Distribution, ecology, and taxonomy of recent freshwater Ostracoda of Lake Mendota, Wisconsin. University of Wisconsin-Madison, Madison, WI. Publication of the Natural History Council, University of Wisconsin-Madison, Wisconsin Geological and Natural History Survey Number 1: 24 pp.
Külköylüoglu, O., 2004. On the usage of ostracods (Crustacea) as bioindicators species in different aquatic habitats in the Bolu region, Turkey. Ecological Indicators 4: 139–147.
Külköylüoglu, O. & M. Dügel, 2004. Ecology and spatiotemporal patterns of Ostracoda (Crustacea) from Lake Gölcük (Bolu, Turkey). Archiv für Hydrobiologie 160: 67–83.
Kummel, B. & D. Raup, 1965. Handbook of Paleontologic Techniques. W. H. Freeman and Co., San Francisco, CA.
Löffler, H., 1979. Ostracode analysis. In Berglund, B. E. (ed.), Paleohydrological Changes in Temperate Zone in the Last 15,000 Years. Lake and Mire Environments. Vol. II. Specific Methods. UNESCO, International Correlation Programme, Project 158: 329–340.
Löffler, H., 1997. The role of ostracods for reconstructing climate change in Holocene and Late Pleistocene lake environment in Central Europe. Journal of Paleolimnology 18: 29–32.
Martens, K., I. Schön, C. Meisch & D. J. Horne, 2008. Global diversity of ostracods (Ostracoda, Crustacea) in freshwater. In Balian, E. V., C. Lévêque, H. Segers & K. Martens (eds), Freshwater Animal Diversity Assessment: Developments in Hydrobiology. Springer, Netherlands: 185–193.
McCune, B. & M. J. Mefford, 1999. PC-ORD for Windows – Multivariate Analysis of Ecological Data. MjM Software, Gleneden Beach, OR.
McCune, B., J. B. Grace & D. L. Urban, 2002. Analysis of Ecological Communities. MjM Software Design, Gleneden Beach, OR. 300 pp.
Meisch, C., 2000. Freshwater Ostracoda of Western and Central Europe. Spektrum Akademischer Verlag, Heidelberg, Berlin. 522 pp.
Mezquita, F., J. R. Roca, J. M. Reed & G. Wansard, 2005. Quantifying species-environment relationships in non-marine Ostracoda for ecological and palaeoecological studies: examples using Iberian data. Palaeogeography, Palaeoclimatology, Palaeoecology 225: 93–117.
Miller, B. B., A. F. Schneider, A. J. Smith & D. F. Palmer, 2000. A 6000 year water level history of Europe Lake, Wisconsin, USA. Journal of Paleolimnology 23: 175–183.
Mittelbach, G. G., C. F. Steiner, S. M. Scheiner, K. L. Gross, H. L. Reynolds, R. B. Waide, M. R. Willig, S. I. Dodson & L. Gough, 2001. What is the observed relationship between species richness and productivity? Ecology 82: 2381–2396.
Morris, K., P. I. Boon, P. C. Bailey & L. Hughes, 2002. Alternative stable states in the aquatic vegetation of shallow urban lakes. I. Effects of plant harvesting and low-level nutrient enrichment. Marine and Freshwater Research 54: 185–200.
Panuska, J. C. & J. C. Kreider, 2002. Wisconsin lake modeling suite: program documentation and user’s manual. PUBL-WR-363-94, Wisconsin Department of Natural Resources, Madison, WI.
Peterjohn, W. T. & D. L. Correll, 1984. Nutrient dynamics in an agricultural watershed: observations on the role of a riparian forest. Ecology 65: 1466–1475.
Prepas, E. E., 1983. Total dissolved solids as predictor of lake biomass and productivity. Canadian Journal of Fisheries and Aquatic Sciences 40: 92–95.
Rosenzweig, M. L., 1995. Species Diversity in Space and Time. Cambridge University Press, Cambridge. 436 pp.
Ruiz, R., M. Abad, A. M. Bodergat, P. Carbonel, J. Rodriguez-Lazaro & M. Yasuhara, 2005. Marine and brackish-water ostracods as sentinels of anthropogenic impacts. Earth Science Reviews 72: 89–111.
Saros, J. E., S. C. Fritz & A. J. Smith, 2000. Shifts in mid- to late-Holocene anion composition in Elk Lake (Grant County, Minnesota): comparison of diatom and ostracode inferences. Quaternary International 67(1): 37–46.
Scheffer, M., S. Carpenter, J. A. Foley, C. Folke & B. Walker, 2001. Catastrophic shifts in ecosystems. Nature 413: 591–596.
Schwalb, A. & W. E. Dean, 2002. Reconstruction of hydrological changes and response to effective moisture variations from North-Central USA lake sediments. Quaternary Science Reviews 21: 1541–1554.
Smith, A. J., 1993. Lacustrine ostracodes as hydrochemical indicators in lakes of the north-central United States. Journal of Paleolimnology 8: 121–134.
Smith, G. L., 1997. Late Quaternary climates and limnology of the Lake Winnebago basin, Wisconsin, based on ostracodes. Journal of Paleolimnology 18: 249–260.
Smith, C. S. & M. S. Adams, 1986. Phosphorus transfer from sediments by Myriophyllum spicatum. Limnology and Oceanography 31: 1312–1321.
Smith, A. J. & B. Dai, 1993. The Ostracode record from Europe Lake, Wisconsin the past 6600 years. In Schneider, A. F. (ed.), Pleistocene Geomorphology and Stratigraphy of the Door Peninsula, Wisconsin, Midwest Friends of the Pleistocene 40th Annual Meeting. University of Wisconsin-Parkside, Kenosha: 63–66.
Smith, A. J. & D. J. Horne, 2002. Ecology of marine, marginal marine and nonmarine ostracodes. Geophysical Monographs 131: 37–64.
Smith, A. J., J. J. Donovan, E. Ito & D. R. Engstrom, 1997. Ground-water processes controlling a prairie lake’s response to middle Holocene drought. Geology 25: 391–394.
StatSoft, Inc., 2000. STATISTICA for Windows [computer program manual]. StatSoft, Inc., Tulsa, OK. http://www.statsoft.com.
StatSoft, Inc., 2007. STATISTICA (data analysis software system), version 8.0. http://www.statsoft.com.
Staplin, F. L., 1963. Pleistocene Ostracoda of Illinois: Part II. Subfamilies Cyclocyprinae, Cypridopinae, Ilyocyprinae; Families Darwinulidae and Cytheridae. Stratigraphic ranges and assemblage patterns. Journal of Paleontology 37(6): 1164–1203.
Swain, F. M., 1999. Stratigraphic distribution of Paleozoic nonmarine Ostracoda Devonian. In Frederick, M. S. (ed.), Developments in Palaeontology and Stratigraphy. Elsevier, Amsterdam: 3–271.
Tressler, W. L., 1959. Ostracoda. In Edmondson, W. T. (ed.), Freshwater Biology, Chapter 28. Wiley, New York: 657–734.
Umbanhower, C. E., 2004. Interactions of climate and fire at two sites in the northern Great Plains, USA. Palaeogeography, Palaeoclimatology, Palaeoecology 208: 141–152.
Welschmeyer, N. A., 1994. Fluorometric analysis of chlorophyll a in the presence of chlorophyll b and pheopigments. Limnology and Oceanography 39: 1985–1992.
Wisconsin Department of Natural Resources (WDNR), 1999a. Digital databases [GeoDisc 3.0. 1999]. Wisconsin Department of Natural Resources. Madison, WI.
WDNR, 1999b. Land Cover of Wisconsin, User’s Guide to WISCLAND Land Cover Data. Wisconsin Department of Natural Resources, Madison, WI: 27 pp.
Williams, M., D. Siveter, M. Salas, J. Vannier, L. Popov & P. M. Ghobadi, 2008. The earliest ostracods: the geological evidence. Palaeobiodiversity and Palaeoenvironments 88: 11–21.
Wolock, D. M., T. C. Winter & G. McMahon, 2004. Delineation and evaluation of hydrologic-landscape regions in the United States using geographic information system tools and multivariate statistical analyses. Environmental Management 34: S71–S88.
Xia, J., D. R. Engstrom & E. Ito, 1997. Geochemistry of ostracode calcite: Part 2. The effects of water chemistry and seasonal temperature variation on Candona rawsoni. Geochimica et Cosmochimia Acta 61(2): 383–391.
Acknowledgments
This research was funded primarily by the Department of Zoology at the University of Wisconsin, Madison, including the Emlen and A. G. Birge Foundations. Additional funds were received from the Milwaukee Zoological Society, the Lois Almon Foundation and the Wisconsin Department of Agriculture, Trade and Consumer Protection. Volunteers made this effort possible. Special thanks are due to Patricia Sanford and Marge Winkler at the Paleoecology Laboratory at the Center for Climate Research for LOI testing and assistance sampling Lulu Lake; Paul Garrison, Wisconsin Department of Natural Resources, for sampling advice and help in sampling Lulu and Moose Lakes; Susan Will-Wolf for help with ordination techniques; Brandon Curry for reviewing ostracod reference materials; Maliha Nash, USEPA, for statistical advise and encouragement; and Dr. Allen’s dissertation committee (Lou Maher, Lee Clayton, David Mladenoff, and Emily Stanley). We would also like to thank several anonymous reviewers whose thoughtful comments greatly improved the manuscript. This research was also funded in part by the U.S. Environmental Protection Agency. It has been subjected to the Agency’s peer and administrative review and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: H. J. Dumont, J. E. Havel, R. Gulati & P. Spaak / A Passion for Plankton: a tribute to the life of Stanley Dodson
Stanley I. Dodson died on 23 August 2009.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Allen, P.E., Dodson, S.I. Land use and ostracod community structure. Hydrobiologia 668, 203–219 (2011). https://doi.org/10.1007/s10750-011-0711-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-011-0711-7