Abstract
Physical soil disturbance and the hydrology of temporary pools affect the biomass, species composition and richness of plant communities. Disturbance liberates sites for the random recruitment of new individuals. The addition of seeds modifies the structure of the communities. In order to verify these hypotheses concerning the vegetation of temporary pools, an experiment was carried out using 72 soil samples collected from a pool in Western Morocco and placed in containers. Three types of laboratory treatments were applied, each combined with control treatments: soil disturbance (control/disturbed), hydrology (flooded, saturated and dry) and seed addition (sowing/no sowing). The total biomass, the annual and perennial species richness were calculated for each sample to test the effects of disturbance, hydrology and seed addition on the biomass and species richness of the various plant communities. The results show that disturbance reduces the total biomass, especially of perennials, but without significantly increasing the richness of annuals. Seed addition does not affect the total biomass and reduces total richness only in saturated soil, where biomass production is high. The most extreme stress conditions (drought and flooding) limit the abundance of species and therefore competition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In aquatic ecosystems, the establishment, development and species diversity of plant communities are often controlled by a combination of internal and external forces (Mitsch & Gosselink, 2000). The most important external forces include periodic disturbances (fire, herbivores, etc.) and hydrological stress (long-term drought-flooding cycles) which differentially favour particular species (Keddy & Fraser, 2000; Trémolières, 2004; Bornette et al., 2008). Stress is defined as external constraints that limit the rate of biomass production of some or all plant species and disturbance as a mechanism that limits biomass through varying levels of destruction (Grime, 2001). The effects of stress and disturbance are accentuated in habitats that are deficient in resources and, consequently, have a low competition capacity (Gaudet & Keddy, 1995; Van Eck et al. 2005).
The importance of these two factors (stress and disturbance) in the regulation of species distribution and community dynamics is widely known (Koning, 2005; Devictor et al., 2007). Disturbance leads to a considerable change to the structure of communities which can in turn affect population dynamics (Elderd & Doak, 2006). Vegetation responds to disturbance and stress in function of their intensity (Bisigato et al., 2008). Disturbance can act as a force for maintaining biodiversity in aquatic systems (Lepori & Hjerdt, 2006). However, if the disturbance increases in intensity and frequency, it leads to local extinction and a reduction in species richness, whereas an increase in the frequency of stress leads to an increase in richness due to the limitation of competition (Tabacchi & Planty-Tabacchi, 2005).
Trampling is a form of disturbance that affects vegetation both directly and indirectly (Liddle, 1975; Kobayashi et al., 1997); it has a selective impact on the abundance of species (Gallet & Rozé, 2001) and modifies their composition and structure (Liddle, 1997) by creating regeneration patches where seeds can germinate and become established without any significant competition effect (Chambers, 1995; Siemann & Rogers, 2003). The regeneration of plant communities within patches can also occur by means of the storage organs (stolons and rhizomes) from individuals in the area of the disturbance (Barrat-Segretain & Bornette, 2000). The mechanisms enabling species occurrence in disturbed systems involve a trade-off between species’ competition capabilities and their tolerance of disturbance (Kneitel & Chase, 2004).
The availability of seeds is a determining factor in the regeneration of disturbed habitats (Tilman, 1997; Zobel et al., 2000; Foster & Tilman, 2003; Foster & Dickson, 2004). In aquatic habitats, the presence of durable seed stocks makes easy regeneration (Van der Valk & Davis, 1978), nevertheless the density of seed banks fluctuates from year to year in function of reproduction phenology and environmental constraints (Leck & Simpson, 1995, Capon & Brock, 2005). After a favourable year, the seed banks of certain species increase in the sediment (Bonis et al., 1995). It remains debatable how the over-abundance of a particular species within the seed bank affects the vegetation composition. Variable results have been obtained in experiments involving the addition of seeds to terrestrial habitats. The addition of seeds to grassland communities had a positive effect on vegetation cover (Tilman, 1997) and biomass production (productivity), whereas no effect was shown in other studies (Wilsey & Polley, 2003; Mouquet et al., 2004).
In the Mediterranean region, temporary pools are wetland habitats with high biodiversity and numerous rare species, particularly in Morocco (Grillas et al., 2004). They are submitted to multiple pressures, both natural and from anthropogenic origin, influencing the structure and composition of their plant communities (Deil, 2005). The interannual rainfall variability of the Mediterranean climate results in considerable variations in the depth and duration of the flooding of the pools (Thiéry, 1991; Grillas et al., 2004). The disturbances caused by the alternating flooded and dry phases that occur during the course of the annual cycle lead to the destruction of many individual plants and result in a high level of species diversity (Médail et al., 1998). In addition, the low productivity resulting principally from summer drought enables the coexistence of plant species that are typically annual, relatively uncompetitive and small in size (Médail et al., 1998). Disturbances generally lead to a reduction in the biomass of perennials, thus, favouring higher species richness, especially annuals, in the plant communities (Rhazi et al., 2005).
In temporary pools, the interannual hydrological variations determine community composition and seed production (Bliss & Zedler, 1998, Grillas & Battedou, 1998). Seed banks are continually modified by the cumulative effects of environmental pressures that have differential impacts on the germination, development and reproduction of species (Bonis et al., 1993, 1995; Brock & Rogers, 1998).
The aim of this study was to test experimentally the following hypotheses:
-
1.
Physical disturbances of the soil affect the structure of communities by reducing the biomass of perennials and increasing the richness of annual species;
-
2.
The abundance of seeds in the seed bank influences the composition of the vegetation;
-
3.
Hydrology (water level variation) plays an essential role in the selection of species.
Materials and methods
Experimental design
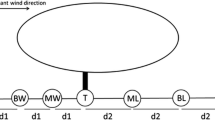
The study was conducted on the vegetation of a temporary pool located at Benslimane, Western Morocco (W33°38′697″; N007°05′215″; elevation: 257 m; area: 3760 m2) in a Cork-oak forest. A set of 72 soil samples (18 cm × 18 cm; 5 cm depth) were randomly collected in December 2006, using a metallic frame (18 cm × 18 cm) within two distinct belts (Rhazi, 2001), the edge and the centre (Fig. 1). Each soil sample was carefully transferred into a plastic container (18 cm × 18 cm × 10 cm depth), to maintain the whole clod soil intact (avoiding mixing). Each container was considered as containing a representative sample of the plant community (Grillas et al., 1992) where the dormant forms dominated (seed bank and bulbs). The samples (72) were placed in the laboratory, subdivided into two sets of 36 samples which were, respectively, submitted to the following treatments (Fig. 1):
Disturbed
In the top soil, 5 cm of soil was mixed manually to disturb the vertical structure of the seed bank and to homogenise the soil. This treatment is a simulation of the disturbance made by herbivores by trampling on wet soils and burrowing (wild boar).
Control
Sediment core remained intact (no mixing). Each set of 36 samples was secondly separated into three subsets of 12 samples, which were, respectively, submitted to one of the following treatments from early January 2006 to late June 2007:
Flooded (F)
During the whole experiment, the soil was kept flooded with 3 cm of water above soil surface.
Wet (W)
Samples were watered daily to maintain high humidity of soil (minimum of 95% saturation).
Dry (D)
Soil was watered twice a week (15% of saturation).
Finally, each subset of 12 samples was separated into two groups of six samples that were submitted, respectively, to one of the following treatments:
Sowing (seed addition)
Under a binocular microscope, 100 seeds of either Illecebrum verticillatum or Spergularia salina were carefully selected and added into each sample. I. verticillatum (an amphibious annual species) seeds were added to the flooded and wet treatments and S. salina seeds (a terrestrial annual species) to the dry treatment. The seeds of both species were collected in the previous autumn in the same pool (September 2005) at the end of plant life cycle.
No sowing (control)
The samples were randomly placed in the laboratory and randomized weekly during the experiment.
The vegetation of each sample was harvested on three successive dates (March, April and June), respectively, on one randomly selected third of the surface area (3 squares 6 cm × 6 cm, selected from a grid of nine squares). The vegetation was cut at soil surface, separated by species and dried at 60°C for 48 h and the dry weight of each species was measured. The biomass considered for each species was the maximum biomass of the three dates. The total biomass and the contribution of annuals and perennials to the total biomass were calculated. The annual and perennial life span was determined according to the flora of North Africa (Maire, 1952–1987) and flora of Morocco (Fennane et al., 1999, 2007). For each sample and each sampling date (March, April and June), the species richness, as well as the annual and perennial species richness, were calculated.
Data analysis
The differences between the biomass of the ‘control’ and ‘disturbed’ treatments and between the species richness of the different hydrological treatments were tested using analysis of variance (ANOVA) (n = 72). The effects of hydrology, disturbance, sowing and there interaction on the biomass (total, annuals and perennials) and the species richness (total, annuals and perennials) were tested using multiple regressions excluding the biomass and richness of the sowed species (I. verticillatum and S. salina) (n = 72).
The effects of ‘hydrology’ and ‘sowing’ and their interaction on species richness were tested by multiple regressions. The effects of sowing on the species richness (total, annuals and perennials) and on the biomass production (total, annuals and perennials) were tested using ANOVA tests for the three hydrological treatments together (n = 72) and also separately (flooded, wet, dry) (n = 24). The biomass and richness of the sowed species (I. verticillatum and S. salina) were not included in the analysis.
The effect of the ‘disturbed’ treatment on the abundance of each occurring species was tested using Kruskal–Wallis non-parametric tests for each hydrological treatment. The abundance per sample for each species was calculated as the ratio of the species biomass over the total biomass.
Results
Biomass production
The total biomass produced (324 cm2 area) was significantly affected (multiple regression F = 14.43; df = 6; P < 0.01) by hydrology (F = 32.56; df = 2; P < 0.01) and by disturbance (F = 19.5; df = 1; P < 0.01). No significant effect, however, was found for sowing and the interaction between the three factors (P > 0.05). The hydrology factor explains 65% of the total variance and 23% of the disturbance. The perennial species biomass was significantly affected (multiple regression F = 18.3; df = 6; P < 0.01) by hydrology (F = 48.3; df = 2; P < 0.01) and by disturbance (F = 9.5; df = 1; P < 0.01); but no significant effect of sowing and of the interaction between these factors (P > 0.05). Concerning the biomass of annuals (multiple regression F = 5.5; df = 6; P < 0.01), a significant effect was found only for hydrology (F = 4.17; df = 2; P < 0.01).
The total species biomass per sample was significantly higher (25%) in the control than in the disturbed treatment (Table 1). There was no significant difference between the biomass of annual species in the two treatments (P > 0.05); however, the biomass of perennials was significantly lower in the ‘disturbed’ treatment (Table 1) than in the control. No significant correlation was found between the biomass of annual and perennial species (P > 0.05).
No significant difference was found between the ‘sowing’ treatment and the control (no sowing) on the biomass of annuals, of perennials and total, even when considering each hydrological treatment separately (Table 2).
Community richness
During the experiment, 28 species germinated, most of them being annuals (71%) (Table 3). The characteristic of aquatic and amphibious species represented 82% and the terrestrial species, 18%. The total number of species recorded in each treatment was 20 (14 annuals and 6 perennials) in the ‘control’ treatment and 25 (18 annuals and 7 perennials) in the ‘disturbed’ treatment. Seventeen species occurred in both treatments, three were only found in the ‘control’ samples (Agrostis salmantica, Sagina apetala and Scilla autumnalis) and eight were solely present in the ‘disturbed’ samples (Lotus hispidus, Myosotis sicula, Apium inundatum, Baldellia ranunculoides, Pilularia minuta, Eleocharis palustris, Daucus carota and Chara sp.).
The hydrology factor had a significant effect on total species richness, as well as on the perennial and annual species richness. No significant effect on species richness was found for the ‘disturbed’ and ‘sowing’ treatment or their interaction with the hydrology factor (Table 4). Hydrology explains 78% of the total variance. Total, annual and perennial species richness were significantly higher in the ‘wet’ treatment when compared to the ‘flooded’ and ‘dry’ treatments (Fig. 2).
Species response to disturbance
Within the ‘wet’ treatment, the disturbance had a significant effect only on the abundance of Pulicaria arabica (χ2 = 4.02; df = 1; P = 0.04) and Glyceria fluitans (χ2 = 4.66; df = 1; P < 0.05), both species being less abundant in the ‘disturbed’ treatment than in the ‘control’ (Table 5). Within the ‘flooded’ treatment, Bolboschoenus maritimus was the only one species with a significantly lower abundance in the disturbed treatment than in the control (χ2 = 6.43; df = 1; P = 0.01). The disturbance factor had no significant effect on the abundance of any species of the ‘dry’ treatment (Table 5).
Effect of sowing on community richness
The total richness was not significantly affected by sowing (F = 1.23; df = 1; P > 0.05), whereas hydrology significantly affected total richness (F = 4.87; df = 2; P = 0.01) as did the interaction of hydrology and sowing (F = 4.30; df = 2; P < 0.05). Within the ‘wet’ treatment, the species richness of the annuals, perennials and total was significantly lower in the ‘sowing’ treatment (I. verticillatum) (Table 2) than in the control (no sowing). The addition of I. verticillatum seeds in the ‘flooded’ treatment did not have a significant effect on the total, perennial and annual species richness (Table 2). There was no effect on the species richness with the addition of S. salina in the ‘dry’ treatment also (Table 2).
The abundance of I. verticillatum was significantly higher in the ‘wet’ treatment (χ2 = 7.97; df = 1; P < 0.01) when compared to the ‘flooded’ treatment. No significant difference was found in the abundance of Illecebrum between the disturbed and control treatments (χ2 = 1.59; df = 1; P > 0.05). I. verticillatum abundance was significantly higher within the sowed samples (χ2 = 17.12; df = 1; P < 0.01). The abundance of I. verticillatum in the disturbed samples was not significantly different between the ‘wet’ and ‘flooded’ treatments (χ2 = 0.94; df = 1; P = 0.33). In the control samples, the abundance of I. verticillatum was significantly higher in the ‘wet’ treatment than in the ‘flooded’ treatment (χ2 = 8.93; df = 1; P<0.05).
Discussion
Effect of disturbance and hydrology on the community
The experiments carried out in the laboratory resulted in the expression of 28 species, mostly annuals (71%). The predominance of annual species in the plant community is characteristic of temporary pools (Zedler, 1987; Grillas et al., 2004; Rhazi et al., 2006; Fraga i Arguimbau, 2008). Annual species frequently predominate in case of unpredictable periods and durations of flooding and drought (Mitchell & Rogers, 1985; Brock, 1986; Médail et al., 1998). The total number of species expressed (28) in the experiment was close to the number of species (31) found in the (30 × 30 cm) quadrats used to monitor the vegetation of the pool (Rhazi, 2001). This slight difference can be explained by the low surface area sampled for the experiment (18 × 18 cm) (Grillas et al., 1992), but it can still be assumed that the number of microcosms used in this study give an accurate picture of the field.
The species richness of the communities in the microcosms can be explained by hydrology alone (78% of variance explained); this factor is often critical for the diversity of plant communities in wetland zones (Keddy & Reznicek, 1986). The richness of communities was higher in saturated soil, with respect to both annuals and perennials, and low in dry or flooded soils (Fig. 2). The alternate conditions of drought and flooding that characterise the annual cycle of temporary pools limit potentially the growth of plants (Bonis et al., 1993; Brewer et al., 1997; Bornette et al., 1998; Trémolières, 2004; Koning, 2005; Bornette et al., 2008). Conversely, saturated conditions are temporarily highly favourable to the development of a large number of species within the communities (Koning, 2005); hence, the high species richness was found in Mediterranean temporary pools.
Biomass production in the microcosms was explained first by hydrology (65% of variance explained) and second by mechanical disturbance of soil (23% of variance explained). The biomass levels produced remained low, in particular, with regard to annual plants, which were predominant in the community in terms of the number of species. The low biomass of annuals could either be due to their small size or to light conditions in the laboratory being lower than natural conditions.
Soil disturbance led to a significant reduction in biomass, particularly of perennials. Physical disturbances are known to alter the storage organs generally responsible for high biomass production (Fahrig et al., 1994; Winkler & Fischer, 2001). The species negatively affected by disturbance were the perennials B. maritimus and P. arabica, together with the annual G. fluitans. The expected result was an increase in the species richness of annuals following the reduction in the biomass of perennials. However, no significant effect of soil disturbance on the species richness of annuals was revealed (Table 4). This could be due to:
-
1.
The burying of seeds after mixing of the sediment. Burial considerably reduces the germination of seeds not only because of the lack of light but also by raising to the surface older seeds with lower germinating power (Bonis et al., 1993; Devictor et al., 2007).
-
2.
The biomass of perennials was too low to induce competition between species (production too low and experiment duration too short).
Effect of seed addition on community structure
Adding seeds of Illecebrum and Spergularia did not affect the total biomass produced in the microcosms but, in the case of saturated soil only, it did have a significant effect on the total richness of the community. The total biomass of Illecebrum was significantly higher in saturated soil and lower in flooded and dry soils. The addition of Illecebrum seeds combined with the saturated soil treatment led to a reduction in total species richness due to the high growth rate of the Illecebrum competing with the community species. This finding is in agreement with the results of Brewer et al. (1997) and Lenssen et al. (1999), who found that competition only plays an important role in the rarely flooded parts of wetland habitats. Conversely, in case of flooding or drought, the production of the community is low. Consequently, the growth of seed-added species remains limited and does not affect total richness or competition intensity.
These results suggest that fluctuations in the reproductive success of species in Mediterranean temporary pools (simulated experimentally by seed addition) only affect the richness of communities during the course of the subsequent cycle if the hydrological conditions are favourable (saturated).
Implications for the conservation of temporary pools
The results obtained in this study confirm that in Mediterranean temporary pools, as in wetland in general, hydrology is the primordial factor that structures and selects the species of the community. The hydrology modifies any disturbance effects and influences competition intensity through its impact on primary production. Competition only plays a role in the structuring of communities when production conditions are favourable (saturation of the sediment by water). Such conditions in the pools are transitory and their duration varies considerably from year to year, thus reducing the role of this factor in the long term.
The results of these experiments show that local disturbance (simulating that caused by herbivores) does not seem to affect the richness of temporary pool communities. This can be explained by a low degree of seed bank stratification resulting from the high frequency of this type of disturbance. Although surface seeds are buried, the previously buried seeds thus exposed still conserve high germinating power. This result does not mean that disturbance by herbivores, especially during the plants’ growing season, is not likely to have a considerable impact on the vegetation and its richness, but rather that its impact can be modulated.
References
Barrat-Segretain, M. H. & G. Bornette, 2000. Regeneration and colonization abilities of aquatic plant fragments: effects of disturbance seasonality. Hydrobiologia 421: 31–39.
Bisigato, A. J., P. E. Villagra, J. O. Ares & B. E. Rossi, 2008. Vegetation heterogeneity in Monte Desert ecosystems: a multi-scale approach linking patterns and processes. Journal of Arid Environments 73: 182–191.
Bliss, S. A. & P. A. Zedler, 1998. The germination process in vernal pools: sensitivity to environmental conditions and effects on community structure. Oecologia 113: 67–73.
Bonis, A., P. Grillas, C. Van Wijck & J. Lepart, 1993. The effect of salinity on the reproduction of coastal submerged macrophytes from experimental communities. Journal of Vegetation Science 4(4): 461–468.
Bonis, A., P. Grillas & J. Lepart, 1995. Seed bank dynamics and coexistence of annual macrophytes in temporary and variable habitat. Oikos 74: 81–92.
Bornette, G., C. Amoros & N. Lamouroux, 1998. Aquatic plant diversity in riverine wetlands: the role of connectivity. Freshwater Biology 39: 267–283.
Bornette, G., E. Tabacchi, C. Hupp, S. Puijalon & J. C. Rostan, 2008. A model of plant strategies in fluvial hydrosystems. Freshwater Biology 53: 1692–1705.
Brewer, J. S., J. M. Levin & M. D. Bertness, 1997. Effects of biomass removal and elevation on species richness in a New England salt marsh. Oikos 80: 333–341.
Brock, M. A., 1986. Adaptation to fluctuations rather than to extremes of environmental parameters. In de Deckker, P. & W. D. Williams (eds), Limnology in Australia. CSIRO/Dr W. Junk, Melbourne/Netherlands: 131–140.
Brock, M. A. & K. H. Rogers, 1998. The regeneration potential of the seed bank of an ephemeral floodplain in South Africa. Aquatic Botany 61: 123–135.
Capon, S. J. & M. A. Brock, 2005. Flooding, soil seed bank dynamics and vegetation resilience of a hydrologically variable desert floodplain. Freshwater Biology 51(2): 206–223.
Chambers, J. C., 1995. Relationship between seed fates and seedling establishment in an alpine ecosystem. Ecology 76: 2124–2133.
Deil, U., 2005. A review on habitats, plant traits and vegetation of ephemeral wetlands – a global perspective. Phytocoenologia 35: 533–705.
Devictor, V., J. Moret & N. Machon, 2007. Impact of ploughing on soil seed bank dynamics in temporary pools. Plant Ecology 192(1): 45–53.
Elderd, B. D. & D. F. Doak, 2006. Comparing the direct- and community-mediated effects of disturbance on plant population dynamics: flooding, herbivory and Mimulus guttatus. Journal of Ecology 94(3): 656–669.
Fahrig, L., D. P. Coffin, W. K. Lauenroth & H. H. Shugart, 1994. The advantage of long-distance clonal spreading in highly disturbed habitats. Evolutionary Ecology 8(2): 172–187.
Fennane, M., M. Ibn Tattou, J. Mathez, A. Ouyahya & J. El Oualidi (eds), 1999. Flore pratique du Maroc. Manuel de détermination des plantes vasculaires, Vol. 1. Travaux de l’Institut Scientifique, Série Botanique No. 36, Rabat.
Fennane, M., M. Ibn Tattou, A. Ouyahya & J. El Oualidi (eds), 2007. Flore Pratique du Maroc, Manuel de détermination des plantes vasculaires, Vol. 2. Travaux de l’Institut Scientifique, Série Botanique No. 38. Rabat.
Foster, B. L. & T. Dickson, 2004. Grassland diversity and productivity: the interplay of resource availability and propagule pools. Ecology 85: 1541–1547.
Foster, B. L. & D. Tilman, 2003. Seed limitation and the regulation of community structure in Oak Savanna grassland. Journal of Ecology 91: 999–1007.
Fraga i Arguimbau, P., 2008. Vascular flora associated to Mediterranean temporary ponds on the island of Minorca. Anales del Jardín Botánico de Madrid 65(2): 393–414.
Gallet, S. & F. Rozé, 2001. Resistance of Atlantic heathlands to trampling in Brittany (France): influence of vegetation type, season and weather conditions. Biological Conservation 97: 189–198.
Gaudet, C. L. & P. A. Keddy, 1995. Competitive performance and species distribution in shoreline plant communities: a comparative approach. Ecology 76: 280–291.
Grillas, P. & G. Battedou, 1998. Effects of the date of flooding on the biomass, species composition and seed production of submerged macrophyte beds in temporary marshes in the Camargue (S. France). Proceedings of the Intecol Conference, Perth, September 1996. In Wetlands for the Future (McComb A.J. & J.A. Davis, eds), INTECOL’S V International Wetland Conference, pp: 207–218.
Grillas, P., V. Boy & C. Van Wijck, 1992. Transferring sediment containing seed bank: a method for studying plant community ecology. Hydrobiologia 228(1): 29–36.
Grillas, P., P. Gauthier, N. Yavercovski & C. Perennou, 2004. Mediterranean temporary pools: issues relating to conservation functioning and management, Vol. 1. Tour du Valat, Arles.
Grime, J. P., 2001. Plant Strategies, Vegetation Processes, and Ecosystem Properties. Wiley, Chichester.
Keddy, P. A. & L. H. Fraser, 2000. Four general principles for the management and conservation of wetlands in large lakes: the role of water levels, nutrients, competitive hierarchies and centrifugal organization. Lakes and Reservoirs: Research and Management 5: 177–185.
Keddy, P. A. & A. A. Reznicek, 1986. Great Lakes vegetation dynamics: the role of fluctuating water levels and buried seeds. Journal of Great Lakes Research 12: 25–36.
Kneitel, J. M. & J. M. Chase, 2004. Trade-offs in community ecology: linking spatial scales and species coexistence. Ecology Letters 7: 69–80.
Kobayashi, T., Y. Hori & N. Nomoto, 1997. Effects of trampling and vegetation removal on species diversity and micro-environment under different shade conditions. Journal of Vegetation Science 8: 873–880.
Koning, C., 2005. Vegetation patterns resulting from spatial and temporal variability in hydrology, soils and trampling in a northern basin marsh, New Hampshire, USA. Wetlands 25(2): 239–251.
Leck, M. A. & R. L. Simpson, 1995. Ten years seed bank and vegetation dynamics of tidal freshwater marsh. American Journal of Botany 82: 1547–1557.
Lenssen, J. P. M., F. B. J. Menting, W. H. van der Putten & C. W. P. M. Blom, 1999. Control of plant species richness and zonation of functional groups along a freshwater flooding gradient. Oikos 86: 523–534.
Lepori, F. & N. Hjerdt, 2006. Disturbance and aquatic biodiversity: reconciling contrasting views. Bioscience 56: 809–818.
Liddle, M. J., 1975. A selective review of the ecological effects of human trampling on natural ecosystems. Biological Conservation 7: 17–36.
Liddle, M. J., 1997. Recreation Ecology. Chapman & Hall, London.
Maire, R., 1952–1987. Flore de L’Afrique du Nord, Vol. 16. Paul Lechevalier, Paris.
Médail, F., H. Michaud, J. Molina, G. Paradis & R. Loisel, 1998. Conservation de la flore et de la végétation des mares temporaires dulçaquicoles et oligotrophes de France Méditerranéenne. Ecologia Mediditerranea 24(2): 119–134.
Mitchell, D. S. & K. H. Rogers, 1985. Seasonality/aseasonality of aquatic macrophytes in Southern hemisphere inland waters. Hydrobiologia 125: 137–150.
Mitsch, W. J. & J. G. Gosselink, 2000. Wetlands, 3rd edn. Wiley, New York, NY, USA.
Mouquet, N., P. Leadley, J. Meriguet & M. Loreau, 2004. Immigration and local competition in herbaceous plant communities: a three-year seed-sowing experiment. Oikos 104: 77–90.
Rhazi, L., 2001. Etude de la végétation des mares temporaires et l’impact des activités humaines sur la richesse et la conservation des espèces rares au Maroc. Thèse de Doctorat, Université Hassan II Faculté des Sciences Ain Chock, Casablanca.
Rhazi, M., P. Grillas, F. Médail & L. Rhazi, 2005. Consequences of shrub dynamics on the richness of aquatic vegetation in oligotrophic seasonal pools in Southern France. Phytocoenologia 35: 489–510.
Rhazi, L., M. Rhazi, P. Grillas & D. El Khyari, 2006. Richness and structure of plant communities in temporary pools from Western Morocco: influence of human activities. Hydrobiologia 570: 197–203.
Siemann, E. & W. E. Rogers, 2003. Reduced resistance of invasive varieties of the alien tree Sapium sebiferum to a generalist herbivore. Oecologia 135: 451–457.
Tabacchi, E. & A. M. Planty-Tabacchi, 2005. Exotic and native plant community distributions within complex riparian landscapes: a positive correlation. Ecoscience 12: 423–434.
Thiéry, A., 1991. Multispecies coexistence of branchiopods (Anostraca, Notostraca & Spinicaudata) in temporary ponds of Chaouia plain (Western Morocco): sympatry or syntopy between usually allopatric species. Hydrobiologia 212: 117–136.
Tilman, D., 1997. Community invisibility, recruitment limitation, and grassland biodiversity. Ecology 78: 81–92.
Trémolières, M., 2004. Plant response strategies to stress and disturbance: the case of aquatic plants. Journal of Bioscience 29: 461–470.
Van der Valk, A. G. & C. B. Davis, 1978. The role of seed banks in the vegetation dynamics of prairie glacial marshes. Ecology 59: 322–335.
Van Eck, W. H. J. M., H. M. Van de Steeg, C. W. P. M. Blom & H. de Kroon, 2005. Recruitment limitation along disturbance gradients in river floodplains. Journal of Vegetation Science 16: 103–110.
Wilsey, B. J. & H. W. Polley, 2003. Effects of seed additions and grazing history on diversity and productivity of subhumid grasslands. Ecology 84: 920–931.
Winkler, E. & M. Fischer, 2001. The role of vegetative spread and seed dispersal for optimal life histories of clonal plants: a simulation study. Evolutionary Ecology 15: 281–301.
Zedler, P. H., 1987. The ecology of Southern California vernal pools: a community profile. U.S. Fish and Wildlife Service Biological Report 85: 7–11.
Zobel, M., M. Otsu, J. Tiira, M. Moora & T. Mols, 2000. Is small-scale species richness limited by seed availability or microsite availability? Ecology 81: 3274–3282.
Acknowledgements
We thank Deirdre Flanagan for help in English, Dr S. D. Muller (University of Montpellier 2) for supporting the project, Florence Daubigney for her logistical and technical support and two anonymous referees for constructive comments which helped in a significant improvement of manuscript. This project has been achieved with the financial support of the Egide Volubilis programme AI N° MA/07/172 and was partly funded by the Fondation Tour du Valat and Fondation MAVA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: B. Oertli, R. Cereghino, A. Hull & R. Miracle
Pond Conservation: From Science to Practice. 3rd Conference of the European Pond Conservation Network, Valencia, Spain, 14–16 May 2008
Rights and permissions
About this article
Cite this article
Sahib, N., Rhazi, L., Rhazi, M. et al. Experimental study of the effect of hydrology and mechanical soil disturbance on plant communities in Mediterranean temporary pools in Western Morocco. Hydrobiologia 634, 77–86 (2009). https://doi.org/10.1007/s10750-009-9894-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-009-9894-6