Abstract
The trophic structure of benthic communities in the Tagus estuary and adjacent coastal shelf was characterized according to a functional guild approach, based on sampling surveys conducted between 1987 and 2000. Macrobenthic organisms were assigned to seven distinct trophic groups (herbivorous, filter feeders, surface deposit feeders, subsurface deposit feeders, carnivores, filter feeders/detritivores, carnivores/detritivores) and the dominance of these groups was related to environmental variables using multivariate ordination techniques. Surface-deposit feeders were numerically dominant in the Tagus estuary, making up 52% of the benthic communities, while in the adjacent coastal shelf the assemblage was dominated by both surface-deposit feeders and filter feeders (37% and 33%, respectively). When biomass was considered, filter feeders and filter feeders/detritivores were the dominant groups in the estuary, while for the adjacent coastal shelf filter feeders represented 83% of the total biomass. Salinity, depth and sediment composition were the main factors structuring spatial distribution. Surface-deposit feeders were the most abundant macrobenthos of the upper estuary. Surface deposit feeders also dominated the middle and the lower estuary but the proportion of filter feeders as well as other trophic groups increased with salinity. Generally, a more even distribution of trophic structure was found at stations with high salinity. In the adjacent coastal shelf, the trophic diversity decreased with depth. The trophic structure revealed that filter feeders dominated in abundance and biomass in shallow sandy sediments (<25 m), while in deeper sandy mud and muddy habitats (>50 m to 260 m), deposit feeders and carnivores were the most important groups in abundance and biomass, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Macrozoobenthic communities are key components in the functioning of estuarine systems. Benthic organisms produce considerable changes in physical and chemical conditions of the sediment, especially in the water-sediment interface. They also promote the decomposition of organic matter, the nutrient recycling and the energy transfer to other links within the food web (Rhoads & Young, 1970; Day et al., 1989).
A large number of studies on the structure and dynamics of estuarine and coastal benthic assemblages have been conducted worldwide (e.g., Jones et al., 1986; Junoy & Viéitez, 1990; Warwick et al., 1991; Davoult et al., 1998; Desmond et al., 2002). Benthic invertebrates are also commonly used in monitoring studies, since this group reflects anthropogenic impacts (Rosenberg & Resh, 1993). However, it is often difficult to distinguish between natural and anthropogenic changes. Benthic communities vary considerably according to environmental conditions and the majority of the species have highly aggregated small-scale distribution patterns induced especially by substrate type, sediment composition, salinity, food availability and predation (e.g., Boesch, 1973; Chester et al., 1983; Flint & Kalke, 1985; McLusky & Elliott, 2004). Contaminants in the sediment may have also a marked influence on the structure of benthic communities. Toxic compounds can be lethal to benthic organisms or my lead to reduced condition, disease resistance or reproductive potential (e.g., McLusky & Elliott, 2004).
Some studies have been focused on the functional characterization of benthic assemblages rather than species composition, outlining that this approach is useful when interpreting estuarine gradients and assessing ecosystem functioning (e.g., Gaston & Nasci, 1988; Gaston et al., 1995; Weston, 1990). The assignment of a trophic group to a certain species may be particularly difficult, since a large number of macrobenthic organisms can change their feeding habits according to environmental conditions (Pearson & Rosenberg, 1978; Wolff, 1983). Nonetheless, it is usually possible to obtain a general pattern of assemblage structure using certain criteria and to establish relationships between spatial and temporal distribution of trophic groups and environmental variables, such as hydrodynamics, salinity and sediment composition, among many others (e.g., Rhoads & Young, 1970; Pearson, 1971; Gaston, 1987; Mancinelli et al., 1998; García-Arberas & Rallo, 2002; Ysebaert et al., 2003).
The main goal of the present study was to evaluate the trophic structure of macrobenthos in the Tagus estuary and adjacent coastal shelf and its relationships with environmental conditions, using this functional guild approach. Former studies on the benthic communities of the Tagus estuary are relative to only few areas of the estuary or to a particular taxonomic group and, thus, knowledge about the structure and dynamics of benthic invertebrate assemblages in this estuary is scarce.
Materials and methods
Study area
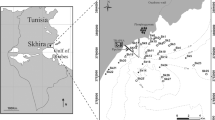
The Tagus estuary has an area of ca. 320 km2 and is approximately 50 km long (Fig. 1). Its width varies from 15 km, at the upper area, to 2 km, near the mouth. The Tagus River is the main fresh water supplier of the estuary with average flow rates of ca. 300 m3 s−1. The circulation inside the estuary is mainly driven by the tidal flow. It’s a mesotidal estuary, with mean tidal amplitude of 2.6 m, ranging from 4.1 m in spring tides to 1.3 m in neap tides. In the entrance channel the velocity may reach 2 m s−1 in spring tides (Portela, 1996).
The adjacent coastal shelf is relatively flat and approximately 30 km long from the mouth of the estuary to the 200 m depth limit, parallel to the coast. In the zone where an important submarine outfall is located (2,000 m depth), the wind and density currents also play an important role in driving the flow, along with the tide (Leitão, 2002).
The upstream area of the estuary is very shallow (<5 m) with extensive intertidal flats that represent 40% of the total estuarine area. Downstream, the depth reaches a maximum of 40 m in the entrance channel.
The Tagus estuary has long been subjected to industrial development and urbanization. Several hundred industries labour along the estuarine borders. The upper part is bordered by land used intensively for agricultural purposes (Fernandes et al., 1995). Fishing occurs throughout the estuary, mainly using beam-trawl and gill-nets.
The Lisbon metropolitan area is heavily populated. At the present ca. two million people live around the estuary. Water quality control and monitoring in the river Tagus basin is recent. Holistic overviews of pollution estimates are particularly scarce, but the values determined for discharges in the river are generally higher in comparison with the estuary.
The estuary has an important role as an overwintering area and feeding ground for birds. Part of the estuarine area is a nature reserve (Tagus Estuary Nature Reserve).
Sampling surveys and data analysis
Sampling surveys were carried out irregularly from 1987 until 2000, in the Tagus estuary and adjacent coastal shelf. Nonetheless, overall numbers of samples collected in different periods of time and space are quite balanced. During this period, 363 sites were sampled: 245 throughout the estuary and 118 in the adjacent coastal area (Fig. 1). Although performed irregularly, sampling occurred throughout the year and the number of samples per estuarine area and season was relatively well distributed, which strongly support the community analysis that was performed.
At each sampling site, three replicates were collected with a Smith-McIntyre grab (0.1 m2), two for the benthic macrofaunal analysis and one for sediment characterisation. In order to characterize the macrofaunal assemblages, sediment samples were sieved through a 500 μm mesh sieve and the individuals retained in the sieve preserved in 4% buffered formaldehyde. Macrobenthos were identified to the lowest possible taxonomic level, counted and wet weighed.
The granulometric analysis was performed by dry sieving using the detailed description given by Gaudêncio et al. (1991). The sediment was classified according to the Wentworth scale (Buchanan, 1984) and characterized by its percentage of silt and clay (<63 μm), very fine sand (63–125 μm), fine sand (125–250 μm), medium sand (250–500 μm), coarse sand (500–1,000 μm), very coarse sand (1,000–2,000 μm), gravel (>2,000 μm) and by the median of grain size diameter. Total organic matter of the sediment was obtained by loss on ignition (details given by Castro, 1997). Water content of the sediment was also determined based on differences between wet and dry (24 h at 60°C) weight of the sediment.
Macrobenthic species were assigned to 7 trophic groups based on their diet and feeding habits, as described in the literature. The trophic guilds were adapted from Hunt (1925), Pearson (1971), Gaston (1987), Le Bris (1988), Rodrigues, (1992) and Sanchez-Mata et al. (1993) and are described as follows: Herbivorous (H)—feed mainly on algae and benthic diatoms; filter feeders (FF)—filter feeding organic matter and plankton in the water column; surface deposit feeders (SDF)—feed on organic matter, bacteria, detritus and benthic algae in the interface water-sediment; subsurface deposit feeders (SSDF)—feed on the same items as SDF but below the water-sediment interface; carnivores (C)—predators or necrophagous species; filter feeders/detritivores (F/SD)—alternate their feeding habits between filter feeders and detritivores; carnivores/detritivores (C/SD)—alternate their feeding habits between carnivores and detritivores.
Macrobenthic species richness, abundance (density, in individuals m−2) and wet weight biomass (g m−2) were determined for each trophic group. Canonical Correspondence Analyses (CCA) were used to outline the main patterns of assemblage structure and to evaluate its relationship with environmental variables. This multivariate technique allows one to directly relate the pattern of community variation to environmental variables (Ter Braak & Prentice, 1988). According to Ter Braak and Prentice (1988), it has several advantages when compared with other direct gradient analysis techniques, in particular when species have nonlinear and unimodal relationships to environmental gradients.
These CCA were performed using species richness, density and biomass data sets for the Tagus estuary and for the adjacent coastal shelf separately. The environmental variables that were considered in these analyses were depth, distance to estuary mouth (Bugio_d), total organic matter of the sediment (TOM), percentage of silt and clay (Fines), very fine sand (Vfs), fine sand (Fs), medium sand (MS), coarse sand (Cs), very coarse sand (Vsf) and gravel (G). All these analyses were performed using CANOCO v.4.0 (Ter Braak & Smilauer, 1998).
Results
Environmental conditions in study areas
A longitudinal gradient can be outlined when analysing depth, salinity and sediment characteristics of sampling areas, with increasing salinity and depth from the inner estuarine area towards the coastal shelf area (Table 1). For some of the sediment components that were considered, the mean values were similar for all sampling areas, namely for coarse sedimentary components. Total organic matter and water content of the sediments were higher in sediments of the middle estuary, where percentages of fines were also high. On the continental shelf, sediment was dominated by fine sands (Table 1).
Taxonomic composition
In the present study, 544 species were identified among the 131,987 specimens collected, which resulted in a total biomass of 65.4 kg. Annelida, Mollusca and Arthropoda were the main groups, representing more than 90% of the species richness, density and biomass.
Arthropoda (37%) was the most important group in number of species, followed by Annelida (30%) and Mollusca (27%), while in terms of abundance and biomass the dominant taxonomic groups were Mollusca (51% and 75%, respectively), followed by Annelida (34% and 12%, respectively) and Arthropoda (13% and 6%, respectively).
The most abundant species were also the most frequent and included Ervilia castanea (Montagu, 1803), Barnea candida (Linnaeus, 1758), Venerupis pullastra (Montagu, 1803), Abra alba (Wood, 1802), Corbula gibba (Olivi, 1792), Pomatoceros lamarcki (Quatrefages, 1866), Melinna palmata Grube, 1870, Mediomastus capensis Day, 1961, Nephtys hombergii Savigny, 1818, Heteromastus filiformis (Claparède, 1864) and Sabellaria spinulosa Leuckart, 1849, that represented 63% of the total number of individuals caught. More than 60% of the total biomass was relative to only 6 species: B. candida, V. pullastra, Chaetopleura angulata (Spengler, 1797), Nassarius reticulatus Linnaeus, 1758, Lanice conchilega (Pallas, 1766) and Sagartia troglodytes (Price, 1847).
Trophic groups
The macrobenthic assemblage of the Tagus estuary and adjacent coastal shelf was dominated by carnivores (C), surface deposit-feeders (SDF) and filter feeders (FF), whose relative importance varied when assessed using species richness, density or biomass (Table 2).
In the Tagus estuary and in the adjacent coastal area, carnivores (28% and 26%, respectively) and surface deposit-feeders (23% and 22%, respectively) were the trophic groups with the highest number of species, while herbivores exhibited the lowest values (1% and 0.3%, respectively). When the mean number of individuals was considered, surface-deposit feeders were dominant in the Tagus estuary, representing about 52% of the benthic organisms. Filter feeders (19%) were the second major trophic group followed by filter feeders/detritivores (11%), carnivores (8%), subsurface-deposit feeders (6%), carnivores/detritivores (2%) and the herbivores (1%). In terms of biomass, filter feeders/detritivores (35%) were the most important functional group followed by filter feeders (27%), carnivores (20%), surface-deposit feeders (12%), herbivores (5%), carnivores/detritivores and subsurface-deposit feeders (both represented 1%) (Table 2).
In the adjacent coastal area, the dominant groups were surface-deposit feeders and filter feeders (that accounted for 37% and 33% of the total number of individuals). Filter feeders were the dominant group in biomass (83%), followed by carnivores (16%). The other trophic groups accounted only for 4% of the total biomass.
Relationships between trophic structure and environmental variables
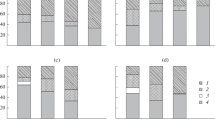
The ordination diagrams of the canonical correspondence analyses (CCA), used to evaluate the relationships between the dominant trophic groups per sampling site and environmental conditions, presented a similar pattern for species richness, density and biomass (Fig. 2). The first two ordination axes accounted for ca. 20% of the total variance in the analyses relative to species richness, density and biomass datasets.
Ordination diagrams based on the canonical correspondence analyses performed to data on the dominant trophic group per sampling station, according to species richness, density and biomass. Each station is represented by the symbol of its dominant trophic group: H (■), FF (O), SDF (□), SSDF (✦), C (Δ), C/SD (▼), F/SD (◆) (○)—stations that have different trophic groups with the same percentage; (▶)—weighted average coordinates obtained for each of the seven trophic groups; environmental variables are represented by vectors: depth, distance to estuary mouth (Bugio_d), total organic matter of the sediment (TOM), percentage of silt and clay (Fines), very fine sand (Vfs), fine sand (Fs), medium sand (MS), coarse sand (Cs), very coarse sand (Vsf) and gravel (G)
For the Tagus estuary, F/SD, C/SD and H were correlated with distance to the estuary mouth and percentage of mud and organic matter in the sediment, and dominated the benthic assemblage in the upper estuary (Fig. 2). FF, although occurring throughout the estuary, presented a strong relationship with sandy areas, depth and salinity. The sites where SSDF was the most important trophic group were mainly characterized by heterogeneous sediment with high mud content. Sites dominated by SDF and C were positioned along both axes of the ordination diagrams, which reflect the dominance of these groups in sites distributed throughout the estuary.
The CCA diagrams relative to the coastal shelf adjacent to the Tagus estuary showed a similar ordination pattern (Fig. 2). The separation between sites where FF and SD dominated is particularly evident: the first was mainly correlated with mud content of the sediment and depth; the second group was associated with sandy grounds and lower depths. Sites where C was the most important trophic group were mainly located at higher depths and in more shallow areas where sediment have a high mud and organic matter content.
The location of sites where H was the most important group is not particularly related to any of the environmental variables analysed. C/SD predominated at sites with a high content of fine sand in the sediment. Sites dominated by SSDF presented a high content of large sand and gravel or, on the contrary, a high percentage of mud. F/SD dominated in sites relatively disperse, related with muddy sand.
Discussion
Macrobenthic communities in the Tagus estuary were numerically dominated by surface deposit-feeders, while in the adjacent coastal shelf surface deposit-feeders and filter feeders were almost equally represented. These groups also represent a large proportion of the total biomass. The dominance of surface deposit-feeders has been reported for a large number of estuarine systems worldwide, since detritus generally present a great influence on estuarine food webs (Day et al., 1989; D’Avanzo & Valiela, 1990; Lastra et al., 1991; Muniz & Pires, 1999; García-Arberas & Rallo, 2002).
The main distribution pattern found in the Tagus estuary reflected a longitudinal gradient: filter feeders were particularly abundant in the adjacent coastal shelf and presented a low occurrence in the upper estuary, while filter feeders and detritivores dominated in the mesohaline area. Surface deposit feeders presented an increasing importance from the coastal shelf to the middle estuary, and then a decreasing importance towards upstream areas. As outlined by several authors the dominance of some of these trophic groups was related to several environmental variables, namely salinity, depth and mud and organic matter content of the sediment. In several estuarine systems in the North Sea, Hummel et al. (1988), Hamerlynk et al. (1993) and Ysebaert et al. (1998) reported that the trophic structure of benthic assemblages was dominated by suspension feeders in the polyhaline areas and by deposit feeder in the meohaline areas.
Besides salinity, sediment composition has also been pointed out as a major factor inducing spatial heterogeneity of benthic invertebrates. Deposit feeders are generally abundant in muddy and muddy sand low energy sediments while suspension feeders prevail in sediments with a low content of fine fractions (e.g., Dewarumez, 1983; García-Arberas & Rallo, 2002).
The relationships between the dominance of trophic groups and the environmental factors that were established for the Tagus estuary using multivariate ordination techniques only accounted for a small fraction of the total variability. Several authors have emphasized the difficulties in establishing these relationships between spatial distribution patterns and environmental factors, being particularly hard to distinguish between natural and anthropogenic induced changes. Hydrodynamic conditions (Davould, 1990), sediment composition (Gaston, 1987; Probert, 1984) and, especially, mud content of the sediment (Pearson, 1971; Chardy & Clavier, 1988; Eleftheriou & Basford, 1989) have been pointed out as key factors in the structuring of benthic communities, but particular combinations of these factors may produce a pattern difficult to interpret or may be masked by other factors such as biological interactions or pollutants.
Probet (1984), Gaston et al. (1995) and Muniz & Pires (1999) pointed out that food availability is responsible, in a large extent, for the spatial distribution pattern of macrobenthos. Sandy sediment communities usually present a more balanced proportion among different feeding types, which could occur because of a higher diversity of microhabitats in coarse sediments (Carrasco & Carbajal, 1998) and because higher food items diversity may occur in sediment interspaces (Muniz & Pires, 1999).
Relationships between trophic group dominance and food availability were also evident in the benthic communities of the Tagus estuary: filter feeders/detritivores were more abundant in the turbidity maximum zone and filter feeders dominated near the estuary mouth, where the highest values of particulate organic carbon were registered (Vale & Sundby, 1987; Vale et al., 1990). In the coastal shelf adjacent to the Tagus estuary, the spatial distribution of dominant trophic groups can also be related to food availability, being the main pattern similar to that described for the estuarine area. Filter feeders mainly occurred in shallow sandy areas, where the access to particulate organic matter can be enhanced (Pearson, 1971; Pearson & Rosenberg, 1978; Weston, 1990). On the contrary, surface deposit feeders were distributed along the shelf, in muddy grounds, up to 260 m deep, since they require high levels of organic matter in the sediment. Carnivores occurred throughout the coastal shelf, suggesting that carbon fluxes are strong enough to support the occurrence of the highest trophic levels (Wijsman et al., 1999).
For some highly impacted estuaries, such as those in the North Sea, the effects of pollution on benthic communities have been reported. Some of these effects are reflected in an impoverishment of the diversity of benthic organisms, mainly due to elevated concentrations of pollutants, especially organic pollutants that cause oxygen deficiency (Ysebaert et al., 1998). García-Arberas & Rallo (2002), in the Gulf of Biscay, also found differences in feeding groups among estuarine areas that could be attributed to organic enrichment of the sediment. An increase in deposit-feeders is usually reported in areas subjected to eutrophication (Beukema, 1991). However, as outlined by Gray et al. (1988) and García-Arberas & Rallo (2002), the utility of feeding group approaches in detecting effects of pollution are limited, since the separation of pollution gradients from natural gradients in estuarine and coastal environments is not evident. Furthermore, the synergistic, additive, or antagonistic interactions among contaminants are poorly understood, which makes it difficult to predict biological responses simply based on knowledge of the types and concentrations of contaminants present in a given area.
The integrative approach used in the present study proved to be efficient in characterizing the trophic structure of benthic assemblages along environmental gradients. Further developments considering abiotic and biotic interactions of benthic macroinvertebrate species are needed to better understand the functioning of benthic communities.
References
Beukema, J. J., 1991. Changes in composition of bottom fauna of a tidal-flat area during a period of eutrophication. Marine Biology 111: 293–301.
Boesch, D. F., 1973. Classification and community structure of macrobenthos in the Hampton Roads area, Virginia. Marine Biology 21: 226–244.
Buchanan, J. B., 1984. Sediment analysis. In Holme, N. A. & A. D. McIntyre (eds), Methods for the study of marine benthos. Blackwell Scientific, London.
Carrasco, F. D. & W. Carbajal, 1998. The distribution of polychaete feeding guilds in organic enriched sediments of San Vincente Bay, Central Chile. International Review of Hydrobiology 83: 233–249.
Castro, M., 1997. Comportamento dos PCBs e DDTs no estuário do Sado e bioacumulação em peixes. [PhD Thesis]. Universidade Técnica de Lisboa, Lisbon.
Chardy, P. & J. Clavier, 1988. Biomass and trophic structure of the macrobenthos in the South-West lagoon of New Caledonia. Marine Biology 99: 105–202.
Chester, A. J., R. L. Fergursen & G. W. Thayer, 1983. Environmental gradients and benthic macroinvertebrate distribution in a shallow North Carolina estuary. Bulletin of Marine Science 33: 282–295.
D’Avanzo, C. & I. Valiela, 1990. Use of detrital foods and assimilation of nitrogen by coastal detritivores. Estuaries 13: 20–24.
Davoult, D., 1990. Biofaciès et structure trophique du peuplement des cailloutis. Oceanologica Acta 13: 345–345.
Davoult, D., J. M. Dewarumez & A. Migne, 1998. Long-term changes (1979–1994) in two coastal benthic communities (English Channel): analysis of structural developments. Oceanologica Acta 21:609–617.
Day, J. W. Jr., C. A. S. Hall, W. M. Kemp & A. Yañez-Arancibia, 1989. Estuarine Ecology. John Wiley, New York.
Desmond, J. S., D. H. Deutscman & J. B. Zedler, 2002. Spatial and temporal variation in estuarine fish and invertebrate assemblages: analysis of an 11-year data set. Estuaries 25: 552–569.
Dewarumez, J. M., 1983. Évolution de populations de mollusques intertidaux en relation avec les modifications sédimentologiques sur le littoral de la Mer du Nord. Haliotis 13: 3–12.
Eleftheriou, A. & D. J. Basford, 1989. The macrobenthic infauna of the offshore Northern North Sea. Journal of the Marine Biological Association of the United Kingdom 69: 123–143.
Fernandes, T. F., M. Elliott & M. C. da Silva, 1995. The management of European estuaries: a comparison of the features, controls and management framework of the Tagus (Portugal) and the Huber (England). Netherlands Journal of Aquatic Ecology 29: 459–468.
Flint, R. W. & R. D. Kalke, 1985. Benthos structure and function in a South Texas estuary. Contributions in Marine Science 28: 33–53.
García-Arberas, L. & A. Rallo, 2002. The intertidal soft-bottom infaunal macrobenthos in three Basque estuaries (Gulf of Biscay): a feeding guild approach. Hydrobiologia 475–476: 457–468.
Gaston, G. R., 1987. Benthic polychaete of the Middle Atlantic Bight: feeding and distribution. Marine Ecology Progress Series 36: 251–262.
Gaston, G. R., S. S. Brown, C. F. Rakocinski, R. W. Heard & J. K. Summers, 1995. Trophic structure of macrobenthic communities in Northern Gulf of Mexico estuaries. Gulf Research Reports 9: 111–116.
Gaston, G. R. & J. C. Nasci, 1988. Trophic structure of macrobenthic communities in the Calcasieu estuary, Louisiana. Estuaries 11: 201–211.
Gaudêncio, M. J., M. T. Guerra, P. R. Almeida, F. M. Moreira, J. L. Costa & C. A. Assis, 1991. Povoamentos Macrozoobentónicos. Faculdade de Ciências da Universidade de Lisboa, Lisbon.
Gray, S. J., M. Aschan, M. R. Carr, K. R. Clarke, R. H. Green, T. H. Pearson, R. Rosenberg & R. M. Warwick, 1988. Analysis of community attributes of the benthic macrofauna of Frierfjord/Langesundfjord and in a mesocosm experiment. Marine Ecology Progress Series 46: 151–165.
Hamerlynk, O., J. Mees, J. A. Craeymeersch, K. Soetaert, K. Hostens, A. Cattrijsse & P. A. van Damme, 1993. The Westerschelde estuary: two food webs and a nutrient rich desert. Progress in Belgian Oceanographic Research, 217–234.
Hummel, H., G. Moerland & C. Bakker, 1988. The concomitant existence of a typical coastal and a detritus food chain in the Westerschelde estuary. In Hummel, H., C. Bakker, A. Huiskes & A. G. A. Merks (eds), Hydrobiology and Chemistry of the Schelde and Westerschelde: Proceedings of the Schelde Symposium. Delta Institute for Hydrobiological Research, Terneuzen, 35–41.
Hunt, O. D., 1925. The food of the bottom fauna of the Plymouth fishing grounds. Journal of the Marine Biological association of the United Kingdom 13: 560–599.
Jones, A. R., C. J. Watson-Russell & A. Murray, 1986. Spatial patterns in the macrobenthic communities of the Hawkesbary Estuary, New South Wales. Australian Journal of Marine and Freshwater Research 37: 521–543.
Junoy, J. & J. M. Viéitez, 1990. Macrozoobenthic community structure in the Ría de Foz, an intertidal estuary (Galicia, Northwest Spain). Marine Biology 107: 329–339.
Lastra, M., J. Palacio, A. Sánchez & J. Mora, 1991. Estructura trófica infralitoral de la Bahía de Santander. Cahiers de Biologie Marine 32: 333–351.
Le Bris, H., 1988. Fonctionnement des écosystèmes benthiques côtiers au contact d’estuaires: la rade de Lorient et La Baie de Vilaine. [PhD Thesis]. Université de Bretagne Occidentale, Brest.
Leitão, P., 2002 Integração de Escalas e de processos na modelação do ambiente marinho. [PhD Thesis]. Universidade Técnica de Lisboa, Lisbon.
Mancinelli, G., S. Fazi & L. Rossi, 1998. Sediment structural properties mediating dominant feeding type patterns in soft-bottom macrobenthos of the Northern Adriatic Sea. Hydrobiologia 367: 211–222.
McLusky, D. S. & M. Elliott, 2004. The Estuarine Ecosystem: Ecology, Threats and Management, 3rd ed. Oxford University Press, Oxford.
Muniz, P. & A. M. S. Pires, 1999. Trophic structure of polychaetes in the São Sebastião Channel (Southern Brazil). Marine Biology 134: 517–528.
Pearson, T. H., 1971. Studies on the ecology of the macrobenthic fauna of Lochs Linnhe and Eil, West Coast of Scotland. II. Analysis of the macrobenthic fauna by comparison of feeding groups. Vie et Milieu 1: 53–91.
Pearson, T. & R. Rosenberg, 1978. Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanography and Marine Biology: an Annual Review 16: 229–311.
Portela, L. I., 1996. Modelação matemática de processos hidrodinâmicos e de qualidade da água no Estuário do Tejo. [PhD Thesis]. Universidade Técnica de Lisboa, Lisbon.
Probet, P. K., 1984. Disturbance, sediment stability and trophic structure of soft-bottom communities. Journal of Marine Research 42: 893–921.
Rhoads, D. C. & D. K. Young, 1970. The influence of deposit-feeding organisms on sediment stability and community trophic structure. Journal of Marine Research 28: 150–178.
Rodrigues, A. M. J., 1992. Environmental status of a multiple use estuary, through the analysis of benthic communities: the Sado estuary, Portugal. [PhD Thesis]. University of Stirling, Stirling.
Rosenberg, D. M. & V. H. Resh, 1993. Freshwater Biomonitoring and Benthic Macroinvertebrates. Chapman & Hall, New York.
Sanchez-Mata, A., M. Lastra & J. Mora, 1993. Macrobenthic crustacean characterization of an estuarine area. Crustaceana 64: 337–355.
Ter Braak, C. J. F. & I. C. Prentice, 1988. A theory of gradient analysis. Advances in Ecological Research 18: 271–317.
Ter Braak, C. J. F. & P. Smilauer, 1998. CANOCO Reference manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination (version 4). Microcomputer Power, Ithaca, New York.
Vale, C., M. J. Gaudêncio & M. T. Guerra, 1990. Evaluation of ecological impacts. In Alzieu, C. & B. Gallene (eds), Proceedings of the International Seminar on Environmental Aspects of Dredging Activities, Nantes, 119–128.
Vale, C. & B. Sundby, 1987. Suspended sediment fluctuations in the Tagus estuary on semi-diurnal and fortnightly time scales. Estuarine, Coastal and Shelf Science 25: 495–508.
Warwick, R. M., J. D. Goss-Custard, R. Kirby, C. L. George, N. D. Pope & A. A. Rowden, 1991. Static and dynamic environmental factors determining the community structure of estuarine macrobenthos in SW Britain: why is the Severn Estuary different? Journal of Applied Ecology 28: 329–345.
Weston, D. P., 1990. Quantitative examination of macrobenthic community changes along an organic enrichment gradient. Marine Ecology Progress Series 61: 233–244.
Wijsman, J. W. M., P. M. J. Herman & M. T. Gomoiu, 1999. Spatial distribution in sediment characteristics and benthic activity on the Northwestern Black Sea shelf. Marine Ecology Progress Series 181: 25–39.
Wolff, W. J., 1983. Estuarine benthos. In Ketchum, B. H. (ed.), Ecosystems of the World: Estuaries and Enclosed Seas. Elsevier, New York, 121–132.
Ysebaert, T., P. M. J. Herman, P. Meire, J. Craeymeersch & H. Verbeek, 2003. Large scale spatial patterns in estuaries: estuarine macrobenthic communities in the Schelde estuary. Estuarine, Coastal and Shelf Science 57: 335–356.
Ysebaert, T., P. Meire, J. Coosen & K. Essink, 1998. Zonation of intertidal macrobenthos in the estuaries of Schelde and Ems. Aquatic Ecology 32: 53–71.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gaudêncio, M.J., Cabral, H.N. Trophic structure of macrobenthos in the Tagus estuary and adjacent coastal shelf. Hydrobiologia 587, 241–251 (2007). https://doi.org/10.1007/s10750-007-0686-6
Issue Date:
DOI: https://doi.org/10.1007/s10750-007-0686-6