Abstract
The maximum quantum yield of photosystem II was estimated from variable chlorophyll a fluorescence in samples of phytoplankton collected from the Taihu Lake in China to determine the responses of different phytoplankters to irradiance and vertical mixing. Meteorological and environmental variables were also monitored synchronously. The maximum quantum yield of three phytoplankton groups: cyanobacteria, chlorophytes, and diatoms/dinoflagellates, showed a similar diurnal change pattern. F v/F m decreased with a significant depth-dependent variation as irradiance increased during the morning and increased as irradiance declined in the afternoon. Furthermore, the rates of F v/F m depression were dependent upon the photon flux density, whereas the rates of recovery of F v/F m were dependent upon the historical photon density. Moreover, photoinhibition affected the instantaneous growth rates of phytoplankton. Although at noon cyanobacteria had a higher photoinhibition value (up to 41%) than chlorophytes (32%) and diatoms/dinoflagellates (34%) at the surface, no significant difference in diurnal growth rates among the three phytoplankton groups were observed indicating that cyanobacteria could photoacclimate better than chlorophytes and diatoms/dinoflagellates. In addition, cyanobacteria had a higher nonphotochemical quenching value than chlorophytes and diatoms/dinoflagellates at the surface at noon, which indicated that cyanobacteria were better at dissipating excess energy. The ratios of enclosed bottle samples F v/F m to free lake samples F v/F m showed different responses for the three phytoplankton groups to irradiance and vertical mixing when wind speed was approximately constant at about 3.0 m s−1. When wind speed was lower than 3.0 m s−1, cyanobacteria accumulated mainly at the surface and 0.3 m, because of their positive buoyancy, where diurnal growth rates of phytoplankton were relatively higher than those at 0.6 m and 0.9 m. Chlorophytes were homogenized completely by vertical mixing, while diatoms/dinoflagellates avoided active high irradiance by moving downward at noon, and then upward again when irradiance decreased. These results explain the dominance of cyanobacteria in Taihu Lake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The interception and utilization of light by phytoplankters are among the complex processes determining net productivity, species succession, and the abundance of higher trophic order organisms in the pelagic zone (Brookes et al., 2003). The availability of light is determined by solar insolation and the optical properties of the water body. Light interception is mainly a function of antenna pigments of the phytoplankters in the water column, which are different among different phytoplankters and are also critical factors affecting their light interception and utilization, even further species composition. In addition, light interception is also partially affected by the vertical positions of the phytoplankters in the water column, which is in turn determined by mixed characteristics of phytoplankton, especially, in a shallow lake waterbody where turbulent mixing is more frequent. Phytoplanktons are often exposed to a widely fluctuating light environment and exhibit a broad range of physiological acclimation strategies. These acclimation strategies allow the cells to maximize photosynthetic rates at light levels that range from limiting to photodamaging (Cullen &Lewis, 1988; Cullen & MacIntyre, 1998).
Generally, phytoplankton cells exposed to high light intensities show a photoinhibitory decline in the quantum efficiency of photosynthesis, the decline being a function of both irradiance intensity and time (Long et al., 1994; Anderson et al., 1997; Oliver et al., 2003). Furthermore, when phytoplanktons are grown under different irradiance conditions, photoinhibitory extent can change through light adaptation or acclimation progress. The extent depends on the kinetics of the onset and dissipation of inhibition in response to irradiance and its attenuation with depth, and the time and depth scales of vertical mixing (Oliver et al., 2003). In stratified waterbodies, especially, the degree of photoinhibition observed in the natural phytoplankton assemblages is strongly influenced by the extent of vertical mixing in the surface layer. Wind-induced mixing will disrupt the photochemical responses to light (Brookes et al., 2003; Oliver et al., 2003).
The photochemical responses of phytoplankton were mainly evaluated by either classical (14C-uptake or O2 evolution) or recently developed methods on the basis of chlorophyll a fluorescence. Furthermore, the analysis of variable chlorophyll a fluorescence yield has become an important tool for studying photosynthesis in natural phytoplankton populations (Mauzerall, 1972; Schreiber, 1986, 1994; Falkowski & Kolber, 1993; Oliver & Whittington, 1998), which has also made field measurements of fluorescence yields and investigating photoinhibition possible (Anderson et al., 1997; Heraud & Beardall, 2000) because it is sensitive to photon flux density and is reliable as a parameter to offer insight into the immediate past light history of phytoplankton. The technique provides information on the major processes in light capture and electron transport, which together provide an estimate of the rate of photosynthesis (Regel et al., 2004). The ratio of the maximum variable fluorescence to the maximum yield (F v/F m), where the maximum variable fluorescence F v is the difference between the maximum (F m) and minimum (F 0) fluorescence yields, provides a measure of the efficiency of excitation capture by active photosystem II (PSII) reaction centers (Genty et al., 1989; Kolber & Falkowski, 1993). This ratio has been used to estimate changes in the proportion of functional reaction centers and as an indicator of the photosynthetic capacity of phytoplankton (Falkowski & Kolber, 1995).
The evaluation of photosynthetic capacity performed by methods on the basis of chlorophyll a fluorescence has shown wide variations among algal species. For example, the maximal PSII quantum yield, F v/F m, which is proportional to maximal photochemical efficiency, can change from 0.4 to 0.8 for different phytoplankton classes (Buechel & Wilhelm, 1993). Chlorophytes such as Dunaliella tertiolecta and Dunaliella salina have F v/F m values around 0.8, whereas the F v/F m values for diatoms Phaeodactylum tricornutum and Thalassiosira weisfflogii were shown to be between 0.6 and 0.7 (Ting & Owens, 1992; Geel et al., 1997; Casper-Lindley & Björkman, 1998; Lippemeier et al., 2001). Even under the same laboratory conditions, some variations (about 15%) in F v/F m were also detected among different algal species (Juneau & Harrison, 2005). These variations were explained to be the result of different functional and structural properties for the photosynthetic apparatus of different species (Wilhelm, 1990; Buechel & Wilhelm, 1993). In the field, furthermore, the distinctions among species might contribute to the composition of phytoplankton. F v/F m of phytoplankton mixed sample showed depth-dependent decreases as irradiance increased during the morning and increases as irradiance declined in the afternoon. Marine phytoplanktons in surface waters also show midday reductions in F v/F m that are associated with both photoprotective strategies in the antennae of PSII and photodamage of PSII reaction centers. In addition, phytoplankton cells may migrate toward the surface in the morning and descend later in the day to avoid photoinhibition and exhibit a high F v/F m by regulating buoyancy (Blasco, 1978; Harris et al., 1979; Villarino et al., 1995; Whittington et al., 2000). Furthermore, some studies have been done on Microcystis aeruginosa and Peridinium cinctum by linking phytoplankton cell distribution, vertical migration, and photosynthesis using F v/F m as an indicator of phytoplankton’s immediate past light history (Brookes et al., 2003; Regel et al., 2004). Therefore, study of the different photochemical responses to irradiance density and vertical mixing among algal species could facilitate the understanding of how different algal species can contribute to primary productivity in aquatic ecosystems.
The aim of this study was (i) to describe the diurnal change patterns of maximum quantum yield (F v/F m) among different freshwater phytoplankters in Taihu Lake situ, a large subtropical shallow lake; (ii) to estimate different responses of phytoplankters to photoinhibition and vertical mixing by relating these to meteorological influences and mixing conditions, and (iii) to explore the relationships between photochemical response and species composition.
Methods
Lake and site descriptions
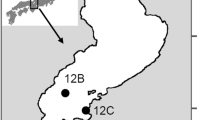
Taihu Lake is located in southeast Jiangsu Province, approximately 100 km west of Shanghai (latitude: 30°55′40″–31°32′58″ N; longitude: 119°52′32″–120°36′10″ E). The lake has a surface area of 2,338 km2, a maximum depth of 2.6 m, an average depth of 1.9 m, and mean water residence time about 264 days. Taihu Lake receives inflows from nearby riverine networks, including over 200 streams, canals, and rivers, mainly Liangxi and Lujiang Rivers. Meiliang Bay is highly eutrophic (Chen et al., 2003). In the past few years, annual Microcystis aeruginosa blooms are initiated in late May or early June. The field site (31°25′53.4″ N, 120°10′42.5″ E) is in an area where annual blooms appear and is between Meiliang Bay and offshore regions of Taihu Lake (Fig. 1).
Sampling and analysis
The experiments were carried out three times between 22 and 24 August 2006. Environmental parameters in the field site, including temperature, dissolved oxygen (DO), pH, oxidation and reduction potential (ORP), and conductivity were monitored in situ on site using a multiparameter meter (model 6600; Yellow Spring Instruments, USA).
To determine the influence of light and vertical mixing on photochemistry of natural phytoplankton community, bottle experiments were performed to assess the variable fluorescence throughout the course of 1 day in static samples incubated in the lake. Integrated samples (surface to 1 m), from the Taihu Lake, were collected prior to dawn and filtered out zooplankton, and then poured into 250 ml PET bottles and suspended at 0, 0.3, 0.6, and 0.9 m depths with three replicates. Photo flux density (PAR 400–720 nm) was measured at each depth with an underwater spectroradiometer (SR9910-PC, Macam Photometrics Ltd., Livingston, Scotland, UK). At 2-h intervals a subsample was taken from each bottle and from the adjacent water column at corresponding depths. The bottled samples showed the response of the population to the vertical light gradient without the influence of physical mixing or migrating cells. The free lake samples were subjected to mixing and vertical migration, and the difference between the two samples was used to infer the influence of mixing and/or migration. Fluorescence was measured using a multiwavelength phytoplankton pulse-amplitude-modulated fluorometry (Phyto-PAM) (Walz, Effeltrich, Germany) equipped with a special Emitter-Detector Unit Phyto-ED. The Phyto-PAM fluorometry can distinguish between differently pigmented algal groups, such as cyanobacteria, chlorophytes, and diatoms/dinoflagellates, by applying four different excitation wavelengths (665, 645, 520, and 470 nm). For example, in chlorophytes chlorophyll fluorescence is much more effectively excited by blue and red light (470, 645, and 665 nm) than by green light (520 nm). In the case of cyanobacteria, almost no chlorophyll fluorescence is excited by blue light (470 nm), while excitation at 645 nm is particularly strong due to phycocyanin and allophycocyanin absorption. On the other hand, in diatoms and dinoflagellates excitation by blue (470 nm) and green (520 nm) is relatively high due to strong absorption by fucoxanthin, chlorophyll c, and carotenoids. The fluorescence signals measured by the 4-wavelength excitation method carry the information to differentiate between the contributions of the main types of phytoplankton with different pigment systems. This allows a separate measurement of the fluorescence signal of each algal group in a mixture. Fluorescence signals from the four wavelengths are deconvoluted to the three algal groups, by inserting the best fitting reference spectra from the test species afterward, using the PHYTO-PAM software Phyto-WIN v1.47. Cells were dark-adapted for 15 min prior to analysis. F 0 was determined as the fluorescence of dark-adapted cells stimulated by a weak probe light immediately following 15 min of darkness. F m was the maximum fluorescence signal following the closure of all reaction centers by a 600-ms pulse of saturating irradiance. Simultaneously, F m′ was the maximum fluorescence signal in the light adapted state. Fluorescence parameters were calculated according to the following equations after subtraction of the blank fluorescence value obtained by measuring the fluorescence of a 0.22-μm filtered sample.
F v/F m is the maximum optical quantum yield (Juneau & Harrison, 2005) and NPQ is the nonphotochemical quenching value (Bilger & Björkman, 1990). Variations of chlorophyll concentrations in subsamples were also monitored on the basis of the current fluorescence yields, which were used to determine the growth rates of phytoplankton.
Monitoring of physical variable was undertaken at a station permanently deployed on the bank immediately near the Meiliang Bay. Wind speed and direction (VAISALA, WAV151, and WAA151) were measured 20 m above the water surface and the 2 min mean value was recorded. When the lake is not losing heat and wind speeds are low, the characteristic velocity of turbulence (u) within the diurnal surface layer can be equated to the water friction velocity u* estimated from the wind speed (Denman & Gargett, 1983):
where ρa is the density of air, ρ is water density, c is the dimensionless drag coefficient (1.3 × 10−3), and U 10 is the wind speed measured 10 m above the surface. For typical densities (ρa = 1.2 kg m−3: ρ = 1,000 kg m−3) the calculation of u* simplifies to 0.001U 10 (Oliver & Ganf, 2000). U 10 was calculated from the wind speed at 20 m (U 20) using the equation:
where Zo is 0.000115.
Statistical analysis
All data were analyzed with Origin 7.0 for Windows. Differences among means were determined using one-way analysis of variance (ANOVA). Differences were considered significant at P < 0.05. But in the present study, only the data on 24 August, which were more integrated than those on 22 and 23 August, were shown because the conditions and results among the 3 days were similar.
Results
Meteorology and environmental parameters
Temperature was recorded to be 29.87 ± 0.61°C, conductivity 0.52 ± 0.00 mS cm−1, dissolved oxygen 10.95 ± 1.25 mg l−1, oxidation reduction potential 12.78 ± 1.35 mV, and pH 9.48 ± 0.09 at the study site, without significant variations within 1-m depth in the whole day (P < 0.05). Surface irradiance presented a typical sinusoidal pattern (Fig. 2). Maximum photosynthetically active irradiance was 1,553 μmol m−2 s−1 at 13:00 h and day length was 12 h. Wind speed was maintained at 2.5–3.5 m s−1 for the whole day, which corresponded to a water friction velocity, u*, of 0.003–0.004 m s−1, except for a period between 17:00 and 20:00 h while maximum wind speed was up to 5.9 m s−1 and u* was 0.007 m s−1 (Fig. 3).
Diurnal changes in F v/F m of bottled and in situ lake samples
Fv/Fm of different types of phytoplankters were measured, respectively, and comparisons were performed between bottled and free lake samples during the day.
Fv/Fm of bottled samples
Cyanobacterial cells suspended in bottles at discrete depths above 0.9 m showed a light-dependent decrease in F v/F m until noon, and a vertical gradient became established as irradiance increased (Fig. 4a). F v/F m at the surface was similar to that at 0.3 m from 7:30 to 9:30 but significantly lower than those of the deeper samples (P < 0.05). Furthermore, from 9:30 to 19:30, F v/F m at the surface was remarkably lower than those of deeper samples (P < 0.05) and reached the lowest value at 13:30 when F v/F m declined by 41%. F v/F m at 0.3 m was lower (P < 0.05) than those of 0.6 m and 0.9 m from 7:30 to 15:30, and reached the lowest value at 11:30 which was similar to those of 0.6 m and 0.9 m. F v/F m at 0.6 m and 0.9 m were indistinguishable from each other throughout the day (P > 0.05). Cells suspended at all depths began to recover at 11:30 and reached to predawn F v/F m value at 15:30 ultimately, except F v/F m at the surface which recovered at 13:30 and did not reach to predawn F v/F m value before dusk.
Fv/Fm of chlorophytes at all depths decreased as irradiance increased til 11:30 when Fv/Fm increased (Fig. 4b). The highest inhibition rate, a 32% reduction in Fv/Fm, occurred at the surface. Fv/Fm at the surface was significantly lower than those of other depths on the whole day (P < 0.05), and from 7:30 to 17:30 Fv/Fm at 0.3 m was lower than those of 0.6 m and 0.9 m which were indistinguishable (P < 0.05). Cells suspended at different depths recovered as irradiance decreased. Only cells at the surface could not recover to predawn Fv/Fm value. Furthermore, recovered values of cells at 0.3, 0.6, and 0.9 m were higher than predawn Fv/Fm value.
Diatoms/dinoflagellates cells displayed a light-dependent decrease in F v/F m til 13:30 when F v/F m recovered (Fig. 4c). However, only F v/F m at 0.9 m recovered to the predawn value. F v/F m at the surface was lower than those from other depths from 7:30 to 17:30, and the lowest F v/F m occurred at 13:30, reducing by 34%. F v/F m at 0.3 and 0.6 m, which were similar, was lower than that at 0.9 m throughout the day (P < 0.05).
Fv/Fm of free lake samples
Fv/Fm of three types of phytoplankton in the lake samples showed a significant decrease as irradiance increased, and reached the lowest value at 11:30. From 11:30 onward, Fv/Fm of phytoplankton cells recovered gradually as irradiance decreased and attained to their initial dawn values ultimately (Fig. 5). Moreover, variations of Fv/Fm of three types of phytoplankton in depth gradient declined and almost disappeared except at 11:30.
Fv/Fm ratios of lake to bottle samples
Fv/Fm ratios of lake to bottle samples in cyanobacteria showed no significant differences at 0.6 m and 0.9 m over the whole day. However, the ratios at the surface and 0.3 m were significantly >1 on the whole day and from 7:30 to 15:30 (Fig. 6a). Fv/Fm ratio of lake to bottle in chlorophytes at the surface was significantly >1 all day long, and the ratios at 0.6 m and 0.9 m were remarkably <1 from 9:30 onward. The ratio of lake to bottle at 0.3 m was fluctuant near 1 with no obvious change (Fig. 6b). Fv/Fm ratios of lake to bottle in diatoms/dinoflagellates at 0.3 m, 0.6 m and 0.9 m had no significant change from 7:30 to 11:30. And the ratio at 0.9 m did not change again after 11:30. The ratios at 0.3 m and 0.6 m were remarkably >1 from 13:30 onward. The ratio at the surface was obviously >1 all day long except that no change occurred at 11:30 (Fig. 6c).
The ratio of the maximum quantum yield of cyanobacteria (a), chlorophytes (b), and diatoms/dinoflagellates (c) sampled from discrete depths to that suspended in bottles at different depths in the Taihu Lake, China, on 24 August 2006 (■ 0 m; ● 0.3 m; ▲ 0.6 m; ▼ 0.9 m, the vertical bars represent standard deviation)
NPQ of different algae at the surface
The nonphotochemical quenching (NPQ) of three types of phytoplankton was analyzed and compared at the surface where irradiance was the most intense (Fig. 7). NPQ of different algae displayed parallel variation patterns: NPQ rose as irradiance increased til 11:30 and then decreased as irradiance dropped. NPQ of cyanobacteria was significantly lower than those of chlorophytes and diatoms/dinoflagellates at 7:30 and 9:30, but obviously higher at 11:30 and no significant variation was found at other times (P < 0.05). Furthermore, there was no distinguishable difference between NPQ of chlorophytes and diatoms/dinoflagellates.
Growth rates of three types of phytoplankton
Diurnal growth rates among different types phytoplankton were found to be insignificant at the same depths (Fig. 8). But at the 0.3-m depth, three types of algae presented maximum diurnal growth rates, and the order of growth rates was 0.3 > 0 > 0.6 > 0.9 m. From 7:30 to 9:30, phytoplankton at the surface showed the highest growth rates, which decreased as irradiance increased, but did not significantly recover when irradiance decreased. From 17:30 to 19:30, growth rates further decreased as it became dark (Fig. 9).
Discussion
Phytoplankton in the waters of Taihu Lake showed significant diurnal variation in the maximum effective quantum yield of PSII, as estimated, in our study, from measurements of the fluorescence variable F v/F m. Additionally, changes in F v/F m with depth were found to be linked to irradiance attenuation at different depths. Similar diurnal changes in F v/F m have been reported for surface populations of marine (Kolber et al., 1990; Greene et al., 1994; Vassiliev et al., 1994) and freshwater phytoplankton (Oliver et al., 2003). When vertical mixing is restricted, the diurnal variability seen in F v/F m is dependent upon the incoming irradiance; midday depressions reflect the light saturation of the photosynthetic reaction centers and subsequent oxidation of the plastoquinone pool (Falkowski & Raven, 1997). Historically, midday decreases in F v/F m have been linked to the inactivation of the photosystem II reaction centers. Despite protective mechanisms, high irradiance will still damage some PSII reaction centers which can then lead to a reduction in carbon fixation (Renger et al., 1989; Jordan, 1996) and lower growth rate that was confirmed by diurnal growth rates seen in different types of phytoplankton. At high levels of incoming irradiance, photosynthetic algae are not able to utilize all the incoming light energy for photosynthesis; the rate of electron transport through PSII is no longer dependent on light absorption (Falkowski et al., 1994). This was also the case in our experiment as evidenced by the decrease in F v/F m. In fact, the quantum efficiency of photosynthetic reaction was lowered by two photoprotective mechanisms which compete with energy trapping in the reaction center at PSII: downregulation mechanisms in the PSII reaction center (Schreiber & Neubauer, 1990) and thermal energy dissipation (Gilmore, 1997; Niyogi, 1999). Therefore, it is important to consider the thermal dissipation of absorbed energy in the analysis of quantum efficiency of photosynthesis. Furthermore, thermal energy of chlorophytes and diatoms/dinoflagellates is dissipated via the fast interconversion of xanthophyll cycle, but cyanobacteria do not perform energy dissipation via the xanthophyll cycle (Wagner et al., 2005). The nonphotochemical quenching of chlorophyll fluorescence is a relative measure of heat dissipation. In this study, the three phytoplankton groups in the water surface showed the highest NPQ value, and NPQ of cyanobacteria was significantly higher than other groups at noon when incoming irradiance energy was the greatest. This indicated that NPQ in cyanobacteria was modified by other mechanisms, one possible mechanism being the redistribution of excitation energy from PSII to PSI via state transitions (Larkum, 2003), Although the exact mechanism for state transition in algae is not yet clarified, it seems that the dissociation of light-harvesting complex II (LHCII) from PSII leads to a decrease in fluorescence and to reduced absorption cross sections (Wagner et al., 2005).
Phytoplanktons have different physiological strategies allowing them to thrive in high light environments. Different types of phytoplankton displayed similar F v/F m diurnal change patterns in bottled samples, but with marked distinction in the present study. For example, F v/F m in cyanobacteria at 0.3 m recovered to predawn F v/F m value at 15:30, which was the fastest among three types of phytoplankton; chlorophytes completely recovered at 17:30, but diatoms/dinoflagellates did not recover to predawn value, which may need more time. Photoinhibition in diatoms/dinoflagellates at 0.6 m was more serious than that in cyanobacteria and chlorophytes, which was in parallel to their value at 0.9 m. All these differences implied that irradiance may have less photoinhibition effect in time and depth scale on cyanobacteria than on chlorophytes and diatoms/dinoflagellates. Although at noon cyanobacteria had a larger photoinhibition value (up to 41%) than chlorophytes (32%) and diatoms/dinoflagellates (34%) at the water surface, no significant difference in growth rates among three types of phytoplankton. Interestingly, inhibition rates of F v/F m in surface cyanobacteria, chlorophytes, and diatoms/dinoflagellates were higher at 16:00 than those at 9:00, although they experienced the same irradiance (750 μmol m−2 s−1) at 9:00 and 16:00. This indicated that changes in F v/F m of cyanobacteria, chlorophytes, and diatoms/dinoflagellates were a function of not only the instantaneous photon flux density but also historical photon density (photo flux density × time). A similar result was also recorded in Microcystis aeruginosa by Brookes et al. (2003).
Furthermore, inhibition of F v/F m in the free-floating population declined and the vertical gradient almost disappeared except at 11:30 in three types of phytoplankton samples. That was due to the mixing of inhibited phytoplankton from the near-surface water down the water column and the movement of cells with less inhibition up to the surface. If mixing were rapid enough or inhibition were not a dynamic process and both were allowed to persist for sufficient time, then parallel F v/F m would be likely to occur in the whole water column. In our study, when wind speed was above 3 m s−1 or water friction velocity was above 0.003 m s−1, the vertical gradient of F v/F m declined and disappeared, which meant the wind-induced mixing entrained the phytoplankton cells within the surface mixed layer. But it is uncertain whether phytoplankton cells have been homogenized completely within the surface mixed layer by the wind-induced mixing entrainment. It needs further evaluation of the chlorophyll vertical distribution during the day. In addition, the F v/F m of surface lake samples was significantly lower than that of 0.9 m at 11:30 (P < 0.05), which might be due to the 2.7 m s−1 wind speed at that time (<3 m s−1). That meant the wind-induced mixing was not able to homogenize completely the phytoplankton cells within the surface mixed layer at 11:30. The previous research in our lab showed when wind speed was about 3.1 m s−1, cyanobacteria tended to be entrained by lake water (Cao et al., 2006). Brookes et al. (2003) also found Microcystis aeruginosa could be entrained by wind speed of 3 m s−1 or friction velocity of 0.003 m s−1 in Torrens Lake, but vertical gradient of F v/F m did not disappear. Moreover, George & Edwards (1976) observed in shallow lake that surface cyanobacteria were entrained entirely at wind speeds above 4 m s−1. The results in the present study confirmed when wind speed was above 3 m s−1 or water friction velocity was above 0.003 m s−1, the wind-induced mixing could entrain the cyanobacterial cells within the surface mixed layer.
Vertical cell distribution was a function of not only friction velocity but also swimming speed. In the present study, the maximum quantum yield (F v/F m) was not only used to monitor photosynthetic capacity of different types of phytoplankton in Taihu Lake, but also the ratio of bottle samples F v/F m to free lake samples F v/F m was calculated to determine the direction of cell movement and whether or not migration and /or water column mixing were enhancing photosynthesis or preventing photoinhibition. The bottled samples defined the F v/F m response of the population to the vertical light gradient without the influence of physical mixing or migrating cells. The free lake samples were subjected to mixing and vertical migration. If the lake samples had a higher F v/F m compared with the bottle samples, this would imply that cells had moved upward; a lower F v/F m would imply the opposite (Regel et al., 2004). In the present study, the ratios of cyanobacteria at the surface and 0.3 m were markedly >1, which indicated cyanobacteria in the deeper layer had moved upward and therefore resulted in higher F v/F m in lake samples than that of bottle samples at the surface and 0.3 m. However, the bottles contained the cells to avoid a continuous exposure to high light intensities. No significant difference between lake samples F v/F m and bottle samples F v/F m in 0.6 m and 0.9 m suggested that the depression of F v/F m of the layers due to low F v/F m cyanobacterial cells from the surface was compensated by high F v/F m cells from deeper layers, and finally there was no effect on the F v/F m of the deep layers. The bidirectional movements under mixing and vertical migration resulted in the disappearance of the vertical gradient of F v/F m in lake samples. However, cyanobacteria, mainly Microcystis aeruginosa in Taihu Lake, have strong positive buoyancy which restricts the cells in the surface like the bottle containment when wind speed is lower than 3 m s−1. Furthermore, cyanobacteria had a relatively high diurnal growth rates in the surface and 0.3 m, which was confirmed by the vertical variability of the primary production in Meiliang Bay (Zhang et al., 2005), although photoinhibition of F v/F m in the depths was greater than in deeper layers. This probably has contributed to the development of cyanobacterial dominance in Taihu Lake, where Microcystis was dominant for a long time (Chen et al., 2003). Buoyancy was proposed as a mechanism by which cyanobacteria can float into a favorable light climate (Sherman & Webster, 1994). However, the persistent positive buoyancy in several cyanobacteria (Walsby et al., 1991; Brookes & Ganf, 2001) indicated cyanobacterial cells could float into the regions of intense illumination and suffered a decrease in F v/F m that is closely associated with the maximum yield of photosynthesis (Ibellings, 1996). This suggests that the avoidance of photoinhibition is not the primary factor governing cyanobacterial dominance development. In contrast, Brookes et al. (2003) pointed out that photoinhibition would result in cyanobacteria decreased carbohydrate accumulation and impaired buoyancy regulation, enhancing the chance to surface. The ratios of chlorophytes implied cells at the surface moved downward and cells at the deeper layer moved up to the surface, which implied chlorophytes had a homogeneous trend in the whole water column under vertical migration and mixing. Compared with cyanobacteria, chlorophytes might have a slower vertical migration speed and were easier to be entrained by the wind-induced mixing because the mixing was identical. The cells of diatoms/dinoflagellates actively avoided high irradiance by moving downward at noon and moved upward when irradiance decreased. Whittington et al. (2000) found dinoflagellates (Ceratium hirundinella) migrated toward the surface in suboptimal irradiance and downward in high light. Dinoflagellate cells accumulated at depths corresponding to light intensities that were optical for photosynthesis, therefore, cells migrated towards the surface in the morning and descend later in high light to avoid photoinhibition (Blasco, 1978; Harris et al., 1979; Villarino et al., 1995). Our results in the present study were consistent with these previous observations. In conclusion, different migration responses to irradiance and vertical mixing among three phytoplankton groups may be one of the reasons for cyanobacterial dominance in Taihu Lake because there were higher diurnal growth rates at the depths where cyanobacteria accumulated easily. Furthermore, appropriate controls/repetitions of the experiment under no and/or high wind conditions would be helpful to confirm the conclusion.
References
Anderson, J. M., Y. I. Park & W. S. Chow, 1997. Photoinactivation and photoprotection of photosystem II in nature. Physiologia Plantarum 100: 214–223.
Bilger, W. & O. Björkman, 1990. Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. Photosynthesis Research 25: 173–185.
Blasco, D., 1978. Observations on the diel migration of marine dinoflagellates off the Baja California coast. Maine Biology 46: 41–47.
Brookes, J. D. & G. G. Ganf, 2001. Variations in the buoyancy response of Microcystis aeruginosa to nitrogen, phosphorus, light. Journal of Plankton Research 32: 1399–1411.
Brookes, J. D., R. H. Regel & G. G. Ganf, 2003. Changes in the photo-chemistry of Microcystis aeruginosa in response to light and mixing. New Phytologist 158: 151–164.
Buechel, C. & C. Wilhelm, 1993. In vivo analysis of slow chlorophyll fluorescence induction kinetics in algae: progress, problems and perspectives. Photochemistry and Photobiology 58: 137–148.
Cao, H. S., F. X. Kong, L. C. Luo, X. L. Shi, Z. Yang, X. F. Zhang & Y. Tao, 2006. Effects of wind and wind-induced waves on vertical phytoplankton distribution and surface blooms of Microcystis aeruginosa in Lake Taihu. Journal of Freshwater Ecology 21: 231–238.
Casper-Lindley, C. & O. Bjoerkman, 1998. Fluorescence quenching in four unicellular algae with different light-harvesting and xanthophylls—cycle pigments. Photosynthesis Research 56: 277–289.
Chen, Y., B. Q. Qin, K. Teubner & M. Dokulil, 2003. Long-term dynamics of phytoplankton assemblages: Microcystis-domination in Lake Taihu, a large shallow lake in China. Journal of Plankton Research 25: 445–453.
Cullen, J. J. & M. R. Lewis, 1988. The kinetics of algal photoadaption in the context of vertical mixing. Journal of Plankton Research 10: 1039–1063.
Cullen, J. & J. MacIntyre, 1998. Behavior, physiology and the niche of depth-regulating phytoplankton. In Anderson, D., A. Cembella & G. Allegraff (eds), The Physiological Ecology of Harmful Algal Blooms. Springer-Verlag, Heidelberg: 559–579.
Denman, K. L. & A. E. Gargett, 1983. Time and space scales of vertical mixing and advection of phytoplankton in the upper ocean. Limnology and Oceanography 28: 801–815.
Falkowski, P. G. & Z. Kolber, 1993. Estimation of phytoplankton photosynthesis by active fluorescence. ICES Marine Science Symposia 197: 92–103.
Falkowski, P. G. & Z. Kolber, 1995. Variations in chlorophyll fluorescence yields in phytoplankton in the world oceans. Australian Journal Plant Physiology 22: 341–355.
Falkowski, P. & J. Raven, 1997. Aquatic Photosynthesis. Blackwell Science, Malden MA: 193.
Falkowski, P., R. Greene & Z. Kolber, 1994. Light utilization and photoinhibition of photosynthesis in marine phytoplankton. In Baker, N. & J. Bowyer (eds), Photoinhibition of Photosynthesis: from Molecular Mechanisms to the Field. BIOS Scientific Publishers Limited, Oxford: 407–432.
Geel, C., W. Versluis & J. F. H. Snel, 1997. Estimation of oxygen evolution by marine phytoplankton from measurement of the efficiency of photosystem II electron flow. Photosynthesis Research 51: 61–70.
Genty, B., J. M. Briantais & N. R. Baker, 1989. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica et Biophysica Acta 990: 87–92.
George, D. G. & R. W. Edwards, 1976. The effect of wind on the distribution of chlorophyll a and crustacean plankton in a shallow eutrophic lake. Journal of Applied Ecology 13: 667–690.
Gilmore, A. M., 1997. Mechanistic aspects of xanthophyll cycle-dependent photoprotection in higher plant chloroplasts and leaves. Physiologia Plantarum 99: 197–207.
Greene, R. M., Z. S. Kolber, D. G. Swift, N. W. Tindale & P. G. Falkowski, 1994. Physiological limitation of phytoplankton photosynthesis in the eastern equatorial Pacific determined from variability in the quantum yield of fluorescence. Limnology and Oceanography 39: 1061–1074.
Harris, G. P., S. I. Heaney & J. F. Talling, 1979. Physiological and environmental constraints in the ecology of the planktonic dinoflagellate Ceratium hirundinella. Freshwater Biology 41: 51–61.
Heraud, P. & J. Beardall, 2000. Changes in chlorophyll fluorescence during exposure of Dunaliella tertiolecta to UV radiation indicate a dynamic interaction between damage and repair processes. Photosynthysis Research 63: 123–134.
Ibellings, B. W., 1996. Changes in photosynthesis in response to combined irradiances and temperature stress in cyanobacterial surface water blooms. Journal of Phycology 32: 549–557.
Jordan, B., 1996. The effects of ultraviolet-B radiation on plants: a molecular perspective. Advances in Botanical Research 22: 97–162.
Juneau, P. & P. J. Harrison, 2005. Comparison by PAM fluorometry of photosynthetic activity of nine marine phytoplankton grown under identical conditions. Photochemistry and Photobiology 81: 649–653.
Kolber, Z. & P. G. Falkowski, 1993. Use of active fluorescence to estimate phytoplankton photosynthesis in situ. Limnology and Oceanography 38: 1646–1665.
Kolber, Z., K. D. Wyman & P. G. Falkowski, 1990. Natural variability in photosynthetic energy conversion efficiency: a field study in the Gulf of Maine. Limnology and Oceanography 35: 72–79.
Larkum, A. M. D., 2003 Light-harvesting systems in algae. In Larkum, A. M. D., S. E. Douglas & J. A. Raven (eds). Photosynthesis in Algae. Kluwer Academic, Dordrecht, The Netherlands, 277–304.
Lippemeier, S., R. Hintze, K. H. Vanselow, P. Hartig & F. Colijn, 2001. In-line recording of PAM fluorescence of phytoplankton cultures as a new tool for studying effects of fluctuating nutrient supply on photosynthesis. European Journal of Phycology 36: 89–100.
Long, S. P., S. Humphries & P. G. Falkowski, 1994. Photoinhibition of photosynthesis in nature. Annual Review of Plant Physiology & Plant Molecular Biology 45: 633–662.
Mauzerall, D., 1972. Light induced changes in Chlorella, and the primary photoreaction for the production of oxygen. Proceedings of the National Academy of Sciences USA 69: 1358–1362.
Niyogi, K. K., 1999. Photoprotection revisited: genetic and molecular approaches. Annual Reviews of Plant Physiology and Plant Molecular Biology 50: 333–359.
Oliver, R.L. & G. G. Ganf, 2000. Freshwater blooms. In: Whitton B. A. & M. Potts (eds), The Ecology of Cyanobacteria. 149–194.
Oliver, R. L. & J. Whittington, 1998. Using measurements of variable chlorophyll-a fluorescence to investigate the influence of water movement on the photochemistry of phytoplankton. In Imberger, J. (ed.), Physical Processes in Lakes and Oceans. Coastal and Estuarine Studies, Vol. 54. American Geophysical Union, Washington, DC: 517–534.
Oliver, R. L., J. Whittington, Z. Lorenz & I. T. Webster, 2003. The influence of vertical mixing on the photoinhibition of variable chlorophyll a fluorescence and its inclusion in a model of phytoplankton photosynthesis. Journal of Plankton Research 25: 1107–1129.
Regel, R. H., J. D. Brookes & G. G. Ganf, 2004. Vertical migration, entrainment and photosynthesis of the freshwater dinoflagellate Peridinium cinctum in a shallow urban lake. Journal of Plankton Research 26: 143–157.
Renger, G., M. Volker, H. Eckert, R. Fromme, S. Hom-Veit & P. Graber, 1989. On the mechanism of photosystem II deterioration by UV-B irradiation. Photochemistry and Photobiology 49: 97–105.
Schreiber, U., 1986. Detection of rapid induction kinetics with a new type of high frequency modulated chlorophyll fluorometer. Photosynthesis Research 9: 261–272.
Schreiber, U., 1994. New emitter detector cuvette assembly for measuring modulated chlorophyll fluorescence of highly diluted suspensions in conjunction with the standard PAM fluorometer. Zeitschrift für Naturforschung C 49: 646–656.
Schreiber, U. & C. Neubauer, 1990. O2-dependent electron flow, membrane energization and the mechanism of non-photochemical quenching. Photosynthesis Research 25: 279–293.
Sherman, B. E. & I. T. Webster, 1994. A model for the light-limited growth of buoyant phytoplankton in a shallow, turbid waterbody. Australian Journal of Marine and Freshwater Research 45(5): 847–862.
Ting, C. S. & T. G. Owens, 1992. Limitations of the pulse-modulated technique for measuring the fluorescence characteristics of algae. Plant Physiology 100: 367–373.
Vassiliev, I. R., O. Prasil, K. D. Wyman, Z. Kolber, A. K. Hanson, J. E. Prentice & P. G. Falkowski, 1994. Inhibition of PSII photochemistry by PAR and UV radiation in natural plankton communities. Photosynthesis Research 42: 51–64.
Villarino, M. L., F. G. Figueias, K. J. Jones, X. A. Alvarez-Salgado, J. Richard & A. Edwards, 1995. Evidence of in situ diel vertical migration of a red-tide microplankton species in Ria de Vigo (NW Spain). Marine Biology 123: 607–617.
Wagner, H., T. Jakob & C. Wilhelm, 2005. Balancing the energy flow from captured light to biomass under fluctuating light conditions. New Phytologist 169: 95–108.
Walsby, A. E., R. Kinsman, B. W. Ibellings & C. S. Reynolds, 1991. Highly buoyant colonies of the cyanobacterium Anabaena lemmermanii form persistent surface waterblooms. Archiv für Hydrobiologie 121: 261–280.
Whittington, J., B. S. Sherman, D. Green & R. L. Oliver, 2000. Growth of Ceratium hirundinella in a sub-tropical Australian reservoir: the role of vertical migration. Journal of Plankton Research 22: 1025–1045.
Wilhelm, C., 1990. The biochemistry and physiology of lightharvesting processes in chlorophyll b- and chlorophyll c-containing algae. Plant Physiology and Biochemistry 28: 293–306.
Zhang, Y. L., B. Q. Qin, W. M. Chen, Y. W. Chen & G. Gao, 2005. Phytoplankton primary production in spring Meiliang bay, Taihu. Journal of Lake Sciences 17(1): 81–86.
Acknowledgments
We thank Taihu Lake Ecosystem Research Station for providing some necessary apparatus and Dr. Cao at Fordham University for his correction of the manuscript. The work was supported by the special program for Taihu Lake water pollution governing of Jiangsu Province (BK2007748), National Natural Science Foundation of China (40471045) and the 100-Researcher Program of CAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: L. Naselli-Flores
Rights and permissions
About this article
Cite this article
Zhang, M., Kong, F., Wu, X. et al. Different photochemical responses of phytoplankters from the large shallow Taihu Lake of subtropical China in relation to light and mixing. Hydrobiologia 603, 267–278 (2008). https://doi.org/10.1007/s10750-008-9277-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-008-9277-4