Abstract
Low-head dams and weirs can greatly limit the distribution and abundance of Atlantic salmon and other migratory salmonids in streams. Weirs can significantly increase the vulnerability of migratory fish to anglers, alter natural migration patterns, and exacerbate the effects of opportunistic predators. Overcrowding of fish at downstream pools can also facilitate the spread of parasites and infectious diseases, magnify the impact of pollution incidents, and increase the risk of mass mortalities, particularly at low flows. Not surprisingly, augmenting the accessible stream area constitutes one of the best ways to restore depleted salmonid populations. In this context, the removal of unused or illegal weirs can be an efficient, cheap solution to increase stream accessibility. Here, I examine some impacts of weirs on Atlantic salmon populations, and document with case studies the removal and breaching of weirs in several Iberian streams.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Along with water abstraction and pollution, damming is probably one of the greatest stressors affecting the integrity of running waters (Pielou, 1998; Heinz Center, 2002). Dams can interfere or even stop the transport of sediment and nutrients along waterways, reduce (or amplify) fluctuations in natural discharge levels, prevent inundation of floodplains and create wider and shallower rivers. Such changes in riverine processes can lead to increased algal blooms, increased bank erosion and reduced water quality (Kondolf, 1997). Impounded waters can also trigger important changes in the composition of stream fauna, favouring lentic over lotic species (Raymond, 1979; Lewis, 2001). Indeed, impounding can result in the loss of native species and their habitats (Vaughn & Taylor, 1999), and facilitate the colonization and spread of invasive species such as water weeds and pest fish (Havel et al., 2005).
Dams can block or delay the movements of migratory fish, and these are responsible for the decline or extirpation of many native salmon populations in both the Atlantic (Netboy, 1968; Mills, 1989) and the Pacific (Meehan, 1991; Frisell, 1993; Levin & Tolimieri, 2001). Difficulty of migration also explains the distribution and abundance of other migratory species such as the eel (Ibbotson et al., 2002). However, the effects of low head dams and small weirs (i.e. those that do not represent a permanent or insurmountable barrier to fish migration) are less well understood. Even small weirs (<5 m) can have significant effects on flow and temperature regimes, sediment transport, biogeochemistry, animal movements and stream habitat (Larinier, 2001; Hart et al., 2002), so it should come as no surprise that the modification of flow caused by such weirs can also alter the structure of communities and function of river ecosystems (Baumgartner, 2007). For example, weirs can prevent natural gravel recruitment along the river, leading to a reduction in the quality and extension of downstream gravel spawning areas (Kondolf, 2000, 2001).
Here, I examine the abundance of weirs and other obstacles in rivers of N. Spain and assess some of their impacts upon Iberian Atlantic salmon populations. In particular, I consider the effect of weirs on (1) upstream migrations, (2) exploitation by anglers, (3) predation risk, and (4) spread of infectious diseases. Finally, I document with case studies the practicalities and challenges of weir removal in France and Spain (online supplemental material).
Impact of weirs on salmon populations in the Iberian Peninsula
Abundance and distribution of weirs
The density of artificial obstacles (chiefly weirs) in the salmon rivers of the northern Spanish provinces ranges from 0.15 km−1 of accessible stream length to a maximum of 1.16 obstacles km−1 depending on the river (Fig. 1). The average for 31 rivers is 0.46 weirs km−1. In general, there is a higher density of weirs in tributaries than in main stems (Alvarez et al., 2003; Tamés et al., 2003), possibly reflecting the higher gradients of the former. Consequently, a few tributaries are now accessible to salmon and other migratory species in Iberian streams, and even then the length of accessible stream has been greatly reduced. For example, in the R. Asón, 30% of the weirs are located within a 10 km radius from the tidal limit, and this can greatly restrict the passage of fish, particularly the weakest swimmers such as the Allis shad (Alosa alosa (L.)).
Density of dams, weirs and other artificial obstacles in salmon streams of N. Spain, expressed as number of weirs per km of accessible stream length. Adapted from data in Alvarez et al. (2003) and unpublished sources
Characteristics of weirs
Most of the barrier that impede or that make difficult the passage of migratory salmonids in Iberian streams are man-made, and only a small percentage (6%) are natural obstacles such as water falls. Artificial obstacles consist of dams (3%), low-head weirs (87%), gauging stations (4%) and culverts or other structures (5%, Alvarez et al., 2003). Over half of the dams and weirs located in the salmon rivers are typically 1–3 m in height (Fig. 2); large dams (>10 m), usually for the production of electricity, are also found in some rivers, especially in the Atlantic rivers of the NW provinces, but these are relatively rare; median weir height is 2.2 m.
Many of the old weirs that are still found in the salmon rivers of the Iberian Peninsula were built to divert water to mills and foundries in the nineteenth century, and some were later reconverted to generate electricity. These were mostly made of masonry and loose slabs and are today in poor condition, constituting a flooding hazard during spates. A survey of 70 weirs in the salmon rivers of Cantabria indicates that 66% are now abandoned and out of use. A similar proportion of unused weirs was also found in other regions (Alvarez et al., 2003; Tamés et al., 2003). Few, if any, weirs were fitted with a fish pass or with downstream fish screens and during the smolt migration large numbers of smolts may get stranded or delayed in water abstraction canals, this being an additional source of mortality.
Impact of weirs on river connectivity and salmon movements
Weirs can impact upon migratory fish by completely blocking their access to spawning grounds or the sea, but also by delaying their passage or by making it more difficult. The cumulative effect of many small weirs (even those that can be overcome) is particularly insidious (Williams, 1998; Naughton et al., 2005) since small weirs are the most numerous and low-head dams are not necessarily regarded by some fisheries managers as barriers to fish movements. However, weir height is a poor indicator of difficulty of fish passage and low weirs can be as difficult or insurmountable as much higher dams, depending on the hydraulic characteristics, water temperature, river flow and type and size of fish (Larinier, 2001). For example, radio-tracking studies have shown that weirs as small as 0.5 m can delay the passage of adult Atlantic salmon and sea trout, while weirs of only 1.2 m may constitute impassable obstacles, depending on flow and water temperatures (Gerlier & Roche, 1998). Discharge in the salmon rivers of N. Spain follows a marked monthly trend, with flows typically peaking in March and April (where the largest salmon tend to enter these rivers) followed by severe droughts from July to September (Fig. 3). At least four migratory fish species are present in many of these rivers (Atlantic salmon, Salmo salar (L.); sea trout, Salmo trutta (L.); European eel, Anguilla Anguilla (L.); and Allis shad, Alosa alosa) and these can enter the rivers throughout the year. Thus, fish passage must be maintained even during low flows.
Seasonal pattern in monthly discharge in the river Asón (Cantabria), typical of many of the salmon streams of northern Spain, expressed as flow coefficients (FC) with respect to the annual mean (FC = 1.0). The migration and spawning periods of several migratory fish are indicated (adapted from Serdio et al., 2001a)
Weirs can force adult Atlantic salmon to fall-back and to spawn away from their home river (Gerlier & Roche, 1998; Thorstad & Heggberget, 1998), thereby negating the benefits of homing behaviour. Even when there are provisions for fish passage, adult salmon may take a long time to ascend fish ladders (Gowans et al., 1999; Rivinoja, 2005) or may not ascend at all (Solomon et al., 1999; Solomon & Sambrook, 2004). A lengthened migration period can deplete energy reserves and result in reduced spawning success or increased pre-spawning mortality (Gerlier & Roche, 1998; Geist et al., 2003). This is because injured or exhausted spawners may be forced to spawn in suboptimal areas or to deposit their eggs at too shallow depths (Berg et al., 1986). Since egg burial in salmonids is directly related to embryo survival (Steen & Quinn, 1999; De Gaudemar et al., 2000), offspring of exhausted females may be more vulnerable to floods, redd overcutting and bed scouring.
Weirs can result in fragmented salmonid populations, decreasing their effective population size (Meldgaard et al., 2003), increasing genetic isolation (Heggenes & Røed, 2006) and compromising their evolutionary potential (Morita & Yamamoto, 2002). Paradoxically, weirs can also increase gene flow among population components. In the case of Atlantic salmon this could affect age at maturity since larger, older multi-seawinter fish tend to enter the rivers earlier and spawn higher upstream than younger grilse, which tend to enter later in the season and spawn lower in the system (Summers, 1996a, b; Youngson & Hay, 1996; Økland et al., 2001). Weirs, hence, can force both age classes to interbreed, thus negating the presumed adaptive benefits of assortative mating (i.e. Taggart et al., 2001) and leading to population homogenization.
Today, Atlantic salmon can only exploit between 5% and 89% of the stream length historically accessible to the species in different Spanish rivers (Alvarez & Lamuela, 2001; Serdio et al., 2001a; Alvarez et al., 2003) and the total loss of salmon habitat due to artificial barriers has been estimated to represent 86% of the stream length historically accessible to the species (Alvarez et al., 2003). Longitudinal profiles of salmon rivers reveal that the cumulative effect of many weirs can be considerable. For example in the R. Asón and its tributaries, the cumulative height of weirs can be in excess of 25 m over a relatively short reach (Fig. 4), making it difficult or even impossible for fish to reach the best spawning grounds located upstream (Garcia de Leaniz et al., 1987). As a result, salmon and sea trout spawners are largely confined to spawn in the lower reaches of the main rivers, where survival of embryos and juveniles is likely to be lower due to increased siltation and greater vulnerability to predation. Radio-tracking of brown trout in the River Bidasoa (Spain) indicates that weirs prevented fish from reaching the spawning grounds, and caused significant reproductive isolation within the watershed (Gosset et al., 2006). In general, weirs in the salmon rivers of N. Spain have disrupted natural community structures and resulted in a significant reduction of fish species diversity (Reyes-Gavilán et al., l996).
Although the effects of weirs on upstream fish passage have traditionally received more attention, impacts upon downstream migrants can be just as damaging (O’Connor et al., 2006). Weirs can delay smolt migrations and this can result in increased mortality. For example, Aarestrup & Koed (2003) reported a 53% loss of Atlantic salmon smolts due to weir passage compared to control smolts. They attributed smolt losses to increased predation, and delays that may have resulted in desmoltification. The weirs they studied ranged in height from 0.6 to 2.5 m, indicating that even small structures can negatively impact upon smolt passage. In the Spanish rivers studied, a few abstraction canals were equipped with effective fish screens, and this constitutes an additional source of potential juvenile mortality, particularly among smolts.
Overexploitation
Weirs and other obstacles can greatly increase angling mortality. For example, using data from radio tagged individuals, Karppinen et al. (2002) estimated that anglers fishing downstream of an impassable waterfall fitted with a fish pass exploited 39% of adult Atlantic salmon attempting to swim through the pass. Similarly, in the River Blanda (Iceland) Gudjónsson (1988) estimated exploitation rates by anglers of 36–77% (mean 50%) downstream of an obstacle and fish pass, compared to 21–31% (mean 26%) upstream of the same obstacle, indicating that barriers can almost double fishing mortality induced by anglers, even when they are fitted with a fish pass.
In Spanish rivers, fishing effort for salmon can be high and tends to target the largest females which enter early in the fishing season (Garcia de Leaniz et al., 2001), resulting in significant phenotypic and genetic changes in the populations (Consuegra et al., 2005). Many popular salmon pools in Iberian rivers tend to be located downstream of weirs or other obstacles, and rod catches in these pools can make up more than 30% of the entire river catch (Table 1), despite the existence of daily catch quotas per angler.
Fine-scale analysis of the spatial distribution of catches downstream of one of these weirs (Pte Viesgo weir) on the River Pas, indicates that the barrier effect is most noticeable immediately downstream of the weir, but that its effect can extend up to 1.5 km downstream (Fig. 5). Thus, existing fishing regulations that prohibit fishing within 50 m of weirs may need to be modified to achieve effective protection from over-exploitation, an important factor in the historical decline of Iberian salmon populations (Netboy, 1968, 1974), and one which has been evidenced recently by the high concentration of lead sinkers found in traditional fishing pools (Guitart & Thomas, 2005).
Spatial distribution of salmon catches in relation to the Pte Viesgo weir (2.9 m) in the R. Pas. Shown are the aggregated rod and line catches in different salmon pools downstream of the weir during the period 1988–2000 (adapted from Serdio et al., 2001a)
Predation
Weirs can facilitate predation in a number of ways. First, crowding of migrants around weirs can increase predation by opportunistic predators such as the otter (Lutra lutra (L.)), particularly during the spawning or smolt migrations when salmonids are most vulnerable (e.g. Carss et al., 1990; Garcia de Leaniz et al., 2006). Weirs also force fish to swim over relatively shallow crests and these provide vantage points for bird predators, particularly during low flows. In addition, the existence of still and relatively deep waters upstream of weirs facilitates foraging by diving birds and negates the refuge afforded by the turbulence of riffles. Two important bird predators in the salmon rivers of the Iberian Peninsula include the great cormorant (Phalacrocorax carbo (L.)) and the heron (Ardea cinera L.), both of which can have a significant effect on salmonid populations (Kennedy & Greer, 1988; Dieperink et al., 2001; Serdio et al., 2003). Data from the R. Asón indicates that the distribution of great cormorants along the river course is closely related to the location of weirs (Fig. 6), suggesting that weirs offer vantage points for bird predators.
Spatial distribution of cormorant sightings along the length of the R. Asón during 1996–2000 and location of main weirs (indicated by arrows; adapted from Serdio et al., 2001a)
Stress and diseases
Infectious diseases represent an important source of adult salmon mortality in Iberian rivers (Martin-Ventura, 1988; Garcia de Leaniz et al., 2001). Delays in upstream passage caused by weirs can result in overcrowding in downstream pools, and this can in turn facilitate the spread of infectious disease especially in the summer months, when temperatures are high and the flows are lowest. Furunculosis (causative agent Aeromonas salmonicida) and ERM (causative agent Yersinia ruckeri) are two infectious diseases commonly found in Atlantic salmon in Iberian rivers (Marquez, 1999; Garcia de Leaniz et al., 2001; Consuegra et al., 2003) which can cause substantial mortalities in some years. Data from the R. Asón indicates that the distribution of dead and moribund adult salmon (mostly collected before the spawning season) closely matches the location of weirs, perhaps suggesting that both are casually linked (Fig. 7).
Spatial distribution of adult salmon mortalities along the length of the R. Asón during 1996 and location of main weirs (indicated by arrows; adapted from Serdio et al., 2001a)
Issues in weir removal
Although dam removal is not new—more than 600 dams have been removed in North America in the last century including 56 during 2005 (Heinz Center, 2002; American Rivers, 2005; http://www.irn.org)—it is still a relatively new restoration technique elsewhere. Nevertheless, weir demolition is quickly becoming widespread in Europe (e.g. Finland, Jormola, 2001; Denmark, Riber, 2001; France, van Ast, 2000, Steinbach, 2001; Spain, Garcia de Leaniz et al., 2001, Brufao & Rodriguez, 2003, Brufao, 2006; Germany, Redeker et al., 2004) and Australia (WWF Australia, 2004, 2005). CEMAGREF (http://www.lyon.cemagref.fr/bea) provides a useful bibliography on dam removal with more than 60 journal articles, books and reports covering projects on several countries.
Reasons for dam removal
Causes for considering dam removal are numerous, but dam removal was originally driven chiefly by structural and safety reasons, and to minimize flooding hazards (Doyle et al., 2003). For example, rivers in many parts of Southern Europe are particularly prone to droughts and extreme changes in discharge (Nijland & Cals, 2001), which can sometimes result in catastrophic flooding events (e.g. Zacharias, 2001) and which are exacerbated by the presence of weirs and other structures opposing the flow of water. As most dams have an expected life span of c. 50 years (Heinz Center, 2002), ageing is also a major issue in dam decommissioning, and thousands of dams built during the 1970s (‘the dam golden area’) will need to be decommissioned in the next few years, offering unprecedented opportunities for river restoration. For this and other reasons, dam removal is rapidly becoming an important management tool in river restoration (Hart & Poff, 2002), though information is still dominated by relative large projects, especially in the United States.
River fragmentation is one of the key impacts of dams on aquatic ecosystems (Nilsson et al., 2005), and restitution of river connectivity is therefore one of the first objectives in dam removal (Pejchar & Warner, 2001; American Rivers, 2002; Roni et al., 2002). In the case of migratory fishes (or those that simply alternate between lentic and riverine habitats), river connectivity is essential for completing their life cycle, and augmenting the area available to these species is tantamount to increasing population numbers (WWF, 2001). For example, 1 year after the Edwards Dam (USA) was demolished, large numbers of migratory fish returned to previously impounded parts of the Kennebec River (Maine), including millions of alewife, a migratory species which had disappeared for 160 years (Meadows, 2001). Likewise, fish diversity in the Baraboo River (Wisconsin) increased from 11 to 24 species less then 2 years following dam removal (Catalano et al., 2001).
Perhaps not surprisingly, the removal of barriers is often the most cost effective instream restoration technique in most situations (Pejchar & Warner, 2001; Roni et al., 2002); in the case of the Atlantic salmon, mathematical models suggest that dam removal and other improvements in freshwater habitats are also the best management options for restoring endangered populations (Robertson, 2005).
Legal framework regulating water concessions and weir removal
Epple (2000) has summarized the legal and regulatory framework governing the concessions of dams in France and most other European nations. Concessions in most European countries last for 40–60 years, a period usually smaller than the expected lifetime of the dam. In the case of Spain, concessions are normally given for 75 years, but these can be easily renewed and Water Authorities have traditionally been reluctant to impose any environmental constraint on water abstraction, let alone for compensatory flows or fish passage requirements (Zataraín, 2001; Brufao & Rodriguez, 2003). As a consequence, a few dams and weirs built in Spain before the 1970s were fitted with fish passes, and those in existence are notoriously inadequate (Garcia de Leaniz et al., 1987, 2001; Alvarez et al., 2003).
In most countries, concessions granted by the Water Authorities include details of the owner of the dam and the beneficiary of the concession, the type and nature of the water abstraction, the duration of the concession, the methods of exploitation, the power capacity, the minimum flow releases, the fish passage facilities and in general any environmental control measures. In most countries, concession details can be reviewed through public consultation, at least in theory. In practice, however, this can be a difficult and time consuming process, which is a complaint frequently lodged with the European Union (see Zataraín, 2001). Also, concession details of small or very old weirs, are often incomplete or nonexistent (Epple, 2000).
In Spain, weirs can be removed for a variety of reasons (Brufao, 2001; Brufao & Rodriguez, 2003; Brufao, 2006), the most common of which include the following:
-
(1)
Lack of concession. Illegal weirs that lack a water concession are common in many situations, and developers or promoters may fail to provide evidence of a permit to abstract or impound water with a weir.
-
(2)
End of concession. Water concessions are not eternal, and although concessions can be renewed, when a concession expires this allows the authorities to decommission many dams and weirs.
-
(3)
Breach of concession terms. Concessions can be terminated if water is abstracted for a purpose different than that authorized or if it fails to comply with the restrictions imposed in the concession (typically some form of compensatory flow or provisions for fish passage).
-
(4)
Lack of use. A concession can be declared extinct if it has been out of operation for three or more consecutive years for reasons attributed to the concession holder. In theory, this clause allows the public to request from the authorities the removal of any weir which is in ruins, or simply out of use.
In addition the fishery service in Spain can also undertake all the necessary works to facilitate fish passage in any weir at the weir concession holder’s expense.
Considerations for removing weirs
Weir removal can have physical, biological and societal implications that need to be taken into account in the planning process (Doyle et al., 2000; Heinz Center, 2002; Hart et al., 2002), and which include the following:
-
1.
Stability of riparian margins;
-
2.
Sediment and gravel transport;
-
3.
Flood risks;
-
4.
Potential transport of toxic sediments;
-
5.
Reduction of stream width upstream of the weir;
-
6.
Other changes in the river channel;
-
7.
Societal and cultural issues.
Although the long-term consequences of dam removal are still poorly understood (Stanley et al., 2002), particularly in the case of large dams (Grant, 2001; Stanley & Doyle, 2003), the potential impact of sudden sediment mobilization is probably the biggest concern (Doyle et al., 2000, Stanley et al., 2002, Stanley & Doyle, 2003). Bednarek (2001) has summarized the ecological impacts of dam removal and concluded that any increase in sediment load following dam removal is generally short-lived and that the benefits greatly outweigh the temporary impacts, from which the stream quickly recovers (but see Stanley & Doyle, 2003). In most situations, river conditions are little affected by weir removal and appear to return quickly to pre-impoundment conditions (Ashley et al., 2006; Velinsky et al., 2006). However, post-removal monitoring is important (Grant, 2001) since not all dams are likely to have the same effects on the stream ecosystem (Levin & Tolimieri, 2001) and contaminated or toxic sediments may require specific pre-removal studies.
Cultural considerations may also need to be taken into account when considering dam removal (Marmulla, 2001), as some weirs may have high historical or societal value. For example, in S. Europe a number of weirs date back to Roman times (Arenillas & Castillo, 2003) or may have other archaeological interests, and this may prevent their demolition or constrain how they are removed.
People’s attitudes to the impact or benefits of weirs in streams can vary markedly between stakeholders (e.g. Østdahl et al., 2001; Pejchar & Warner, 2001), and these should be incorporated whenever possible into the decision-making process (Babbitt, 2002), particularly in the case of weirs and low-head dams which may not be viewed as detrimental by all (Johnson & Graber, 2002). For example, weirs (some up to 7 m high) continue to be constructed in many streams under the umbrella of ‘river restoration’, allegedly for sediment retention, reduction of peak flows and creation of fish habitat (e.g. Portugal, Machado & Alves, 2001; Spain, Schmidt et al., 2001; Norway, Østdahl et al., 2001), suggesting that weir removal will always be a contentious issue (Grant, 2001).
Cost of weir removal
Costs of removing old, unused weirs are quite variable but in general are considerably lower than the cost of repairing the structure, or of building fish passages. Costs of removing weirs have been estimated to vary between 20,000 and 90,000 € in several projects in Spain reviewed by Brufao (2006). In the region of Cantabria, actual costs (1999 prices) ranged from just under 2,000 € for small weirs (<1 m) that were removed in a few days to 16,000 € for the demolition of a large weir (6.5 m) that took 3 weeks. In the United States, the cost of removing small weirs (max 10 feet in height) was $69,000 on average, or $23,000 per metre height (range $2,000–$126,000 per metre height) according to data provided in Heinz Center (2002). In general we have estimated that for small to medium sized weirs (up to 3 m in height), the cost of weir removal was typically less than 20% of the cost of building a Denil fish pass, and less than 12% of the cost of building a pool and weir fish ladder (de la Fuente & Araujo, 2001). Thus, weir removal will often be the most direct, cost-effective option for improving fish passage, and for eliminating the negative effects of low-head dams on stream integrity (Hart et al., 2002).
For some weirs, particularly the highest ones, safety considerations should also be factored in when costing dam removal, as dam removal typically costs only a fraction of the costs of repairing an unsafe dam (Hjorth, 2001). Moreover, even if these costs are comparable, dam removal eliminates the need (and cost) for continued maintenance and repairs in the future. In Wisconsin (USA), the cost of removing low-head dams was 20–50% of the estimated repair costs (American Rivers, 1999).
Decision-making and prioritisation in the removal of weirs
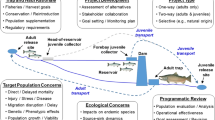
Not all barriers can be removed easily, thus some form of evaluation criteria and prioritization strategy will be required to optimize weir removal (Pejchar & Warner, 2001). One commonly used prioritizing method employs a scoring and ranking approach (e.g. Roni et al., 2002; Clarkin et al., 2005) which is easily implemented in most situations and which can benefit from the availability of specialized software such as FishXing (Love, 1999). An alternative decision-making approach for weir removal which does not rely on subjective scoring has recently been developed by O’Hanley & Tomberlin (2005) based on dynamic programming methods.
A simple decision flow chart adopted to demolish unused weirs in the salmon rivers of N. Spain is shown in Fig. 8. The process begins with a field inventory of all obstacles in target watersheds, including the main stems of river and their tributaries. The inventory provides information on the location, characteristics and impact of each barrier based on expert knowledge combined with an in-situ impact assessment based on weir height, crest profile, existence of a holding pool, distance to river mouth, size of impoundment, weir use, water abstraction and ease of fish passage amongst other criteria (Table 2). From this, weirs are classified as limiting or not limiting depending on their estimated impact. Weirs which are deemed to cause a significant impact on salmon populations are then studied in more detail, and information on their current use and legal status is sought from the relevant authorities. Those weirs which are abandoned or in ruins are singled out and a legal case can be initiated for their decommissioning, and eventually, their removal. Those weirs which lack concessions or those whose concessions have expired can also be singled out for removal. Only in those cases where the weir causes an important impact and cannot be removed are other alternatives explored, typically involving the provision or modification of fish passes or the setting of compensatory flows.
Conclusions
Thousands of low-head dams and weirs, many of them in ruins, litter the salmon rivers of the Iberian Peninsula and probably those elsewhere in Europe. While a few of these structures are listed and may retain some archaeological value, most do not. They cause a substantial impact on stream ecosystems, particularly amongst migratory fish.
Results from the Iberian Peninsula and elsewhere show that even relatively small weirs can delay or completely block the passage of upstream and downstream migrants. They also facilitate predation, poaching and overexploitation of salmonid populations, and exacerbate the spread of infectious diseases due to overcrowding and stress during low flows.
The removal of weirs offers considerable advantages over other solutions (typically the provision of fish passage facilities) since it solves simultaneously both upstream and downstream fish passage problems, something difficult or impossible to accomplish by other means (Larinier, 2003). In addition, the cost of weir removal is typically a fraction of the cost of building fish passes or repairing unsafe or old weirs. Weir removal is often the most cost effective option for salmonid river restoration, as it achieves direct, integral stream restoration and does not hinder future options (Peters & Marmorek, 2001; Peters et al., 2001).
However, weir removal also has limitations. It may not always be practical or feasible. Some river systems may take a long time to recover, or may not recover fully, because long-term changes caused by dams may not always be reversible (Doyle et al., 2005). The impact caused by the sudden mobilization of sediments, some potentially toxic, also needs to be taken into account. There is still limited experience in Europe, particularly when compared to the construction of fish passes. Societal and cultural issues need to be considered, while bureaucracy and red tape may mean that the decommissioning process may take a long time. Nevertheless, with many dams in Spain and elsewhere rapidly approaching their life span, increasingly large structures will likely be decommissioned in the next few years (Fig. 9), offering unparallel opportunities for river restoration at an unprecedented scale.
References
Aarestrup, K. & A. Koed, 2003. Survival of migrating sea trout Salmo trutta and Atlantic salmon Salmo salar smolts negotiating weirs in small Danish rivers. Ecology of Freshwater Fish 12: 169–176.
Alvarez, J. & M. Lamuela, 2001. Situación actual del salmón atlántico en Navarra. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 97–110.
Alvarez, J., I. Urrizalki, I. Mendiola, C. Rodriguez, C. Garcia de Leániz, S. Consuegra & A. Serdio, 2003. Densidad de obstáculos y su incidencia sobre el área utilizada por el salmón en los ríos ibéricos. In Lamuela, M. & J. Alvarez (eds), Un Viaje de Ida y Vuelta. IV Jornadas del Salmón Atlántico en la Península Ibérica. Dpto. de Medio Ambiente, Ordenación del Territorio y Vivienda. Gestión Ambiental—Viveros y Repoblaciones de Navarra, Pamplona: 177–186.
American Rivers, 1999. Dam removal success stories. Restoring rivers through selective removal of dams that don’t make sense. Report Prepared in Cooperation with Trout Unlimited and Friends of the Earth. http://www.amrivers.org/successstories.html 12/14/99.
American Rivers, 2002. The Ecology of Dam Removal. A Summary of Benefits and Impacts. American Rivers, Washington, DC.
American Rivers, 2005. Dams Slated for Removal in 2005 and Dams Removed from 1999–2004. American Rivers, Washington, DC.
Arenillas, M. & J. C. Castillo, 2003. Dams from the Roman era in Spain. Analysis of design forms. TRAIANVS. 1st International Congress on Construction History, Madrid.
Ashley, J. T. F., K. Bushaw-Newton, M. Wilhelm, A. Boettner, G. Drames & D. J. Velinsky, 2006. The effects of small dam removal on the distribution of sedimentary contaminants. Environmental Monitoring and Assessment 114: 287–312.
Babbitt, B., 2002. What goes up, may come down. BioScience 52: 656–658.
Baumgartner, L. J., 2007. Diet and feeding habits of predatory fishes upstream and downstream of a low-level weir. Journal of Fish Biology 70: 879–894.
Bednarek, A. T., 2001. Undamming rivers: A review of the ecological impacts of dam removal. Environmental Management 27: 803–814.
Berg, M., B. Abrahamsen & O. K. Berg, 1986. Spawning of injured compared to uninjured female Atlantic salmon, Salmo salar L. Aquaculture & Fisheries Management 17: 195–199.
Brufao, P., 2001. Aprovechamientos hidráulicos y conservación de los ecosistemas fluviales: consideraciones legales. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 181–186.
Brufao, P., 2006. La demolición de presas y otras obras hidráulicas en España. Quercus 241.
Brufao, P. & C. Rodriguez, 2003. Desmantelamiento de presas y otras obras hidráulicas. In Lamuela, M. & J. Alvarez (eds), Un Viaje de Ida y Vuelta. IV Jornadas del Salmón Atlántico en la Península Ibérica. Dpto. de Medio Ambiente, Ordenación del Territorio y Vivienda. Gestión Ambiental—Viveros y Repoblaciones de Navarra, Pamplona: 205–214.
Carss, D. N., H. Kruuk & J. W. H. Conroy, 1990. Predation on adult Atlantic salmon, Salmo salar L., by otters, Lutra lutra L., within the River Dee system, Aberdeenshire, Scotland. Journal of Fish Biology 37: 935–944.
Catalano, M. J., M. A. Bozek & T. D. Pellett, 2001. Fish-habitat relations and initial response of the Baraboo River fish community to dam removal. Bulletin of the North American Benthological Society 18: 177.
Clarkin, K., A. Connor, M. Furniss, B. Gubernick, M. Love, K. Moynan & S. W. Musser, 2005. National inventory and assessment procedure for identifying barriers to aquatic organism passage at road-stream crossings. U.S. Department of Agriculture Forest Service, National Technology and Development Program.
Consuegra, S., C. García de Leániz & A. Serdio, 2003. Estado sanitario del Salmón atlántico en Cantabria: desarrollo y aplicación de métodos moleculares. In Lamuela, M. & J. Alvarez (eds), Un Viaje de Ida y Vuelta. IV Jornadas del Salmón Atlántico en la Península Ibérica. Dpto. de Medio Ambiente, Ordenación del Territorio y Vivienda. Gestión Ambiental—Viveros y Repoblaciones de Navarra, Pamplona: 123–134.
Consuegra, S., C. García de Leániz, A. Serdio & E. Verspoor, 2005. Selective exploitation of early running fish may induce genetic and phenotypic changes in Atlantic salmon. Journal of Fish Biology 67: 130–146.
De Gaudemar, B., S. L. Schroder & E. P. Beall, 2000. Nest placement and egg distribution in Atlantic salmon redds. Environmental Biology of Fishes 57: 37–47.
de la Fuente, J. & J. M. Araujo, 2001. Mejora de la accesibilidad en pequeños azudes. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 251–268.
Dieperink, C., S. Pedersen & M. I. Pedersen, 2001. Estuarine predation on radiotagged wild and domesticated sea trout Salmo trutta L. smolts. Ecology of Freshwater Fish 10: 177.
Doyle, M. W., E. H. Stanley, J. M. Harbor & G. S. Grant, 2003. Dam removal in the United States: Emerging needs for Science and Policy. EOS 84: 29–33.
Doyle, M. W., E. H. Stanley, M. A. Luebke & J. M. Harbor, 2000. Dam removal: Physical, biological, and societal considerations. American Society of Civil Engineers. Joint Conference on Water Resources Engineering and Water Resources Planning and Management, Minneapolis, MN.
Doyle, M. W., E. H. Stanley, C. H. Orrb, A. R. Sellec, S. A. Sethib & J. M. Harbor, 2005. Stream ecosystem response to small dam removal: Lessons from the heartland. Geomorphology 71: 227–244.
Epple, R., 2000. Dam decommissioning in France. World Commission on Dams Report. http://www.dams.org/kbase/submissions/showsub.php?rec=OPT136.
ERN, 2006. European Rivers Network. The Loire-Allier salmon in France: A survivor. http://www.rivernet.org/general/salmon/saumo1_e.htm.
Frissell, C. A., 1993. Topology of extinction and endangerment of native fishes in the Pacific Northwest and California U.S.A. Conservation Biology 7: 342–354.
Garcia de Leaniz, C., D. W. Forman, S. Davies & A. Thomson, 2006. Non-intrusive monitoring of otters (Lutra lutra) using infrared technology. Journal of Zoology (London) 270: 577–584.
Garcia de Leaniz, C., A. D. Hawkins, D. W. Hay & J. J. Martínez, 1987. The Atlantic Salmon in Spain. The Atlantic Salmon Trust, Pitlochry.
García de Leániz, C., A. Serdio & S. Consuegra, 2001. Situación actual del salmón atlántico en Cantabria. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 55–82.
Geist, D. R., R. S. Brown, V. I. Cullinan, M. G. Mesa, S. P. Vanderkooi & C. A. McKinstry, 2003. Relationships between metabolic rate, muscle electromyograms and swim performance of adult chinook salmon. Journal of Fish Biology 63: 970–989.
Gerlier, M. & P. Roche, 1998. A radio telemetry study of the migration of Atlantic salmon Salmo salar L. and sea trout Salmo trutta trutta L. in the upper Rhine. Hydrobiologia 371/372: 283–293.
Gosset, C., J. Rives & J. Labonne, 2006. Effect of habitat fragmentation on spawning migration of brown trout Salmo trutta L. Ecology of Freshwater Fish 15: 247–254.
Gowans, A. R. D., J. D. Armstrong & I. G. Priede, 1999. Movements of adult Atlantic salmon in relation to a hydroelectric dam and fish ladder. Journal of Fish Biology 54: 713–726.
Grant, G., 2001. Dam removal: Panacea or Pandora for rivers? Hydrological Processes 15: 1531–1532.
Gudjónsson, T., 1988. Exploitation of Atlantic salmon in Iceland. In Mills, D. & D. Piggins (eds), Atlantic Salmon: Planning for the Future. Croom Helm, London: 162–178.
Guitart, R & V. G. Thomas, 2005. Is lead used in sports hunting, shooting and angling an underestimated public health problem? Revista Española de Salud Pública 79: 621–632.
Hart, D. D., T. E. Johnson, K. L. Bushaw-Newton, R. J. Horwitz, A. T. Bednarek, D. F. Charles, D. A. Kreeger & D. J. Velinsky, 2002. Dam removal: Challenges and opportunities for ecological research and river restoration. BioScience 52: 669–681.
Hart, D. D. & N. L. Poff, 2002. A special section on dam removal and river restoration. BioScience 52: 653–655.
Havel, J. E., C. E. Lee & M. J. V. Zanden, 2005. Do reservoirs facilitate invasions into landscapes? BioScience 55: 518–525.
Heggenes, J. & K. H. Røed, 2006. Do dams increase genetic diversity in brown trout Salmo trutta? Microgeographic differentiation in a fragmented river. Ecology of Freshwater Fish 15: 366–375.
Heinz Center, 2002. Dam Removal. Science and Decision Making. The Heinz Center, Washington.
Hjorth, P., 2001. Operation, Monitoring and Decommissioning of Dams. World Commission on Dams. Thematic Review IV.5: Operation, Monitoring and Decommissioning of Dams.
Ibbotson, A., J. Smith, P. Scarlett & M. Aprahamian, 2002. Colonisation of freshwater habitats by the European eel Anguilla anguilla. Freshwater Biology 47: 1696–1706.
Johnson, S. E. & B. E. Graber, 2002. Enlisting the social sciences in decisions about dam removal. BioScience 52: 731–738.
Jormola, J., 2001. Problems and possibilities of lowland river restoration in boreal circumstances. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 313.
Karppinen, P., T. S. Mäkinen, J. Erkinaro, V. V. Kostin, R. V. Sadkovskij, A. I. Lupandin & M. Kaukoranta, 2002. Migratory and route-seeking behaviour of ascending Atlantic salmon in the regulated River Tuloma. Hydrobiologia 483: 23–30.
Kennedy, G. J. A. & J. E. Greer, 1988. Predation by cormorants Phalacrocorax carbo on the salmonid populations of an Irish river. Aquaculture and Fisheries Management 19: 159–170.
Kondolf, G. M., 1997. Hungry water: Effects of dams and gravel mining on river channels. Environmental Management 21: 533–551.
Kondolf, G. M., 2000. Assessing salmonid spawning gravel quality. Transactions of the American Fisheries Society 129: 262–281.
Kondolf, G. M., 2001. Historical changes to the San Francisco bay-delta watershed: Implications for ecosystem restoration. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 327–338.
Larinier, M., 2001. Environmental issues, dams and fish migration. In Marmulla, G. (ed.), FAO Fisheries Technical Paper 419. Dams, Fish and Fisheries. Opportunities, Challenges and Conflict Resolution. FAO, Rome: 45–89.
Larinier, M., 2003. L’expérience française dans le domaine du rétablissement de la libre circulation des poissons à la montaison et à la dévalaison. In Lamuela, M. & J. Alvarez (eds), Un Viaje de Ida y Vuelta. IV Jornadas del Salmón Atlántico en la Península Ibérica. Dpto. de Medio Ambiente, Ordenación del Territorio y Vivienda. Gestión Ambiental—Viveros y Repoblaciones de Navarra, Pamplona: 171–176.
Levin, P. S. & N. Tolimieri, 2001. Differences in the impacts of dams on the dynamics of salmon populations. Animal Conservation 4: 291–299.
Lewis, D., 2001. Rescuing a fragile river from over-exploitation—Australia’s River Murray. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 319–326.
Love, M., 1999. FishXing Version 2.1. FishXing Project. Six River National Forest Watershed Interactions Team, USA.
Machado, T. F. & M. H. Alves, 2001. Rehabilitation of a river in an urban area, the Jamor River case. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 277–280.
Marmulla, G., 2001. Dams, Fish and Fisheries. Opportunities, challenges and conflict resolution. FAO Fisheries Technical Paper 419. Dams, Fish and Fisheries. Opportunities, Challenges and Conflict Resolution. FAO, Rome: 1–166.
Martín Ventura, J. A., 1988. The Atlantic salmon in Asturias, Spain: Analysis of catches, 1985–86. Inventory of juvenile densities. In D. Mills & D. Piggins (eds), Atlantic Salmon: Planning for the Future. Croom Helm, London: 210–227.
Márquez, I., 1999. Patógenos de salmónidos detectados en el Principado de Asturias. I Semana del Salmón Atlántico en la Península Ibérica. Libro de Ponencias y Comunicaciones, Oviedo: 135–139.
Meadows, R., 2001. Turning a radical idea into reality: Removing Edwards Dam in Augusta, Maine. Conservation Biology in Practice 2: 32–35.
Meehan, W. R. (ed.), 1991. Influences of Forest and Rangeland Management on Salmonid Fishes and their Habitats. American Fisheries Society, Bethesda, Maryland.
Meldgaard, T., E. E. Nielsen & V. Loeschcke, 2003. Fragmentation by weirs in a riverine system: A study of genetic variation in time and space among populations of European grayling Thymallus thymallus in a Danish river system. Conservation Genetics 4: 735–747.
Mills, D. H., 1989. Ecology and Management of Atlantic Salmon. Chapman and Hall, London.
Morita, K. & S. Yamamoto, 2002. Effects of habitat fragmentation by damming on the persistence of stream-dwelling charr populations. Conservation Biology 16: 1318–1323.
Naughton, G. P., C. C. Caudill, M. L. Keefer, T. C. Bjornn, L. C. Stuehrenberg & C. A. Peery, 2005. Late-season mortality during migration of radio-tagged adult sockeye salmon Oncorhynchus nerka in the Columbia River. Canadian Journal of Fisheries and Aquatic Sciences 62: 30–47.
Netboy, A., 1968. The Atlantic Salmon, a Vanishing Species? Faber and Faber, London.
Netboy, A., 1974. The Salmon, their Fight for Survival. Andre Deutsch, London.
Nijland, H. J. & M. J. R. Cals, (eds), 2001. River Restoration in Europe. Conference on River Restoration, Practical Approaches. Riza Rapport Nr.: 2001.023.
Nilsson, C., C. A. Reidy, M. Dynesius & C. Revenga, 2005. Fragmentation and flow regulation of the world’s large river systems. Science 308: 405–408.
O’Connor, J. P., D. J. O’Mahony, J. M. O’Mahony & T. J. Glenane, 2006. Some impacts of low and medium head weirs on downstream fish movement in the Murray-Darling Basin in southeastern Australia. Ecology of Freshwater Fish 15: 419–427.
O’Hanley, J. R. & D. Tomberlin, 2005. Optimizing the removal of small fish passage barriers. Environmental Modeling and Assessment 10: 85–98.
Økland, F., J. Erkinaro, K. Moen, E. Niemelä, P. Fiske, R. S. McKinley & E. B. Thorstad, 2001. Return migration of Atlantic salmon in the River Tana: Phases of migratory behaviour. Journal of Fish Biology 59: 862–874.
Østdahl, T., T. Taugbøl, O. Andersen & J. Vittersø, 2001. Stakeholder attitudes to river modification and restoration. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 315.
Pejchar, L. & K. Warner, 2001. A river might run through it again: Criteria for consideration of dam removal and interim lessons from California. Environmental Management 28: 561–575.
Peters, C. N. & D. R. Marmorek, 2001. Application of decision analysis to evaluate recovery actions for threatened Snake River spring and summer chinook salmon Oncorhynchus tshawytscha. Canadian Journal of Fisheries and Aquatic Sciences 58: 2431–2446.
Peters, C. N., D. R. Marmorek & R. B. Deriso, 2001. Application of decision analysis to evaluate recovery actions for threatened Snake River fall chinook salmon Oncorhynchus tshawytscha. Canadian Journal of Fisheries and Aquatic Sciences 58: 2447–2458.
Pielou, E. C., 1998. Freshwater. The University of Chicago Press, Chicago.
Raymond, H. L., 1979. Effects of dams and impoundments on migration of juvenile chinook salmon and steelhead from the Snake River, 1966–1975. Transactions of the American Fisheries Society 109: 509–525.
Redeker, M., M. Weyand & E. A. Nusch, 2004. Restoration of fish passage and results of a master plan established for the Ruhr River Basin. 3rd European Conference on River Restoration. Zagreb, Croatia: 305–313.
Reyes-Gavilán, F. G., R. Garrido, A. G. Nicieza, M. M. Toledo & F. Braña, 1996. Fish community variation along physical gradients in short streams of northern Spain and the disruptive effect of dams. Hydrobiologia 321: 155–163.
Riber, H. H., 2001. Skjern River restoration project. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 209–214.
Rivinoja, P., 2005. Migration problems of Atlantic salmon Salmo salar L. in flow regulated rivers. PhD thesis, Swedish University of Agricultural Sciences, Umeå.
Robertson, C. T., 2005. Conservation of endangered Atlantic salmon in Maine. PhD thesis, Department of Zoology, University of Toronto, Toronto.
Roni, P., T. J. Beechie, R. E. Bilby, F. E. Leonetti, M. M. Pollock & G. R. Pess, 2002. A review of stream restoration techniques and a hierarchical strategy for prioritizing restoration in Pacific Northwest watersheds. North American Journal of Fisheries Management 22: 1–20.
Schmidt, G., M. Otoala-Urrutxi, A. Fernández-Lop & B. Gutiérrez-Monzonís, 2001. River restoration practice in Spain. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 309.
Serdio, A., C. García de Leániz & S. Consuegra, 2001a. Efectos de los azudes sobre las poblaciones de salmón Atlántico. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 237–250.
Serdio, A., C. García de Leániz & S. Consuegra, 2001b. Estrategia de recuperación del río Nansa como hábitat salmonero. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 143–153.
Serdio, A., C. García de Leániz & S. Consuegra, 2003. Depredadores piscívoros en los ríos de Cantabria y su efecto sobre poblaciones amenazadas de salmón Atlántico. In Lamuela, M. & J. Alvarez (eds), Un Viaje de Ida y Vuelta. IV Jornadas del Salmón Atlántico en la Península Ibérica. Dpto. de Medio Ambiente, Ordenación del Territorio y Vivienda. Gestión Ambiental—Viveros y Repoblaciones de Navarra, Pamplona: 43–54.
Solomon, D. J. & H. T. Sambrook, 2004. Effects of hot dry summers on the loss of Atlantic salmon, Salmo salar, from estuaries in South West England. Fisheries Management and Ecology 11: 353–363.
Solomon, D. J., H. T. Sambrook & K. J. Broad, 1999. Salmon migration and river flow. Results of tracking radio tagged salmon in six rivers in South West England. Research & Development Publication 4, Environment Agency, Almondsbury.
Stanley, E. H. & M. W. Doyle, 2003. Trading off: The ecological effects of dam removal. Frontiers in Ecology and the Environment 1: 15–22.
Stanley, E. H., M. A. Luebke, M. W. Doyle & D. W. Marshall, 2002. Short-term changes in channel form and macroinvertebrate communities following low-head dam removal. Journal of the North American Benthological Society 21: 172–187.
Steen, R. P. & T. P. Quinn, 1999. Egg burial depth by sockeye salmon Oncorhynchus nerka: Implications for survival of embryos and natural selection on female body size. Canadian Journal of Zoology 77: 836–841.
Steinbach, P., 2001. Situation and restoration of amphihalin migratory fishes in the Loire Basin. Bulletin Français de la Pêche et la Pisciculture 357–360: 263–276.
Summers, D. W., 1996a. Differences in the time of river entry of Atlantic salmon, Salmo salar L., spawning in different parts of the River North Esk. Fisheries Management and Ecology 3: 209–218.
Summers, D. W., 1996b. Environmental influences on the timing of spawning of Atlantic salmon, Salmo salar L., in the River North Esk. Fisheries Management and Ecology 3: 281–283.
Taggart, J. B., I. S. McLaren, D. W. Hay, J. H. Webb & A. F. Youngson, 2001. Spawning success in Atlantic salmon (Salmo salar L.): A long-term DNA profiling-based study conducted in a natural stream. Molecular Ecology 10: 1047–1060.
Tamés, P., F. Alvarez, J. Puebla & D. San Emeterio, 2003. Actuaciones sobre la eliminación de obstáculos y construcción de pasos para peces en los ríos del territorio histórico de Gipuzkoa. In Lamuela, M. & J. Alvarez (eds), Un Viaje de Ida y Vuelta. IV Jornadas del Salmón Atlántico en la Península Ibérica. Dpto. de Medio Ambiente, Ordenación del Territorio y Vivienda. Gestión Ambiental—Viveros y Repoblaciones de Navarra, Pamplona: 197–204.
Thorstad, E. B. & T. G. Heggberget, 1998. Migration of adult Atlantic salmon Salmo salar; the effects of artificial freshets. Hydrobiologia 371/372: 339–346.
van Ast, J. A., 2000. Interactive management of international river basins; experiences in Northern America and Western Europe. Physics and Chemistry of the Earth B 25: 325–328.
Vaughn, C. C. & C. M. Taylor, 1999. Impoundments and the decline of freshwater mussels: A case study of an extinction gradient. Conservation Biology 13: 912–920.
Velinsky, D. J., K. L. Bushaw-Newton, D. A. Kreeger & T. E. Johnson, 2006. Effects of small dam removal on stream chemistry in southeastern Pennsylvania. Journal of the North American Benthological Society 25: 569–582.
Williams, J., 1998. Fish passage in the Columbia River, USA, its tributaries: Problems and solutions. In Jungwirth, M., S. Schmutz & S. Weiss (eds), Fish Migration and Fish Bypasses. Fishing News Book, University Press, Cambridge: 180–191.
WWF, 2001. The Status of Wild Atlantic Salmon: A River by River assessment. WWF.
WWF Australia, 2004. Weir Removal, a New Option for Rehabilitating our Rivers. WWF Australia, Sydney, New South Wales.
WWF Australia, 2005. Duck Creek Crossing Removal Case Study. WWF Australia, Sydney, New South Wales.
Youngson, A. F. & D. W. Hay, 1996. The Lives of Salmon. Swan-Hill Press, Shrewsbury.
Zacharias, I., 2001. An alternative to conventional river management in Greece. In Nijland, H. J. & M. J. R. Cals (eds), River Restoration in Europe. Conference on River Restoration, Practical Approaches Riza Rapport Nr.: 2001.023: 303–307.
Zataraín, J., 2001. El “hábitat jurídico” del salmón en España. In García de Leániz, C., A. Serdio & S. Consuegra (eds), El Salmón, Joya de Nuestros Ríos. Consejería de Ganadería, Agricultura y Pesca, Santander: 173–180.
Acknowledgements
Special thanks are due to all the people who helped and supported the weir demolition projects discussed here, in particular Paco Vaamonde, Pablo Caballero, Manolo Rego, Jesús de la Fuente, José Manuel Araujo, Angel Serdio, Sonia Consuegra, Pedro Brufao and César Rodríguez. Sonia Consuegra and two anonymous referees made useful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: R. L. Welcomme & G. Marmulla
Hydropower, Flood Control and Water Abstraction: Implications for Fish and Fisheries
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garcia de Leaniz, C. Weir removal in salmonid streams: implications, challenges and practicalities. Hydrobiologia 609, 83–96 (2008). https://doi.org/10.1007/s10750-008-9397-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-008-9397-x