Abstract
Leeches (Hirudinea) constitute a relatively small monophyletic group of highly specialized annelids, but may play important roles as invertebrate predators in freshwater, while others are infamous for their ectoparasitic bloodsucking. About 15% of the 680 described species are marine and slightly less have switched to terrestrial life; the rest are freshwater, divided among 91 genera. They are globally distributed on all continents except Antarctica, reaching the highest diversity in the Holarctic region with one-half of all continental species. Known areas of local endemism are the ancient Siberian lake Bajkal and lake Ohrid (about 10 species each) on the Balkan Peninsula, which is an endemicity area in itself. A small number of sanguivorous species known as “medicinal leeches” have played an important role in traditional and modern medicine, most noticeably four Hirudo spp. from the Western Palearctic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Leeches (Hirudinea) used to be considered as derivatives of oligochaetes, Acanthobdella, with few chaetae still present, being the connecting link. It is now clear that Hirudinea (Achaetobdellae, Euhirudinea, Hirudinida) constitute a monophylum that renders the Oligochaeta paraphyletic (Martin, 2001; Siddall et al., 2001). The Acanthobdellida and the Branchiobdellida (crustacean epizoans or parasites traditionally linked with Oligochaeta) are the two closest relatives of true leeches, the latter being the more likely sister group, according to molecular phylogenetic investigations (Siddall et al., 2001).
Leeches are hermaphroditic annelids with totally reduced both parapodia and chetae, with unpaired male and female genital openings in the region of the glandulose belt called clitellum, and with a sucker on both the anterior and posterior end of the body (Fig. 1). Each somite is superficially divided into usually three to five, but sometimes more than 10 annuli. The coelomic cavity is transformed into a contiguous system of channels. Many leeches are blood-sucking on vertebrates or invertebrates; the others are mainly predators, rarely scavengers. Most inhabit freshwaters, but there are marine and terrestrial species, too. They reproduce by eggs deposited in cocoons secreted by the clitellum. Their ontogeny is direct, without larval stages.
Habitus of three typical representatives of major hirudinean taxa: (A) Cystobranchus fasciatus, a fish leech (Piscicolidae), dorsal view; (B) Placobdella costata, a glossiphoniid, dorsal view; (C) – Barbronia assiuti, an erpobdelliform, lateral view; (D) Ventral view of the clitellum with genital and accessory openings; the latter are found only in some species. os––oral sucker, cs––caudal sucker, mgo––male genital opening; fgo––female genital opening. Original drawings are a courtesy of Hasko Nesemann
Species diversity
This compilation was greatly facilitated by some previous local or global monographs, mainly indirectly through Sawyer (1986); beside the latter, such are by Harding & Moore (1927), Klemm (1972), Lukin (1976), Ringuelet (1980), Yang (1996), Nesemann & Neubert (1999), and a series by Soos (1965–1969). Numerous primary sources (not listed in References) were used to complete the work.
Linnaeus (1789) described 14 species of Hirudo, classified as Vermes Intestina. The classical taxonomy used to be based on the general shape, color pattern, position and number of eyes, later also on distance between genital openings. Moquin-Tandon (1846) presented anatomical pictures which were later used extensively; so was also the system of annulation with an appropriate coding, proposed by Moore (1898). Pawlowski (1948) added the male atrium shape. However, nobody assessed the variability level of the morphological and anatomical characters. A study of the hirudinean diversity of the former Yugoslavia area, notorious for its complex biodiversity, showed that neither the anatomy of the reproductive apparatus nor the annulation is stable enough to be reliable, as character above the species level (Sket, 1968). This, and some phylogenetic uncertainties, triggered first molecular studies (Trontelj et al., 1996; Siddall & Burreson, 1998) followed by many others that are nowadays yielding surprising solutions predicting discovery of further unexpected errors of the current taxonomy.
However, morphological means allowed us to describe approximately 680 species till now, some 480 of which are freshwater. Bielecki (1997) introduced new detailed morphological and anatomical standards in the systematics of piscicolids (fish leeches) that gave rise to a manifold increase of European fish leech species. This, along with a considerable ‘cryptic’ diversity among European erpobdellids (own studies, unpublished), suggests that a large number of species has still remained unrecognized even within the traditionally best-studied European hirudinean fauna. The cumulative plot of described species (Fig. 2) shows no sign of a plateau.
These facts also imply that leech taxonomy––at the species level and higher––is in a revolutionary phase right now. In the present overview (Table 1), we mainly follow the system of Sawyer (1986) but we could not ignore some novel findings; therefore, some of our solutions are partial and provisional. Too little care has been taken till now to represent genera in molecular studies by their type species and species by their topo-type populations, as these are the only reliable means for assuring the taxonomical identity of objects. Neither the number of families, nor their generic subdivision, are final.
Species diversity is relatively evenly distributed among four major hirudinean clades, the proboscis-bearing Piscicolidae and Glossiphoniidae, the jawed Hirudiniformes, and the predaceous Erpobdelliformes (see Table 1 for classification and species numbers). The latter two groups are sisters, often referred to as arhynchobdellids for their lack of a proboscis. This leaves the small tropical marine and freshwater family Ozobranchidae with an uncertain position among the paraphyletic group of proboscis-bearing leeches, traditionally known as rhynchobdellids.
Piscicolidae are parasites, mainly on fishes, a large part of them being the only marine leeches. Bound to freshwater only, but ecologically very diverse and globally distributed, are the Glossiphoniidae. They feed as parasites on vertebrates and invertebrates; some may be predators if the prey is small enough. Arhynchobdellids live predominantly in freshwaters, but might also be amphibious or terrestrial. Most hirudiniform species are parasitic on vertebrates; some are also predators of small invertebrates, occasionally even scavengers. Erpobdelliforms are exclusively predatory species with an aquatic or amphibious lifestyle. They swallow their prey as a whole using their muscular pharynx. They may occur in high densities and are among the most important invertebrate predators in freshwater communities.
Traditionally a part of the Hirudinea but now recognized as a clade of their own, the Acanthobdellida are limited to two species at the boreal fringe of the Holarctic. They parasitize fishes. The larger, more than 100 spp. strong Branchiobdellida have been traditionally discussed with Oligochaeta.
Phylogeny and historical processes
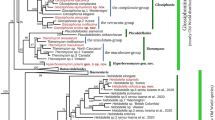
Based on recent molecular phylogenetic hypotheses (e.g., Apakupakul et al., 1999; Trontelj et al., 1999; Borda & Siddall, 2004; Utevsky & Trontelj, 2004), it can be concluded that leeches are primarily and essentially freshwater animals with few switches to marine and terrestrial habitats (Fig. 3). Further, their notorious ectoparasitic bloodsucking might be a sophistication of a less specialized commensalism or parasitism inherited from ancestors shared with branchiobdellids and acanthobdellids. It has been proposed that the first true leech was a proboscis-bearing bloodsucker, and that bloodfeeding facilitated by jaws as in the medicinal leech evolved independently, following a predatory stage (Trontelj et al., 1999; Borda & Siddall, 2004).
Simplified phylogenetic tree of main hirudinean and sister taxa, the height of the triangle reflecting the species richness of each clade. Waves represent the share of marine, bricks the share of terrestrial species, while white areas correspond to freshwater (and amphibian) species. The share of terrestrial erpobdelliforms and marine ozobranchids (two species each) is too small to be shown. Dark vertical bars represent ecto-commensal or parasitic feeding, light bars predatory behavior. Only relationships that have consistently received high support in all molecular phylogenetic studies are drawn as resolved
A second focus of current molecular phylogenetic work is on species-level relationships and alpha taxonomy. A number of studies have indicated that not only much of the traditional low-level taxonomy is mislead by highly homoplastic characters, but also a great deal of species diversity remained overlooked or ignored. Perhaps the most striking example of the former case is the family Erpobdellidae, in which, after molecular scrutiny, virtually all characters used for subfamilial and generic subdivision (e.g., annulation, genital anatomy, and color patterns) turned out to be useless for this purpose (Trontelj & Sket, 2000; Siddall, 2002). Lack of reliable taxonomic characters is the main reason for recently discovered cases of cryptic diversity among erpobdellids. Finally, the most famous of all leeches, the European medicinal leech, is represented by at least three species as demonstrated by phylogenetic analyses of nuclear and mitochondrial DNA sequences (Trontelj & Utevsky, 2005). Their coloration pattern, often rejected as too variable, has ultimately proven to be a reliable identification feature.
Present distribution and main areas of endemicity
Global distribution patterns
Erpobdellidae are evidently limited to the Holarctic. The only species in New Zealand can hardly avoid the suspicion of a taxonomical mistake, while in Mexico the group crosses the border of the Neotropical region. Closely related species are widely distributed across the Palearctic, as well as between Palearctic and Nearctic. A particularly high number of erpobdellid species are known from Europe, but the degree of knowledge is territorially biased. The Haemopidae s.str are mainly Nearctic (Haemopis) and East Palearctic (Whitmania), with a few species of Haemopis distributed in Europe. Generally Holarctic, and mainly Western Palearctic, is the whole freshwater section of Piscicolidae. Myzobdella (Neotropical) and Limnotrachelobdella spp. (E Palearctic) might be secondary invaders from the sea. Limited to the Holarctic are also some genera of Glosiiphoniidae: Glossiphonia, Placobdella with the majority of species in the Nearctis, and Torix limited to the east of the Eastern Palearctis. In unison, they make the Holarctic with one-half of all continental leech species the most diverse biogeographic region (Fig. 4, Table 2).
In the Neotropical region, the glossiphoniid genus Haementeria is mainly tropical and hardly crossing the northern border of the region. Biogeographically similar, and probably closely related, are the smaller genera Gloiobdella and Adaetobdella. The genus Helobdella has, on the other hand, richly speciated (more than 35 species known) in colder (elevated) regions of South America. It developed a couple of ecologically very successful species, one of which spread widely into the Holarctic region, possibly as an accidental passenger on migrating aquatic birds. Two additional species succeeded to spread out of the Neotropical region by some human means. A similar distribution display the hirudiniform sister families Semiscolecidae and Macrobdellidae, the former being limited to the Neotropical region, the latter with at least one genus each in the Neotropics and the Nearctis.
The Oriental region is still relatively rich in leeches but has virtually no endemic groups. The region can be characterized by some smaller genera with predominantly Oriental species and only slight extrusions into the Eastern Palearctic. Such are the glossiphoniid Paraclepsis and hirudiniform Myxobdella, Poecilobdella and Hirudinaria. Most Oriental groups are in fact Paleotropical, occuring also in the Afrotropical region. Oriental and Afrotropical is the genus Asiaticobdella; mainly Oriental, although generally tropical are freshwater Ozobranchidae and the rich glossiphoniid genus Placobdelloides. Limnatis is also present outside tropics in southern parts of Europe. The hirudinid Praobdella seems to be purely African.
Somehow in the warmer East is also the gravity point of the family Salifidae; its main genera, Salifa and Barbronia, are both present in the Oriental region and in the eastern Palearctis, spreading slightly to the Afrotropical, the Western Palearctic, and even into the Australian regions. Since some of its species have clearly demonstrated good spreading abilities, it is questionable whether such a distribution pattern is ancient, and if it is natural at all.
The Australasian region, except for the endemic hirudinid subfamily Richardsonianinae (Bassianobdella, Goddardobdella, Richardsonianus), is inhabited by very few freshwater leech species. They belong to different genera and might be either results of late natural introductions (e.g., by birds) or even of taxonomical errors. The most numerously represented are the snail leeches Alboglossiphonia, a globally distributed genus. Australasia is the domain of the terrestrial Haemadipsidae, which might be most diverse here, while spreading throughout the Oriental region and even crossing into the SE of the Eastern Palearctic.
The distribution type of some phyletic groups is at present not definable, e.g., the glossiphoniid Theromyzon has species distributed across all biogeographical regions except for the Australasian. One could easily explain this by their relation to waterfowl. The explanation of the even distribution of Alboglossiphonia throughout all regions is more enigmatic. In oceans, piscicolids are present around all continents and at all latitudes. Previous biogeographical analyses have been published by Soos (1970), Ringuelet (1980), and Sawyer (1986).
Endemicity areas
Discovery of species flocks and therefore of rich endemic faunas can be predicted for the next future. They may occur in SE Europe (own studies) and in the southern United States (according to Govedich et al., 1999). Most ancient lakes have single or no endemic leech species. Likewise, with only two Caspiobdella spp., the number of endemics in the (brackish) Caspian is surprisingly low, but a number of European species seems to originate from Ponto-Caspian waters. Richer is Lake Bajkal with three piscicolids (Baicalobdella torquata, Codonobdella truncata, C. zelenskiji) and some glossiphoniids (Baicaloclepsis echinulata, B. grubei, Torix baicalensis and probably some Theromyzon spp.). The Balkan lake of Ohrid harbors the richest known endemic leech fauna (Sket, 1968, 1989) with some endemic glossiphoniids (Glossiphonia complanata maculosa, G. pulchella) and a flock of eight erpobdellid species (‘Dina’ ohridana aggregate). The latter are young species according to their mitochondrial DNA divergence (unpublished results), but remarkably differentiated in their body shapes (Sket, 1989). Another probable Lake Ohrid endemic is Piscicola pawlowskii.
Ecological specialists
Some species occur in caves (Sket, 1986; unpublished data). These may be generalists, like Haemopis sanguisuga, or troglobionts, like H. caeca from Dobrogea in Romania. Particularly rich in troglobionts is the family Erpobdellidae. Some described or undescribed cave species are present in southern Europe (N Italy–Balkans–Turkey–Caucasus), their derivative is also the extraordinarily transformed Croatobranchus mestrovi form deep caves in the Croatian Dinaric mountains. Some undescribed species, probably erpobdellids, occur in caves of China and the US.
Another habitat less frequently inhabited by leeches are brackish waters, like lakes and lagoons along the SE Indian coast (lake Chilka) with Pterobdella amara, Aestabdella caeca, Calliobdella olivacea. Both Caspian piscicolids and the Ponto-Caspian Archaeobdella esmonti can be attributed to this group.
Among terrestrial specialists the giant Chilean Americobdella valdiviana, representing its own family, is one of the largest leeches (reportedly measuring more than 20 cm, along with two other giants, the Amazonian freshwater species Haementeria ghiliani and the Antarctic marine Megaliobdella szidati). Ornithobdellinae, a couple of terrestrial hirudinid species, are feeding on sea-birds and can be found in their colonies in Australia and New Zealand. In the Xerobdellidae, Xerobdella spp. are terrestrial predators in temperate to alpine climates of Europe, whereas the neotropical Mesobdella and Diestecostoma spp. are reported as sanguivorous. Entirely terrestrial are also the haematophagous Haemadipsidae and predatory Cylicobdellidae.
Human related issues
Leeches have been intimately connected to humans throughout nearly 2000 years of documented history of Western medicine. While in ancient times the haematophagous medicinal leech (Hirudo medicinalis) was considered as panacea, nowadays mainly its bio-active anticoagulant and anti-inflammatory substances are attracting medical and pharmaceutical attention (Sohn et al., 2001; Whitaker et al., 2004). Moreover, the direct therapeutic application of leeches is experiencing a renaissance, albeit for different purposes, e.g., to restore blood circulation after reconstructive surgery or, recently, to treat osteoarthritis (Pilcher, 2004). Although the leeches are now commercially bred in leech farms, the annual consumption will probably never approach the nineteenth century numbers when up to 100 million leeches per year were imported to France alone. It has only recently become clear that most commercially used leeches are not the species officially declared (H. medicinalis), but rather its congener H. verbana or sometimes H. orientalis. Other species, mainly of the SE Asian genus Hirudinaria have been exploited medically and are sometimes even offered for sale as genuine medicinal leeches. Even some rhynchobdellids (Haementeria officinalis in Mexico, Placobdella costata in Krym) have been in medical use.
The “medicinal leech” is protected and/or listed as endangered species in many European countries. It is not clear to what extent the alleged unfavorable conservation status is a consequence of past over-harvesting and how much of it can be contributed to more recent habitat destruction. Moreover, as long as the new taxonomic knowledge is not taken into account, we will not even know which species we are struggling to preserve.
Through centuries of exploitation and translocation the natural distribution of all Hirudo spp. was probably substantially affected by humans. More conspicuous, however, are transcontinental introductions, like the one of SE Asian Hirudinaria manillensis to the West Indies (Kutschera & Roth, 2006), probably also as a consequence of transport for medical purposes. Accidental transfers of leeches have resulted in several successful invasions of new ranges, most noticeably by the misleadingly named Neotropic glossiphoniid Helobdella europaea to Europe and Australia (e.g., Kutschera 2004), or the salifid Barbronia weberi from Asia to Europe and Australia. Other non-native Helobdella spp. have been reported from Europe, and the Australian Barbronia arcana from Mexico (Oceguera-Figueroa et al., 2005). Although B. weberi has been characterized as invasive (Govedich et al., 2003), the invasiveness of most non-native leech species does not approach the aggressive nature of some invasive crayfishes and fishes.
References
Apakupakul, K., M. E. Siddall & E. M. Burreson, 1999. Higher level relationships of leeches (Annelida: Clitellata: Euhirudinea) based on morphology and gene sequences. Molecular Phylogenetics and Evolution 12: 350–359.
Bielecki, A., 1997. Fish leeches of Poland in relation to the Palearctic piscicolines (Hirudinea: Piscicolidae: Piscicolinae). Genus 8: 223–375.
Borda, E. & M. E. Siddall, 2004. Arhynchobdellida (Annelida: Oligochaeta: Hirudinida): phylogenetic relationships and evolution. Molecular Phylogenetics and Evolution 30: 213–225.
Govedich, F. R., B. A. Bain, M. Burd & R. W. Davies, 2003. Reproductive biology of the invasive Asian freshwater leech Barbronia weberi (Blanchard, 1897). Hydrobiologia 510: 125–129.
Govedich, F. R., D. W. Blinn, R. H. Hevly & P. S. Keim, 1999. Cryptic radiation in erpobdellid leeches in xeric landscapes: a molecular analysis of population differentiation. Canadian Journal of Zoology 77: 52–57.
Harding W. A. & J. P. Moore, 1927. Hirudinea. In Shipley, E. (ed.), The Fauna of British India, including Ceylon and Burma. London.
Klemm, D. J., 1972. Freshwater leeches (Annelida: Hirudinea) of North America. Biota of freshwater ecosystems identification manual no. 8. Water Pollution Control Research Series. U.S. Environmental Protection Agency, Washington, D.C.
Kutschera, U., 2004. The freshwater leech Helobdella europaea (Hirudinea: Glossiphoniidae): an invasive species from South America? Lauterbornia 52: 153–162.
Kutschera, U. & M. Roth, 2006. Notes on the ecology of the Asian medicinal leech Hirudinaria manillensis (Hirudinea: Hirudinidae). Lauterbornia 56: 9–13.
Linnaeus, C., 1789. Systema Naturae. Vermes.
Lukin, E. I., 1976. Pijavki. Tom I. Pijavki presnyh i solonovatyh vodoemov, Izdatel’stvo “Nauka”, Leningrad.
Martin, P., 2001. On the origin of the Hirudinea and the demise of the Oligochaeta. Proceedings of the Royal Society of London Series B––Biological Sciences 268: 1089–1098.
Moore, J. P., 1898. The leeches of the U. S. National Museum. Proceedings US National Museum 21(1160): 543–563.
Moquin-Tandon, A., 1846. Monographie de la famille des Hirudinees. Paris.
Nesemann, H. & E. Neubert, 1999. Annelida: Clitellata: Branchiobdellida, Acanthobdellea, Hirudinea. In: Süßwasserfauna von Mitteleuropa, 6/2. Spektrum Akademischer Verlag, Heidelberg, Berlin.
Oceguera-Figueroa, A., V. León-Règagnon & M. E. Siddall, 2005. Phylogeny and revision of the Erpobdelliformes (Annelida, Arhynchobdellida) from Mexico based on nuclear and mitochondrial gene sequences. Revista Mexicana de Biodiversidad 76: 191–198.
Pawlowski, L. K., 1948. Contributions á la systématique des sangsues dú genre Erpobdella de Blainville. Acta zoologica et oecologica Universitatis Lodziensis, sect. III 8: 1–55.
Pilcher, H., 2004. Medicinal leeches: stuck on you. Nature 432: 10–11.
Ringuelet, R. A., 1980. Biogeografia de los hirudineos de America del Sur y de Mesoamerica. Obra del Centenario del Museo de La Plata 6: 1–27.
Sawyer, R. T., 1986. Leech biology and behaviour. Clarendon Press, Oxford.
Siddall, M. E. & E. M. Burreson, 1998. Phylogeny of leeches (Hirudinea) based on mitochondrial cytochrome c oxidase subunit I. Molecular Phylogenetics and Evolution 9: 156–162.
Siddall, M. E., 2002. Phylogeny of the leech family Erpobdellidae (Hirudinea: Oligochaeta). Inveretbrate Systematics 16: 1–6.
Siddall, M. E., K. Apakupakul, E. M. Burreson, K. A. Coates, C. Erséus, S. R. Gelder, M. Källersjö & H. Trapido-Rosenthal, 2001. Validating Livanow: molecular data agree that leeches, Branchiobdellidans and Acanthobdella peledina form a monophyletic group of oligochaetes. Molecular Phylogenetics and Evolution 21: 346–351.
Sket, B., 1968. K poznavanju faune pijavk (Hirudinea) v Jugoslaviji (Zur Kenntnis der Egel–Fauna Jugoslawiens). Razprave SAZU, Cl. IV., 11(4): 127–197.
Sket B., 1986. Hirudinea. In Botosaneanu, L. (ed.), Stygofauna Mundi, pp. 250–253.
Sket, B., 1989. Intralacustrine speciation in the genus Dina (Hirudinea, Erpobdellidae) in Lake Ohrid (Yugoslavia). Hydrobiologia 182: 49–58.
Sohn, J. H., H. A. Kang, K. J. Rao, C. H. Kim, E. S. Choi, B. H. Chung & S. K. Rhee, 2001. Current status of the anticoagulant hirudin: its biotechnological production and clinical practice. Applied Microbiology and Biotechnology. 57: 606–613.
Soós, Á., 1965. Identification key to the leech (Hirudinoidea) genera of the world, with a catalogue of the species. I. Family: Piscicolidae. Acta zoologica Academiae scientiarum Hungaricae 11: 417–463.
Soós, Á., 1969. Identification key to the leech (Hirudinoidea) genera of the world, with a catalogue of the species. VI. Family: Glossiphoniidae. Acta zoologica Academiae scientiarum Hungaricae 15: 397–454.
Soós, Á., 1970. A zoogeographical sketch of the fresh-water and terrestrial leeches (Hirudinoidea). Opuscula zoologica Budapest 10(2): 313–324.
Trontelj, P., B. Sket, P. Dovč & G. Steinbrueck, 1996. Phylogenetic relationship in European erpobdellid leeches (Hirudinea: Erpobdellidae) inferred from restriction site data of the 18S ribosomal gene and ITS2 region. Journal of Zoological Systematics and Evolutionary Research 34: 85–93.
Trontelj, P., B. Sket & G. Steinbrück, 1999. Molecular phylogeny of leeches: congruence of nuclear and mitochondrial rDNA data sets and the origin of bloodsucking. Journal of Zoological Systematics and Evolutionary Research 37: 141–147.
Trontelj, P. & B. Sket, 2000. Molecular re-assessment of some phylogenetic, taxonomic and biogeographic relationships between the leech genera Dina and Trocheta (Hirudinea: Erpobdellidae). Hydrobiologia 438: 227–235.
Trontelj, P. & S. Y. Utevsky, 2005 Celebrity with a neglected taxonomy: molecular systematics of the medicinal leech (genus Hirudo). Molecular Phylogenetics and Evolution 34: 616–624.
Utevsky, S. & P. Trontelj, 2004. Phylogenetic relationships of fish leeches (Hirudinea, Piscicolidae) based on mitochondrial DNA sequences and morphological data. Zoologica Scripta 33: 375–385.
Whitaker, I. S., J. Rao, D. Izadi & P. E. Butler, 2004. Hirudo medicinalis: ancient origins of, and trends in the use of medicinal leeches throughout history. British Journal of Oral & Maxillofacial Surgery 42: 133–137.
Yang, T., 1996. Annelida Hirudinea. In Fauna Sinica. Science Press, Beijing.
Acknowledgments
We thank Hasko Nesemann for kindly providing his drawings, and Gregor Bračko for his help with copying out some literature data. This work was in part supported by the Slovenian Research Agency.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: E. V. Balian, C. Lévêque, H. Segers & K. Martens
Freshwater Animal Diversity Assessment
Rights and permissions
About this article
Cite this article
Sket, B., Trontelj, P. Global diversity of leeches (Hirudinea) in freshwater. Hydrobiologia 595, 129–137 (2008). https://doi.org/10.1007/s10750-007-9010-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-007-9010-8