Abstract
Subterranean or groundwater estuaries occur in porous and cavernous substrates where groundwater abuts the ocean. Like surface estuaries, they are strongly stratified, temporally and hydrochemically heterogeneous environments that support complex hydrogeochemical and biological processes and ecological communities. Here, we contend that groundwater estuaries also occur where groundwater flow approaches salt lakes and provide evidence in the context of groundwater (valley or phreatic) calcretes in palaeovalleys of the arid western plateau of Australia. The calcrete groundwater estuaries display marked and complex physico-chemical gradients along, across and through the groundwater flow path. From the first principles and the density differences between water bodies, we may expect the form and dynamics of the saltwater front to mimic that of marine estuaries but with the dynamic and temporal response to changing hydrology heavily dampened, and driven by the episodic groundwater recharge and lake filling typical of arid regions. The calcrete aquifers support diverse biological communities of obligate groundwater animals, largely endemic to a given calcrete body. These communities comprise both macro and microinvertebrates, predominantly a suite of crustacean higher taxa, and a great diversity of diving beetles (Dytiscidae) isolated in the calcrete aquifers between ca. 5 and 8 million years ago.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
…salt lakes are more akin to small-scale, locally controlled, and transient analogues of the ultimate salt lake, the world’s ocean (Torgersen et al., 1986).
Typical [river] estuaries are semi-enclosed coastal bodies of water with free connection with the open sea and within which seawater mixes with freshwater. Subterranean estuaries also form in groundwater where it enters the sea. In this article, we will develop the concept of groundwater estuaries associated with salt lakes and, as with their coastal counterparts, those on the western plateau of Australia support a rich subterranean biodiversity, suggesting, by analogy, that important biological processes may occur associated with the hydrogeochemical evolution of the groundwater. Williams (1986) defended the inclusion on both fresh and saline waters under the umbrella of limnology; here, we broaden this view to encompass also the study of groundwaters, both fresh and saline.
Nature of estuaries
River estuaries comprise a region of mixing between seawater and freshwater, and a tide is a necessary component to maintain a dynamic relationship between the two waters. Owing to the influx of freshwater, salinity gradients form along the length of the estuary, the form of which are determined by its hydrological characteristics. Vertical gradients also form owing to the density differences between the mixing waters, with the seawater forming a wedge beneath the freshwater (Moore, 1966). Changing hydrological heads on semi-diurnal and seasonal timescales occur as a result of oceanic tides and it changes according to the flux of the river. These properties interact with the density structure of the estuary to form the complex spatial and temporal gradients in physico-chemical conditions characteristic of estuaries, and which do much to influence their biological attributes. The forms of these gradients change with location in the estuary at a given time, and with time at a given location, depending on the state of the hydrological flux within the estuary.
Where groundwater abuts the oceanic coast, subterranean estuaries that have complexity similar to that of surface estuaries may be formed, but, owing to the relatively low flow rates in groundwater systems, they characteristically have a much longer time base and concomitantly a slower response rate, such as the changes in the vector and dynamics of salt water flux in response to seasonal groundwater recharge (Michael et al., 2005). The existence of subterranean or groundwater estuaries in porous aquifers was proposed by Moore (1999), who recognised the mixing zone between seawater and groundwater to be a region of considerable chemical reactivity. This work received a wider audience with the ‘iron curtain’ allusion (Charette, 2001), which described the precipitation of ferrous iron from groundwater at the groundwater–seawater interface. The resulting accumulation of iron oxides onto subsurface sands serves to act as strong adsorbers and concentrators of many dissolved chemical species (Charette & Sholkovitz, 2002; Testa et al., 2002). This is consistent with the broad recognition of significant biological interaction with the hydrogeochemical evolution along groundwater flowpaths (Humphreys, 2008b).
Groundwater estuaries, however, have been studied for much longer, especially in a biological context, in substrates with open conduit flow. The latter is characteristic of some regions of volcanic or limestone bedrock, respectively, typified by lava tubes in Lanzarote, Canary Islands, and limestone karst of carbonate platforms, such as those on the Bahamas Banks, and on Quintana Roo on the Yucatan Peninsula, Mexico (Iliffe, 2000). In these situations, the groundwater estuaries have been studied under the guise of anchialine (variously anchihaline) systems. Anchialine habitats consist of bodies of haline waters, usually with a restricted exposure to open air, always with more or less extensive subterranean connections to the sea, and showing noticeable marine as well as terrestrial influences (Stock et al., 1986). Anchialine waters are typically stratified, with freshwater or brackish water overlying seawater and separated by a mixing zone. These waters typically have complex vertical profiles, in which the redox values drop sharply across the chemocline, have very low concentrations of oxygen at depth, layers of hydrogen sulphide, and a cascade of nitrogen species (Humphreys, 2006; Seymour et al., 2007). Together, these conditions support a complex aerobic and anaerobic microbiological community providing some chemoautotrophic energy fixation (Pohlman et al., 1997, 2000; Humphreys, 1999a; Seymour et al., 2007). Anchialine systems are mostly found in tropical and subtropical regions where they support significant biodiversity, with taxa that are often endemic at a higher taxonomic level (Sket, 1996; Iliffe, 2000). While such anchialine systems are mostly known from the Northern Hemisphere, Bundera Sinkhole, in northwestern Australia (Humphreys, 1999a), fulfils the complete Tethyan distribution of this type of ecosystem (Jaume et al., 2001; Page et al., 2008). These various studies are leading to the increasing recognition that significant hydrogeochemical and biological processes are associated with the salt front in groundwater estuaries. While the role of groundwater in the evolution of salt lakes has not long been appreciated (Hammer, 1984; Torgersen et al., 1986), there is now a considerable body of work on the groundwater mixing zone associated with salt lakes (e.g. Lyons et al., 1995; English et al., 2001).
Phreatic calcretes and salt lake groundwater estuaries
We take as our model, the salt lakes associated with the palaeodrainage systems of the Australian western plateau (Fig. 1), including the Ngalia Basin, Northern Territory (Watts & Humphreys, 2006), and the Yilgarn, Western Australia, north of 30° S (Humphreys, 2001). Here, we address salt lake groundwater estuaries, particularly those associated with groundwater calcretes (variously termed valley or phreatic calcrete, hereafter termed calcrete). These calcretes are carbonate deposits whose formation is directly associated with groundwater, rather than with soil development (pedogenic calcrete). Groundwater calcretes sometimes develop typical karst features (Sanders, 1974; Barnett & Commander, 1985) with sinkholes serving as major recharge zones for the aquifers. Well-developed karst has interconnected phreatic conduits providing attributes that make calcrete aquifers suitable both as potential habitat for subterranean aquatic fauna (hereafter termed stygofauna) and for the exploitation of groundwater.
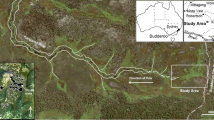
Distribution of groundwater calcretes (shaded) in the palaeovalleys of the northern Yilgarn region, Western Australia. From Hydrogeological Map of Western Australia 1989, 1:2,500,000, Geological Survey of Western Australia, Perth
These carbonate deposits, generally in the order of 10 m, but up to 30 m thick, form near the water table of shallow aquifers in arid lands as a result of concentration processes by near-surface evaporation (Jacobson and Arakel, 1986). They are especially important in the Australian context as they form in arid climates (annual rainfall <200 mm) with high potential evaporation (>3,000 mm per year: Mann and Horwitz, 1979), such as developed widely in Australia during the Tertiary (Byrne et al., 2008). They occur where the movement of the groundwater is slow and where the rainfall is episodic resulting in substantial fluctuations in the water table. In this respect, Simon (2000) has shown that regular changes in water level may be important in maintaining the trophic dynamics of some karst aquifers.
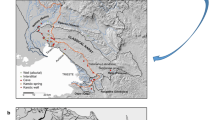
The Hinkler Well calcrete—the type section for the hydrogeological model for groundwater evolution leading to the formation of groundwater calcretes—is a narrow linear carbonate deposit draining to Lake Way in the Yilgarn region, Western Australia (Fig. 2; Mann & Deutscher, 1978; Mann & Horwitz, 1979). From these studies, it was recognised that there is a succession of chemical precipitates associated with increasing salinity, as the lake is approached (Fig. 3). Carbonate is deposited in the mid-line of the drainage with associated silcrete and celestite. Downstream aeolian deposits of gypsum occur with sepiolite and aragonite on the shores of the salt lake, on the bed of which halite is found. On the upstream of the ‘chemical delta’, the calcrete is underlain in places by ferricrete and laterite, which is thought to have formed during the Miocene in more humid times (Mann & Horwitz, 1979; English et al., 2001). The main channel calcretes are formed at the downstream end of an individual hydrochemical system and immediately upstream of an evaporation outflow area forming a salt lake.
Image of the region around Lake Way salt lake, Northern Goldfields, Western Australia, showing the juxtaposition of the major calcrete aquifers (areas enclosed by pale lines) formed from the several palaeodrainages entering the salt lake. Several large open cut mine pits are in the area, the largest visible north of the Lake Violet calcrete. The palaeovalley draining from the northwest towards Lake Violet leads to Bubble Well and Paroo (see text). The calcretes each support an endemic fauna (Table 1). LV, Lake Violet calcrete; LU, Uramurdah Lake calcrete, HW, Hinkler Well calcrete. The image depicts an area about 40 by 60 km. The eastern end of the Hinkler Well calcrete is at position 26°41′ S; 120°13′ E. Base image derived from World Wind 1.3.3 (NASA)
Change in salinity along the length of the Hinkler Well calcrete, from Dawsons Well (left) to Lake Way (right), the type section of calcrete for the study of the hydrogeochemical evolution of groundwater on the approach to salt lakes. Data are derived from Mann & Deutscher (1978)
Morgan (1993) considered that a separate geochemical system is associated with the formation of each salt lake along a palaeoriver, with a well-defined change in the common ion ratio developing with increasing salinity. This observed increase in salinity and relative chloride/sulphate content has both spatial and temporal components because the changes occur between widely separate intake and outflow locations, and thus the changes occur well separated in time. This hydrochemical trend commences at the headwaters of each recharge system and completes its cycle at the evaporation outlet shown by the lower boundary of the calcrete with salt lake. The low salinity waters near the intake (500–2,000 mg l−1 total dissolved solids) are alkaline and rich in bicarbonates. Groundwater interacts with sediments along its flow path and as a result becomes less alkaline, increases markedly in salinity and changes to a chloride–sulphate type. The calcretes pass downstream into dolomites, then into gypsum and halites in waters from 20,000 to over 200,000 mg l−1 TDS (Morgan, 1993). Several similar hydrochemical cycles may occur along a single palaeodrainage system (Humphreys, 2001: Fig. 2). We would argue that a series of independent systems are involved on the larger lakes, such as Lake Way, namely Uramurdah Lake, Hinkler Well and Lake Violet inflows, each contributing a separate body of calcrete (Fig. 2) and each supporting a unique stygofauna community (Table 1). In other areas, calcrete formation has been associated with palaeolakes, for example Lake Lewis, Northern Territory, a salt lake more than 1,000 km from the nearest coast and fed by groundwater discharge, now has a 10-km wide aureole of phreatic and vadose calcrete (English et al., 2001).
Groundwater transports allochtonous chemical compounds (Wanty & Schoen, 1991) to the salt lake (Gray, 2001). By analogy with coastal groundwater estuaries in both anchialine and porous rock aquifers, the salt lake groundwater estuary may be expected to form a particularly chemically dynamic region owing to the interaction of water masses of differing chemical composition that will variously liberate and precipitate the dissolved and solid phases. These chemical reactions, which will be enhanced by the strong redox gradients associated with the salinity stratification (Watts & Humphreys, 2004, 2006; Humphreys, 2008b) (Fig. 4), will free both inorganic and organic compounds, with degradation of the latter likely to occur through various aerobic and anaerobic pathways in the groundwater (Pérez del Villar et al., 2004), a process likely enhanced or facilitated by biotic interactions (Castanier et al., 1999, 2000; Humphreys, 2008b). The complex physico-chemical stratification found in anchialine systems (Humphreys, 1999a) are seen also in salt lakes themselves, as for example in Waldsea Lake, Saskatchewan (Lawrence et al., 1978). Similarly, complex stratification of the water column with sharp redox changes associated with the chemocline may occur in calcrete aquifers (Fig. 4) (Watts & Humphreys, 2000, 2003, 2006). Groundwater estuaries and the freshwater–saltwater interface are regions of substantial biogeochemical activity (Moore, 1999; Pohlman et al., 2000; Charette, 2002; Testa et al., 2002; Seymour et al., 2007) because microbial life concentrates at physical and chemical interfaces where energy and nutrients fluctuate most dramatically (Ash et al., 2002), a theme developed further elsewhere (Humphreys, 2008b). Combined with the high sulphate and nitrate levels (Watts & Humphreys, 2003), these factors warrant research into the potential contribution of chemoautotrophy in the trophic dynamics of the calcrete aquifers.
Depth (m) profile of salinity (TDS g l−1), pH, dissolved oxygen (DO, mg l−1), and redox (ORP, mV) in a Main Road bore near Nyung Well, Challa Pastoral Station, Murchison District, Western Australia (27°59′ S, 118°31′ E). Note the markedly negative redox values at depth. After Watts & Humphreys (2006)
Calcrete is commonly thin, spreading into delta-like shapes (Arakel, 1986), even forming ‘chemical deltas’ (Mann & Horwitz, 1979). We contend that such calcretes function as groundwater estuaries that support complex ecosystems containing macro and micro-invertebrate communities. The characteristics of these systems have been discussed in terms of the hydrogeological evolution and their fauna, (Humphreys, 1999b, 2001, 2008b), and the latter is further developed below. There is also a diverse microbiological community in anchialine systems (Seymour et al., 2007), and it is expected in the carbonate precipitating environments (Castanier et al., 1999, 2000) of the calcrete aquifers but which is yet to be formally characterized.
Hydrodynamics of calcrete aquifers
In coastal and groundwater estuaries, tides, density differentials and changes in piezometric head are important to maintain the dynamical relationships between the two merging bodies of water. We have no direct data for the presence of tides in salt lake groundwater estuaries; however, several published accounts and first principles support the view that there are tides that will affect salt lake estuaries but the time base is orders of magnitude longer than that of marine tides, commensurate with the low velocity of groundwater flow. Where salt lakes represent groundwater base level, as in the palaeovalleys of the Yilgarn region (Morgan, 1993), local recharge, lake-fill events, and the slopping of shallow lake waters from sustained wind directions (or anthropogenic drawdown and recharge) can cause surface water to invade groundwater and terminate or reverse the previous sequence of chemical precipitation (Torgersen, 1984). Further, changes in the piezometric surface resulting from groundwater recharge and discharge, variously on seasonal (Michael et al., 2005) and Milankovic cycle time scales (Hatton, 2001), will also maintain the hydrological dynamics of the system.
Models of palaeochannel systems and their associate calcretes tend to be longitudinal (Morgan, 1993), ignoring the much steeper vertical gradients that occur within the groundwater estuary (Humphreys, 1999a, 2006; Watts & Humphreys, 2000, 2003, 2006; Seymour et al., 2007). As the biotic component of the salt lake estuaries are expected to interact with the chemoclines (Castanier et al., 1999, 2000) within the groundwater (Humphreys et al., 1995; Humphreys, 2008b), the study of both may be necessary to understand the depositional models of salt lake systems, as well as the nature and persistence of the biota.
In surface estuaries, the movement of a saline wedge is positively related with the boundary between saline and fresh water in the subsurface aquifer system (Tokuoka et al., 2000). Flooding of salt lakes also is likely to affect conditions in salt lake groundwater estuaries. Although groundwater calcretes are associated with slow moving groundwater, the episodic rainfall, characteristic of the Australian arid zone, causes groundwater levels to fluctuate widely (Jacobson & Arakel, 1986; Morgan, 1993) with associated changes in salinity. For example, in the Lake Austin calcrete, two monitoring bores had annual salinity variation between 5,000 to 9,000 mg l−1 and 5,000 to 22,000 mg l−1, respectively (Watts & Humphreys, 2000). Such large changes in local salinity can be exacerbated by the amplified movement of the freshwater–saltwater interface, which moves vertically by a factor much greater than the change in the level of the groundwater surface owing to the density differences between the water bodies: in freshwater over seawater conditions, this factor is of the order of 40 times, an effect that is explored in the coastal contexts by the application of the Ghyben–Herzberg principle (see Chow, 1964), and seasonally by Michael et al. (2005). Calcrete aquifers have highly variable hydraulic characteristics reflecting the heterogeneous mineralogical and textual characteristics (Carlisle, 1980; Arakel, 1986). The net effect is that the groundwater salinities may vary spatially on quite fine scales (Fig. 5) (Watts & Humphreys, 2004; Humphreys, 2006).
Series of salinity profiles (as specific conductance) on the same date (2 April 2005) showing the wide range of profiles across a small section (~2.3 km2) of Sturt Meadows calcrete (~43 km2), Western Australia, about 3 km north of the Lake Raeside salt lake. The location of the grid of bores and their arrangement is shown in Allford et al. (2008), and the data were acquired as in Watts & Humphreys (2006)
By definition, an estuary is where the freshwater and the saltwater mix and so, functionally, it will shut down if starved of freshwater and potentially challenges the biotic diversity of the aquifer. The ultimate test of the concept of salt lake groundwater estuary would be to deprive it of groundwater flow.
Fauna and endemicity
We have examined, to varying degrees, 107 discrete calcrete bodies found in 15 palaeodrainage systems on the western plateau of Australia, predominantly in the northern Yilgarn, Western Australia, and the Ngalia Basin, Northern Territory (Watts & Humphreys, 1999, 2000, 2001, 2003, 2004, 2006, in press; Taiti & Humphreys, 2001; Cooper et al., 2002, 2007, 2008; Allford et al., 2008; Guzik et al., 2008; Leys & Watts, 2008; Leys et al., 2003; Cho, 2005: Cho et al., 2005, 2006a, b; Humphreys, 2001, 2006, 2008a). The stygofauna (subterranean aquatic animals) found in the calcrete aquifers comprises, almost exclusively, obligate subterranean species (stygobites) (Humphreys, 2008a). These exhibit many of the convergent characteristics (stygomorphies) typical of obligate subterranean animals, namely reduced or absent eyes, lack of pigment, fragility, translucence, and loss or reduction of wings (Langecker, 2000). As is typical of such faunas, it largely comprises a variety of crustacean higher taxa, but includes also the world’s greatest diversity of stygal diving beetles (Dytiscidae) (Watts & Humphreys, 2000, 2001, 2003, 2004, 2006, in press; Balke et al., 2004), annelids, especially the Gondwanan family Phreodrilidae (Pinder & Brinkhurst, 1997; Pinder et al., 2006), hydrobiid gastropods and mites (Humphreys, 2008a), Chiltoniidae, Melitidae, Bogidiellidae and Paramelitidae (Amphipoda), and Bathynellidae and Parabathynellidae (Bathynellacea) (Guzik et al., 2008). Altogether, the fauna includes species from 36 families of invertebrates (Table 2), and aspects of the fauna of the western plateau are described in a number of publications (Poore & Humphreys, 1998, 2003; Watts & Humphreys, 1999, 2000, 2001, 2003, 2004, 2006, in press; Taiti & Humphreys, 2001; Karanovic and Marmonier, 2002; Balke et al., 2004; Karanovic, 2004; Cho, 2005: Cho et al., 2005, 2006a, b). In addition, some elements of the evolution of the subterranean fauna have been addressed using both morphological and molecular methods (Taiti & Humphreys, 2001; Cooper et al., 2002, 2007, 2008; Leys et al., 2003; Guzik et al., 2008; Leys & Watts, 2008). The diverse stygofauna of the Pilbara region, on the Western Shield north of the Yilgarn, is summarised by Eberhard et al. (2005), but published works to date (Karanovic & Marmonier, 2002, 2003; Karanovic, 2003, 2005, 2007; Karanovic, 2006; Finston et al., 2004, 2007; Reeves et al., 2007) do not specifically address the calcrete-related issues discussed herein, except for some (Poore & Humphreys, 1998; Humphreys, 1999b, 2001; Eberhard et al., 2005: review).
Only one taxon associated with calcretes in salt lake groundwater estuaries has clear affinity with salt lakes themselves, namely oniscidean isopods of the genus Haloniscus. The genus is represented in salt lakes right across the southern Australia (Williams, 1983) by H. searli Chilton, a fully eyed, epigean species, which is one of the most tolerant halobionts known (Bayly & Ellis, 1969). This distribution is remarkable owing to the isolated nature of salt lakes, and the lack of an aerial phase or resistant eggs in the species. Immediately to the north, numerous eyeless stygobiont Haloniscus occur in calcrete aquifers, each species being restricted to a single calcrete aquifer and some calcretes having sympatric species (Taiti & Humphreys, 2001; Cooper et al., 2008). In contrast, the calcretes support a very diverse fauna of Bathynellacea, typically an interstitial taxon of freshwaters (Schminke, 1981) with a Pangaean distribution (Schminke, 1974). In some calcrete aquifers, notably around Lake Way, very large species (6.3 mm long) occur that swim within the water column in the water of marine salinity (Table 1; Fig. 6) (Watts & Humphreys, 2000; Humphreys, 2006), and which resemble Schminke’s (1973) hypothetical parabathynellid (Cho, 2005). Some calcretes contain a mix of near-marine lineages (e.g., Cyclopidae, Halicyclops, the genera Nitocrella and Parapseudoleptomesochra of the marine family Ameiridae: Harpacticoida) (Karanovic, 2004), and chiltoniid and melitid amphipods, alongside ancient freshwater lineages (Parastenocaris, Parastenocarididae and crangonyctoid amphipods) (Karanovic, 2004; Cooper et al., 2007). The great diversity of stygal diving beetles (Dytiscidae) is treated below.
Depth profile through the water column in a mineral exploration bore (designated bore 267) at the Lake Urumurdah calcrete aquifer. Salinity as TDS (g l−1), temperature (°C) and dissolved oxygen (DO mg l−1) (after Humphreys, 2006). This site supports stygal oniscideans, bathynellaceans, harpacticoid copepods, amphipods and dytiscid diving beetles
Age of systems
The ‘Western Shield’ (Hocking et al., 1987) of Australia, which comprises the Pilbara and Yilgarn cratons and the related orogens, lies at the western rim of the western plateau of Australia. It ranks amongst the oldest non-marine landmasses on Earth—although the erosion surface means the landscape is younger (Vasconcelos et al., 2008)—and it supports many of the higher taxa considered to be ancient freshwater lineages, such as bathynellaceans, tainisopidean and phreatoicidean isopods, crangonyctoid amphipods and candonine ostracods (Bradbury, 1999; Wilson & Johnson, 1999; Humphreys, 2001a; Wilson, 2001; Karanovic, 2003). The calcrete estuaries discussed encompass fresh to hypersaline waters and many of the lineages occur through a wide range of salinity, as shown, for example by the fauna found in Lake Uramurdah calcrete adjacent to the Lake Way (Table 1; Fig. 6) (Watts & Humphreys, 2000; Taiti & Humphreys, 2001) and sites in the Carey palaeodrainage system progressively upstream at Bubble Well (5,363 mg l−1, 26°34′ S, 120°02′ E;) to Paroo (530–1,380 mg l−1, 26°24′ S, 119°46′ E; Watts & Humphreys, 2000, in press) (Fig. 2). The occurrence of ancient lineages typical of salt lakes, for example oniscidean isopods of the genus Haloniscus (Table 1; Fig. 6) (Taiti and Humphreys, 2001; Cooper et al., 2008) locally endemic in shallow aquifers within the palaeovalleys of the western plateau, suggests that such saline conditions, independent of evidence derived from salt lakes (De Deckker, 1983), have a long history in the Australian landscape, one amenable to investigation using molecular phylogenies, for example Cooper et al. (2008).
Athalassic (‘inland’) waters are considered impermanent (Bayly, 1967) and unstable, subject to rapid changes in physico-chemical conditions. In contrast, groundwater systems are highly stable and of extremely long duration. A variety of evidence supports the long-term persistence of the calcrete aquifers. The calcretes are deposited from groundwater, and their position in the landscape suggests that they are a Tertiary phenomenon (Morgan, 1993). The prolonged hydrogeochemical evolution of groundwater leading to the deposition of calcrete en route to salt lakes is well documented (e.g. Hinkler Well: Mann & Deutscher, 1978; Mann & Horwitz, 1979, and Lake Lewis: Arakel, 1986; English et al., 2001). The groundwater systems in semi-arid Australia are in a state of net discharge owing to global climatic changes in the order of 103–105 years (Hatton, 2001). Groundwater residence time near the playa lakes is often considerably in excess of 80 ka and consequently, reflected by the dominance of sulphate and chloride (Jacobson & Wischusen, 2001) with samples from groundwater calcretes having Na > Mg > Ca > K and Cl > SO4 > HCO3 > CO3, as typical of salt lakes (Williams, 1984). Data from Lake Lewis, Northern Territory, concurs with information from elsewhere in arid and semi-arid inland Australia that bears witness to widespread oscillating climatic and fluctuating hydrologic conditions after the last interglacial (English et al., 2001; review in Hesse et al., 2004). Despite these long temporal flow paths, these groundwaters bordering the playas contain a rich stygofauna (Humphreys, 2008a). The firmest evidence for the longevity of the calcrete systems generally comes from studies of the distribution of the numerous obligate stygal lineages inhabiting the calcrete aquifers, and from the molecular phylogeny of the diving beetles (Dytiscidae) (Cooper et al., 2002; Leys et al., 2003), and potentially from the molecular phylogenies of other stygal lineages inhabiting the calcretes (Cooper et al., 2007, 2008; Guzik et al., 2008; Leys & Watts, 2008).
The higher taxa examined to date show that each calcrete contains a unique assemblage of species even when separated by short distances (Table 3). In the Haloniscus (Oniscidea) lineage, for example each species is restricted to a single calcrete but several species may occur in a single calcrete (Table 1: Taiti & Humphreys, 2001; Cooper et al., 2008). The calcretes also support more than 100 species of subterranean diving beetles, and each species is restricted to a single calcrete containing up to four sympatric species forming a size series (Leys et al., 2003; Leys & Watts, 2008). In 13 cases, the sympatric species are each other’s closest relatives (sister species) and they provide a firm base for the timing of the speciation events (Leys et al., 2003; Leys & Watts, 2008; our unpublished data). This molecular evidence suggests that the beetles are speciated between eight and five million years ago, and, because stygal animals are obligate groundwater dwellers, this provides a minimum age for the permanence of calcrete aquifers. The data seem to have sufficient resolution to detect the southward onset of increasing aridity (Leys et al., 2003). Even lineages typical of small interstitial voids, such a bathynellaceans, seemingly are restricted to single calcrete occurrences (Guzik et al., 2008).
Salinity and fauna distribution
Anchialine systems are mixohaline with salinities between freshwater and seawater, whereas calcrete estuaries are athalassic with salinities ranging from freshwater to >100,000 mg l−1 TDS (Fig. 6). The salt in Australian inland waters is of marine origin, presumably as aerosols (McArthur et al., 1989), and so the major ion composition of young groundwater may map that of marine waters. Salinity itself, however, defined, even of quite major extent, is not a prime determinant of the distribution of many species in marine estuaries (Wolff, 1973) or in salt lakes, at least in Australia (Williams et al., 1990). Bayly (1967) considered that salinity changes might be more important than absolute salinity per se, although species dominance has been reported to increase with the rising salinity (De Los Rios & Crespo, 2004). Therefore, the wide range of groundwater salinities (Figs. 3–6) (Watts & Humphreys, 2000, 2003, 2006, in press) occupied by the diverse stygofauna (Table 2) in the calcrete aquifers is not unexpected. In contrast, however, a large proportion (72%) of the potentially available fauna seems to be excluded from salt lake waters by even moderate salinities (23,000 mg l−1). In an examination of the fauna of 79 (3 afaunate) lakes in western Victoria, Australia, 41 of the 147 species recorded (27.9%) were never found in waters exceeding 23,000 mg l−1 (26% of 62 species of insects, 42% of 19 species of copepods, and 33% of 24 species of ostracods: Williams et al., 1990). While there was a negative relationship between salinity and species richness, there was only weak correlation at intermediate salinities—for salinities in the range of 3,000–100,000 mg l−1, species richness was not markedly lower than that found in the freshwater lakes—indicating that salinity is not the only, or not the most important, determinant of the occurrence of a particular species in a lake (Williams et al., 1990).
Whereas divalent cations and bicarbonate tend to be dominant in the standard composition of freshwater, Na and Cl tend to dominate in Australian inland waters, including salt lakes (Williams, 1984). The history of the waterbody is relevant (Bayly, 1967), as the present ionic ratio will depend on whether the present salinity was derived by a process of dilution or of evaporation. Pora (1969) considered that the ratios between various ions in brackish water, which he termed the rhopic factor, might be the determining factor in the species distribution in brackish water even when salinity is comparable. The relative amounts of Ca2+, Mg2+, SO42− and HCO3− in solution determines how mineral precipitation causes branch points in solute evolutionary pathways in evaporative systems (Hardie & Eugster, 1970; Eugster & Jones, 1979). This focus has been applied to groundwater/salt lake research to great effect, particularly using ostracods as palaeoclimate indicators (Radke, 2000; Radke et al., 2003). It is notable that candonine ostracods are mostly absent in the more saline waters of the Yilgarn, but extraordinarily diverse in the freshwater calcretes of the Pilbara region (Humphreys, 2001, 2008a; Karanovic, 2003; Karanovic & Marmonier, 2003; Reeves et al., 2007).
Williams et al. (1990) argued that once a salt lake species solves the physiological problem of osmoregulatory stress, it is able to occupy a wide salinity range, and that chance [stochasticity] is probably an important factor in the colonization of inland saline waters from where biological interactions—prior occupancy (competition), predation and parasitism—will determine their persistence. Dominant factors of inland salt lakes are the short-term (episodic rainfall) and long-term changes (Milankovic cycles) that determine the persistence of suitable habitat. However, as we argued above, and discussed elsewhere (Humphreys, 2000a) groundwater estuaries, unlike the ephemeral waters at the surface, have had a long-term permanence measured in millions of years. As a result, biological interactions will have been finely honed by the prolonged and enforced joint occupancy of obligate stygal lineages within the calcretes, an ecosystem especially amenable to study owing to the simplified community structure typical of subterranean ecosystems (Gibert & Deharveng, 2002).
Despite the evidence for the long persistence of the calcrete aquifers, at a given location, there is evidence of considerable variability, both in respect of the physicochemical conditions referred to above, and in the numbers and diversity of the stygofauna (Allford et al., 2008). Physicochemical profiles can be similar in bores that are 480 m apart (Taiti & Humphreys, 2001), or distinctly different between similar bores (Watts & Humphreys, 2006) separated by only 30 m, including differences in the stygofauna (Watts & Humphreys, 2004). Similarly, the bore-monitoring data show marked temporal changes in the water level and in the salinity of calcrete aquifers (e.g. Dames & Moore, 1984; Barnett & Commander, 1985; Watts & Humphreys, 2000). Such marked temporal differences within an ecosystem that is perceived to be constant, suggest that a fruitful line of research may be to link such changes with the tidal changes, proposed herein, that may result from the episodic recharge of the aquifers and affect the dynamics of the groundwater estuaries.
Functional role
By analogy with surface ecosystems, the invertebrates have numerous potential functional roles in groundwater systems, but experimental evidence is still largely lacking (see Boulton, 2000: Table II; Humphreys, 2002; Hancock et al., 2005; Boulton et al., 2008). Of particular relevance, here is the role of stygofauna—suggested also for protozoa (Haack & Bekins, 2000)—in the maintenance of voids (Danielopol, 1989), processing organic carbon (Fenwick et al., 2004), and the alteration of redox gradients through their metabolic intakes and products. Since microorganisms can significantly affect groundwater quality by controlling mineral solubility (Castanier et al., 1999, 2000) and surface reactivity, as discussed by Humphreys (2008b), the presence of stygofauna within calcretes may be a significant factor in these processes. However, while it is recognised that micro-organisms can work as consortia (Haack & Bekins, 2000), it has yet to be recognized whether stygofauna interact with biofilms, for example, by grazing (Simon et al., 2003), serving to promote or to restrict their activity and the community diversity in groundwaters (Humphreys, 2000b), issues discussed recently by Boulton et al. (2008) and Humphreys (2008b).
Conclusions
The hydrogeochemical evolution of the groundwater flowing into salt lakes on the Australian western plateau results in the deposition of carbonates to form phreatic calcretes. The large void space, partly through karstification, forms suitable habitat for subterranean invertebrates. These calcretes occur in a groundwater zone with steep biogeochemical gradients, analogous to those found in surface estuaries and coastal groundwater estuaries (anchialine systems). These salt lake groundwater estuaries have complex gradients in their physico-chemical properties, which suggest that a cascade of microbiological communities may be found there, as found in other complex stratified systems such as lakes and anchialine systems.
The calcretes are effectively isolated from each other in respect of stygofauna, and each calcrete, even those separated by only a few hundred metres, contain separate species. After evolving stygobitic lifestyles, the lineages are obligate dwellers in the groundwater and are unable to disperse. As such, they represent a sample of the surface fauna present at the time they became isolated underground, and the fauna is still present in the same relative position in the landscape, whereas the surface relatives may have been displaced by the changing climate. In this respect, they truly are ‘living fossils’ and any information that can be derived from these faunas, for example from molecular phylogenies, can capture the time and place of that isolation event.
Salt lake researchers often use the extraordinary array of physical, chemical and biological diversity of salt lakes as a window through which to look at the past, either the immediate past with limnological studies, or the extended past using the methods of palaeolimnology to extend studies beyond individual lifetimes (Bowler, 1981). By contrast, studies of groundwater estuaries combine information from hydrogeochemistry and that of extant communities to determine the origin of faunas in place and time in a manner with potential to provide very long-term palaeoenvironmental information.
References
Allford, A., S. J. B. Cooper, W. F. Humphreys & A. D. Austin, 2008. The ecology and distribution of groundwater fauna in a limestone aquifer: does sampling alter the story? Invertebrate Systematics 22: 127–138.
Arakel, A. V., 1986. Evolution of calcrete in palaeodrainages of the Lake Napperby area, Central Australia. Palaeogeography, Palaeoclimatology, Palaeoecology 54: 283–303.
Ash, C., B. Hanson & C. Norman, 2002. Earth, air, fire, and water. Science 296: 1055.
Balke, M., C. H. S. Watts, S. J. B. Cooper, W. F. Humphreys & A. P. Vogler, 2004. A highly modified stygobitic diving beetle of the genus Copelatus (Coleoptera, Dytiscidae): taxonomy and cladistic analysis based on mtDNA sequences. Systematic Entomology 29: 59–67.
Barnett, J. C. & D. P. Commander, 1985. Hydrogeology of the Western Fortescue Valley, Pilbara Region, Western Australia. Geological Survey Record 1986/8. Western Australia Geological Survey, Perth.
Bayly, I. A. E., 1967. The general biological classification of aquatic environments with special reference to those of Australia. In Weatherley, A. H. (ed.), Australian Inland Waters and Their Fauna: Eleven Studies. ANU Press, Canberra: 78–104.
Bayly, I. A. E. & P. Ellis, 1969. Haloniscus searlei Chilton: an aquatic “terrestrial” isopod with remarkable powers of osmotic regulation. Comparative Biochemistry and Physiology 31: 523–528.
Boulton, A. J., 2000. River ecosystem health down under: assessing ecological condition in riverine groundwater zones in Australia. Ecosystem Health 6: 108–118.
Boulton, A. J., G. D. Fenwick, P. J. Hancock & M. S. Harvey, 2008. Biodiversity, functional roles and ecosystem services of groundwater invertebrates. Invertebrate Systematics 22: 103–116.
Bowler, J. M., 1981. Australian salt lakes a palaeohydrologic approach. Hydrobiologia 82: 431–444.
Bradbury, J. H., 1999. The systematics and distribution of Australian freshwater amphipods: a review. In Schram, F. R. & J. C. von Vaupel Klein (eds), Crustaceans and the Biodiversity Crisis. Proceedings of the Fourth International Crustacean Congress, Amsterdam, The Netherlands, July 20–24, 1998. Brill, Leiden: 533–540.
Bradbury, J. H. & W. D. Williams, 1997. Amphipod (Crustacea) diversity in underground waters in Australia: an Aladdin’s Cave. Memoirs of Museum Victoria 56: 513–519.
Byrne, M., D. K. Yeates, M. Kearney, J. Bowler, M. A. Williams, S. J. B. Cooper, S. C. Donnellan, S. Keogh, R. Leijs, J. Melville, D. Murphy, N. Porch & K.-H. Wyrwoll, 2008. Birth of a biome: synthesizing environmental and molecular studies of the assembly and maintenance of the Australian arid zone biota. Molecular Ecology 17: 4398–4417.
Carlisle, D., 1980. Possible variations on the calcrete-gypcrete uranium model. Report Prepared for the United States Department of Energy Under Subcontract Number 76-022-E between Bendix Field Engineering Corporation and The Regents of the University of California.
Castanier, S., G. Le Métayer-Levrel & J. P. Perthuisot, 1999. Ca-carbonates precipitation and limestone genesis—the microbiogeologist point of view. Sedimentary Geology 126: 9–23.
Castanier, S., G. Le Métayer-Levrel & J. P. Perthuisot, 2000. Bacterial roles in the precipitation of carbonate minerals. In Riding, R. E. & S. M. Awramik (eds), Microbial Sediments. Springer Verlag, NY, USA: 32–39.
Charette, M., 2001. Submarine groundwater discharge creates “Iron Curtain”. Woodshole Oceanographic Institute, Annual Report 2001: 23–24.
Charette, M. A. & E. R. Sholkovitz, 2002. Oxidative precipitation of groundwater-derived ferrous iron in the subterranean estuary of a coastal bay. Geophysical Research Letters 29(10), doi:10.1029/2001GLO14512.
Cho, J.-L., 2005. A primitive representative of the Parabathynellidae (Bathynellacea, Syncarida) from the Yilgarn Craton of Western Australia. Journal of Natural History 39: 3423–3433.
Cho, J.-L., J.-G. Park & W. F. Humphreys, 2005. A new genus and six new species of the Parabathynellidae (Bathynellacea, Syncarida) from the Kimberley Region, Western Australia. Journal of Natural History 39: 2225–2255.
Cho, J.-L., W. F. Humphreys & S.-D. Lee, 2006a. Phylogenetic relationships within the genus Atopobathynella Schminke, 1973 (Bathynellacea, Parabathynellidae): with the description of six new species from Western Australia. Invertebrate Systematics 20: 9–41.
Cho, J.-L., J.-G. Park & Y. Ranga Reddy, 2006b. Brevisomabathynella gen. nov. with two new species from Western Australia (Bathynellacea, Syncarida): the first definitive evidence of predation in Parabathynellidae. Zootaxa 1247: 25–42.
Chow, V. T. (ed.), 1964. Handbook of Applied Hydrology: A Compendium of Water-Resources Technology. McGraw-Hill, New York.
Cooper, S. J. B., S. Hinze, R. Leys, C. H. S. Watts & W. F. Humphreys, 2002. Islands under the desert: molecular systematics and evolutionary origins of stygobitic water beetles (Coleoptera: Dytiscidae) from central Western Australia. Invertebrate Systematics 16: 589–598.
Cooper, S. J. B., J. H. Bradbury, K. M. Saint, R. Leys, A. D. Austin & W. F. Humphreys, 2007. Subterranean archipelago in the Australian arid zone: mitochondrial DNA phylogeography of amphipods from central Western Australia. Molecular Ecology 16: 1533–1544.
Cooper, S. J. B., K. M. Saint, S. Taiti, A. D. Austin & W. F. Humphreys, 2008. Subterranean archipelago II: mitochondrial DNA phylogeography of stygobitic isopods (Oniscidea: Haloniscus) from the Yilgarn region of Western Australia. Invertebrate Systematics 22: 195–206.
Dames & Moore, 1984. Millstream water management programme. Unpublished report. Public Works Department, Western Australia. ISBN: 0 7244 9536 3.
Danielopol, D. L., 1989. Groundwater fauna associated with riverine aquifers. Journal of the North American Benthological Society 8: 18–35.
De Deckker, P., 1983. Australian salt lakes: their history, chemistry and biota—a review. Hydrobiologia 105: 231–244.
De Laurentiis, P., G. L. Pesce & W. F. Humphreys, 1999. Copepods from ground waters of Western Australia, IV. Cyclopids from basin and craton aquifers (Crustacea: Copepoda: Cyclopidae). Records of the Western Australian Museum 19: 243–257.
De Laurentiis, P., G. L. Pesce & W. F. Humphreys, 2001. Copepods from ground waters of Western Australia, VI. Cyclopidae (Crustacea: Copepoda) from the Yilgarn Region and the Swan Coastal Plain. Records of the Western Australian Museum Supplement 64: 115–131.
De Los Rios, P. & J. E. Crespo, 2004. Salinity effects on the abundance of Boeckella poopoensis (Copepoda, Calanoida) in saline ponds in the Atacama Desert, northern Chile. Crustaceana 77: 417–423.
Eberhard, S. M., S. A. Halse & W. F. Humphreys, 2005. Stygofauna in the Pilbara region, north-west Western Australia: a systematic review. Journal of the Royal Society of Western Australia 88: 167–176.
English, P., N. A. Spooner, J. Chappell, D. G. Questiaux & N. G. Hill, 2001. Lake Lewis basin, central Australia: environmental evolution and OSL chronology. Quaternary International 83–85: 81–101.
Eugster, H. P. & B. F. Jones, 1979. Behaviour of major solutes during closed-basin brine evolution. American Journal of Science 279: 609–631.
Fenwick, G. D., H. R. Thorpe & P. A. White, 2004. Groundwater systems. In Harding, J., P. Mosely, C. Pearson & B. Sorrell (eds), Freshwaters of New Zealand. New Zealand Hydrological Society and New Zealand Limnological Society: Christchurch, New Zealand 29: 1–18.
Finston, T. L., J. H. Bradbury, M. S. Johnson & B. Knott, 2004. When morphology and molecular markers conflict: a case history of subterranean amphipods from the Pilbara, Western Australia. Animal Biodiversity and Conservation 27: 83–94.
Finston, T. L., M. S. Johnson, W. F. Humphreys, S. Eberhard & S. Halse, 2007. Cryptic speciation in two widespread subterranean amphipod genera reflects historical drainage patterns in an ancient landscape. Molecular Ecology 16: 355–365.
Geological Survey, 1989. Hydrogeological Map of Western Australia, 1:2,500,000. Geological Survey of Western Australia, Perth.
Gibert, J. & L. Deharveng, 2002. Subterranean ecosystems: a truncated functional biodiversity. Bioscience 52: 473–481.
Gray, D. J., 2001. Hydrogeochemistry in the Yilgarn Craton. Geochemistry: Exploration, Environment, Analysis 1: 253–264.
Guzik, M. T., S. J. B. Cooper, W. F. Humphreys, J.-L. Cho & A. Austin, 2008. Phylogeography of the ancient Parabathynellidae (Crustacea: Bathynellacea) from the Yilgarn region of Western Australia. Invertebrate Systematics 22: 205–216.
Haack, S. K. & B. A. Bekins, 2000. Microbial populations in contaminated plumes. Hydrogeology Journal 8: 63–76.
Hammer, U. T., 1984. The saline lakes of Canada. In Taub, F. B. (ed.), Ecosystems of the World: 23. Lakes and Reservoirs. Elsevier, Amsterdam: 521–540.
Hancock, P. J., A. J. Boulton & W. F. Humphreys, 2005. Aquifers and hyporheic zones: towards an ecological understanding of groundwater. The Future of Hydrogeology. Hydrogeology Journal 13: 98–111.
Hardie, L. A. & H. P. Eugster, 1970. The evolution of closed basin brines. Mineralogical Society of America Special Publication 3: 273–290.
Hatton, T., 2001. Land use and catchment water balance. CSIRO Land and Water Technical Report 18/01, Perth.
Hesse, P. P., J. W. Magee & S. van der Kaars, 2004. Late Quaternary climates of the Australian arid zone: a review. Quaternary International 118–119: 87–102.
Hocking, R. M., H. T. Moors & W. J. E. van de Graaff, 1987. Geology of the Carnarvon Basin, Western Australia. Geological Survey of Western Australia, Bulletin 133: 1–289.
Humphreys, W. F., 1999a. Physico-chemical profile and energy fixation in Bundera Sinkhole, an anchialine remiped habitat in north-western Australia. Journal of the Royal Society of Western Australia 82: 89–98.
Humphreys, W. F., 1999b. Relict stygofaunas living in sea salt, karst and calcrete habitats in arid northwestern Australia contain many ancient lineages. In Ponder, W. & D. Lunney (eds), The Other 99%. The Conservation and Biodiversity of Invertebrates. Transactions of the Royal Zoological Society of New South Wales, Mosman 2088: 219–227.
Humphreys, W. F., 2000a. Karst wetlands biodiversity and continuity through major climatic change—an example from arid tropical Western Australia. In Gopal, B., W. J. Junk & J. A. Davis (eds), Biodiversity in Wetlands: Assessment, Function and Conservation, Vol. 1. Backhuys Publishers, Leiden: 227–258.
Humphreys, W. F., 2000b. First in, last out: should aquifer ecosystems be at the vanguard of remediation assessment? In Johnston, C. D. (ed.), Contaminated Site Remediation: From Source Zones to Ecosystems, Vol. 1. Centre for Groundwater Studies, Wembley, Western Australia: 275–282.
Humphreys, W. F., 2001. Groundwater calcrete aquifers in the Australian arid zone: the context to an unfolding plethora of stygal biodiversity. Records of the Western Australian Museum, Supplement 64: 63–83.
Humphreys, W. F., 2002. Keynote address: groundwater ecosystems in Australia: an emerging understanding. In Proceedings of the International Association of Hydrogeologists Conference, Darwin, Australia, 12–17 May 2002. CD-ROM from steven.tickell@nt.gov.au.
Humphreys, W. F., 2006. Aquifers: the ultimate groundwater dependent ecosystems. In Eamus, D. (ed.) Special Edition on Groundwater Dependent Ecosystems. Australian Journal of Botany 54: 115–132.
Humphreys, W. F., 2008a. Rising from down under: developments in subterranean biodiversity in Australia from a groundwater fauna perspective. Invertebrate Systematics 22: 85–101.
Humphreys, W. F., 2008b. Hydrogeology and groundwater ecology: does each inform the other? Hydrogeology Journal. doi:10.1007/s10040-008-0349-3.
Humphreys, W. F., S. M. Awramik & M. H. P. Jebb, 1995. Freshwater biogenic tufa dams in Madang Province, Papua New Guinea. Journal of the Royal Society of Western Australia 78: 43–54.
Iliffe, T. M., 2000. Anchialine cave ecology. In Wilkens, H., D. C. Culver & W. F. Humphreys (eds), Ecosystems of the World: 30. Subterranean Ecosystems. Elsevier, Amsterdam: 59–76.
Jacobson, G. & A. V. Arakel, 1986. Calcrete aquifers in the Australian arid zone. In Proceedings of the International Conference on Groundwater Systems Under Stress, Brisbane, Australian Water Resources Council: 515–523.
Jacobson, G. & J. Wischusen, 2001. Groundwater for Aboriginal communities in central Australia: the Western Water Study (Wiluraratja Kapi), Northern Territory. In Gostin, V. A. (ed.), Gondwana to Greenhouse: Australian Environmental Geoscience. Geological Society of Australia Special Publication 21: 171–178.
Jaume, D., G. A. Boxshall & W. F. Humphreys, 2001. New stygobiont copepods (Calanoida; Misophrioida) from Bundera sinkhole, an anchialine cenote on north-western Australia. Zoological Journal of the Linnean Society London 133: 1–24.
Karanovic, I., 2003. Towards a revision of Candoninae (Crustacea: Ostracoda): descriptions of two new genera from Australian groundwaters. Species Diversity 8: 353–383.
Karanovic, T., 2004. Subterranean copepods (Crustacea: Copepoda) from arid Western Australia. Crustaceana Supplement 3: 1–366.
Karanovic, I., 2005. Towards a revision of Candoninae (Crustacea, Ostracoda): Australian representatives of the subfamily, with description of three new genera and seven new species. New Zealand Journal of Marine and Freshwater Research 39: 29–75.
Karanovic, T., 2006. Subterranean copepods (Crustacea, Copepoda) from the Pilbara region in Western Australia. Records of the Western Australian Museum Supplement 70: 1–239.
Karanovic, I., 2007. Candoninae ostracodes from the Pilbara region in Western Australia. Crustaceana Monographs 7: 1–432.
Karanovic, I. & P. Marmonier, 2002. On the genus Candonopsis (Crustacea: Ostracoda: Candoninae) in Australia, with a key to the world recent species. Annals of Limnology 38: 199–240.
Karanovic, I. & P. Marmonier, 2003. Three new genera and nine new species of the subfamily Candoninae (Crustacea, Ostracoda, Podocopida) from the Pilbara region (Western Australia). Beaufortia 53: 1–51.
Kiefer, F., 1967. Cyclopiden aus salzhaltigen Binnengewassern australiens (Copepoda). Crustaceana 12: 292–302.
Langecker, T. G., 2000. The effects of continuous darkness on cave ecology and cavernicolous evolution. In Wilkens, H., D. C. Culver & W. F. Humphreys (eds), Ecosystems of the World, Vol. 30. Amsterdam, Elsevier: 581–601.
Lawrence, J. R., R. C. Hynes & U. T. Hammer, 1978. Contribution of photosynthetic green sulfur bacteria to total primary production in a meromictic saline lake. Verhandlungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 20: 201–207.
Leys, R. & C. H. Watts, 2008. Systematics and evolution of the Australian subterranean hydroporine diving beetles (Dytiscidae), with notes on Carabhydrus. Invertebrate Systematics 22: 217–225.
Leys, R., C. H. S. Watts, S. J. B. Cooper & W. F. Humphreys, 2003. Evolution of subterranean diving beetles (Coleoptera: Dytiscidae: Hydroporini, Bidessini) in the arid zone of Australia. Evolution 57: 2819–2834.
Lyons, W. B., S. W. Tyler & H. E. Gaudette, 1995. The use of strontium isotopes in determining the groundwater mixing and brine salt-fingering in a playa discharge zone, Lake Tyrrell, Australia. Journal of Hydrology 167: 225–239.
Mann, A. W. & R. L. Deutscher, 1978. Hydrogeochemistry of a calcrete-containing aquifer near Lake Way, Western Australia. Journal of Hydrology 38: 357–377.
Mann, A. W. & R. C. Horwitz, 1979. Groundwater calcrete deposits in Australia: some observations from Western Australia. Journal of the Geological Society of Australia 26: 293–303.
McArthur, J. M., J. Turner, W. B. Lyons & M. F. Thirlwall, 1989. Salt sources and water–rock interactions on the Yilgarn Block, Australia: isotopic and major element tracers. Applied Geochemistry 4: 79–92.
Michael, H. A., A. E. Mulligan & C. F. Harvey, 2005. Seasonal oscillations in water exchange between aquifers and coastal ocean. Nature 436: 1145–1148.
Moore, H. B., 1966. Marine Ecology. Wiley, New York.
Moore, W. S., 1999. The subterranean estuary: a reaction zone of ground water and sea water. Marine Chemistry 65: 111–125.
Morgan, K. H., 1993. Development, sedimentation and economic potential of palaeoriver systems of the Yilgarn Craton of Western Australia. Sedimentary Geology 85: 637–656.
Page, T. J., W. F. Humphreys & J. M. Hughes, 2008. Shrimps down under: evolutionary relationships of subterranean crustaceans from Western Australia (Decapoda: Atyidae: Stygiocaris). PLoS ONE 3(2): e1618. doi:10.1371/journal.pone.0001618.
Pesce, G. L., P. De Laurentiis & W. F. Humphreys, 1996. Copepods from ground waters of Australia, II. The genus Halicyclops (Crustacea: Copepoda: Cyclopidae). Records of the Western Australian Museum 18: 77–85.
Pérez del Villar, L., A. Garralón, A. Delgado, E. Reyes, J. S. Cózar, P. Gómez, R. Núñez, L. Sánchez & J. Raya, 2004. Hydrogeochemical evolution and C isotope study of groundwaters from “Mina Fe” U deposit (Salamanca, Spain): implications for processes in radwaste disposal. Applied Geochemistry 20: 465–485.
Pinder, A. M. & R. O. Brinkhurst, 1997. Review of the Phreodrilidae (Annelida: Oligochaeta: Tubificida) of Australia. Invertebrate Taxonomy 11: 443–523.
Pinder, A. M., E. M. Eberhard & W. F. Humphreys, 2006. New phallodrilines (Annelida: Clitellata: Tubificidae) from Western Australian groundwater. Zootaxa 1304: 31–48.
Pohlman, J. W., L. A. Cifuentes & T. M. Iliffe, 2000. Food web dynamics and biogeochemistry of anchialine caves: a stable isotope approach. In Wilkens, H., D. C. Culver & W. F. Humphreys (eds), Ecosystems of the World: 30. Subterranean Ecosystems. Elsevier, Amsterdam: 345–357.
Pohlman, J. W., T. M. Iliffe & L. A. Cifuentes, 1997. A stable isotope study of organic cycling and the ecology of an anchialine cave ecosystem. Marine Ecology Progress Series 155: 17–27.
Poore, G. C. B. & W. F. Humphreys, 1998. First record of Spelaeogriphacea from Australasia: a new genus and species from an aquifer in the arid Pilbara of Western Australia. Crustaceana 71: 721–742.
Poore, G. C. B. & W. F. Humphreys, 2003. Second species of Mangkurtu (Spelaeogriphacea) from north-western Australia. Records of the Western Australian Museum 22: 67–74.
Pora, E. A., 1969. L’importance du facteur rhopique (équilibre ionique) pour la vie aquatique. Verhandlungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 17: 970–986.
Radke, L., 2000. Solute divides and chemical facies in south-eastern Australian salt lakes and the response of ostracods in time (Holocene) and space. Ph.D. thesis, Australian National University, Canberra.
Radke, L., S. Juggins, S. A. Halse, P. De Deckker & T. Finston, 2003. Chemical diversity in south-eastern Australian saline lakes II: biotic implications. Marine and Freshwater Research 54: 895–912.
Reeves, J. M., P. De Deckker & S. A. Halse, 2007. Groundwater ostracods from the arid Pilbara region of northwestern Australia: distribution and water chemistry. Hydrobiologia 585: 99–118.
Sanders, C. C., 1974. Calcrete in Western Australia. Western Australia Geological Survey Annual Report 1973: 12–14.
Schminke, H. K., 1973. Evolution, System und Verbreitungsgeschichte der Familie Parabathynellidae (Bathynellacea, Malacostraca). Mikrofauna des Meeresbodens 24: 1–192.
Schminke, H. K., 1974. Mesozoic intercontinental relationships as evidenced by bathynellid crustacea (Syncarida: Malacostraca). Systematic Zoology 23: 157–164.
Schminke, H. K., 1981. Adaptation of Bathynellacea (Crustacea, Syncarida) to life in the interstitial (“Zoea Theory”). Internationale Revue gesamten Hydrobiologie 66: 575–637.
Seymour, J. R., W. F. Humphreys & J. G. Mitchell, 2007. Stratification of the microbial community inhabiting an anchialine sinkhole. Aquatic Microbial Ecology 50: 11–24.
Simon, K. S., 2000. Organic dynamics and trophic structure in karst groundwater. Ph.D. thesis. Faculty of Biology, Virginia Polytechnic Institute and State University.
Simon, K. S., E. F. Benfield & S. A. Macko, 2003. Food web structure and the role of epilithic biofilms in cave streams. Ecology 84: 2395–2406.
Sket, B., 1996. The ecology of anchihaline caves. Trends in Ecology and Evolution 11: 221–255.
Stock, J. H., T. M. Iliffe & D. Williams, 1986. The concept ‘anchialine’ reconsidered. Stygologia 2: 90–92.
Taiti, S. & W. F. Humphreys, 2001. New aquatic Oniscidea (Crustacea, Isopoda) from groundwater calcretes of Western Australia. In Humphreys, W. F. & M. S. Harvey (eds), Subterranean Biology in Australia 2000. Records of the Western Australian Museum, Supplement No. 64: 133–151.
Testa, J. M., M. A. Charette, E. R. Sholkovitz, M. C. Allen, A. Rago & C. W. Herbold, 2002. Dissolved iron cycling in the subterranean estuary of a coastal bay: Waquoit Bay, Massachusetts. Biological Bulletin 203: 255–256.
Tokuoka, T., Y. Sampei, K. Nishimura, S. Suzaki, S. Matsuda, S. Kubota, S. Suzuki, H. Ueno & T. Ikeda, 2000. Saline water intrusion at estuary river and its relation to the underground water: a case study at R. Gonokawa, Shimane Pref., Japan. Proceedings of the Techno Ocean Symposium 3: 715–720.
Torgersen, T., 1984. Wind effects on water and salt loss in playa lakes. Journal of Hydrology 74: 137–149.
Torgersen, T., P. De Deckker, A. R. Chivas & J. M. Bowler, 1986. Salt lakes: a discussion of processes influencing palaeoenvironmental interpretation and recommendations for future study. Palaeogeography, Palaeoclimatology, Palaeoecology 54: 7–19.
Vasconcelos, P. M., K. M. Knesel, B. E. Cohen & J. A. Heim, 2008. Geochronology of the Australian Cenozoic: a history of tectonic and igneous activity, weathering, erosion, and sedimentation. Australian Journal of Earth Sciences 55(6): 865–914.
Wanty, R. B., & R. Schoen, 1991. A review of the chemical processes affecting the mobility of radionuclides in natural waters, with applications. In Gundersen, L. C. S. & R. B. Wanty (eds), Field Studies of Radon in Rocks, Soils, and Water. US Geological Survey Bulletin 1971: 183–194.
Watts, C. H. S. & W. F. Humphreys, 1999. Three new genera and five new species of Dytiscidae (Coleoptera) from underground waters in Australia. Records of the South Australian Museum 32: 121–142.
Watts, C. H. S. & W. F. Humphreys, 2000. Six new species of Nirridessus and Tjirtudessus (Dytiscidae; Coleoptera) from underground waters in Australia. Records of the South Australian Museum 33: 127–144.
Watts, C. H. S. & W. F. Humphreys, 2001. A new genus and six new species of Dytiscidae (Coleoptera) from underground waters in the Yilgarn palaeodrainage system of Western Australia. Records of the South Australian Museum 34: 99–114.
Watts, C. H. S. & W. F. Humphreys, 2003. Twenty-five new Dytiscidae (Coleoptera) of the genera Tjirtudessus Watts & Humphreys, Nirripirti Watts & Humphreys and Bidessodes Regimbart, from underground waters in Australia. Records of the South Australian Museum 36: 135–187.
Watts, C. H. S. & W. F. Humphreys, 2004. Thirteen new Dytiscidae (Coleoptera) of the genera Boongurrus Larson, Tjirtudessus Watts & Humphreys and Nirripirti Watts and Humphreys, from underground waters in Australia. Transactions of the Royal Society of South Australia 128: 99–129.
Watts, C. H. S. & W. F. Humphreys, 2006. Twenty-six new Dytiscidae (Coleoptera) of the genera Limbodessus Guignot and Nirripirti Watts and Humphreys, from underground waters in Australia. Transactions of the Royal Society of South Australia 130: 123–185.
Watts, C. H. S. & W. F. Humphreys, in press. Fourteen new Dytiscidae (Coleoptera) of the genera Limbodessus Guignot, Paroster Sharp and Exocelina Broun, from underground waters in Australia. Transactions of the Royal Society of South Australia 133.
Williams, W. D., 1983. On the ecology of Haloniscus searlei (Isopoda, Oniscoidea), an inhabitant of Australian salt lakes. Hydrobiologia 105: 137–142.
Williams, W. D., 1984. Australian lakes. In Taub, F. B. (ed.), Ecosystems of the World: 20. Lakes and Reservoirs. Elsevier, Amsterdam: 499–519.
Williams, W. D., 1986. Limnology, the study of inland waters: a comment on perceptions of studies of salt lakes, past and present. In De Deckker, P. & W. D. Williams (eds), Limnology in Australia. CSIRO Melbourne & Dr W. Junk Publishers, Dordrecht: 471–484.
Williams, W. D., A. J. Boulton & R. G. Taaffe, 1990. Salinity as a determinant of salt lake fauna: a question of scale. Hydrobiologia 197: 257–266.
Wilson, G. D. F., 2001. Australian groundwater-dependent isopod crustaceans. In Humphreys, W. F. & M. S. Harvey (eds), Subterranean Biology in Australia 2000. Records of the Western Australian Museum, Supplement No. 64: 239–240.
Wilson, G. D. F., 2003. A new genus of Tainisopidae fam. nov. (Crustacea: Isopoda) from the Pilbara, Western Australia. Zootaxa 245: 1–20.
Wilson, G. D. F. & R. T. Johnson, 1999. Ancient endemism among freshwater isopods (Crustacea, Phreatoicidea). In Ponder, W. & D. Lunney (eds), The Other 99%. The Conservation and Biodiversity of Invertebrates. Transactions of the Royal Zoological Society of New South Wales, Mosman: 264–268.
Wolff, W. J., 1973. The estuary as a habitat: an analysis of data on the soft-bottom macrofauna of the estuarine area of the rivers Rhine, Meuse, and Scheldt. Zoologische Verhandelingen 126: 1–242.
Acknowledgements
We thank Stefano Taiti and Joo-Lae Cho for their determinations and continued involvement in the calcrete studies, Jessica Reeves for processing ionic data, and Andy Austin and the Environmental Futures Network working group on short-range endemic taxa associated with troglobitic and groundwater ecosystems for support and valuable discussion. Elements of this work were supported by grants from the Australian Biological Resources Study and the Australian Research Council (A00106441). We thank Brian Timms and an anonymous referee, whose discerning comments served to improve the lucidity of the article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: J. John & B. Timms
Salt Lake Research: Biodiversity and Conservation—Selected papers from the 9th Conference of the International Society for Salt Lake Research
An erratum to this article can be found at http://dx.doi.org/10.1007/s10750-009-9864-z
Rights and permissions
About this article
Cite this article
Humphreys, W.F., Watts, C.H.S., Cooper, S.J.B. et al. Groundwater estuaries of salt lakes: buried pools of endemic biodiversity on the western plateau, Australia. Hydrobiologia 626, 79–95 (2009). https://doi.org/10.1007/s10750-009-9738-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-009-9738-4