Abstract
Cascade effects of an exotic predator, the rainbow trout (Oncorhynchus mykiss), on periphyton and leaf litter were analysed in a headwater, forested stream of Andean Patagonia (Argentina). We conducted seasonal field sampling and two field experiments measuring leaf litter mass, periphyton biomass and macroinvertebrate biomass in relation to the presence and absence of rainbow trout. In the field survey, the presence of trout influenced resource mass: leaf litter (60% decrease in summer, P = 0.024) and periphyton (tenfold increase in chlorophyll a, P < 0.001) were affected, which were mediated by a decrease in the biomass of shredders (95% decrease in summer, P < 0.001) and scrapers (90% decrease, P < 0.001). There was an effect on leaf litter biomass only in the summer, whereas fish presence reduced periphyton biomass all year except in the winter. In the field experiments, we observed that leaf litter breakdown and periphyton development were effectively controlled by consumers in the absence of fish. In contrast, the presence of fish caused a release of herbivory and detritivory resulting in a significant increase in periphyton biomass (100% increase, P < 0.001) and a decrease in leaf litter decay (40% decrease, P < 0.001). Our results suggest that in low order streams and in the presence of visual predators, trophic cascades may operate both on detritus and algae, but with different timing.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A trophic cascade occurs when top predators regulate the abundance or behaviour of their prey populations resulting in dramatic changes in the abundance and/or biomass of non-adjacent lower trophic levels (Carpenter et al., 1985; Schmitz et al., 2004). In freshwater environments, this phenomenon commonly occurs in systems controlled by fish predation (Townsend, 2003; Peckarsky et al., 2008). Most studies have evaluated autotrophic-based food webs in which the top predators negatively affect the herbivores, which in turn has positive effects on primary producers (Bechara et al., 1992; Katano et al., 2003; Meissner & Muotka, 2006).
Aquatic ecosystems have heterotrophic and autotrophic resources that may be temporally or spatially important in terms of matter and energy fluxes (Power, 1992; Gaedke et al., 1996; Hall et al., 2000; Eggert & Wallace, 2003), and these heterotrophic systems are donor-controlled (i.e. invertebrate consumers do not regulate resource renewal) (Woodward et al., 2005). In these streams, light is a major abiotic constraint on photosynthesis; however, primary production may be important seasonally, such as before spring leaf-out (Mulholland et al., 2001), or spatially, such as in gaps created by tree falls. Consequently, due to the seasonality of temperate systems, the abundance and identity of basal food resources that sustain a stream food web may change in space and time. In addition, predatory effects on non-adjacent trophic levels may vary in the same manner (Marchand et al., 2002; Kishi et al., 2005; McIntosh et al., 2005). However, the simultaneous impacts of top predators on different trophic compartments of a food web (i.e. detritivory and herbivory) have not yet been addressed (Schmitz et al., 2004; Shurin et al., 2006). It has been suggested that trophic cascades in streams are more likely to occur in algal-based than in detritus-based food webs (Rosenfeld, 2000). This is because grazers are associated with exposed substrate surfaces where periphyton grows, and consequently, they are more vulnerable to predation than shredders, which inhabit the interstices where detritus accumulates. In an extensive study of 21 paired fishless/trout streams in the Sierra Nevada of California, Herbst and co-workers (Herbst et al., 2009) reported strong cascading effects of trout on periphyton but not on detritus standing stocks. Whilst grazer and shredder densities in their study were similar in fishless streams, the abundance of grazers, but not shredders, was heavily reduced in the presence of trout. Nevertheless, emerging evidence indicates that predators can also influence shredder abundance by exerting indirect impacts on the dynamics of detritus breakdown (Ruetz et al., 2002; Greig & McIntosh, 2006). Predators can act as keystone species by selecting highly competitive prey and exerting top-down effects within food webs (Carpenter et al., 1985). On the other hand, this selective predation may also determine compensation effects by releasing competition within other functional components of the web, which may reduce cascade effects (Meissner & Muotka, 2006).
In small, high-altitude Andean Patagonian streams, the deciduous, endemic beech Nothofagus pumilio (P. et E.) Krasser is responsible for most of the leaf litter input. Its timing and breakdown rates are the key components of stream functioning (Albariño & Balseiro, 2002). However, these stream systems also support an important benthic algal community dominated by diatoms that serve as food resource for grazing invertebrates (Díaz Villanueva & Albariño, 1999). In Patagonian freshwater systems, salmonid species have been extensively introduced, and three of them [Oncorhynchus mykiss (Walbaum), Salmo trutta L. and Salvelinus fontinalis (Mitchill)] dominate fish fauna (Pascual et al., 2002). In particular, rainbow trout (O. mykiss) seems to be a top predator in headwater streams, and waterfalls act as natural barriers to upstream trout dispersion, which results in fishless sections of headwaters in which large shredders and scrapers dominate total community biomass (Buria et al., 2007). A significant reduction in large shredders and grazers as a consequence of fish predation is likely to alter ecosystem processes; for instance, large shredders have been shown to increase leaf litter breakdown (Albariño & Balseiro, 2002). Based on this, we hypothesised that trout would exert a trophic cascade effect on leaf litter and primary producer biomass by reducing both shredder and scraper biomass. Therefore, our main objective was to assess the simultaneous occurrence of trophic cascades, and for this purpose, we carried out a seasonal field sampling and field experiments in two contiguous reaches (with and without fish) in a headwater deciduous-forested stream in Andean Patagonia.
Materials and methods
Study site
This study was conducted in the upper section of Challhuaco catchment (41°08′S; 71°W) in the Nahuel Huapi National Park, Northwestern Patagonia (Argentina). The catchment drains an area densely covered by N. pumilio forest, a deciduous beech that dominates the upper vegetation belt. The climate is cold temperate and has a bimodal hydrological regime with higher precipitation (rain and snow) in autumn and winter, which determines peak discharges in autumn–winter and late spring. The stream contains a waterfall that acts as a physical barrier to the upstream movement of salmonids, and consequently, we were able to establish two continuous reaches for the study. In the section downstream of the waterfall, a single fish species was present: the exotic rainbow trout, O. mykiss (Buria et al., 2007). The absence of fish in the section above the waterfall was seasonally confirmed by extensive electrofishing (covering over 300 m2).
The stream bottom was dominated by a mixture of boulder-cobble substrates. Physicochemical features indicated that both stream sections were similar (Table 1). The pH was circumneutral (annually), and dissolved oxygen concentration was always at saturation levels. Phosphorus concentration did not vary between sections (TDP in fishless section = 2.46 ± 0.48 μg l−1, in section with fish = 5.20 ± 1.27 μg l−1).
Field survey
The field survey was carried out seasonally from March 2003 to February 2004 in two contiguous sections separated by a waterfall. The upper section was fishless, whilst the lower one contained the exotic rainbow trout (O. mykiss). Benthic organic matter and invertebrates were sampled with a Surber sampler (0.09 m2, 250 μm mesh size). In each section, ten samples were randomly collected at erosional habitats (runs and riffles) within a ~30 m reach. The samples were preserved in 5% formalin until processing. In the laboratory, samples were washed through 1 and 0.25 mm sieves to facilitate detritus and invertebrate sorting. Invertebrates were identified to the lowest taxonomic unit and assigned to the corresponding functional feeding group (FFG) based on our own invertebrate gut content analysis and on Merrit & Cummins (1996), Díaz Villanueva & Albariño (1999) and Albariño (2001). Macroinvertebrate total body length was measured in a stereomicroscope with an ocular micrometer to the nearest 0.1 mm, and biomass was estimated based on length–mass regressions (Albariño & Díaz Villanueva, 2006). When regressions were unavailable, a set of fresh individuals of the selected species was measured, dried at 80°C for 24 h, weighed at the nearest 0.01 mg and used to estimate biomass versus body length relationships.
Organic matter was also collected with a Surber sampler and classified into four major categories: fine particulate organic matter (FPOM) (0.25 mm < FPOM < 1.00 mm), leaf litter (≥5 mm, entire or fragments), coarse woody matter (≥5 mm, twigs, bark and woody chunks) and miscellaneous material (≥1 mm, mostly unrecognizable fragments of the previous categories, moss fragments, algal filaments and colonies and insect exuviae). Only FPOM and leaf litter were considered for quantitative analysis because they are the basic resources for collector–gatherers and shredders, respectively. FPOM and leaf litter were dried at 80°C for 48 h before weighing to the nearest 0.01 mg. Ash free dry mass (AFDM) was obtained after incineration at 550°C for 1.5 h.
Periphyton biomass was estimated as chlorophyll a concentration (Chl a) and AFDM from nine cobbles sampled at each sampling section. The substrates were individually stored in plastic containers and immediately carried to the laboratory under dark conditions in thermally isolated containers. In the laboratory, substrates were scraped with a nylon brush and rinsed with distilled water to obtain a sample of approximately 100 ml. After homogenizing, a 1-ml aliquot was used for Chl a determination. Extraction was performed with hot 90% ethanol (Nusch, 1980), and measurements were carried out with a fluorometer (Turner designs 10-AU). Periphyton AFDM was determined by filtering an aliquot of 5 ml onto pre-weighed and pre-combusted Whatman GF/F filters and dried at 80°C for 48 h. The filters were weighed and then combusted at 550°C for 1 h and re-weighed, and AFDM was calculated as the difference in mass before and after incineration (American Public Health Association, 2005). The individual substrate surface of each cobble was estimated from three lengths measured along the three main axes (Graham et al., 1988). To estimate the colonised surface, only 2/3 of the cobble surface was considered to be available for algal growth (Biggs & Close, 1989).
A two-way ANOVA was used to assess the effects of fish presence, season and their interaction on benthic resources mass (leaves, FPOM, periphyton Chl a and AFDM). Differences in shredders, scrapers and collector–gatherers biomass were tested with two-way ANOVAs. Previously, we normalised invertebrate AFDM biomass by the AFDM of their food resource (mg shredders g−1 leaf litter, mg collector–gatherer g−1 benthic FPOM, mg grazers μg−1 Chl a). Tukey’s test was applied for a posterior multiple comparisons.
Field experiments
We conducted two field experiments in the two sections (fish and fishless) to test trophic cascades on the detritus-based food web (Experiment 1) and on the autotrophic-based food web (Experiment 2).
Experiment 1
Experiment 1 was carried out from late spring (2005) to early autumn (2006) and was finished before autumnal leaf litter inputs occurred. To measure leaf litter decomposition, we placed two sets of litter bags in the fish and fishless reaches (25 bags per site) on December 13 (late spring). Litter bags were 18 × 12 × 2 cm and contained 10-mm mesh (25% of bag surface) and 1-mm mesh (remaining 75%). They were placed in the substrate with the large mesh size facing upstream. Using this setup, all sizes of invertebrates could gain access to leaves, and leaf fragments (>1 mm) produced by breakdown would not be washed out (Albariño & Balseiro, 2002). Physical abrasion was assumed to be minor because of the physical protection provided by the bags. Therefore, the loss of leaf mass inside the bags was attributed to leaching, microbial decomposition and macroinvertebrate feeding.
Undamaged, freshly fallen leaves of N. pumilio were collected from the riparian floor in the same catchment, and each bag was filled with non-dried leaves (corresponding to 2 g dry mass). Each bag was placed randomly in a run-riffle habitat fastened to the stream bottom by a large steel nail. Five bags (replicates) of each treatment (fish versus fishless site) were removed after 16, 35, 65, 111 and 135 days of exposure. In the laboratory, all invertebrates were separated from the remaining leaf material, taxonomically identified and assigned to their corresponding FFG. The invertebrates were measured to estimate their biomass as previously described. We focussed our attention on shredders because they are the functional group that is directly associated with leaf litter breakdown. The mass loss of leaves was determined after drying the remaining leaf material at 60°C for 48 h and weighing. CPOM decaying rates were measured as leaf mass loss, and CPOM decaying rates between fish and fishless sections were compared with an ANCOVA with time as covariate.
To compare the “shredders independent” leaf litter decay in both stream conditions (fish and fishless), a set of five 1-mm mesh bags was placed in each section on December 13, 2005, following the methodology previously described. Leaf bags were collected at the end of the experiment (day 135) and represented control groups of leaf litter processing without large shredders.
A two-way ANOVA was used to assess the effects of fish presence, bag mesh size and their interaction on the decaying rates of the leaf litter. When significant differences were obtained, a multiple comparison Tukey test was applied.
Experiment 2
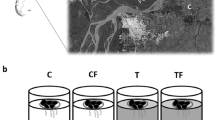
Experiment 2 was performed to determine the predatory effect of trout on periphyton biomass. Trout predation was evaluated by comparing grazing effects in sections with and without fish.
Unglazed ceramic tiles (64 cm2 each) were placed in two sections of the Challhuaco stream (with trout and fishless) on January 25 (austral summer). A total of four treatments were established with two treatments in each stream section: one exposed to grazers and the other not. To prevent grazing in the latter, tiles were elevated approximately 15 cm from the bottom and at least 2 cm under the water surface with J-shaped metal rods. There were five replicates for each treatment. All tiles were placed in riffles where the velocity of the current was near 45 cm s−1. All elevated tiles were checked every third day, and macroinvertebrates were removed. After 54 days, tiles were carried to the laboratory and periphyton biomass was estimated using Chl a and AFDM. Invertebrates colonising the underside of tiles placed on the stream bottom were collected when tiles were removed by placing a small D-frame net downstream of the tile. In the laboratory, macroinvertebrates that were still attached to the tiles were collected with forceps and preserved in 70% ethanol with those collected with the D-frame net. Afterwards, macroinvertebrates were identified and biomass was calculated. In this case, we focussed on grazers. The net grazing effect (NGE%) (removal by feeding and movement activities) was calculated as
where UnG is the Chl a or AFDM in ungrazed tiles and G is the Chl a or AFDM in grazed tiles. Differences in grazing between sites and grazed and ungrazed tiles were analysed with a two-way ANOVA was used to assess the effects of fish presence, grazer effect (grazed and ungrazed) their interaction on benthic Chl a and AFDM.
In all cases, if data did not meet normality or homoscedasticity assumptions, the data were log transformed.
Results
Field survey
There was a seasonal pattern to the standing stocks of benthic organic matter, which is typical of deciduous-forested streams with larger amounts of leaf litter accumulations during autumn–winter and lower amounts in the summer (Fig. 1). Leaf litter samples were mainly composed by N. pumilio leaves (90%). There were seasonal differences in leaf litter and FPOM in both the fish and fishless sections (leaf litter Table 2; Fig. 1). Leaf litter mass was lowest in the summer (Tukey’s test, P = 0.047) and higher in the section with fish (Tukey’s test, P = 0.024). On the contrary, FPOM did not differ significantly between sections (Table 2).
Differences in periphyton biomass between sites depended on the season (Table 2); Chl a concentration and AFDM were significantly higher in the site with fish in all seasons except winter (Tukey’s test winter, Chl a P = 0.718 and AFDM P = 0.827) (Fig. 2). In the fishless site, periphyton was entirely composed by diatoms (100% of taxa) throughout the year. The dominant species was Gomphonema angustum Ag., which accounted for between 50 and 90% of total cell abundance. In contrast, in the section with fish, the proportion of diatoms decreased to 30% because of the development of a thick mat of the Chrysophyta Hydrurus foetidus (Vill.) Trev. and filamentous Chlorophyta (mainly Ulotrix sp.). Cyanophyta (mainly Lyngbya sp.) were present in very low relative abundances (<1%).
The trophic structure of the macroinvertebrate assemblage differed between sections, and the magnitude of the differences varied throughout the year (Fig. 3). The biomass of shredders per unit of leaf litter mass in the section with fish was lower than in the fishless section (Table 2), but this difference was significant only in the summer (Tukey’s test, P < 0.001; Fig. 3a). Conversely, the biomass of collector–gatherers per unit FPOM mass differed between sites depending on the season, where it was significantly lower in the fishless site in the summer (Table 2; Tukey’s test, P < 0.001; Fig. 3b) but was not different in the other seasons (Tukey’s test, winter P = 0.928; autumn P = 0.156; spring P = 0.442). The biomass of scrapers per unit of periphyton Chl a mass was significantly lower in the section with fish year round (Table 2; Fig. 3c). The shredders were dominated by the plecopterans Klapopteryx kuscheli and Antarctoperla michaelseni and the dipteran Tipula sp., whilst the dominant scrapers were the plecopterans Notoperla archiplatae and Aubertoperla illiesi and the mayfly Meridialaris chiloeensis. Collector–gatherers were mainly chironomids.
Experiments
In experiment 1, leaf litter breakdown differed between sections. The decay rate was significantly lower in the fish section than in the fishless one (ANCOVA, F (1,4) = 6.140, P = 0.019), and this was more evident at the end of the experiment (Fig. 4a). On the contrary, leaf litter losses in the fine mesh bags did not differ between sections (21.6% ± 9.3 in the fishless section, 27.1% ± 8.8 in the section with fish; t test, P = 0.677), suggesting that the difference in decomposition rate was due to changes in shredder feeding.
Invertebrate biomass in the fishless section was dominated by the large shredders Klapopteryx kuscheli and Tipula sp. In the section with fish, collector–gatherers (oligochaetes and chironomids) also colonised the bags. The normalised shredder biomass increased in the fishless section only in the last two sampling dates (111 and 135 days) (Table 3; Tukey’s test P < 0.001) (Fig. 4b).
In Experiment 2, grazers were successfully removed from elevated tiles in both sections, and this resulted in significant differences in periphyton growth between grazed and ungrazed tiles (Table 3), which was lower on grazed than ungrazed tiles. In addition, we found significant differences between sites (Table 3), where periphyton growth was lower in the fishless one (Fig. 5a). The AFDM was also significantly different between treatments and sites (Table 3; Fig. 5b). The NGE% on the active photosynthetic fraction (Chl a) was significantly higher in the fishless section than the section with fish (t test, P = 0.02; Fig. 6a). On the other hand, the NGE% on AFDM did not differ between stream sections (t test, P = 0.96; Fig. 6b). The biomass of scrapers on the tiles normalised by Chl a concentration was significantly lower in the section with fish than in the fishless section (t test, P = 0.016) (Fig. 6c). Tiles on the streambed were colonised mainly by the scrapers Meridialaris chiloeensis and Aubertoperla illiesi, which comprised nearly 85% of biomass. Finally, there was an increase in small collector–gatherers, mainly chironomids, in the grazed tiles of the section with fish.
Discussion
Our combined field and experimental studies in a headwater Patagonian stream showed that the presence of exotic O. mykiss strongly reduced macroinvertebrate biomass and changed community functional structure, and these effects cascaded down to lower trophic levels. The sections with and without fish of the studied forested stream differed both in resource abundance (leaf litter and periphyton) and consumer biomass (shredders and scrapers). The visually hunting fish, such as trout, are size-selective consumers that prefer large and active prey and feed less on small prey (McIntosh, 2000; Blanchet et al., 2008). A previous study in the Challhuaco system indicated that predation by rainbow trout was size selective, where populations of large shredders (i.e. Klapopteryx kuscheli, Tipula sp.) and grazers (i.e. Notoperla archiplatae, Meridialaris chiloeensis) were efficiently reduced (Buria et al., 2007). The consumption of large amounts of terrestrial arthropods may potentially reduce the impact of fish on benthic prey thereby weakening top-down control in aquatic systems (Nakano et al., 1999). However, studies on the diet of rainbow trout in Patagonia streams suggest that they do not depend significantly on such terrestrial subsidy (Palma et al., 2002; Buria et al., 2009). On the other hand, it has been also shown that the large plecopterans K. kuscheli and N. archiplatae, which belong to different FFGs, have narrow feeding habits, as growth rates were nearly zero when food resources were experimentally swapped (Albariño & Díaz Villanueva, 2006). As a consequence of the non-plastic feeding behaviour of consumers and the high efficiency of rainbow trout in capturing large aquatic prey, fishes exert a strong cascade effect on the two resource compartments as observed in this study.
Trophic cascades on the autotrophic-based food web occur when fish reduce grazing activity leading to an increase in algal biomass (Forrester et al., 1999; Biggs, 2000). Our experiment examined the grazing effect on the periphyton-based food web and showed that rainbow trout produces a cascade effect on active autotrophic biomass (Chl a). However, no cascading effect was observed on the organic matter content (AFDM) of the biofilm. One explanation is that ungrazed tiles in the section with fish had a thicker periphytic mat with more detritus than the fishless section. In the section with fish, grazed tiles had a high density of chironomids. Most chironomids were collector–gatherers feeding from (and living inside) the lower portions of the periphytic mat. On the other hand, it has been shown that the dominant grazers Notoperla archiplatae and Meridialaris chiloeensis may reduce diatoms with an erect habit, thereby favouring the prostrate ones (Díaz Villanueva & Albariño, 1999; Díaz Villanueva et al., 2004). As a result, the distal portion of the biofilm, which contains more photosynthetic organisms, is more efficiently removed by grazers. Consequently, the cascade effect was only significant in the autotrophic portion and not in the whole biofilm. Nevertheless, the field data suggest that the cascade effect occurred in the entire biofilm because the standing stock of both AFDM and Chl a differed between stream sections. Additionally, the differences in algal composition between fish and fishless sites were also correlated with grazing release. In the section with fish, the substrates developed filamentous algae (Chrysophyta, Chlorophyta and Cyanophyta) that are vulnerable to grazing (Biggs, 2000). These differences in the algal assemblage influenced the total AFDM of the biofilm, where the effect on this parameter resulted from the accumulated biomass of filamentous algae.
Seasonal variation in light, nutrients and temperature affects autotrophic resources, which can modify cascade effects (Rosemond et al., 2000). If the periphyton were a limiting resource, an increase in production due to higher irradiance would lead to an increase in herbivore biomass (Hart & Robinson, 1990; Hill et al., 1995). In our field study, grazer biomass increased substantially in spring and summer in the fishless section. In contrast, grazers were not able to control algal biomass in the section with fish; this allowed a pronounced increase in autotrophic biomass, which indicated the strength of the trophic cascade. In streams with high herbivore abundances, weak seasonal variation in periphyton biomass has been reported (Rosemond, 1994). In fact, our field survey showed a low annual variation in the fishless site that may be attributed to the high herbivore abundances. Additionally, the significant interaction between season and site in periphyton biomass (both Chl a and AFDM) showed that seasonal variations in periphyton production affect the strength of grazing pressure.
In forested streams, differences in leaf litter mass and standing stocks have been linked to the detrimental effects of fish predation on shredders, thereby demonstrating that trophic cascades also affect coarse detritus processing (Ruetz et al., 2002; Greig & McIntosh, 2006). In the Challhuaco stream, the presence of trout affected shredders like Klapopteryx kuscheli and Tipula sp., thereby favouring litter accumulation. On the other hand, it has been shown that increases in species less vulnerable to predators may mask trophic cascades (Persson, 1999; Duffy, 2002; Meissner & Muotka, 2006). In our litter bag experiment, the lower abundance of large shredders and grazers in the section with fish was associated with an increase in the abundance of small, less-vulnerable species (chironomids and oligochaetes). The significant interaction between time and section suggests a cumulative effect in the colonisation of the litter bags by the large shredders in the fishless section. Introduced trout greatly increased collector–gatherers in an originally fishless stream in the Sierra Nevada, California (Herbst et al., 2009). However, in our study, as well as in that of North America, small species were unable to functionally compensate the reduction of large consumers by fish. Although we did not experimentally explore the cascading effect of predation through collector–gatherers, FPOM standing stock tended to be lower in the site with fish in autumn and summer. Low FPOM standing stocks were also registered in New Zealand streams stocked with salmonids (Nystrom et al., 2003), and as in our study, the biomass of shredders was lowered by trout. The lower amount of FPOM could result from an indirect effect of trout preying on large shredders, which could cause a net reduction of their feeding activity, in turn affecting FPOM production (Ruetz et al., 2002; Nystrom et al., 2003).
Small, forested streams are heterotrophic systems in which invertebrate consumers do not regulate resource renewal; therefore, they are donor-controlled (Woodward et al., 2005). In our study, seasonal differences in leaf litter accumulation were associated with the deciduous phenology of dominant riparian plants with most inputs in autumn and winter but with differences between sites only observed in summer. There is evidence that invertebrates in detrital food webs may be temporally limited by their resources (Dobson & Hildrew, 1992; Wallace et al., 1999). Therefore, in periods when vegetal debris is scarce, as before autumnal leaf shedding, top-down effects should be particularly pronounced. In our study, the greatest shredder biomass was observed in summer in the fishless section. The greatest shredder biomass was concurrent with low leaf litter-standing stock, which would determine resource limitation for consumers. In the presence of trout, shredder biomass was lower which decreased the breakdown of leaf litter. On the contrary, the absence of a cascade effect during the winter may be related to three factors: (1) an increase in leaf litter availability due to the input from the riparian vegetation; (2) an absence of a numerical response of shredders, as reproduction occurs in other seasons such that there is no population increase associated with the resource increase, resulting in no top-down control of leaf litter in this period and (3) the concealing of shredders between leaves when litter-standing stock in the aquatic system is high.
The two different food sources considered in this study, detritus and primary producers, have very different renewal rates (Brönmark et al., 1997). Whilst the renewal rate of primary producers is controlled within the stream, the renewal of detritus is independent of it, as it is produced and controlled outside the system (Persson, 1999). Despite these differences in resource dynamics, our results in an Andean low order stream showed that in the presence of the introduced rainbow trout, trophic cascades may operate both on the detritus and algae but with different timing. Before autumnal leaf litter input occurs in these systems, shredders would be food-limited in the fishless reach, and this would promote a cascade effect on leaf litter processing. On the contrary, in the autotrophic food web, a strong cascade effect occurs all year except in winter.
References
Albariño, R. J., 2001. Food habit and mouthpart morphology of a South Andes population of Klapopteryx kuscheli (Plecoptera: Austroperlidae). Aquatic Insects 23: 171–181.
Albariño, R. J. & E. G. Balseiro, 2002. Leaf litter breakdown in Patagonian streams: native versus exotic trees and the effect of invertebrate size. Aquatic Conservation: Marine and Freshwater Ecosystems 12: 181–192.
Albariño, R. J. & V. Díaz Villanueva, 2006. Feeding ecology of two plecopterans in low order Andean-Patagonian streams. International Review of Hydrobiology 91: 122–135.
American Public Health Association, 2005. Standard Methods for the Examination of Water and Wastewater. American Public Health Association, AWWA, Washington, DC.
Bechara, J. A., G. Moreau & D. Planas, 1992. Top-down effects of brook trout (Salvelinus fontinalis) in a boreal forest stream. Canadian Journal of Fisheries and Aquatic Sciences 49: 2093–2103.
Biggs, B. J. F., 2000. Eutrophication of streams and rivers: dissolved nutrient–chlorophyll relationships for benthic algae. Journal of the North American Benthological Society 19: 17–31.
Biggs, B. J. E. & M. E. Close, 1989. Periphyton biomass dynamics in gravel bed rivers: the relative effects of flow and nutrients. Freshwater Biology 22: 209–231.
Blanchet, S., G. Loot & J. J. Dodson, 2008. Competition, predation and flow rate as mediators of direct and indirect effects in a stream food chain. Oecologia 157: 93–104.
Brönmark, C., J. Dahl & L. A. Greenberg, 1997. Complex trophic interactions in freshwater benthic food chains. In Streit, B., T. Städler, C. M. Lively (eds) Evolutionary Ecology of Freshwater Animals: Concepts and Case Studies. Birkhäuser, Boston: 55–88.
Buria, L., R. Albariño, V. Díaz Villanueva, B. Modenutti & E. Balseiro, 2007. Impact of exotic rainbow trout on the benthic macroinvertebrate community from Andean-Patagonian headwater streams. Fundamental and Applied Limnology 168: 145–154.
Buria, L. M., R. J. Albarino, B. E. Modenutti & E. G. Balseiro, 2009. Temporal variations in the diet of the exotic rainbow trout (Oncorhynchus mykiss) in an Andean-Patagonian canopied stream. Revista Chilena de Historia Natural 82: 3–15.
Carpenter, S. R., J. F. Kitchell & J. R. Hodgson, 1985. Cascading trophic interactions and lake productivity. BioScience 35: 634–638.
Díaz Villanueva, V. & R. Albariño, 1999. Feeding habit of Notoperla archiplatae (Plecoptera) larvae in a North Patagonia Andean stream, Argentina. Hydrobiologia 412: 43–52.
Díaz Villanueva, V., R. Albariño & B. Modenutti, 2004. Grazing impact of two aquatic invertebrates on periphyton from an Andean-Patagonian stream. Archiv fur Hydrobiologie 159: 455–471.
Dobson, M. & A. G. Hildrew, 1992. A test of resource limitation among shredding detritivores in low order streams in southern England. Journal of Animal Ecology 61: 69–77.
Duffy, J. E., 2002. Biodiversity and ecosystem function: the consumer connection. Oikos 99: 201–219.
Eggert, S. L. & J. B. Wallace, 2003. Litter breakdown and invertebrate detritivores in a resource-depleted Appalachian stream. Archiv für Hydrobiologie 156: 315–338.
Forrester, G. E., T. L. Dudley & N. B. Grimm, 1999. Trophic interactions in open systems: effects of predators and nutrients on stream food chains. Limnology and Oceanography 44: 1187–1197.
Gaedke, U., D. Straile & C. Pahl-Wostl, 1996. Trophic structure and carbon flow dynamics in the pelagic community of a large lake. In Polis, G. & K. Winnemiller (eds), Food Webs: Integration of Patterns and Dynamics. Chapman and Hall, New York: 60–71.
Graham, A. A., D. J. McCaughan & F. S. McKee, 1988. Measurement of surface area of stones. Hydrobiologia 157: 85–87.
Greig, H. S. & A. R. McIntosh, 2006. Indirect effects of predatory trout on organic matter processing in detritus-based stream food webs. Oikos 112: 31–40.
Hall, R. O., J. B. Wallace & S. L. Eggert, 2000. Organic matter flow in stream food webs with reduced detrital resource base. Ecology 81: 3445–3463.
Hart, D. D. & C. T. Robinson, 1990. Resource limitation in a stream community: phosphorus enrichment effects on periphyton and grazers. Ecology 71: 1494–1502.
Herbst, D. B., E. L. Silldorff & S. D. Cooper, 2009. The influence of introduced trout on the benthic communities of paired headwater streams in the Sierra Nevada of California. Freshwater Biology 54: 1324–1342.
Hill, W. R., M. G. Ryon & E. M. Schilling, 1995. Light limitation in a stream ecosystem: responses by primary producers and consumers. Ecology 76: 1297–1309.
Katano, O., Y. Aonuma, T. Nakamura & S. Yamamoto, 2003. Indirect contramensalism through trophic cascades between two omnivorous fishes. Ecology 84: 1311–1323.
Kishi, D., M. Murakami, S. Nakano & K. Maekawa, 2005. Water temperature determines strength of top-down control in a stream food web. Freshwater Biology 50: 1315–1322.
Marchand, F., P. Magnan & D. Boisclair, 2002. Water temperature, light intensity and zooplankton density and the feeding activity of juvenile brook charr (Salvelinus fontinalis). Freshwater Biology 47: 2153–2162.
McIntosh, A. R., 2000. Habitat- and size-related variations in exotic trout impacts on native galaxiid fishes in New Zealand streams. Canadian Journal of Fisheries and Aquatic Sciences 57: 2140–2151.
McIntosh, A. R., H. S. Greig, S. A. McMurtrie, P. E. R. Nystrom & M. J. Winterbourn, 2005. Top-down and bottom-up influences on populations of a stream detritivore. Freshwater Biology 50: 1206–1218.
Meissner, K. & T. Muotka, 2006. The role of trout in stream food webs: integrating evidence from field surveys and experiments. Journal of Animal Ecology 75: 421–433.
Merritt, R. W. & K. W. Cummins, 1996. An Introduction to the Aquatic Insects of North America. Kendall/Hunt Publishing Company, Dubuque, IA.
Mulholland, P. J., C. S. Fellows, J. L. Tank, N. B. Grimm, J. R. Webster, S. K. Hamilton, E. Marti, L. Ashkenas, W. B. Bowden, W. K. Dodds, W. H. McDowell, M. J. Paul & B. J. Peterson, 2001. Inter-biome comparison of factors controlling stream metabolism. Freshwater Biology 46: 1503–1517.
Nakano, S., H. Miyasakan & N. Kuhara, 1999. Terrestrial-aquatic linkages: riparian arthropod inputs alter trophic cascades in a stream food web. Ecology 80: 2435–2441.
Nusch, E. A., 1980. Comparison of different methods for chlorophyll and phaeopigment determination. Archiv fur Hydrobiologie Beiheft Ergebnisse der Limnologie 14: 14–36.
Nystrom, P., A. R. McIntosh & M. J. Winterbourn, 2003. Top-down and bottom-up processes in grassland and forested streams. Oecologia 136: 596–608.
Palma, A., R. Figueroa, V. H. Ruiz, E. Araya & P. Berríos, 2002. Composición de la dieta de Oncorhynchus mykiss (Walbaum 1792) (Pisces: Salmonidae) en un sistema fluvial de baja intervención antrópica: Estero Nonguén, VIII Región, Chile. Gayana 66: 129–139.
Pascual, M., P. Macchi, J. Urbanski, F. Marcos, C. Riva Rossi, M. Novara & P. Dell’Arciprete, 2002. Evaluating potential effects of exotic freshwater fish from incomplete species presence-absence data. Biological Invasions 4: 101–113.
Peckarsky, B. L., B. L. Kerans, B. W. Taylor & A. R. McIntosh, 2008. Predator effects on prey population dynamics in open systems. Oecologia 156: 431–440.
Persson, L., 1999. Trophic cascades: abiding heterogeneity and the trophic level concept at the end of the road. Oikos 85: 385–397.
Power, M. E., 1992. Top-down and bottom-up forces in food webs – do plants have primacy. Ecology 73: 733–746.
Rosemond, A. D., 1994. Multiple factors limit seasonal variation in periphyton in a forest stream. Journal of the North American Benthological Society 13: 333–344.
Rosemond, A. D., P. J. Mulholland & S. H. Brawley, 2000. Seasonally shifting limitation of stream periphyton: response of algal populations and assemblage biomass and productivity to variation in light, nutrients, and herbivores. Canadian Journal of Fisheries and Aquatic Sciences 57: 1–10.
Rosenfeld, J., 2000. Effects of fish predation in erosional and depositional habitats in a temperate stream. Canadian Journal of Fisheries and Aquatic Sciences 57: 1369–1379.
Ruetz, C. R., R. M. Newman & B. Vondracek, 2002. Top-down control in a detritus-based food web: fish, shredders, and leaf breakdown. Oecologia 132: 307–315.
Schmitz, O. J., V. Krivan & O. Ovadia, 2004. Trophic cascades: the primacy of trait-mediated indirect interactions. Ecology Letters 7: 153–163.
Shurin, J. B., D. S. Gruner & H. Hillebrand, 2006. Review all wet or dried up? Real differences between aquatic and terrestrial food webs. Proceedings of the Royal Society Series B Biological Sciences 273: 1–9.
Townsend, C., 2003. Individual, population, community, and ecosystem consequences of a fish invader in New Zealand Streams. Conservation Biology 17: 38–47.
Wallace, J. B., S. L. Eggert, J. L. Meyer & J. R. Webster, 1999. Effects of resource limitation on a detrital-based ecosystem. Ecological Monographs 69: 409–442.
Woodward, G., R. Thompson, C. R. Townsend & A. G. Hildrew, 2005. Pattern and process in food webs: evidence from running waters. In Belgrano, A., U. M. Scharler, J. Dunne & R. E. Ulanowicz (eds), Aquatic Food Webs: An Ecosystem Approach. Oxford University Press, Oxford.
Acknowledgements
We thank the authorities of Nahuel Huapi National Park for permission to conduct the field survey and experiments. This study was supported by Fondo para la Investigación Científica (FONCyT) PICT 2007-01256, PICT 2007-01747, PICT 2007-01258, CONICET PIP 112-200801-01702 and Universidad Nacional del Comahue B141. RA, VDV, BM and EB are CONICET Researchers.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Robert Bailey
Rights and permissions
About this article
Cite this article
Buria, L., Albariño, R., Díaz Villanueva, V. et al. Does predation by the introduced rainbow trout cascade down to detritus and algae in a forested small stream in Patagonia?. Hydrobiologia 651, 161–172 (2010). https://doi.org/10.1007/s10750-010-0293-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-010-0293-9