Abstract
It is becoming increasingly evident that jellyfish (Cnidaria: Scyphozoa) play an important role within marine ecosystems, yet our knowledge of their seasonality and reproductive strategies is far from complete. Here, we explore a number of life history hypotheses for three common, yet poorly understood scyphozoan jellyfish (Rhizostoma octopus; Chrysaora hysoscella; Cyanea capillata) found throughout the Irish and Celtic Seas. Specifically, we tested whether (1) the bell diameter/wet weight of stranded medusae increased over time in a manner that suggested a single synchronised reproductive cohort; or (2) whether the range of sizes/weights remained broad throughout the stranding period suggesting the protracted release of ephyrae over many months. Stranding data were collected at five sites between 2003 and 2006 (n = 431 surveys; n = 2401 jellyfish). The relationship between bell diameter and wet weight was determined for each species (using fresh specimens collected at sea) so that estimates of wet weight could also be made for stranded individuals. For each species, the broad size and weight ranges of stranded jellyfish implied that the release of ephyrae may be protracted (albeit to different extents) in each species, with individuals of all sizes present in the water column during the summer months. For R. octopus, there was a general increase in both mean bell diameter and wet weight from January through to June which was driven by an increase in the variance and overall range of both variables during the summer. Lastly, we provide further evidence that rhizostome jellyfish may over-wintering as pelagic medusa which we hypothesise may enable them to capitalise on prey available earlier in the year.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Within temperate coastal marine systems there is a classic axiom of spring and autumn peaks in phytoplankton and zooplankton biomass, linked to seasonal stratification and hence nutrient supply to surface waters (Parsons et al., 1983). However, there is increasing evidence that this distinct seasonal pattern may become less defined in line with current and predicted shifts in climate (Reid et al., 1998; Hays et al., 2005). For example, rising water temperatures around the UK have altered the synchronicity of larval cod with its planktic prey (match–mismatch hypothesis: e.g., Cushing, 1990) in a way that reduces the survival of young fish (Beaugrand et al., 2003). There is also concern that the trophic dynamics of coastal marine ecosystems may be altered by increases in gelatinous zooplankton which again have been linked to climate change (Lynam et al., 2005), eutrophication (Arai, 2001) and over-fishing (Möller, 1984; Pauly et al., 1998; Purcell and Arai, 2001; Lynam et al., 2006). Such community level shifts have obvious ecological and economic consequences given that both larval and juvenile fish often feed upon the same prey as jellyfish, with fears that energy previously directed to fishes may be diverted leading to irreversible shifts within many marine food webs (Hay et al., 1990; Mills, 1995, 2001).
On a regional scale (the North Sea), pronounced interannual variability in the abundance of jellyfish medusae may also be driven by hydro-climatic changes (via the North Atlantic Oscillation) induced through atmospheric effects on wind stress, temperature and currents (Lynam et al., 2004). These abiotic changes alter the timing of spring phytoplankton blooms and zooplankton community structure (Lynam et al., 2004), which in turn determine the success of medusae in any given year. As such interannual variations will inevitably determine events at an ecosystem level (via increased or decreased predation by medusae upon zooplankton and ichthyoplankton), there is an urgent need to better understand the seasonality and reproductive strategies of many common and abundant jellyfish species (Mills, 2001; Lynam et al., 2006). For schyphozoan jellyfish (Cnidaria: Scyphozoa), this lack of information may be traced to their complex life history which, in simple terms, comprises a benthic asexually-reproducing polyp (scyphistoma) that constitutes the over-wintering phase of many species and a planktic sexually reproducing phase (medusa) that exists in the water column from spring to autumn (e.g., Arai, 1997). The process by which new medusae are formed is termed strobilation and involves partial metamorphosis of the benthic polyp into an ephyrae; an immature medusa that detaches and swims away (Arai, 1997). In general, strobilation may be triggered by physical changes within the water column (Kingsford et al., 2000). For example, in Aurelia aurita, a minimum water temperature is required to stimulate strobilation (e.g., Omori et al., 1995), whilst in Nemopilemia nomurai the reverse is true with a thermal increase thought to be the principal trigger (Kawahara et al., 2006). Alternatively, temperature had no effect on strobilation of Chrysaora quinquecirrha at low salinities, yet at higher salinities a significant temperature increase had a proportionate impact on the release of ephyrae (Purcell et al., 1999). Depending on the species, a single polyp may produce one or many ephyrae at once, over a period of time, or at different intervals. The ephyrae subsequently develop into mature medusae over a period of weeks to months (Arai, 1997). Logically, the timing and duration of ephyrae release will drive the temporal patterns in the size of medusae at any particular site. Therefore an examination of medusae size over protracted periods of time may provide some insight into the seasonality and reproductive plasticity of schyphozoan species (Lucas, 2001).
In a previous study (Doyle et al., 2006), we proposed that the seasonality of jellyfish stranding events in the Irish and Celtic Seas was highly variable with species such as Chrysaora hysoscella and Cyanea lamarckii washing ashore during a brief summer period whilst Rhizostoma octopus stranded throughout the entire year. Here we critically assess these findings in the broader context of scyphozoan life history by examining previously unpublished measurements of bell diameter (and estimates of wet weight) collected over the same period. For three schyphozoan species (R. octopus; C. hysoscella; Cyanea capillata) we tested whether (1) the bell diameter or wet weight of stranded medusae increased over time in a uniform manner suggesting the existence of a single synchronised reproductive cohort; or (2) whether the size range of stranded medusae remained broad throughout the stranding period suggesting the protracted release of ephyrae over many months. Secondly, we hypothesised that the year round stranding of Rhizostoma octopus throughout the Irish and Celtic Seas provided further evidence of temperate scyphozoan jellyfish over-wintering as pelagic medusa as previously reported by Kawahara et al. (2006). By examining temporal shifts in the size and weight of species over time, we were able to empirically test these hypotheses providing valuable insights to the seasonality and persistence of jellyfish medusae in temperate marine environments.
Methods
Jellyfish stranding data
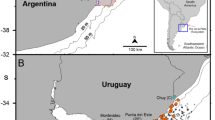
Data were collected in conjunction with broader beach survey programme (2003–2006; 158 beaches; >1200 individual surveys) to map the distribution of five scyphozoan species throughout the southern Irish and Celtic Seas (51.0°N to 53.5°N and −3.0°W to −11.0°W) (Doyle et al., 2006). Size and size range data for stranded medusae were not included in this previous, complimentary study. These novel data (bell diameter) were recorded for the three largest and most robust species (C. capillata, C. hysoscella and R. octopus) at five sites (identified as areas of high abundance) over the three-year period (Fig. 1). Bell diameter was measured for all freshly stranded specimens found close to the waters edge at low tide, except during mass stranding events when the first 50 jellyfish encountered were taken as a random sample. The exception to this rule was site 4 (Fig. 1) on the east coast of Ireland where size data were reported as a range (i.e., maximum and minimum size recoded on a particular day). At all sites, specimens found towards the top of the beach (i.e., at high tide), or those which appeared to have stranded on a previous incoming tide were not measured as shrinkage through water loss rendered estimates of size inaccurate. Diameter was measured by laying the medusa on a flat surface and measuring the maximum distance across the top of the bell. If the bell of a particular individual was damaged or malformed in any obvious manner it was excluded from the study.
Converting bell diameters to wet weight
In a previous study of jellyfish energy density (Doyle et al., 2007) we provided raw data on the bell diameter and wet weight for three species: C. capillata, C. hysoscella and R. octopus. Using these raw data we explored the relationship between bell diameter and wet weight so that equations might be derived allowing estimates of wet weight to be made for stranded jellyfish (Fig. 2). Non-linear, quadratic functions revealed significant relationships between bell diameter and wet weight for two species (Fig. 2) (C. capillata F 2,23 = 68.10, r 2 = 0.84, P < 0.0001; R. octopus F 2,26 = 817.17, r 2 = 0.98, P < 0.0001). A significant power relationship was found for C. hysoscella (F 1,8 = 64.05, r 2 = 0.89, P < 0.0001), although the difference in this relationship may have resulted from a smaller sample size. Therefore, to test the validity of the C. hysoscella equation a comparison was made with previously published data for the species (Buecher et al., 2001). Figure 3b from this earlier study (showing bell diameter versus wet weight) was digitised and a power regression fitted. 95% confidence intervals were generated for this relationship and our data for C. hysoscella superimposed. This revealed a marked consistency between the two studies with the data from the present study falling well within the generated confidence intervals.
Bell diameter and wet weight for three study species. Raw data were obtained from Doyle et al. (2007)
Bell diameter of stranded jellyfish and estimated wet weights versus day of year for (a, b) C. capillata; (c, d) C. hysoscella and (e, f) R. octopus. Data for (e, f) is reproduced from online supplementary material in Houghton et al. (2006a). Direct measurements of C. capillata (North Wales) made during 2004 are shown as (o). For the east coast of Ireland, error bars show the range of size recorded on each survey day at during 2004 and 2005 combined. C. hysoscella data are shown as direct measurements made in North Wales during 2004 (Δ) and the SW coast of Ireland during 2006 (o). For R. octopus data are shown as size range for each day. Years can be differentiated by mid-points marked on each range: 2003 (◊); 2004 (o); 2005 (∇)
Results
Between June 2003 and June 2006, the bell diameter of 2,401 stranded jellyfish was measured at the five study sites combined, during 431 individual beach surveys. A summary of survey effort, bell diameters and estimated wet weights of jellyfish is given in Table 1.
Figure 3 combines data from different years to give bell diameter and estimated wet weight for all three jellyfish species versus day of year. Stranding data for R. octopus were presented in graphical form to illustrate the availability of prey for foraging leatherback turtles (Houghton et al., 2006a). No critical assessment of the size or size range of medusae was conducted in this previous study. Here, bell diameters versus time of year (and subsequent estimates of wet weight) are alternatively shown as simplified ranges (max–min).
Direct measurements (i.e., not just range) of bell diameter for C. capillata were only available for two beaches at study site 1 (Fig. 1) for a single season (2004). No significant relationship could be detected between bell diameter and day of year when considered as either raw data or weekly means (date of first through to the last recorded stranding event). Care should however be taken in the interpretation of this finding, given the restricted time scale over which C. capillata appears to strand (Fig. 3). Moreover, given that bell diameter does not scale with mass in a linear fashion (Fig. 2a), a consideration of wet weight throughout the stranding period is a more robust proxy of events in the water column. Again, over the short time scale in question no discernable trend could be detected. For C. hysoscella, the size of stranded medusae displayed no distinct trend over the stranding period with individuals of all sizes present throughout. Care should be taken with the interpretation of these findings given the spatial separation of study sites for this species and the confined seasonality of stranding events (Doyle et al., 2006).
Lastly, and consistent with the other two species examined, the bell diameter of R. octopus (sites 2 and 3, 2003–2005) did not increase in a consistent manner over time when considered as raw data or weekly means (3rd January–24th December; n = 524; P > 0.05). Nevertheless, the protracted time scale over which R. octopus appears to strand (Fig. 3e) did allow for a more thorough assessment of seasonal patterns. For example, both the mean bell diameter and mean wet weights were greatest during the summer months (Table 2). The increase in bell diameter was a function of both the standard deviation (F 1,7 = 12.2, r 2 = 0.63, P < 0.05) and overall range of sizes recorded (F 1,7 = 13.28, r 2 = 0.65, P < 0.05) which both showed a marked increase from June through to October. An even more distinct pattern was evident for the estimated wet weights of stranded R. octopus over time, with standard deviation (F 1,7 = 79.39, r 2 = 0.91, P < 0.0001) and range (F 1,7 = 73.77, r 2 = 0.91, P < 0.0001) again driving an increase in mean weight during the summer.
Stranding events at individual sites
As the study area spanned several degrees of latitude, there was an intrinsic problem when considering all sites in conjunction given that variations in water temperature may lead to differences in the timing of ephyrae release and growth rate, resulting in a broad size range of medusae in the water column at the same time. To account for this possible bias, analyses were again conducted for bell diameter and wet weight data over the duration of respective stranding periods. Both the bell diameter and wet weight of C. capillata were again independent of day of year with variable sizes and weights evident throughout (site 1; 2004). Alternatively, C. hysoscella (site 5, 2006) displayed a modest, yet highly significant, negative relationship between bell diameter and day of year (F 1,505 = 78.39, r 2 = 0.13, P < 0.0001). Wet weight also appeared to decrease in a linear fashion with smaller individuals appearing more prominent towards the end of the stranding period (F 1,505 =65.23, r 2 =0.11, P < 0.0001). Finally, as with all sites combined, the bell diameter of R. octopus at site 2 (2004) was not correlated with day of year. However, mean bell diameter did again increase during the summer months with typically smaller individuals washing ashore during the winter (e.g., June: mean diameter (cm) = 47.1 (SD 13.9); December: mean diameter (cm) = 23.5 (SD 8.5). This pattern was not driven by a linear increase in bell diameter but by seasonal changes in both standard deviation (F 1,4 =11.62, r 2 =0.74, P < 0.05) and range (i1,4 = 11.21, r 2 =0.74, P < 0.05) throughout 2004 (Fig. 4e). Similarly, wet weight displayed no direct correlation with day of year, but displayed an increase in mean weight during the summer months [e.g., June: mean wet weight (Kg) = 10.6 (SD 3.1); December mean wet weight (Kg) = 3.4 (SD 2.1)]. This could again be explained by an increase in standard deviation (F 1,4 = 169.46, r 2 = 0.97, P < 0.0001) and range (F 1,4 = 125.47, r 2 = 0.97, P < 0.0001), which were strongly correlated.
Jellyfish stranding events at discrete sites during individual years. (a, b) C. capillata stranding data as bell diameter and estimated wet weights recorded for two proximate beaches (Benllech and Llanddona) at site 1 (the Isle of Anglesey) during 2004. (c, d) Bell diameter and estimated wet weight of stranded C. hysoscella recorded for a single beach at site 5 (Inch Strand), during 2006. For both species, stranded individuals of varied sizes and weights were found throughout. (e, f) Bell diameter and estimated wet weight of stranded individuals given as range for a single beach (Llanbedrog) at site 2 during 2004. Gaps earlier in the season reflect survey effort and should not be taken as evidence of no-stranding events
Accounting for variable survey effort
Lastly, we tested whether the number of surveys conducted in a particular week at a particular site was independent of the respective data collected. For example, the probability of finding jellyfish of a particular size in a particular week must logically increase with the number of surveys conducted. To test this hypothesis, the stranding data for each species given in Fig. 3 were divided into weeks. The number of surveys conducted in each week was then tested against stranded bell diameters given as (1) mean; (2) standard deviation; (3) range and (4) maximum and minimum sizes. In each case (and for each species) our data appeared independent of the numbers of surveys conducted (P < 0.05), validating the use of stranding events to assess changes in size and weight on an intra-seasonal scale.
Discussion
If we are to accurately predict the present and future role of schyphozoan jellyfish within marine ecosystems (Mills, 1995; Buecher et al., 2001; Purcell, 2005; Lynam et al., 2005) it is vital that we understand the temporal backdrop of life history events (Lucas, 2001). To this aim we put forward a number of hypotheses to explore the seasonality and life history of schyphozoan jellyfish in the Irish and Celtic Seas. Initially we tested if the size of stranded medusae for three species showed a general increase with time, or whether individuals of varying size where present in the water column at all times suggesting the protracted release of ephyrae over many months. The brief stranding period of C. capillata and C. hysoscella prevented a thorough analysis of temporal trends, although individuals of all sizes were recorded throughout providing possible evidence of protracted ephyrae release, or alternatively markedly different growth rates at population level.
For C. hysoscella the simultaneous presence of broad size ranges within the water column has been reported before (Buecher et al., 2001; Brierley et al., 2001; Brierley et al., 2005). Net tows conducted in the Benguela ecosystem conducted over a five day period revealed a size range of medusae from 6–66 cm, centering around a mean bell diameter of 26.8 cm (SE = 0.6 cm) (Brierley et al., 2001). For the Irish and Celtic Seas, C. hysoscella displays the briefest stranding period with 95% of all specimens washing ashore between 9th-July to 3rd August (2003–2005) (Doyle et al., 2006). Despite this temporal constraint, the size of medusae (when considered for a single site) displayed a slight negative correlation with day of year local scale (Fig. 4). Shoreline surveys on the Dingle Peninsula (site 5) gave some insight to this with a marked shift in the size frequency apparent throughout the summer. For example, at Inch Strand the proportion of medusae ≤10 cm increased from 7% on 9th June (N = 27) to 50% by 31st July (N = 49). A possible explanation for why such small individuals might strand comes from previous studies of A. aurita (Lucus and Lawes, 1998). For this species the onset of maturation occurs at the upper end of the size range for the population on a given date (Rasmussen, 1973; Brewer, 1989) followed by a period of spawning after which most adult medusae die (Lucas, 2001). The inference here is that stranded C. hysoscella may have undergone sexual maturation and gamete-release (independent of their bell diameter) after which they underwent senescence and washed ashore. Anecdotally, this idea is supported by the observation that even small freshly stranded individuals rarely possessed oral arms or peripheral tentacles suggesting they did not merely wash ashore as healthy specimens, but died previously at sea. The predominance of small individuals at the end of the season may also reflect the inability of such medusae to move against prevailing currents (Edwards et al., 1999; Mills, 2001) although this would result in the stranding of fully intact small individuals which was rarely the case.
The most thorough assessment of seasonality could be made for R. octopus with both range and overall variance (expressed as standard deviation) in size and estimated wet weight peaking around the end of June (sites 2 and 3) whilst only small and mid-sized individuals (<50 cm bell diameter) appeared to strand in the late autumn, winter and early spring. Studies of another rhizostome jellyfish, Rhopilema esculentum, in Japanese waters (Nakano, 1980 cited in Omori, 1981; Omori and Nakano, 2001) also suggested a summer peak in medusae size with an increase in average bell diameter and wet weight from 17 mm to 700 mm and 0.61 g–27 kg, respectively, between the middle of May and early September. These previous studies also suggested a general increase in the mean size of medusae over the same time frame, with the overall inference of an annual cohort increasing in size. The data presented here differ slightly form this orthodox pattern with the largest individuals washing ashore in June, with a subsequent decrease in mean bell diameter during the late summer/early autumn. This particular finding may reflect a number of possibilities. Firstly, the decreased size of medusae during the autumn logically reflects the removal of large individuals that tended to strand during the summer and should not be taken as evidence of medusae shrinkage. Indeed, if we consider the mean size and weight of medusae from January (32.2 cm, 3.3 kg) through to June (46.0 cm, 10.1 kg) (Table 2) there is some evidence of an overall increase in size throughout the year as previously reported (Nakano, 1980 cited in Omori, 1981; Omori and Nakano, 2001). Lastly, the release of ephyrae may differ in its timing and duration between the relevant studies, with the typically broad size ranges reported here masking the presence of a single, annual cohort. This pattern was most evident during mass strandings of R. octopus which appeared almost as ‘snapshot’ mortality events with individuals of all sizes dying at sea in close succession and washing ashore with the incoming tide (Fig. 5). However, unlike C. hysoscella there was no consistency in the condition of medusae with robust gamete-replete individuals simultaneously washing ashore with reproductively spent, malformed or small individuals with no visible evidence of sexual maturation. Although only a few studies exist on growth rates of rhizostome jellyfish (Omori and Nakano, 2001), some consideration must be given to the possibility of differential growth rates within population. Indeed, given that R. octopus is found in coastal embayments (Houghton et al., 2006b) where variations in containment, tidal flow, water depth, temperature and salinity and trophic conditions are considerable (Lucas, 2001) this possibility is not unfounded. Nevertheless, the size ranges given here (e.g., site 1: 14–77 cm, 24th June 2003; Fig. 2) would require extraordinary and prolonged variability to be driven entirely by such factors. More likely is that sharp thermal variations within such estuarine / coastal environments may themselves trigger multiple releases of ephyrae, as shown for the giant jellyfish Nemopilema nomurai (>2 m bell diameter) under laboratory conditions (Kawahara et al., 2006). Indeed, it is well established that thermal elevation is one of the triggers inducing strobilation in rhizostome jellyfish (Kawahara et al., 2006), as previously reported for Cephea cephea (Sugiura, 1966), Rhopilema esculentum (Chen and Ding, 1983) and Rhopilema nomadica (Lotan et al., 1994). As the scyphistoma of rhizostome jellyfish (Nemopilema nomurai) are capable of asexual reproduction for >1.5 years (Kawahara et al., 2006) the notion of protracted and repeated ephyrae release in the Irish Sea is certainly possible, although bespoke investigations of this claim are clearly required.
(a) Mass stranding of R. octopus on Whitford sands, Gower Peninsula, South Wales. (b) Freshly stranded C. capillata at River Nanny to Mosney Station, County Meath, Ireland. (c) Large stranded R. octopus on Whitford Sands, June 2003. (d) Measurements of diameter were taken across the top of the bell; figure shows a large C. hysoscella collected at sea in Ventry Bay, Dingle Peninsula during July 2006
The second hypotheses put forward was that year round stranding events of medusae provided empirical evidence that temperate schyphozoans did not exclusively over-winter as scyphistoma. Given that C. hysocella and C. capillata strand predominantly during summer months (Doyle et al., 2006) this question can only really be considered for R. octopus. For this species there was substantial evidence of over-wintering medusae with medium sized individuals washing ashore in the winter/early spring (Fig. 2). This pattern appears quite different to the findings of Pérez-Ruzafa et al. (2002) who used Bongo and Juday-Bogorov nets to record densities of Rhizostoma pulmo in the Mar Menor coastal lagoon in Spain. This revealed a more orthodox pattern of zooplankton abundance (e.g., Parsons et al., 1983) with R. pulmo present in the water column between June and September. However, the year round persistence of rhizostome medusae has been reported elsewhere, as in the case of Nemopilema nomurai along the coast of the Sea of Japan (Kawahara et al., 2006). In this study the majority of medusae had died off by mid-winter, possibly as a result of low water temperature (i.e., ∼10°C), starvation (Lucas and Lawes, 1998) or simply genetically determined senescence (Möller, 1980; Lucas, 2001). Nevertheless, a very small number of medusae did survive until the following March in a manner consistent with our findings. The reasons for such year-round persistence in the Irish Sea remain unclear although it is feasible that medusae undergo a winter diapause in deeper water as suggested for A. aurita at northerly Scandinavian sites (Hernroth and Gröndahl, 1983, 1985). For example, the rhizostomid Rhopilema esculentum in the Liaodong Bay, northeast China required 2–3 months for ephyra of 1.5–3.0 mm in diameter to reach maturity a bell 250–450 mm in diameter (Ding and Chen, 1981) implying that medusae stranding during the winter/early spring may have been present in the water column for some time prior to this. If so, then such behaviour must in some way be advantageous to the partially phyto-planktivorous R. octopus (Pérez-Ruzafa et al., 2002) when compared to the more carnivorous C. capillata and C. hysoscella (Russell, 1970; Purcell, 1991). Potentially, it might enable them to capitalise on the spring phytoplankton bloom from its onset as reported for other over-wintering zooplankton (Conover, 1988). For example, Calanus finmarchicus over-winters as the immature copepodite stage 5 (C5) in deep water (1000 m+), and then as the population ascend at the end of the winter they mature into C6s (Heath et al., 2000; Harris et al., 2000; Irigoien, 2004). The theory behind this is that they arrive in surface waters “ready to go” and release eggs which can feed on the spring bloom. Alternatively, a year round presence may simply imply that suitable prey is available to medusae beyond the summer growth period (Omori, 1981; Omori and Nakano, 2001), thus negating the need for a winter diapause. However, despite recent changes in the seasonality of phytoplankton in the north Atlantic (Reid et al., 1998), the herbivorous preferences of R. octopus render this unlikely unless medusae were able to completely shift predation towards over-wintering zooplankton. The presence of copepods and veliger larvae within the gut of Rhizostoma pulmo (Pérez-Ruzafa et al., 2002) suggest this is feasible, with over-wintering copepodites serving as potential prey items (Hirche, 1983; Heath et al., 2000) as recently suggested for zooplanktivorous basking sharks (Cetorhinus maximus) in the region (Sims et al., 2003). Such a strategy would be less effective for C. capillata given that newly released ephyrae were shown not to grow within a copepod dominated zooplankton but grew with an average rate of 16.5% day−1 (maximum of 30.6% day−1) when presented with ctenophores (Båmstedt et al., 1997). Indeed, the availability of gelatinous zooplankton for newly released ephyrae was suggested as a bottleneck in the development of C. capillata, placing constraints on the population succession and partially explaining inter-annual variations in the abundance of this large schyphozoan (Båmstedt et al., 1997). Consequently, given that the ctenophore Pleurobrachia pileus has a summer peak in British and Irish waters, (Frid et al., 1994) it would serve little purpose for C. capillata to be present as medusae until seasonal populations of such species were established later in the season. Similar reasoning could be applied to C. hysoscella given that the species is also known to prey on gelatinous zooplankton (Russell, 1970; Purcell, 1991) although further research is again required to verify such claims.
To summarise, stranding data for three scyphozoan jellyfish were explored over broad temporal and spatial scales. Using equations derived from previously gathered data we were able to estimate the wet weight of stranded medusae based on bell diameter alone. These data allowed us to explore a number of life history questions. In terms of reproductive cohorts, the broad size and weight ranges of stranded jellyfish implied that the release of ephyrae may be protracted (albeit to different extents) in each species, with individuals of all sizes present in the water column during the summer months. For R. octopus, there was a general increase in both mean bell diameter and wet weight from January through to June which was driven by increase in the variance and overall range of both variables during the summer. Consistent with previous studies, we also provided evidence that rhizostome jellyfish are capable of over-wintering as pelagic medusa which we hypothesise may enable them to capitalise on prey available earlier in the year.
References
Arai, M. N., 1997. A functional biology of Scyphozoa. Chapman & Hall, London.
Arai, M. N., 2001. Pelagic coelenterates and eutrophication: a review. Hydrobiologia 451: 69–87.
Båmstedt, U., H. Ishii & M. B. Martinussen, 1997. Is the scyphomedusa Cyanea capillata dependent on gelatinous prey for its early development. Sarsia 82: 269–273.
Beaugrand, G., K. M. Brander, A. Lindley, S. Souissi & P. C. Reid, 2003. Plankton effect on cod recruitment in the North Sea. Nature 426: 661–664.
Brewer, R. H., 1989. The annual pattern of feeding, growth, and sexual reproduction in Cyanea (Cnidaria: Scyphozoa) in the Niantic River estuary, Connecticut. Biological Bulletin of the Marine Biological Laboratory, Woods Hole 176: 272–281.
Brierley, A. S., B. E. Axelsen, E. Buecher, C. A. J. Sparks, H. Boyer & M. J. Gibbons, 2001. Acoustic observations of jellyfish in the Namibian Benguela. Marine Ecology Progress Series 210: 55–66.
Brierley, A. S., D. C. Boyer, B. E. Axelsen, C. P. Lynam, C. A. J. Sparks, H. J. Boyer & M. J. Gibbons, 2005. Towards the acoustic estimation of jellyfish abundance. Marine Ecology Progress Series 295: 105–111.
Buecher, E., C. Sparks, A. Brierley, H. Boyer & M. Gibbons, 2001. Biometry and size distribution of Chrysaora hysoscella (Cnidaria, Scyphozoa) and Aequora aequora (Cnidaria, Hydrozoa) off Namibia with some notes on their parasite Hyperia medusarum. Journal of Plankton Research 23: 1073–1080.
Chen, J. & G. Ding, 1983. Effect of temperature on the strobilation of edible medusa, Rhopilema esculenta Kishinouye Scyphozoa, Rhizostomeae). Acta Zoologia Sin 29: 195–206.
Conover, R. J., 1988. Comparative life histories in the genera Calanus and Neocalanus in high latitudes of the northern hemisphere. Hydrobiologia 167–168: 127–142.
Cushing, D. H., 1990 Plankton production and year-class strength in fish populations: an update of the match/mismatch hypothesis. Advances in Marine Biology 26: 249–292.
Ding, G. W. & J. K. Chen, 1981. The life history of Rhopilema esculenta Kishinouye. Journal of Fisheries, China 5: 93–102.
Doyle, T. K., J. D. R. Houghton, S. M. Buckley, G. C. Hays & J. Davenport, 2006. The distribution of five scyphozoan jellyfish species across a temperate coastal environment. Hydrobiologia 579: 29–39.
Doyle, T. K., J. D. R. Houghton, R. McDevitt, G. C. Hays & J. Davenport, 2007. The energy density of jellyfish: estimates from bomb-calorimetry and proximate-composition. Journal of Experimental Marine Biology and Ecology, DOI: 10.1016/j.jembe.2006.12.010.
Edwards, M., A. W. G. John, H. G. Hunt & Lindley J. A., 1999. Exceptional influx of oceanic species into the North Sea late 1997. Journal of the Marine Biological Association of the United Kingdom 79: 737–739.
Frid, C. L. J., L. C. Newton & J. A. Williams, 1994. The feeding rates of Pleurobrachia (ctenophore) and Sagitta (chetognatha) with notes on the seasonal role of planktonic predators in the dynamics of North Sea zooplankton communities. Aquatic Ecology 28: 181–191.
Harris, R. P., X. Irigoien, R. N. Head, C. Rey, B. H. Hygum, B. W. Hansen, B. Niehoff, B. Meyer-Harms & F. Carlotti, 2000. Feeding, growth and reproduction in the genus Calanus. ICES Journal of Marine Science 57: 1708–1726.
Hay, S. J., J. R. G. Hislop & A. M. Shanks, 1990. North Sea scyphomedusae: summer distribution, estimated biomass and significance particularly for O-group gadoid fish. Netherlands Journal of Sea Research 25: 113–130.
Hays, G. C., A. J. Richardson & C. Robinson, 2005. Climate change and marine plankton. Trends in Ecology & Evolution 20: 337–344.
Heath, M. R., J. G. Fraser, A. Gislason, S. J. Hay, S. H. Jonasdottir & K. Richardson, 2000. Winter distribution of Calanus finmarchicus in the northeast Atlantic. ICES Journal of Marine Science 57: 1628–1635.
Hernroth, L. & F. Gröndahl, 1983. On the biology of Aurelia aurita (L.): 1. Release and growth of Aurelia aurita (L.) ephyrae in the Gullmarfjorden, western Sweden. Ophelia 22: 189–199.
Hernroth, L. & F. Gröndahl, 1985. On the biology of Aurelia aurita (L.): 2. Major factors regulating the occurrence of ephyrae and young medusae in the Gullmarfjorden, western Sweden. Bulletin of Marine Science 37: 567–576.
Houghton, J. D. R., T. K. Doyle, J. Davenport & G. C. Hays, 2006a. Jellyfish aggregations and leatherback turtle foraging patterns in a temperate coastal environment. Ecology 98: 1967–1972.
Houghton, J. D. R., T. K. Doyle, J. Davenport & G. C. Hays, 2006b. Developing a simple, rapid method for identifying and monitoring jellyfish aggregations from the air. Marine Ecology Progress Series 314: 159–170.
Hirche, H. J., 1983. Overwintering of Calanus finmarchicus and C. helgolandicus. Marine Ecology Progress Series 11: 281–290.
Irigoien, X., 2004. Some ideas about the role of lipids in the life cycle of Calanus finmarchicus. Journal of Plankton Research 26: 259–263.
Kawahara, M., S.-I. Uye, K. Ohtsu & H. Lizumi, 2006. Unusual population explosion of the giant jellyfish Nemopilema nomurai (Scyphozoa: Rhizostomeae) in East Asian waters. Marine Ecology Progress Series 307: 161–173.
Kingsford, M. J., K. A. Pitt & B. M. Gillanders, 2000. Management of jellyfish fisheries, with special reference to the Order Rhizostomeae. Oceanography and Marine Biology 38: 85–156.
Lotan, A., M. Fine & R. Ben-Hillel, 1994. Synchronization of the life cycle and dispersal pattern of the tropical invader scyphomedusan Rhopilema nomadica is temperature dependent. Marine Ecology Progress Series 109:59–65.
Lucas, C. H. & S. Lawes, 1998. Sexual reproduction of the scyphomedusa Aurelia aurita in relation to temperature and variable food supply. Marine Biology 131:629–638.
Lucas, C. H., 2001. Reproduction and life history strategies of the common jellyfish, Aurelia aurita, in relation to its ambient environment. Hydrobiolgia 451: 229–246.
Lynam, C. P., S. J. Hay & A. S. Brierley, 2004. Interannual variability in abundance of North Sea jellyfish and links to the North Atlantic Oscillation. Limnology and Oceanography 49: 637–643.
Lynam, C. P., S. J. Hay & A. S. Brierley, 2005. Jellyfish abundance and climatic variation: contrasting responses in oceanographically distinct regions of the North Sea, and possible implications for fisheries. Journal of the Marine Biological Association of the United Kingdom 85: 435–450.
Lynam, C. P., M. J. Gibbons, B. E. Axelsen, C. A. J. Sparks, J. Coetzee, B. G. Heywood & A. S. Brierley, 2006. Jellyfish overtake fish in a heavily fished ecosystem. Current Biology 16(13): R492.
Möller, H., 1980. Population dynamics of Aurelia aurita medusae in Kiel Bight, Germany (FRG). Marine Biology 60:123–128.
Möller, H., 1984. Reduction of larval herring population by jellyfish predator. Science 224: 621–622.
Mills, C. E., 1995. Medusae, siphonophores and ctenophores as planktivorous predators in changing global ecosystems. ICES Journal of Marine Science 52: 575–581.
Mills, C. E., 2001. Jellyfish blooms: are populations increasing globally in response to changing ocean conditions. Hydrobiologia 451: 55–68.
Omori, M., 1981. Edible jellyfish (Scyphomedusae: Rhizostomeae) in the Far East waters: a brief review of the biology and fishery. Bulletin of the Plankton Society of Japan 28: 1–11.
Omori, M., H. Ishii & A. Fujinaga, 1995. Life history strategy of Aurelia aurita (Cnidaria, Scyphomedusae) and its impact on the zooplankton community of Tokyo Bay. ICES Journal of Marine Science 52: 597–603.
Omori, M. & E. Nakano, 2001. Jellyfish fisheries in Southeast Asia. Hydrobiologia 451: 19–26.
Pauly, D., V. Christensen, J. Dalsgaard, R. Froese & F. Torres, 1998. Fishing down marine food webs. Science 279: 860–863.
Parsons, T. R., M. Takahashi & B. Hargrave, 1983. Biological Oceanographic Processes. Pergamon Press, Oxford.
Pérez-Ruzafa, A., J. Gilabert, J. M. Gutiérrez, A. I. Fernández, C. Marcos & S. Sabah, 2002. Evidence of a planktic food web response to changes in nutrient input dynamics in the Mar Menor coastal lagoon, Spain. Hydrobiologia 475/476: 359–369.
Purcell, J. E., 1991. A review of cnidarians and ctenophores feeding on competitors in the plankton. Hydrobiologia 216/217: 335–342.
Purcell, J. E., 2005. Climate effects on formation of jellyfish and ctenophore blooms: a review. Journal of the Marine Biological Association of the United Kingdom 85: 461–476.
Purcell, J. E & M. N. Arai, 2001. Interactions of pelagic cnidarians and ctenophores with fish: a review. Hydrobiologia 451: 27–44.
Purcell, J. E., J. R. White, D. A. Nemazie & D. A. Wright, 1999. Temperature, salinity and food effects on asexual reproduction and abundance of the scyphozoan Chrysaora quinquecirrha. Marine Ecology Progress Series 180: 187–196.
Rasmussen, E., 1973. Systematics and ecology of the Isefjord marine fauna (Denmark). Ophelia 11: 41–46.
Reid, P. C., M. Edwards, H. G. Hunt & A. J. Warner, 1998. Phytoplankton change in the northern Atlantic. Nature 391: 546.
Russell, F. S., 1970. The Medusae of the British Isles. II. Pelagic Scyphozoa with a Supplement to the First Volume on Hydromedusae. Cambridge University Press, Cambridge: 284.
Sugiura, Y., 1966. On the life history of rhizostome medusae. 4. Cephea cephea. Embryologia 9:105–122.
Sims, D. W., E. J. Southall, A. J. Richardson, P. C. Reid & J. D. Metcalfe, 2003. Seasonal movements and behaviour of basking sharks from archival tagging: no evidence of winter hibernation. Marine Ecology Progress Series 248: 187–196.
Acknowledgements
Funding was provided by: (1) INTERREG IIIA (European Regional Development Fund); (2) the Countryside Council for Wales Species Challenge Fund (3) the Marine Conservation Society; (4) National Parks and Wildlife Service (Ireland) and (5) The Marine Institute (Ireland). Special thanks to David Jones, Vincent, Sean and Christina Rooney, Jim and Rose Hurley, Kevin McCormack, Eithne Lee, Maria Doyle, Kate Williamson, Irena Kruszona and colleagues, Vernon Jones, Tom Stringell, Ailish Murphy and Irene Fitzgerald and everyone else who assisted with the shoreline surveys.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: K. Martens
Rights and permissions
About this article
Cite this article
Houghton, J.D.R., Doyle, T.K., Davenport, J. et al. Stranding events provide indirect insights into the seasonality and persistence of jellyfish medusae (Cnidaria: Scyphozoa). Hydrobiologia 589, 1–13 (2007). https://doi.org/10.1007/s10750-007-0572-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-007-0572-2