Abstract
To propose a concept of their mutual diversity, twenty-nine permanent shallow floodplain pools and oxbows in the river Lužnice floodplain were analysed for area, depth, shape, flooding, and shading by terrestrial vegetation, and sampled in all seasons for their water chemistry, phytoplankton composition and biomass, and zooplankton composition. The sites are regularly flooded, eutrophic, and often shaded by surrounding vegetation. Cryptophyceae, Chrysophyceae and Euglenophyceae dominated the phytoplankton, while Cyanophytes were rare. Within the rich zooplankton assemblage (63 species), cladocerans and rotifers dominated. Correlation matrices and multivariate analyses indicated that shaded and relatively deeper sites had lower oxygen saturation and higher concentrations of PO4–P and NH4–N. Shade and relative depth correlated negatively with phytoplankton biomass and number of phytoplankton taxa, and positively with Cryptophytes and large cladocerans—thus indicating poor mixing, poor light availability and low fish pressure on herbivores. Decomposition of leaf litter increased oxygen consumption, while shade from terrestrial vegetation restricted photosynthesis and decreased oxygen production. Larger sites were more species-rich in phytoplankton and supported Euglenophyceae, green algae and rotifers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In a floodplain with a natural hydrological regime, aquatic ecosystems are characterised by their transience between being either marshy, lotic or lentic in character. Their mutual connectivity remains relatively high because water flowing through the underlaying sediments influences the lotic subsystems biogeochemically and ecologically from the adjacent river, and from the soil and bedrock sediments (Jones & Mulholland, 2000). Moreover, flood events enhance the connectivity, accelerating the exchange of plankton communities, plant seeds and fish among the various pools and oxbows. After each flood event, a process of diversification begins again as the water level declines, the water current diminishes, and finally the floodplain lakes and oxbows become disconnected (Pithart, 1999).
Water level fluctuations and the transient character of the many aquatic parts of the floodplain intensify the interactions between the floodplain’s terrestrial and aquatic environments (Prach, 1996). As the volume of each water body decreases, the ratio between its surface and volume increases, enhancing the intensity of exchange between the sediment and water column. Similarly, the relative impact of terrestrial vegetation—with its effect of shading and leaf litter—increases with decreasing area. The small, shallow lakes and pools can be relatively deep, with a tendency towards intense stratification and oxygen deficits, influencing the whole aquatic community, and in particular the fish (Pithart & Pechar, 1995; Pechar et al., 1996).
The aim of this study is to describe and explain the spatial and temporal diversity of the permanent lotic ecosystems of the hydrologically well-preserved Lužnice river floodplain. We focus on the environmental factors specific to these pools and oxbows, and try to maintain a multi-dimensional viewpoint that incorporates both spatial as well as temporal changes (Ward, 1989). We use the term diversity sensu lato as the overall heterogeneity of our studied subsystems; one that includes environmental variables as well as species occurence, composition and biomass.
Long-term limnological monitoring of two floodplain pools has been carried out for about 20 years, resulting in several papers being published: about the plankton of the Lužnice floodplain, describing the plankton dynamics (Hrbácek et al., 1994; Pechar et al., 1996); flagellate vertical migrations (Pithart, 1997) or and phytoplankton composition (Kylbergerová et al., 2002). This more synthetic paper is based on repeated synoptical samplings of a larger set of different localities.
Sampling sites
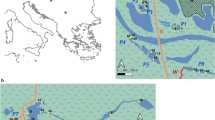
The Lužnice floodplain within the study area remains one of the last floodplains in the Czech Republic whose natural hydrological regime has not been altered substantially by any massive river regulation. The river channel is still meandering and changing its course after every major flood event, including the cutting of new meanders. The studied area is flooded for several weeks every year, mostly in March and April, with occasional floods in winter or summer. The area, about 25 km long and 1–2 km wide, lies to the south of the Czech Republic between the Austrian border and the town of Suchdol nad Lužnicí (Fig. 1). The average (50 years) river discharge is 5.8 m3s−1. The floodplain area consists of: the main stream and standing water bodies (15% of area, about 200 permanent pools and oxbows); meadows, both maintained and abandoned (the latter prevailing, mainly overgrown by Phallaris arundinacea and Urtica dioica); pastures; and floodplain softwood forest (15% dominated by willow and white poplar with an admixture of oak and alder). In winter, the pools regularly freeze over, the ice being mostly covered by snow. Water levels fluctuate within a range of 1.5 m.
The 29 permanent oxbows and pools chosen for this study represent a range of parameters typical of the studied area in terms of their surface area, depth, shape, level of shading by shore vegetation, distance from the river, and macrophyte vegetation.
In this paper, we use the term “oxbow lake” for water bodies with a length many times exceeding their width; they originate in former meanders that have been cut off from the active river channel. The term “pools” we use for circular or oval-shaped water bodies that originated from oxbows but were reshaped by heterogeneous silting-up and erosion caused by floods.
Methods
Sampling was carried out five times to cover different parts of the seasonal development: September 6–7 1994, October 25–26 1994, January 24–25 1995, May 2–3 1995, and August 19–20 1997.
During each of these samplings, water temperature, dissolved oxygen concentration, pH and conductivity were measured in situ by WTW field probes and Multi-line P4 measuring set.
Total alkalinity (TA) was determined by end-point (pH = 4.5) titration with 0.1 M HCl. For nutrient and phytoplankton analysis, an integrated water sample (3 l) was taken with a Plexiglas tube from the first 1–1.5 m of the water column. Concentrations of NH4-N, NO3-N, and PO4-P were determined using a Tecator flow injection analyser (FIA Star 5010, Tecator, Sweden; Ruzicka & Hansen, 1981) in samples filtered through Whatman GF/C filters. Total nitrogen (TN) and total phosphorus (TP) were determined in samples filtered through 100 μm mesh after mineralisation by persulphate as NO3-N and PO4-P (for details, see Pithart & Pechar, 1995). Chlorophyll-a concentration was estimated spectrophotometrically or fluorometrically after extraction in an acetone–methanol mixture (Pechar, 1987).
For enumeration, 100 ml of the sample was preserved by Lugol solution, sedimented in chambers and analysed under the inverted microscope. Algal species were counted in random fields at a magnification of 200× or 400× up to the number of 300 individuals for each dominant species; cell volume was calculated as the volume of corresponding geometrical body (Javornický, 1978). Macrophytes in the pools were determined to species, and their coverage was estimated.
Zooplankton was sampled in a qualitative way. One or two mixed samples were taken from each of the pools, two samples in the case of a separate free-water area and nearshore macrophyte area. From free-water areas (without macrophytes), several bottom-up tows by a conical plankton net (mesh size 100 μm) were taken and mixed together. Zooplankton from macrophyte beds was sampled from throughout the water column using a plastic vessel, and filtering through a plankton net (mesh size 100 μm). All samples were preserved by formalin. Later, 300–400 individuals were determined and counted. Zooplankton was not sampled in August 1997. In zooplankton samples, cladocerans and rotifers were determined mostly to species, or at least to genus level. Copepods were determined only to groups: nauplii, copepodids, diaptomids and harpacticids (Straškraba, 1964).
The dimensions of pools were measured during a period of average water level to calculate their area and shape. Maximum depth was measured from the ice cover. Relative depth was calculated according to the equation z r = 50·z max·√π/√A (without dimension, usually <2 for shallow lakes; Hutchinson, 1957) and Landscape Shape Index (equalling 1 for a circle) according to the formula LSI = p/(2√π·A) (McGarigal & Marks, 1995); where A—area, z max—maximum depth, z r—relative depth, and p—perimeter. Position along the longitudinal river course (river km) and distance from river was estimated from aerial photographs.
Shading was measured during clear-sky days by simultaneous measurement of relative PhAR by Li-Cor 150 with SA 190—sensors at an unshaded site outside the locality, and on the water surface of the water body, at each point of a virtual network consisting of 2 × 2, 3 × 3 or 5 × 5 m squares (according to pool area; total number of points per water body ranged from 20 to 40). Average values from all points of the network and two measurements (morning and afternoon) were used.
An index of flooding was developed as an estimate of probability of flooding for each of the pools, i.e., direct surface contact of a pool with main stream water. We had observed that pools underwent more flooding depending on their proximity to the river, and the more they were upstream. Also, we found that NO3-N concentration strongly correlated linearly in the same way, i.e., negatively with distance from river and positively with river kilometre. Thus, we used NO3-N concentration as an indicator of flooding. Using our dataset (distance from river, river kilometre and average NO3-N concentration) we developed a linear function for calculating the flood index:
Correlation between the measured and predicted NO3-N concentration was 0.30, P = 0.009. From this analysis 6 pools were excluded, as these pools clearly obtained most of their water supply from terrace runoff and hence their NO3-N values were very high.
For data analyses, correlation matrices were calculated for all parameters for each pool using all sampling data (total), from the average value of parameters, and finally for particular seasonal samplings separately. For multivariate statistics we used CANOCO software; DCA method was tried first and finally RDA or CCA. Only the most relevant environmental factors were used for models. For phytoplankton, only relative contributions of selected important groups to the total biovolume were used; for zooplankton, only relative contributions of large pelagial cladocerans and pelagial rotifers to total zooplankton biomass. To make the biplots more understandable, the environmental factors were divided into two groups: (1) morphology, flooding, shading and distance to the river; and (2) water chemistry parameters.
Results
Morphology, distance to the river, flooding and shading
The studied set of water bodies are small, shallow lakes (Table 1) of different shapes, from almost circular pools to elongated oxbows (LSI = 3.7). Some of them were permanently connected to the river (parapotamon-type side arms). Despite most pools maximum or average depth being rather shallow, relative depths can reach rather extreme values (maximum 17). Due to their small area and bankside woody vegetation, pools can be substantially shaded (up to 79%). Some morphological parameters were correlated (see Fig. 2); shape index was correlated significantly with area because the elongated oxbows were also the largest water bodies. All oxbows were also close to the river, therefore their relative depth correlated with distance from river.
Water chemistry
According to total nitrogen (TN, mean value 3.7 mg l−1), total phosphorus (TP, 130 μg l−1), and chlorophyll a (67 μg l−1) concentrations, the studied sites would be classified as eutrophic or hypertrophic. Ranges between minimum and maximum values showed extreme variations in most parameters regardless of sampling date and site. Conductivity fluctuated mostly between 200 and 300 μS cm−1. Alkalinity below 1 meq l−1 was found in 70% of cases; values higher than 2.00 meq l−1 were recorded in less then 5% of measurements. Values of pH ranged mostly from 6.5 to 7.
Some chemical parameters were asociated with shading (Fig. 2), which was positively and significantly correlated with PO4 and NH4 and negatively with oxygen saturation. Area was correlated with chlorophyll a concentration, pH, temperature and shape index and associated with oxygen. Flooding was associated with NO3-N, alkalinity and conductivity.
NO3-N was the prevailing component in the nitrogen budget for most sites: on average nitrate represented about 60% of TN. No significant correlations between NO3-N and PO4-P or NH4-N were found. Also ratios of dissolved inorganic nitrogen to phosphate (DIN/PO4-P) and TN/TP were determined mainly by the nitrate concentrations (not shown).
At some sites, NO3-N concentration exceeded the six-year river maximum (monitored monthly). These pools (6 in all) were considered to have an important source of water from some origin other than the river. The highest values of conductivity, sulphates and high alkalinity as well as lower temperatures in summer in these 6 pools indicate an intensive throughflow of underground water. The remaining 23 water bodies were assumed to be sourced by infiltrating river water. The flood index of these 23 pools correlated with biomass of diatoms (0.58, P = 0.004) abundant in the main stream plankton, and lotic species of diatoms (Achnanthes lanceolata, Navicula lanceolata, and Navicula gregaria; Poulíčková, 1997) were found there.
Some parameters also showed seasonal aspects (Fig. 3). High NO3-N and oxygen were associated with spring (time of flooding), whereas high NH4-N concentrations were associated with winter. Summer was characterised by higher chlorophyll a, PO4-P and alkalinity.
Plankton composition
Phytoplankton composition was very different compared to that of other types of shallow or deep lakes (Kylbergerová et al., 2002). Typical was the year-round prevalence of Cryptophyceae, (70% of algal biomass in the whole set of localities in September, 67% in January), namely species such as: Cryptomonas curvata, C. marssonii, C. reflexa and Rhodomonas minuta; abundance of Euglenophyceae (Trachelomonas volvocina, T. volvocinopsis and a variety of species of Euglena and Phacus); Dinophyceae; and Chrysophyceae (namely Synura spp., Mallomonas spp. and Chrysococcus spp.) In contrast, except for diatoms, the phytoplankton groups typical of eutrophic shallow lakes were either quite rare (Cyanophyceae contributed up to 2%) or were remarkably less abundant (green chlorococcal algae contributed up to 10%). The species richness for the whole set of water bodies was high, especially for green algae (66 species) and the flagellate groups—Euglenophytes (34 species) and Chrysophytes (16 species). Species richness per particular locality varied considerably from 4 to 42 species.
Zooplankton was collected in four sets of samples; 33 taxa of cladocerans and 30 taxa of rotifers were determined (copepods were not identified to species level). For the purpose of this analysis, the species were grouped in several distinguishable categories to simplify the dataset. In general the most relatively abundant were pelagial rotifers: species of genera Anureopsis, Brachionus, Conochilus, Filinia, Gastropus, Hexarthra, Keratella, Notholca, Polyarthra, Pompholyx, Asplanchna, Synchaeta, nauplii and copepodids, contributing together 75–90% to total individuals in zooplankton. Rotifers dominated in late summer (September, 39% of total count of zooplankton) and autumn (October 32%), whereas copepods including nauplii were most numerous in winter and spring (about 40%). Large pelagial cladocerans (Daphnia longispina, Daphnia pulicaria, and Daphnia pulex), littoral and surface cladocerans (Sida sp., Simocephalus sp., Ceriodaphnia megops, C. reticulata, C. rotunda, Megafenestra sp., and Chydoridae) were virtually absent in January, whereas small pelagic cladocerans (Bosmina sp., Ceriodaphnia affinis, C. pulchella, Daphnia cucullata, Daphnia parvula, Diaphanosoma sp. and Moina sp.) were at least present in May and otherwise held a stable share in the zooplankton assemblage.
Multivariate analysis of environmental parameters and plankton
RDA (Fig. 4) showed that shaded and relatively deep sites were associated with abundant Cryptophytes, significantly in the total dataset, January and May (r = 0.49; P = 0.01) and large cladocerans, whereas large sites were associated with chlorophyll a. Area significantly correlated with chlorophyll a in August and September (r = 0.44; P = 0.008), with euglenophytes (August, r = 0.67; P = 0.00), and was positively associated with green algae and Cyanophytes (significantly in total dataset, r = 0.79; P = 0.00), and rotifers.
RDA analysis of morphometry, shading, flooding and distance from river (explaining variables, dashed vectors) and plankton variables (relative contribution of groups to total biomass (phytoplankton) or count (zooplankton) as explained parameters. First axis explains 12% of variability, second 5%. Monte Carlo permutation test for first axis: F-ratio = 13.821, P = 0.102
CCA (not shown) tested the impact of season on phytoplankton groups and showed that Euglenophytes, diatoms and green algae were typical for summer, whereas Cryptophyceae and large cladocera were more typical in autumn.
RDA used to model the relations of water chemistry parameters and plankton is shown in Fig. 5. A more detailed seasonal analysis (not shown) showed that surface oxygen saturation correlated in May with chlorophyll a (r = 0.43; P = 0.02) and with pH (r = 0.58; P = 0.001) but not with shade (at the beginning of May most leaves had not yet developed), indicating that the rapid growth of the algal spring bloom was the main source of oxygen in the water bodies. The correlation between the biomass of Chrysophytes (0.44; P = 0.02) and Cryptophytes (r = 0.62; P = 0.017) as they reached their spring peak, supports this explanation. On the other hand, in summer, oxygen saturation correlated significantly with pH, chlorophyll a and negatively with shade, whereas in October it correlated only with shade (r = −0.66; P = 0.00), indicating the reduction of photosynthesis as the day shortened and enhancing the role of sediment decomposition in the oxygen dynamics.
RDA analysis of water chemistry and temperature (explaining variables, thick lines) and plankton variables—relative contribution of groups to total biomass (phytoplankton) or count (zooplankton)—as explained variables. First axis explains 25% of variability, second 0.4%. Monte Carlo permutation test for first axis: F-ratio = 45.5, P = 0.002
Concentrations of NH4-N and PO4-P (mostly mutually correlated, Fig. 2) were apparently bound to the oxygen regime as well. These variables were also associated with shade, a stronger correlation for PO4-P (significantly correlated in September, January and October, and also for all seasons) than that for NH4-N (significant only in September).
Also a correlation between large pelagic daphniids and shade was repeatedly significant (in September r = 0.66, P = 0.02), indicating weak fish predation at these sites.
Cryptophyceae was the only algal group positively correlated with shade (significantly in September, r = 0.58; P = 0.008, and in the total and average dataset); the other algal groups, both in terms of biomass and proportion, showed a tendency to correlate negatively with shade (mostly not significantly). Not surprisingly, shade restricted algal species number (r = −0.53; P = 0.015 in September).
Discussion
Concept of spatial and temporal heterogeneity
The data analysis indicated the mutual interplay of the different mechanisms that result in the ecosystem diversity of the studied water bodies. Even if it is possible to explain the observed phenomena through the action of only one mechanism, it is more likely that other different mechanisms share a contribution to every phenomena, in different proportions from case to case or from time to time. To explain such a network of relations, we divide the factors driving the diversity among the studied localities into three groups (Fig. 6): (i) source of water: infiltration and flooding; (ii) morphology: area, shape and depth; and (iii) terrestrial vegetation: shading and leaf litter.
Source of water: infiltration and flooding
Ground water seeping to the floodplain water bodies through the permeable sandy alluvial sediments may flow in from either the main river stream or laterally from the elevated river terrace. The nutrient load of these two water sources is markedly different and thus their relative proportion in each case influences the alluvial water bodies substantially. Due to the adjoining intensive agriculture, the water seeping laterally has a much higher conductivity and concentration of HCO −3 , NO −3 , and a relatively lower concentration of PO4-P.
Perhaps somewhat surprisingly the river water is not the most important source of N and P for the studied sites. Correlation between water chemistry in the pools and the river is low most of the year, which indicates the prevalence of some other autonomous processes (Pechar et al., 1996). The high level of nutrients (N, P) in the pools can be considered the result of natural input and accumulation of allochtonous, mainly organic, matter from terrestrial vegetation.
An increasing discharge results in surface flow from the river channel to the adjacent ditches, oxbows and pools; later, the flooding of the whole floodplain connects the subsystems into one water body. The flood events influence the basic chemistry of floodplain waters, especially the concentrations of major ions. During a high discharge, their concentrations are usually diluted—with the exception of nitrates. With decreasing discharge and water levels, and even before the surface separation of water subsystems, a horizontal heterogeneity of water chemistry and plankton already appears, being accelerated by the subsequent surface separation of water bodies. Thus, the different spatial and temporal course of each flood causes a time shift in water chemistry and plankton development in different parts of the floodplain (Pithart, 1999).
Morphometry: area, shape and relative depth
The morphometry of any water body has a crucial impact on the mixing of the water column. Mixing was generally supposed to be poor throughout the entire set of studied localities. Even the relatively large oxbows have an elongated shape, curved character and elevated banks, preventing wind action. Larger and/or relatively shallower water bodies were better mixed -enhancing the growth of a wider spectrum of algal species.
Cryptophyceae, being well adapted to poor light conditions, also prospered well in the very stabilized water column, there occurring no water mixing to move the phytoplankton upwards to the light (Reynolds, 1984). As efficient swimmers among flagellates, they are able to locate themselves according to light conditions and nutrient gradients (Jones, 1988). Cryptophyceae dominate throughout most of the year in waters whose trophic status are very different, but which have a poorly mixed (and often shaded) water column (e.g., in Finish forest lakes: Smolander & Arvola, 1988; Lužnice river floodplain: Pithart, 1997; karstic lakes in Catalonia: Pedrós-Alió & Guerrero, 1993; and lakes covered with macrophytes: Blinn & Green, 1986, Izaguirre et al., 2004).
Planktic cyanobacteria frequently predominate in shallow eutrophic lakes all over the world (Scheffer, 1998), just as in the Třeboň basin (Pechar et al., 2002). They are well adapted to low light conditions and, similarly as with Cryptophycea, they are able to regulate their position in the water column. However, unlike Cryptophyceae and other phytoplankton groups, Cyanophyceae were much less abundant in the alluvial pools, a fact that can be explained by the low pH of the alluvial waters. A similar disappearance of blue-greens as a result of a decrease in pH has been described by Shapiro (1984). Moreover, the decomposition of different plant materials, as for example barley straw (Barret et al., 1999), can restrict the development of Cyanophyceae and may favour the mixotrophic flagellates (Jones, 1988).
The impact of fish should also differ substantially between larger and smaller water bodies. Oxygen in large oxbows is never depleted throughout the whole water column; a better oxygen saturation in winter and more frequent flooding (long oxbows have a closer connectivity with the river) can enhance the fish population. The distinctive zooplankton structure in large pools may serve as indirect evidence of the impact of fish: large pelagic cladocerans are virtually absent in the pelagial and small cladocerans contribute very little. In the littoral zone, however, small pelagial cladocerans significantly correlate positively with area, indicating the growing fish impact that results in the change of spatial distribution of zooplankton, as they seek refuge among macrophytes (Timms & Moss, 1984).
Pelagic rotifers most contribute to the zooplankton in large pools, correlating significantly and consistently with chlorophyll a (for the whole set of localities). This phenomenon seems to be the effect of both a release from competition, when large filtrators are not present (Lampert & Rothhaupt, 1991, Urabe, 1992) and simultaneously the elevated concentration of suitable food (Merriman & Kirk, 2000).
Similarly, the absence of any significant correlation between those nutrients occurring in high concentrations and phytoplankton biomass may also indicate the frequent light limitation (Scheffer, 1998) and therefore the importance of such mechanisms as mixing.
Terrestrial vegetation: shading and leaf litter
Terrestrial vegetation has a crucial impact on the chemistry and life in these water bodies. Permanent shading and nutrient-rich leaf litter affect the oxygen regime and water chemistry as a whole, macrophyte development, fish survival, and consequently the whole structure of the plankton community (Fig. 6). Height and density of terrestrial vegetation on the banks and a pool’s total area, determine the relative proportion of shaded area.
Shade restricts algal photosynthesis, resulting in lower oxygen production. At the same time, oxygen is consumed by decomposition: the amount of organic-rich leaf litter in the sediment would be expected to enhance this process. These two mechanisms have not been experimentally separated, but analysis of the seasonal aspects can help towards understanding which one is more important in which situation for determining the final oxygen concentration.
Oxygen tends to be depleted under the ice cover in shaded small pools; therefore fish cannot survive in these localities and large filtrators are not consumed. On the other hand, larger pools with a longer, more twisted shoreline may support more fish, which would relieve the pressure of food competition for smaller cladocerans as fish graze on larger species. Poor mixing, shade, and humic substances leaching from the leaf litter, are all different mechanisms that again result in poor light conditions in the water column, to which most algal species are unable to adapt.
References
Barrett, P. R. F., J. W. Littlejohn & J. Curnow, 1999. Long-term algal control in a reservoir using barley straw. Hydrobiologia 415: 309–313.
Blinn, D. W. & J. Green, 1986. A pump sampler study of microdistribution in Walker Lake, Arizona, U.S.A.: a senescent crater lake. Freshwater Biology 16: 175–185.
Hrbácek, J., L. Pechar & V. Dufková, 1994. Anaerobic conditions in winter shape of the seasonal succession of Copepoda and Cladocera in pools in forested inundations. Verhandlungen Internationale Verein Limnologie 25: 1335–1336.
Hutchinson, G. E., 1957. A treatise on limnology v.1. Geography, Physics and Chemistry, Wiley, 1015 pp.
Izaguirrre, I., I. O`Farrell, F. Urein, R. Sinitro, M. dos Santos Afonso & G. Tell, 2004. Algal assemblages across a wetland, from a shallow lake to relictual oxbow lakes (Lower Paraná River, South America). Hydrobiologia 511: 25–36.
Javornický, P., 1978. Metódy kvantitatívného štúdia fytoplanktónu (Methods of phytoplankton quantitative study). In Hindák, F. (ed.), Sladkovodné riasy (Freshwater algae). ŠPN, Bratislava, Czechoslovakia, 35–57.
Jones, J. B. & P. J. Mulholland (eds), 2000. Streams and ground waters. Academic Press, San Diego, 425 pp.
Jones, R. I., 1988. Vertical distribution and diel migration of flagellated phytoplankton in a small humic lake. Hydrobiologia 161: 75–87.
Kylbergerová, M., D. Pithart & M. Rulík, 2002. Algological survey of small floodplain backwaters. Algological Studies 104: 169–187.
Lampert, W. & K. O. Rothhaupt, 1991. Alternating Dynamics of Rotifers and Daphnia Magna in a Shallow Lake. Archiv für Hydrobiologie 120: 447–456.
McGarigal, K. & B. J. Marks, 1995. FRAGSTATS: spatial pattern analysis program for quantifying landscape structure. Program documentation. Gen Tech Rep US Dep Agric For Serv Pac Northwest Res Stn 351, Portland.
Merriman, J. L. & K. L. Kirk, 2000. Temporal patterns of resource limitation in natural populations of rotifers. Ecology 8: 141–149.
Pedros-Alió, C. & R. Guerrero, 1993. Microbial ecology of Lake Cisó. In Jones, J. G. (ed.), Advances in microbial ecology, Volume 13. Plenum Press, New York, 155–207.
Pechar, L., 1987. Use of an acetone-methanol mixture for the extraction and spectrophotometric determination of chlorophyll a in phytoplankton. Archiv für Hydrobiologie, Suppl. (Algological Studies 46) 78: 99–117.
Pechar, L., J. Hrbáček, D. Pithart & J. Dvořák, 1996. Ecology of pools in the floodplain. In Prach, K., J. Jeník & A. R. G. Large (eds), Floodplain ecology and management. The Lužnice River in the Třeboň Biosphere Reserve, Central Europe., Backhuys Publishers Amsterdam, 209–226.
Pechar, L., I. Přikryl & R. Faina, (2002). Hydrobiological evaluation of Třeboň fishponds since the end of 19th century. In Květ, J., J. Jeník & L. Soukupová (eds), Freshwater wetlands and their sustainable future: A case study of the Třeboň basin biosphere reserve, Czech Republic. Man and the Biosphere Series 28, UNESCO & The Parthenon Paris, 31–62.
Pithart, D., 1997. Diurnal vertical migration study during a winter bloom of Cryptophyceae in a floodplain pool. Internationale Revue der Gesamten Hydrobiologie 82: 33–46.
Pithart, D., 1999. Phytoplankton and water chemistry of several alluvial pools and oxbows after the flood event–a process of diversification. Algological Studies 95: 93–113.
Pithart, D. & L. Pechar, 1995. The stratification of pools in the alluvium of the river Lužnice. Internationale Revue der Gesamten Hydrobiologie 80: 61–75.
Poulíčková, A., 1997. Rozsivková flóra povodi Lužnice. (Diatom flora of Lužnice River catchment). Preslia 68: 257–264.
Prach, K., 1996. The river Lužnice and its floodplain. In Prach, K., J. Jeník & A. Large (eds), Floodplain Ecology and Management. The Lužnice River in the Třeboň Biosphere Reserve, Central Europe, SPB Academic Publishing, Amsterdam, 11–18.
Reynolds, C. S., 1984. The ecology of freshwater phytoplankton. Cambridge University Press, Cambridge, New York, Melbourne, 384 pp.
Ruzicka, J. & E. H. Hansen, 1981. Flow injection analysis. John Wiley, New York, 207 pp.
Shapiro, J., 1984. Blue-green dominance in lakes: The role and management significance of pH and CO2. Internationale Revue der Gesamten Hydrobiologie 69: 765–780.
Scheffer, M. (ed.), 1998. Ecology of shallow lakes. Chapman & Hall London, Weinheim, 357 pp.
Straškraba, M., 1964. Preliminary results of a new method for the quantitative sorting of freshwater net plankton into main groups. Limnology and Oceanography 9: 268–270.
Smolander, U. & L. Arvola, 1988. Seasonal variation in the diel vertical distribution of the migratory alga Cryptomonas marssonii (Cryptophyceae) in a small, highly humic lake. Hydrobiologia 161: 89-98.
Timms, R. M. & B. Moss, 1984. Prevention of growth of potentially dense phytoplankton populations by zooplankton grazing in the presence of zooplanktivorous fish in a shallow wetland ecosystem. Limnology and Oceanography 29: 472–486.
Urabe, J., 1992. Midsummer succession of rotifer plankton in a shallow eutrophic pond. Journal of Plankton Research 14: 851–866.
Ward, J. V., 1989. The four dimensional nature of lotic ecosystems. Journal of North American Benthological Society 8: 2–8.
Acknowledgements
This study was financially supported by Research Projects AV0Z60870520 and MSM 6007665806 of the Ministry of Education, Youth and Sport and projects of the Czech Grant agency GAČR 206/01/1113 and Ministry of Environment VaV - SL/1/6/04. We are obliged to Martina Kylbergerova for help with phytoplankton counting and Steve Ridgill for language corrections.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pithart, D., Pichlová, R., Bílý, M. et al. Spatial and temporal diversity of small shallow waters in river Lužnice floodplain. Hydrobiologia 584, 265–275 (2007). https://doi.org/10.1007/s10750-007-0607-8
Issue Date:
DOI: https://doi.org/10.1007/s10750-007-0607-8