Abstract
The physical and chemical variabilities as well as the distribution of diatoms of six boreal lakes in the Laurentian Mountains (southern Québec, Canada) were studied. The lakes are located along an altitudinal gradient and were sampled at a biweekly resolution from May through October, 2002. In general, we found later onset and weaker lake stratification under colder climates. Lake circulation and SiO2 are strongly correlated and together significantly explain the distribution of diatoms of the individual lakes. Diatoms that accumulated in the sediment traps were mostly composed of benthic species, suggesting resuspension. However, diatom flux and lake circulation were not significantly correlated, the diatom assemblages in the sediment traps were similar in two consecutive years, and species–environment relationships were comparable among lakes, which indicates that the effects of resuspension were minimal. In addition, we found that one lake was more productive due to forest logging. The forest in the catchment of Lake Maxi was entirely clear-cut shortly prior to our sampling. Mean total phosphorus, dissolved organic carbon, and chlorophyll a concentrations were significantly higher when compared to the other five study lakes. This study seeks to improve our understanding of how diatoms in boreal lakes respond to climate change and forest clear-cut.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climate warming is having a profound impact on the Earth’s environment, including the earlier seasonal ice break-up of rivers and lakes (Smol et al., 2005; Duguay et al., 2006; Intergovernmental Panel on Climate Change, 2007). Many paleoclimate studies have focused on annual climate reconstructions to constrain models of future climate change (Hughes & Ammann, 2009). However, there is a growing interest in higher resolution subannual climate reconstructions (Sorvari et al., 2002; Hausmann & Pienitz, 2007). Siliceous remains of diatoms (class Bacillariophyta) that are archived in lake sediments can provide such high-resolution seasonal quantitative climate records (Stoermer & Smol, 1999; Hausmann & Pienitz, 2007). However, to achieve these high-resolution reconstructions, there must be a greater understanding of modern seasonal processes in lakes. A recent study by Nonaka et al. (2007) found that the spring ice break-up is occurring earlier on boreal lakes and rivers in south-eastern Canada due to climate warming. The changes in lake boundary conditions as a response to climate change should affect lake circulation patterns, nutrient distribution, and diatom assemblages, but need to be quantified (Salonen et al., 1984; Reynolds, 1993). Regularly sampled sediment traps (designed to collect diatoms) deployed in lakes together with water chemistry sampling have the capacity to capture seasonal species–environment relationships under specific climate conditions. Therefore, by monitoring a series of lakes along an altitudinal gradient, seasonal species–environmental relationships as a response to climate change can be quantified. This study explores the impact of altitude on the seasonality of lakes by analyzing diatom-environment responses in southern Québec.
A major requirement for climate reconstructions using lake sediments is that the lake and its biota are sensitive recorders of changes in the lake environment driven by climate (Birks, 1995). A climate gradient can be latitudinal or altitudinal. An altitudinal gradient has the advantage that the insolation among lakes is comparable. At first sight, boreal (taiga) lakes in southern Quebec may appear to be less suited to climate change studies than remote arctic lakes because they have a potentially higher exposure to anthropogenic influences, such as forest harvesting (Desjardins & Monderie, 1999; Pienitz et al., 2004). However, the proximity of these lakes to a population center allows for year-round biweekly sampling, a logistical constraint that often limits high-resolution sampling in the Arctic (Lotter et al., 1999). In addition, diatoms in boreal lakes have been shown to respond sensitively to changes in lake catchment vegetation as a response to climate warming (Pienitz & Smol, 1993; Fallu et al., 2000). In addition, Schindler et al. (1996) found lower SiO2 concentrations of boreal lakes in the Experimental Lake District Area related to recent climate warming.
This study documents the seasonal changes in the water chemistry and diatom assemblages, which are measured and sampled biweekly using sediment traps in six boreal forest lakes located along a climate gradient in the Laurentian Mountains (southern Québec, Canada). We explored species–environment relationships using multivariate statistics. We also discuss uncertainties related to the use of sediment traps in shallow lakes.
Study sites
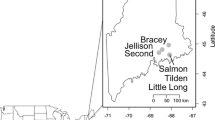
The six study lakes (Cyprès, Joachim, Moreau, du Sommet, Elysée, and Maxi) are of glacial origin and are located in a natural reserve in the Laurentian Mountains on granitic–gneissic bedrock of the Canadian Precambrian Shield (Fig. 1). The study lakes are distributed along an elevation gradient (280–950 m asl, Table 1). The maximum lake depth varied between 3 and 16 m, catchment area ranged from 0.41 to 24.67 km2, and lake surface area ranged from 0.02 to 0.27 km2 (Table 1). Along the elevation gradient, the lake catchment vegetation changed from Betula papyrifera (paper birch) to Abies balsamea (balsam fir), which is replaced by Picea mariana (black spruce) above 800 m asl. Within the study region, permafrost occurs sporadically at an elevation of 960 m asl, reflecting the strong continental character of the regional climate (Allard & Fortier, 1990). Except for a narrow 20-m-wide zone along the lake perimeter, the catchment of Lake Maxi was completely clear-cut shortly before sampling in 2002, when the soils were still lacking a vegetation cover.
Location of the study lakes in southern Québec superimposed on a digital elevation map (http://atlas.nrcan.gc.ca)
Materials and methods
Collection of water samples and limnological analysis
Over the period May to October 2002, water samples were taken every 2 weeks from each lake at 1 m depth with a Wildco® Kemmerer sampler, and were filtered on the day of sampling through cellulose acetate filters (pore size of 45 μm) for the analysis of dissolved organic carbon (DOC), soluble reactive phosphorus (SRP), nitrate (NO3), nitrite (NO2), ammonium (NH4), chloride (Cl), and silicate ions (SiO2). Samples for DOC analyses were kept in brown glass bottles and those for SiO2 analyses in polyethylene bottles. Total phosphorus (TP) and total nitrogen (TN) were analyzed from unfiltered water. Water samples were stored at 4°C prior to chemical analysis by Environment Canada laboratories, Aquatic Ecosystem Protection Research Division (Burlington, Ontario). For detailed analytical procedures and methods, consult the Analytical Methods Manual (Environment Canada, 1994).
Subsequent to water sampling, light penetration was measured using a Secchi disk (diameter = 20 cm). Temperature, pH, conductivity, and oxygen profiles were measured using a Quanta Hydrolab®. Water temperatures of each study lake were recorded in 1.5 h intervals by two Hobo® dataloggers circa 10 cm below the lake surface and circa 50 cm above the lake bottom. A ratio calculated between temperatures measured at the bottom and near the surface of the water column was used to estimate the degree of water column stratification and lake circulation (Köster & Pienitz, 2006).
For analysis of chlorophyll a (Chl a), 300 ml aliquots of water was filtered in dimmed light conditions using Whatman® GF/F glass micro fiber filters. The Chl a samples were stored in aluminum foil at −15°C until Chl a fluorescence measurements were performed according to standard procedures (Nusch, 1980). Chl a was corrected for phaeopigments using the equations of Jeffrey & Welschmeyer (1997). Lake Elysée was also investigated at monthly intervals in 2003.
Sediment trap samples
Glew sediment traps (Paleoecological Environmental Assessment and Research Laboratory, Queen’s University, Ontario) were installed in mid-April 2002 under the lake ice cover, which had a maximum thickness of 80 cm. The upper edges of the traps were positioned approximately 1 m above the lake floors. The deposits were collected in removable glass jars of 110-ml capacity at the bottom of each of three black plastic tubes, with an internal diameter of 5.2 cm and a length of 44 cm (8.5:1 aspect ratio). Three traps were mounted together on a frame that provided nominal equal center-to-center spacing of 520 mm. Submerged vertical stability was provided by an integral central float along with a mooring system similar to that described by Larsson et al. (1986). Lugol’s solution was added to the jars at the beginning of each sampling interval to prevent grazing of algae by zooplankton. Sediment traps were collected biweekly from May 2002 to the end of October in 2002. In Lake Elysée, sampling continued in 2003 at monthly intervals until October 2003.
For diatom analysis, frustules were cleaned with heated 30% H2O2. Afterwards, the glass beakers (250 ml) were filled with distilled water, and the cleaned diatom frustules were allowed to settle for at least 12 h at 4°C. In order to calculate fluxes, a known number of microspheres (plastic beads; University College London) with a diameter of 6 μm were added to the diatom suspension (Battarbee & Kneen, 1982). Aliquots of the diatom-microsphere suspensions were transferred onto ethanol-cleaned cover glasses, and after drying at room temperature, mounted with Naphrax® mounting medium onto microscope slides. Approximately, 400 diatoms per sample were analyzed at a magnification of 1000× using differential interference contrast (DIC) optics. The taxonomy mostly followed Krammer & Lange-Bertalot (1986, 1988, 1991a, b), Camburn and Charles (2000), and Fallu et al. (2000).
Numerical analysis
For each lake, coefficients of correlation between environmental variables (TP, SRP, TN, NO2, NO3, NH4, DOC, SiO2, Si:TP ratio, Si:SRP ratio, SO4, DIC, pH, Secchi depth, temperature, circulation, and conductivity) were calculated and tested for statistical significance (Table 2). For each lake, the significance of each environmental variable for the variance in the diatom data during the open water period was tested by Monte Carlo permutation tests (999 permutations) implemented in canonical correspondence analysis and redundancy analysis (CCA and RDA; Table 2; ter Braak, 1986). In order to decide whether unimodal (CCA) or linear (RDA) ordination methods were appropriate, the species turnovers were assessed as lengths of the floristic gradients using a detrended correspondence analysis (DCA). Relative abundances of diatoms were square-root transformed, and the frequencies of rare taxa down-weighted. If the length of the floristic gradient was above two standard deviation (SD) units, a CCA with the focus on inter-sample distance scaling was used (Birks, 1995). If the species turnover was < 2 SD, a RDA, with the focus on inter-sample correlation scaling and centering by species, was applied.
In addition, to determine which environmental variables explained most of the variance among the lakes, a principal component analysis (PCA) was performed. Environmental variables were normalized prior to analysis.
A CCA was performed to determine which environmental variables best explained the distribution of diatom taxa from lakes along the altitudinal gradient. For this purpose, ter Braak & Šmilauer (1998) recommend to remove highly intercorrelated environmental variables, indicated by variance inflation factors (VIF) above 20. After screening for redundant variables, the VIFs of the set of environmental variables of our CCA were between 1.4 and 14.6. CCA was performed, with scaling focused on inter-sample distances, square-root transformation of relative species abundances, down-weighting of rare species, and normalizing environmental variables prior to ordination. All ordinations were performed using the computer program CANOCO (version 4.0; ter Braak and Šmilauer, 1998).
Results
Physical and chemical properties of the six study lakes
Five of six lakes (Lake Cyprès, Lake Joachim, Lake Moreau, Lake du Sommet, and Lake Elysée) were oligotrophic and showed no signs of lake acidification. They had mean TP concentrations of less than 10 μg l−1, Chl a concentrations of around 2 μg l−1, mean TN concentrations of at most 200 μg l−1, and Secchi depths between 3 and 4 m (Fig. 2). Lake Maxi had significantly higher TP, Chl a, and DOC concentrations, as well as higher turbidity (Secchi depth 3× shallower; Fig. 2).
Mean water chemistry, Secchi depth, and Chl a concentrations of the six study lakes in the Laurentian Mountains (C Cyprès, J Joachim, M Moreau, S Sommet, E Elysée, M Maxi). The samples were taken from May through October 2002 at biweekly intervals. The error bars represent the standard deviation. For number of samples, consult Table 2
In all lakes, seasonal SiO2 fluctuations were significantly correlated with lake circulation or temperature (Table 2). The overall SiO2 concentrations increased during enhanced lake circulation (r = 0.39, n = 65, P < 0.001; Table 3). In all lakes, except Lake Elysée, SiO2 and DOC concentrations were positively correlated (Table 2). In four of the six lakes, the Si:TP ratio was highest in autumn; in Lake Moreau, it was highest in spring and in Lake Maxi, it remained low (Fig. 4). Concentrations of NH4 were highest in June and July, and SO4 concentrations were highest during late summer.
a Surface temperatures of the six boreal lakes during 2002 (Cyprès: filled square, Joachim: filled circles, Moreau: empty circles, du Sommet: empty squares, Elysée: filled triangles, and Maxi: empty triangles). b–g Ratios of top and bottom temperature (filled squares) and SiO2 concentrations (empty diamonds) of the six study lakes during 2002. The horizontal bars indicate ice cover. In each lake, and along the climate gradient, the two variables are highly statistically significantly correlated (compare Tables 2 and 3)
The onset of thermal stratification was in mid-May in Lake Cyprès (elevation 280 m asl; Fig. 3g). Approximately 2 weeks later in early June, thermal stratification formed in Lake Joachim (370 m asl), Lake Moreau (620 m asl), and Lake du Sommet (830 m asl; Fig. 3d–f). In mid-June, thermal stratification developed in Lake Elysée (920 m asl; Fig. 3c). Lake Maxi (950 m asl) cannot be directly compared, as sampling was not possible until mid-July. Lake Cyprès, the lowest elevation lake, was polymictic (Fig. 3g). The two next highest lakes, Lake Joachim and Lake Moreau, had stable thermal stratification of the water column and are considered dimictic (Fig. 3f + e). At higher elevation, Lake du Sommet and Lake Elysée were also polymictic during several periods of uniform water temperature (Fig. 3c + d). Lake Maxi, the highest elevation lake, was stratified during June and July. The autumn water column circulation of the dimictic lakes started in the lower elevation Lake Joachim at the beginning of October, in Lake Moreau in mid-September, and in the highest elevation lake, Lake Maxi, in mid-August (Fig. 3f + e + b).
Relative abundance of most abundant diatom taxa with N2 > 10 deposited in sediment traps, diatom flux, water chemistry, and stratification of the water column sampled from May to October in 2002 at biweekly intervals in the six lakes and in Lake Elysée in 2003. The lowest elevation Lake Cyprès is shown at the bottom, starting with samples taken in spring
The duration of ice cover in general increased with lake elevation (r = 0.83, n = 6, P < 0.0005). The ice break-up began in the lowest elevation lake (Lake Cyprès, 280 m asl) at the end of April. Ice-out occurred at Lake Joachim (370 m asl) at the beginning of May, Lake Moreau (619 m asl) and Lake Elysée (920 m asl) during the third week of May, and in Lake du Sommet at the end of May (Table 1; Fig. 3). There is no ice-out data from Lake Maxi. All lakes froze over during the second half of October (Fig. 3).
In general, lakes with longer ice cover had significantly weaker stratification of the water column (r = 0.380, n = 65, P = 0.005; Table 3). However, Lake Cyprès, a shallow lake at the lowest elevation of the altitudinal gradient, behaved in a non-linear fashion (Fig. 3). Lake Cyprès did not stratify for the entire open water period, despite having the shortest duration of ice cover (Fig. 3g). Lake Maxi also had a non-linear response as a result of the forest clear-cut in its catchment. Significantly higher DOC concentrations caused by forest clear-cut darkened the water color and led to surface temperatures similar to those in the lower elevation lakes. In order to determine the driving factors of ice cover on lake stratification and the distribution of diatoms, the non-linear response Lakes Cyprès and Maxi were not included in the CCA (Fig. 5b). As a result, the correlation between the duration of the ice cover and lake circulation increased from 0.380 to 0.602 (Table 3). Stratification of the water column was largely independent of water depth (r = 0.04, n = 65, P = 0.79; Table 3). Elevation and depth were also not significantly correlated (r = 0.26, n = 6, P > 0.05).
Indirect and direct ordinations of biweekly samples from six shallow lakes in the Laurentian Mountains from May to October, 2002: a Sample–environment biplot of a principal components analysis (PCA), time-series trajectories are shown as solid lines, and b species–environment biplot of a canonical correspondence analysis (CCA). Symbols for the lakes are the same as in the PCA
Diatoms
Between 61 and 78 diatom taxa were identified in each lake. The relative abundances of the most common taxa (effective number of occurrences (N2) >10) and diatom fluxes are shown in Fig. 4. The seasonal species turnover was >2 SD units in the four higher elevation lakes (Moreau, du Sommet, Elysée, and Maxi) and <2 SD units in the two lower elevation lakes (Cyprès and Joachim). Typically, the highest diatom flux was recorded in spring and/or autumn (Fig. 4). Only the polymictic Lake Cyprès had a third peak in diatom flux during the summer in July (Fig. 4). The diatom taxa were predominantly benthic. Contrary to our expectation, in all study lakes, diatom flux in the traps was uncorrelated to water column circulation (Table 2). Overall diatom flux was highest in lakes with higher SRP (r = 0.42, n = 65, P < 0.0005). As expected, lake circulation was highly correlated to SiO2, Si:TP, and/or Si:SRP ratios, which significantly explained diatom composition in the individual study lakes (Table 2). A noteworthy observation is that in all but the two lowest elevation lakes, the seasonal diatom distributions were significantly related to SRP.
In spring, during lake circulation, Fragilaria virescens var. exigua GRUNOW dominated the diatom assemblages (40%) in Lake du Sommet (Fig. 4). F. virescens occurred, in spring, also in Lake Cyprès, Lake Joachim, Lake Moreau, and Lake Elysée. In general, it was significantly correlated with lake circulation (r = 0.24, n = 65, P < 0.05, Fig. 5b). From Lake Maxi, no diatom samples during spring are available (Fig. 4).
In Lake Cyprès, the Spring samples consisted mainly of Fragilaria brevistriata GRUNOW (35%) and Fragilaria pinnata EHRENBERG (20%). Also, F. pinnata was present in May, July, October, and November in Lake Cyprès (Fig. 4). In general, F. pinnata was significantly more dominant during lake circulation (r = 0.26, n = 72, P < 0.025). In Spring, the lotic diatom Meridion circulare (GREVILLE) C. AGARDH was found in Lake Moreau (10%) and was probably washed in during snowmelt.
In early summer, when the lakes started to stratify, Tabellaria flocculosa (ROTH) KÜTZING significantly increased in all lakes with formation of thermal stratification (r = 0.34, n = 72, P < 0.005; Figs. 4 + 5b). In August, Cyclotella bodanica aff. lemanica MÜLLER EX SCHRÖTER was found during lake stratification in lakes Moreau and Elysée. In August, while the lake was still stratified, Frustulia rhomboides EHRENBERG was deposited in the traps of the warmer, low elevation Lake Joachim (Fig. 4). In Lake Joachim, F. rhomboides declined with the onset of circulation of the water column (Fig. 4). In general, F. rhomboides abundance was significantly higher during lake stratification (r = 0.43, n = 72, P < 0.0005).
In almost all the study lakes, Achnanthes minutissima KÜTZING represented approximately 10% of the assemblage during the open water period (Fig. 4). In Lake Cyprès, Aulacoseira distans (EHRENB.) SIMONSEN was also prominent, and made up 15% of the diatom assemblage during the entire open water period.
In the mesotrophic Lake Maxi, Asterionella formosa HASSALL was the dominant diatom (70%) at the beginning of July and in October at low Si:TP ratios (r = −0.54, n = 7, P = 0.029; Fig. 4). In Lake Maxi, the relative abundance of A. formosa increased significantly during periods of elevated sulfate concentrations (r = 0.86, n = 8, P < 0.005; Fig. 4). A. formosa occurred also in Lake Cyprès, Lake Joachim, Lake Moreau, Lake Elysée, and Lake Maxi during low Si:TP ratios (Fig. 4). In general, the relative abundance of A. formosa was significantly higher at low Si:TP ratios (r = −0.32, n = 72, P < 0.005).
Ordinations of all samples
Principal components analysis axis 1 was correlated most strongly to Si:TP ratio, SiO2, DOC, TP, TN concentrations, and conductivity, and explained 26.8% of the variance in the physical and chemical variables among all samples (Fig. 5a). PCA axis 2 explained 16.6% of the variance and was negatively correlated to the duration of ice-cover, surface water temperature, SO4, NH4, and depth (Fig. 5a). PCA axis 1 was correlated to nutrients and PCA axis 2 to climate.
The selected environmental variables explained 44.3% of the overall diatom distribution, with CCA axis 1 and 2 explaining 13% and 6.6% of the overall diatom variance, respectively. Axis 1 was highly correlated to SiO2 (r = 0.84; Fig. 5b). The CCA results show that Fragilaria virescens var. exigua and Fragilaria pinnata were associated with higher Si:TP ratios and enhanced water column circulation (Fig. 5b), whereas Tabellaria flocculosa was related to Si:TP ratio and stronger stratification (Fig. 5b).
Discussion
Climate
Lake circulation was stronger in lakes with longer ice cover (controlled by climate) and led to increase of SiO2, as seen through significant negative correlations between SiO2 and thermal stratification, in our boreal study lakes (Table 2). In five out of six study lakes, nutrients and/or SiO2 concentrations significantly explained the distribution of diatoms. In general, the study lakes had low SiO2 concentrations. Therefore, any shifts in these nutrient-deprived systems will likely be reflected in diatom assemblage responses (Tilman, 1982). Our observation that Fragilaria species were associated with higher Si:TP ratios has been previously reported by Kilham et al. (1986) who demonstrated that Fragilaria species have the highest demand of silica among lacustrine diatoms. We conclude that the climatic control on diatoms in our lakes was linked to the redistribution of SiO2. For a review about indirect climate impact on diatoms, compare Anderson (2000) and references within.
We identified Fragilaria virescens as an indicator species for lake circulation and Tabellaria flocculosa as indicator species for thermal stratification. In a sediment trap study in Elk Lake conducted in 1979 and 1983, Bradbury (1988) found that during the year with 10 days longer ice cover, Fragilaria species dominated as a result of later onset of thermal stratification and higher Si:TP ratios. Our results are also supported by another sediment trap study by Köster & Pienitz (2006) with subannual resolution. Fragilaria species were found to be associated with stronger water column circulation. In addition, based on their sediment trap study, Lotter & Bigler (2000) considered the alternation between Fragilaria pinnata and Cyclotella comensis in a sediment core from Lake Hagelseeli (2,339 m asl; Switzerland) to be in response to changes in the duration of ice cover. These results are in correspondence with Smol (1988) who suggested that, in arctic ponds, the ratio of periphytic to planktonic diatoms is closely related to the duration and extent of ice coverage during summer. In surface sediment samples from subarctic lakes distributed across climate gradients, Fragilaria species were usually found at the cold end of the temperature gradient (Pienitz et al., 1995; Lotter et al., 1997; Joynt & Wolfe, 2001). Along an altitudinal gradient, Wunsam and Schmidt (1995) similarly found that Fragilaria species are indicative of colder temperature. Sorvari et al. (2002) compared three thermally stratified lakes with two isothermal lakes in Finnish Lapland, determining that isothermal lakes were dominated by Fragilaria species.
A comparison of a high-resolution diatom record of Lake du Sommet with the instrumental record of the past 35 years showed that the relative abundance of Fragilaria virescens var. exigua was significantly higher in samples encompassing years with colder August temperature (Hausmann & Pienitz, 2007). In the subarctic Lake Saanajärvi (Finnish Lapland), Korhola et al. (2002) interpreted a shift in fossil diatom assemblage composition from Fragilaria to Cyclotella dominance as a response to post-Little Ice Age warming. Solovieva et al. (2008) compared observed June temperatures with a high-resolution diatom record of Lake Vanukty. During recent climate warming, Fragilaria pinnata decreased and Tabellaria flocculosa increased in this boreal forest lake west of the Ural.
The studies discussed above provide validation for our finding that Fragilaria pinnata is an indicator species for lake circulation and Tabellaria flocculosa is an indicator species for thermal stratification. The sediment trap deposits from the boreal lakes in this study, situated along an altitudinal gradient, improve the seasonal paleoclimatological interpretation of lacustrine diatom records.
It is likely that the high positive correlation between SiO2 and lake circulation in four of the six lakes reflects high hypolimnetic SiO2 concentrations caused by SiO2 released from sediments (Wetzel, 2001). In order to better understand the links between lake circulation and SiO2, water samples from different depths should be included in future studies.
Forestry
Water chemistry of Lake Maxi was impacted by forest clear-cut. It had three times higher TP concentrations compared to the other five oligotrophic lakes. According to the lake classification system suggested by Vollenweider (1968), Lake Maxi was mesotrophic. Similarly, Carignan et al. (2000) and Planas et al. (2002) documented significantly increased TP, DOC, and Chl a concentrations in their nine study lakes following forest clear-cuts in Haute-Mauricie, Québec. A. formosa was the dominant diatom of Lake Maxi with 80% representation. Lund (1950) and Tilman (1982) found that Asterionella species can tolerate lower Si:TP ratios. In Lake Maxi, the seasonal distribution of Asterionella formosa was significantly increased when the Si:TP ratios were lower and at higher sulfate concentrations. Algae that are dependent on NO3 and N2 fixation have a competitive disadvantage at high sulfate concentrations, because NO3 uptake is inhibited by sulfate (Cole et al., 1986; Marino et al., 1990). However, A. formosa has a competitive advantage at high sulfate concentrations, because it can use NH4 as nitrogen source (Happey, 1970).
There are some studies that show a negligible long-term impact of forest harvesting. Laird et al. (2001) demonstrated that a 90% cumulative forest clear-cut, distributed over a 40-year period, had only negligible long-term impact on diatom assemblages or content of organic matter. However, the impact of Bronze Age forest clear-cut on lake trophy archived in lake sediments has been described by Wick et al. (2003) and Schmidt et al. (1998). At 3,700 cal. BP (Bronze Age), widespread deforestation in the catchment of Lake Sägistalsee, a small lake at 1,935 m asl in the Swiss Alps, is reflected by high erosion activity in the lake’s catchment (Wick et al., 2003) which led to reduced chironomid abundance probably due to oxygen depletion caused by eutrophic conditions (Heiri & Lotter, 2003). Köster et al. (2005) discussed a nutrient pulse due to deforestation around AD 1750 in New England as cause for a shift in the diatom assemblages of Walden Pond. The change of a domination of Aulacoseira species to A. formosa and an associated darkening of the sediment color of the top 5 cm of a gravity core from Lake Maxi followed the forest clear-cut (Hausmann, unpublished data). Paleolimnological studies contribute to better understanding of the long-term impact of catchment disturbances such as forest clear-cuts.
Sediment traps
The use of sediment traps allows for the study of time-integrated diatom growth, but requires caution when interpreting the data (Cameron, 1995). Diatom assemblages in sediment traps are not only affected by chemical and physical conditions of lakes, but also by their settling rate and resuspension. Many diatoms found in our sediment traps, such as taxa belonging to the Fragilariaceae, are tychoplanktonics and may indicate resuspension of benthic diatoms (Hustedt, 1923). Resuspension can be problematic when subfossil diatoms bias the modern diatom assemblages collected by the traps. However, in shallow lakes, diatoms can have a benthic life-form, and as a consequence, resuspended modern benthic diatoms deposited in the traps would still be related to water chemistry and allow some insights into species–environment relationships. A distinction between dead and living cells by staining or assessing the ratio of valves with and without chloroplasts was not done in this study, but is highly recommended for future studies (Sawai, 2001).
If the diatoms in our trap samples originated mainly from resuspension, then we would expect a significant correlation between diatom flux and water column circulation, with a seasonal homogeneity of taxa. However, this was not the case in either of our study lakes (Table 2), and most lakes showed a high species turnover from May through October. In addition, the seasonal distribution of diatom taxa in Lake Elysée showed very similar patterns in 2002 and 2003 (Fig. 4).
Another uncertainty is that two of the six lakes were only 3 and 4-m deep; thus, only 60–75% of the water column was located above the trap. The entire water column could not be sampled by the trap. Diatom growth on the trap could have biased the diatom assemblages but black tubes without a funnel were used to minimize this effect. The use of sediment traps for the collection of phytoplankton seems to be more promising in deeper lakes when placed below the euphotic zone. However, Köster & Pienitz (2006) used the same type of traps in a 3-m deep temperate lake from New England, and a comparison of plankton and trap samples showed that the shallow water sediment traps provided realistic reflections of the seasonal succession of planktonic diatoms.
Conclusions
Our study revealed that the onset of thermal stratification was later in lakes with later ice break-up. We demonstrated that lake circulation was significantly stronger in colder lakes with longer ice cover. We showed evidence that lake circulation had a strong impact on SiO2, nutrients and SO4 concentrations in boreal lakes, which played a significant role for diatom composition found in the sediment traps. We identified Tabellaria flocculosa as indicator of lake stratification and Fragilaria virescens and Fragilaria pinnata as indicators for lake circulation and elevated Si:TP ratios.
Lake Maxi, the only lake impacted by forest clear-cut, had significantly higher TP, DOC and Chl a concentrations.
Our results contribute to a better paleoclimatological interpretation of diatom records from boreal lakes.
References
Allard, M. & R. Fortier, 1990. The thermal regime of a permafrost body at Mont du Lac des Cygnes, Québec. Canadian Journal of Earth Sciences 27: 694–697.
Anderson, N. J., 2000. Diatoms, temperature and climate change. European Journal of Phycology 35: 307–314.
Battarbee, R. W. & M. J. Kneen, 1982. The use of electronically counted microspheres in absolute diatom analysis. Limnology and Oceanography 27: 184–188.
Birks, H. J. B., 1995. Quantitative Palaeoenvironmental Reconstructions. In Maddy, D. & J. S. Brew (eds), Statistical Modelling of Quaternary Science Data. Quaternary Research Association, Cambridge: 161–254.
Bradbury, J. P., 1988. A climatic-limnologic model of diatom succession for paleolimnological interpretation of varved sediments at Elk Lake, Minnesota. Journal of Paleolimnology 1: 115–131.
Camburn, K. E. & J. C. Charles, 2000. Diatoms of low-alkalinity lakes in the northeastern United States. The Academy of Natural Sciences of Philadelphia. Philadelphia, Philadelphia.
Cameron, N. G., 1995. The representation of diatom communities by fossil assemblages in a small acid lake. Journal of Paleolimnology 14: 185–223.
Carignan, R., P. D’Arcy & S. Lamontagne, 2000. Comparative impacts of fire and forest harvesting on water quality in Boreal Shield lakes. Canadian Journal of Fisheries and Aquatic Sciences 57: 105–117.
Cole, J. J., R. W. Howarth, S. S. Nolan & R. Marino, 1986. Sulfate inhibition of molybdate assimilation by planktonic algae and bacteria: some implications for the aquatic nitrogen cycle. Biogeochemisty 2: 179–196.
Desjardins, R. & R. Monderie, 1999. Forest Alert, National Film Board of Canada, 68 min 3 sec.
Duguay, C. R., T. D. Prowse, B. R. Bonsal, R. D. Brown, M. P. Lacroix & P. Ménard, 2006. Recent trends in Canadian lake ice cover. Hydrological Processes 20(4): 781–801.
Environment Canada, 1994. Manual of Analytical Methods, Vol. 1. Major Ions and Nutrients. Environmental Conservation Service—ECD. Canadian Communications Group, Toronto.
Fallu, M.-A., N. Allaire & R. Pienitz, 2000. Freshwater Diatoms from Northern Québec and Labrador (Canada), Bibliotheca Diatomologica, Vol. 45. J. Cramer, Berlin/Stuttgart.
Happey, C. M., 1970. Some physico-chemical investigations of stratification in Abbot’s Pool, Somerset: the distribution of some dissolved substances. The Journal of Ecology 58(3): 621–634.
Hausmann, S. & R. Pienitz, 2007. Seasonal climate inferences from high-resolution modern diatom data along a climate gradient: a case study. Journal of Paleolimnology 38: 73–96.
Heiri, O. & A. F. Lotter, 2003. 9000 years of chironomid assemblage dynamics in an Alpine lake: long-term trends, sensitivity to disturbance, and resilience of the fauna. Journal of Paleolimnology 30: 273–289.
Hughes, M. K. & C. M. Ammann, 2009. The future of the past—an earth system framework for high resolution paleoclimatology: editorial essay. Climatic Change 94: 247–259.
Hustedt, F., 1923. Süsswasser-Diatomeen Deutschlands, 4th ed. Frankh’sche Verlagsbuchhandung, Stuttgart.
IPCC, 2007. Summary for Policymakers. In Solomon, S., D. Qin, M. Manning, Z. Chen, M. Marquis, K. B. Averyt, M. Tignor & H. L. Miller (eds), Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK and New York.
Jeffrey, S. W. & N. A. Welschmeyer, 1997. Spectrophotometric and fluorimetric equations in common use in oceanography. In Jeffrey, S. W., R. F. C. Mantoura & S. W. Wright (eds), Phytoplankton Pigments in Oceanography: Guidelines to Modern Methods. UNESCO, Paris: 597–621.
Joynt, E. H. & A. P. Wolfe, 2001. Paleoenvironmental inference models from sediment diatom assemblages in Baffin Island lakes (Nunavut, Canada) and reconstruction of summer water temperature. Canadian Journal of Fisheries and Aquatic Sciences 58: 1222–1243.
Kilham, P., S. S. Kilham & R. E. Hecky, 1986. Hypothesized resource relationships among African planktonic diatoms. Limnology and Oceanography 31: 1169–1181.
Korhola, A., S. Sorvari, M. Rautio, P. G. Appleby, J. A. Dearing, Y. Hu, N. Rose, A. Lami & N. G. Cameron, 2002. A multi-proxy analysis of climate impacts on the recent development of subarctic Lake Saanajärvi in Finnish Lapland. Journal of Paleolimnology 28: 59–77.
Köster, D. & R. Pienitz, 2006. Seasonal diatom variability and paleolimnological inferences—a case study. Journal of Paleolimnology 35: 395–416.
Köster, D., R. Pienitz, B. B. Wolfe, S. Barry, D. R. Foster & S. S. Dixit, 2005. Paleolimnological assessment of human-induced impacts on Walden Pond (Massachusetts, USA) using diatoms and stable isotopes. Aquatic Ecosystem Health and Management 8: 117–131.
Krammer, K. & H. Lange-Bertalot, 1986. Bacillariophyceae. 1. Teil: Naviculaceae. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa, Band 2/1. Gustav Fischer Verlag, Stuttgart, New York.
Krammer, K. & H. Lange-Bertalot, 1988. Bacillariophyceae. 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa, Band 2/2. Gustav Fischer Verlag, Jena.
Krammer, K. & H. Lange-Bertalot, 1991a. Bacillariophyceae. 3. Teil: Centrales, Fragilariaceae, Eunotiaceae. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa, Band 2/2. Gustav Fischer Verlag, Jena.
Krammer, K. & H. Lange-Bertalot, 1991b. Bacillariophyceae. 4. Teil: Achnanthaceae, Kritische Ergänzungen zu Navicula (Lineolatae) und Gomphonema, Gesamtliteraturverzeichnis Teil 1-4. In Ettl, H., G. Gärtner, J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa, Band 2/4. Gustav Fischer Verlag, Stuttgart, Jena.
Laird, K., B. Cumming & R. Nordin, 2001. A regional paleolimnological assessment of the impact of clearcutting on lakes from the west coast of Vancouver Island, British Columbia. Canadian Journal of Fisheries and Aquatic Sciences 58: 479–491.
Larsson, U., S. Blomqvist & B. Abrahamsson, 1986. A new sediment trap system. Marine Ecology Progress Series 31: 205–207.
Lotter, A. F. & C. Bigler, 2000. Do diatoms in the Swiss Alps reflect the length of ice-cover? Aquatic Sciences 62: 125–141.
Lotter, A. F., H. J. B. Birks, W. Hofmann & A. Marchetto, 1997. Modern diatom, cladocera, chironomid, and chrysophyte cyst assemblages as quantitative indicators for the reconstruction of past environmental conditions in the Alps. I. Climate. Journal of Paleolimnology 18: 395–420.
Lotter, A. F., R. Pienitz & R. Schmidt, 1999. Diatoms as indicators of environmental change near the arctic and alpine treeline. In: Stoermer E. F. & J. P. Smol (eds), The Diatoms: Applications for the Environmental and Earth Sciences. Cambridge University, Cambridge, New York, Melbourne: 469 pp.
Lund, J. W. G., 1950. Studies on Asterionella. W. Nutrient depletion and the spring maximum. Journal of Ecology 38: 1–5.
Marino, R., R. W. Howarth, J. Shamess & E. E. Prepas, 1990. Molybdenum and sulfate as controls on the abundance of nitrogen-fixing cyanobacteria in saline lakes in Alberta. Limnology and Oceanography 35(2): 245–259.
Nonaka, T., T. Matsunaga & A. Hoyano, 2007. Estimating ice breakup dates on Eurasian lakes using water temperature trends and threshold surface temperatures derived from MODIS data. International Journal of Remote Sensing 28(10): 2163–2179.
Nusch, E. A., 1980. Comparison of different methods for chlorophyll and phaeopigment determination. Archiv für Hydrobiology 14: 14–36.
Pienitz, R. & J. P. Smol, 1993. Diatom assemblages and their relationship to environmental variables in lakes from the boreal forest-tundra ecotone near Yellowknife, Northwest Territories, Canada. Hydrobiologia 269/270: 391–404.
Pienitz, R., J. P. Smol & H. J. B. Birks, 1995. Assessment of freshwater diatoms as quantitative indicators of past climatic change in the Yukon and Northwest Territories, Canada. Journal of Paleolimnology 13: 21–49.
Pienitz R., M. S. V. Douglas & J. P. Smol, 2004. Long-term Environmental Change in Arctic and Antarctic Lakes. Springer, Dordrecht: 562 pp.
Planas, D., M. Desrosiers, S. Groulx, S. Paquet & R. Carignan, 2002. Pelagic and benthic algal responses in eastern Canadian Boreal Shield lakes following harvesting and wildfires. Canadian Journal of Fisheries and Aquatic Sciences 57: 136–145.
Reynolds, C. S., 1993. Scales of disturbance and their role in phytoplankton ecology. Hydrobiologia 249: 157–171.
Salonen, K., L. Arvola & M. Rask, 1984. Autumnal and vernal circulation of small forest lakes in southern Finland. Verhandlungen Internationale Vereinigung für Theoretische und Angewandte Limnologie 22: 103–107.
Sawai, Y., 2001. Distribution of living and dead diatoms in tidal wetlands of northern Japan: relations to taphonomy. Palaeogeography, Palaeoclimatolology and Palaeoecology 173: 125–141.
Schindler, D. W., S. E. Bayley, B. R. Parker, K. G. Beaty, D. R. Cruikshank, E. J. Fee, E. U. Schindler & M. P. Stainton, 1996. The effects of climatic warming on the properties of boreal lakes and streams at the Experimental Lakes Area, Northwestern Ontario. Limnology and Oceanography 41: 1004–1017.
Schmidt, R., S. Wunsam, U. Brosch, J. Fott, A. Lami, H. Löffler, A. Marchetto, H. W. Müller, M. Prazaková & B. Schwaighofer, 1998. Late and post-glacial history of meromictic Längsee (Austria), in respect to climate change and anthropogenic impact. Aquatic Sciences 60: 56–88.
Smol, J. P., 1988. Paleoclimate proxy data from freshwater arctic diatoms. Verhandlungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 23: 837–844.
Smol, J. P., A. P. Wolfe, H. J. Birks, M. S. Douglas, V. J. Jones, A. Korhola, R. Pienitz, K. Rühland, S. Sorvari, D. Antoniades, S. J. Brooks, M.-A. Fallu, M. Hughes, B. E. Keatley, T. E. Laing, N. Michelutti, L. Nazarova, M. Nyman, A. M. Paterson, B. Perren, R. Quinlan, M. Rautio, E. Saulnier-Talbot, S. Siitonen, N. Solovieva & J. Weckström, 2005. Climate-driven regime shifts in the biological communities of arctic lakes. Proceedings of the National Academy of Sciences of the United States of America 102(12): 4397–4402.
Solovieva, N., V. Jones, J. H. B. Birks, P. Appleby & L. Nazarova, 2008. Diatom responses to 20th century climate warming in lakes from the northern Urals, Russia. Palaeogeography, Palaeoclimatology, Palaeoecology 259(2–3): 96–106.
Sorvari, S., A. Korhola & R. Thompson, 2002. Lake diatom response to recent Arctic warming in Finnish Lapland. Global Change Biology 8(2): 171–181.
Stoermer, E. F. & J. P.Smol, 1999. The Diatoms: Applications for the Environmental and Earth Sciences. Cambridge University, Cambridge, New York, Melbourne: 469 pp.
ter Braak, C. J. F., 1986. Canonical Correspondence Analysis: A New Eigenvector. Technique for Multivariate Direct Gradient Analysis. Ecology 67: 1167–1179.
ter Braak, C. J. F. & P. Šmilauer, 1998. CANOCO Reference Manual and User’s Guide to CANOCO for Windows: Software for Canonical Community Ordination (Version 4). Microcomputer Power, Ithaca, NY.
Tilman, D., 1982. Resource Competition and Community Structure. Princeton University Press, Princeton.
Vollenweider, R. A., 1968. Scientific Fundamentals of the Eutrophication of Lakes and Flowing Waters, with Particular Reference to Nitrogen and Phosphorus as Factor in Eutrophication. OECD, Paris.
Wetzel, R. G., 2001. Limnology, 3rd ed. Academic Press, San Diego.
Wick, L., W. O. van der Knaap, J. F. N. Leeuwen & A. F. Lotter, 2003. Holocene vegetation development in the catchment of Sägistalsee (1935 m a.s.l.), a small lake in the Swiss Alps. Journal of Paleolimnology 30: 261–272.
Wunsam, S. & R. Schmidt, 1995. A diatom-phosphorus transfer function for Alpine and prealpine lakes. Memorie dell’Istituto Italiano di Idrobiologia 53: 85–99.
Acknowledgments
This research was supported by a Leopoldina (# BMBF-LPD 9901) stipend from the Academy of Natural Sciences (Federal Ministry of Education and Research, Germany), and a Swiss National Science Foundation (SNSF) postdoctoral fellowship to S. Hausmann, and a Natural Sciences and Engineering Research Council (NSERC) of Canada grant to R. Pienitz. We are grateful to Centre d’études nordiques of Université Laval for logistic support and thank C. Zimmermann for field and laboratory assistance, and D. Muir and X. Wang for water chemistry analyses. This work could only be realized with the field assistance of T. Menninger, L. Laperrière, I. Laurion, D. Köster, K. Roberge, É. Saulnier-Talbot and M. Touazi. We would like to thank M. Rautio, T. Menninger, R. Keveren and J. Dixon for feedback on the manuscript. The manuscript would not exist in its present form without the valuable comments of J. Black. In addition, we would like to thank the two reviewers for their constructive comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: J. Saros
Rights and permissions
About this article
Cite this article
Hausmann, S., Pienitz, R. Seasonal water chemistry and diatom changes in six boreal lakes of the Laurentian Mountains (Québec, Canada): impacts of climate and timber harvesting. Hydrobiologia 635, 1–14 (2009). https://doi.org/10.1007/s10750-009-9855-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-009-9855-0