Abstract
Summer outbreaks of the dinoflagellate Alexandrium taylorii Balech are recurrent events in nearshore waters of Sicily (Italy)—a central region in the Mediterranean Sea—producing dense yellowish–green patches. Beyond the local phenomenon, the problem covers a broader geographic scale, involving also other European localities, mostly in Spain.
Biological, environmental, and molecular data are reported here from a semi-closed bay of Sicily (Vulcano Island, Tyrrhenian Sea, 2000–2003), showing in summer the recurrence of high-biomass blooms and events of water discolouration. Without underestimating the setbacks to the tourism industry, the ecological impact of A. taylorii blooms may be important considering the high levels of biomass produced (West Bay, Vulcano: up to a magnitude order of 107 cells l−1, 50–180 μg-Chla l−1, June 2002 and 2003) and coincident conditions of oxygen supersaturation of the waters (130–170%). Trophic trends in the Tyrrhenian site indicate high amounts of nutrients linked to the increased anthropogenic activity in summer, although recently there has been an apparent shift of the marked eutrophic conditions towards a slighter eutrophy.
Genetic data on isolates of A. taylorii from the Mediterranean Sea are also discussed. Molecular analyses implied the sequencing of target rDNA regions (5.8S rDNA and ITS regions) of several isolates from different Mediterranean localities, as well as the application of species-specific PCR assays for rapid species identification in preserved field samples. The confirmation of the specific identity provided new insights into the biogeography of this species and further evidence of the occurrence of A. taylorii in a number of Mediterranean localities, both in the western side (the Catalan coast of Spain) and the eastern area (Greece). Analyses of the molecular diversity of geographically distinct isolates of A. taylorii from Italy, Spain, and Greece based on the 5.8S rDNA-ITS region sequences showed a high level of similarity, indicating the existence of an unique Mediterranean population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Blooms of phytoplankton organisms are recurrent events in European coastal waters and worldwide (Anderson, 1989), being potentially harmful to human health, marine ecosystems and resources such as tourism, fisheries and aquaculture. The socio-economic impact of HABs (Harmful Algal Blooms) highlights the need for ecological studies addressed to the understanding of HAB dynamics and processes of bloom formation and dispersal. Summer outbreaks of the dinoflagellate Alexandrium taylorii Balech produce dense yellowish–green patches of limited extension in coastal waters of Sicily, Italy (Giacobbe & Yang, 1999; Penna et al., 2002), a central island in the Mediterranean Sea. Beyond the local phenomenon, similar events are also observed in beach areas along the Costa Brava and Balearic Islands (Spain), causing in some localities water discolouration lasting two months (Garcés et al., 1999). Dense aggregates of cells are also found near the sediment, this representing a possible way of avoiding advection from the area and prevent population losses. As a whole, the observations reported in literature suggest that the magnitude of the event is increasing in some places and that this species is in a phase of expansion, probably due to ballast water release and other causes.
Some authors have shown the morphological variability of A. taylori, the high production of temporary cysts having a significant role in the bloom dynamics (Garcés et al., 2002), and the existence of a sexual resting stage as a part of its life cycle (Delgado et al., 1997, Giacobbe & Yang, 1999). Thus, the specific identity may be easily masked when dealing with encysted stages—vegetative or sexual resting cysts.
In this study, we examined a set of biological and environmental data obtained in the last four years (2000–2003, EU Project Strategy, EVK3-CT-2001–00046) from a Tyrrhenian locality of Sicily subject to bloom events. Sequence analyses of different strains of A. taylorii isolated from various Mediterranean areas, as well as results of the application of molecular probes (by PCR assays) to preserved field samples, are also provided.
Study area
The West Bay of Vulcano (Sicily), a tourist locality of the Aeolian Islands (Tyrrhenian Sea), is a shallow, semicircular area extending approximately 300 × 100 m. The site is located at 38°25′04′′–10′′ Lat N and 14°57′14′′–23′′ Long E and covers as a whole about 7 ha. Recurrent summer blooms of A. taylorii take place in some nearshore points of the bay with evident water discolouration due to the high-biomass produced by this dinoflagellate (Penna et al., 2002). Before 2000, there had been no studies on HABs in this Tyrrhenian locality, although popular references on events of red tide date back to 1995.
Materials and methods
Sample collection and analyses
Three nearshore stations (1 m depth) located in the West Bay of Vulcano (Stn 1: 38°25′4′′ N, 14°57′14′′ E; Stn 2: 38°25′6′′ N, 14°57′19′′ E; Stn 3: 38°25′10′′ N, 14°57′23′′ E) were sampled in spring-summer 2000–2003, on 7–19 occasions each year. 100–250 ml of surface seawater samples were collected and fixed with Lugol’s solution (0.4% final concentration). Samples were taken at the same time of day (between 12 and 15 h). The bloom density was determined by cell counts of target species and other phytoplankton groups. The procedure for identification and quantification of phytoplantkon cells involved: sedimentation of a subsample in 10 or 50 ml settling chambers and counts of cells in an appropriate area (Throndsen, 1995) using an inverted Zeiss Axiovert 200 microscope equipped with epifluorescence. The identity of A. taylorii was confirmed by staining the cells with Calcofluor White M2R (Fritz & Triemer, 1985) and using a Zeiss UV filter set Fs 01.
Measures of environmental parameters such as water temperature, salinity and oxygen were taken by a mobile Multiprobe (YSI 556 MPS). Nutrient samples were filtered and frozen immediately after water collection. Concentrations of nitrate, nitrite and phosphate were determined following the Strickland and Parsons (1972) method, and ammonia according to Aminot & Chaussepied (1983).
Clonal cultures of Alexandrium taylorii were established at the Consiglio Nazionale delle Ricerche (CNR, Messina), Institut de Ciències del Mar (CSIC, Barcelona), and Instituto Español de Oceanografia (IEO, Vigo) from unfixed seawater samples taken at various sites of the Mediterranean. All marine cultures were maintained in F/2 or F/20 medium (see http://ccmp.bigelow.org/), at 18 ± 1°C and 12:12 h (light:dark) photoperiod. Illumination was provided by a photon irradiance of 100 μmol m−2 s−1.
Molecular assays
Total DNA extractions of different A. taylorii isolates and mixed phytoplankton populations from seawater samples collected in different localities of the Mediterranean basin—Vulcano (Italy), St. Carles, Catalan Sea (Spain), and Saronikos Gulf (Greece)—were performed by using DNeasy Plant Kit (Qiagen, Valencia, CA, USA), according to the manufacturer’s instructions. PCR amplification conditions of the 5.8S gene and ITS regions were performed as described in Penna & Magnani (1999). PCR amplified products of all A. taylorii isolates used in this study were directly sequenced. The sequences were deposited at EMBL Bank and given in Table 1.
Sequence alignment was done with Clustal X software and subsequently improved by eye. The rDNA-ITS regions were aligned with other ITS sequences of Alexandrium species, listed in GenBank. Sequence alignment analyses were also carried out by including other sequence data of Alexandrium species from GenBank (Table 2). Phylogenetic relationships, based on the 5.8S rDNA-ITS regions, were inferred using the Neighbor-Joining (NJ) (Saitou & Nei, 1987). The sequence of Gymnodinium sanguineum Hirasaka [=Akashiwo sanguinea (Hirasaka) Hansen & Moestrup] (GenBank AF131075) was used as outgroup. Robustness of the phylogenetic tree generated by NJ was tested by using the non-parametric bootstrap (Felsenstein, 1985) with 10,000 pseudoreplicates. The above analysis was performed with the software package MEGA ver. 3.0.
A. taylorii specific PCR assays were carried out on the genomic DNA of mixed phytoplankton population samples by using two species-specific designed primers (Penna et al., 2004). The PCR products were visualized on 2.0% agarose gel.
Results
Spring-summer A. taylorii densities in the last four years (2000–2003) showed exceptionally high values (Fig. 1), compared with the concentrations of phytoplankton usually found in Sicilian coastal waters. On various occasions (2001–2003), the bloom density reached a 107 order of magnitude, with maxima of 2−3×107 cells l−1 and amounts of Chla as high as 50−180 μg l−1 in June 2002 and 2003. During these blooms, the water temperature was 27.5–32°C, salinity 37.1–37.2, dissolved inorganic nitrogen 2.42–2.88 μM DIN, phosphates 2.32–5.48 μM, with a corresponding condition of oxygen supersaturation of the waters (up to 170%). The highest concentrations of DIN were due to nitrates, or alternatively to ammonia, and phosphates in some cases reached values of ≈10 μM (2001, Fig. 1) with a slight decreasing trend through the years.
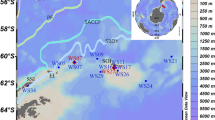
The water discolouration lasted 1–2 months, as confirmed again in 2004 and 2005 (data not shown), gradually disappearing in July or August–September (2001). The bloom persistence and discolouration was apparently favoured by optimal, summer weather conditions. The horizontal distribution of A. taylorii cells within the West Bay (Vulcano) was characterized in all cases by the assembling of cells nearshore, in the most sheltered part of the bay, as shown in Fig. 2, although in some cases a wind-driven advection of cells to NE of the bay was observed.
The percentage of temporary cysts versus motile cells of A. taylorii (Fig. 3), as determined in 2001 in water samples, showed an incidence of resting vegetative stages of 7–11% over the total A. taylorii population, with the highest percentages near the sediment. These values increased to 22% in late summer when the bloom was declining.
The NJ phylogenetic tree of A. taylorii and other Alexandrium species based on the 5.8S rDNA-ITS regions is shown in Fig. 4. In respect to the other species of Alexandrium examined to date, A. taylorii appeared to diverge. Within the genus Alexandrium, the Mediterranean A. taylorii isolates formed a homogeneous clade, distinct from the others. These isolates shared identical nucleotide sequences.
Neighbor-Joining (NJ) tree inferred from the 5.8S rDNA and ITS sequences of Alexandrium species. Sequences of A. taylorii from this study are in bold. G. sanguineum was used as an outgroup. Numbers on the major nodes represent NJ (10000 pseudo-replicates) bootstrap values. Only bootstrap values ≥ than 50% are shown
Species-specific PCR assays of fixed field samples are shown in Fig. 5. By using species-specific primers designed for A. taylorii, PCR amplifications of the 5.8S rDNA-ITS regions yielded products of 297 base pair size for the field samples containing A. taylorii cells. Species-specific PCR amplifications for A. taylorii revealed positive results for the presence of this species in the Vulcano phytoplankton population samples in agreement with the microscopic examination.
A. taylorii specific PCR assays of mixed phytoplankton population samples from different Mediterranean localities. (a) template plasmid containing A. taylorii 5.8S rDNA-ITS regions at different concentrations: lane 1, 10 ng; lane 2, 1 ng; lane 3, 0.1 ng. (b) Saronikos Gulf (Greece) field sample: lane 1, phytoplankton template DNA (1 ng); lane 2, template plasmid containing A. taylorii 5.8S rDNA-ITS regions (1 ng); lane 3, negative control (no template DNA). (c) St. Carles field sample, Catalan Sea (Spain): lane 1, phytoplankton template DNA (1 ng); lane 2, negative control (no template DNA); lane 3, template plasmid containing A. taylorii 5.8S rDNA-ITS regions (1 ng). (d) Vulcano West Bay field samples, Tyrrhenian Sea (Italy), 15th June 2003: lane 1, Station1 phytoplankton template DNA (1 ng); lane 2, Station 3 phytoplankton template DNA (1 ng); lane 3, Station2 phytoplankton template DNA (1 ng); lane 4, Station Capo Testa Grossa phytoplankton template DNA (1 ng); lane 5, template plasmid containing A. taylorii 5.8S rDNA-ITS regions (1 ng). (e) Vulcano field sample, West Bay Station1: lane 1, phytoplankton template DNA (10 ng), 16th June 2003; lane 2, phytoplankton template DNA (0.1 ng), 16th June 2003; lane 3, phytoplankton template DNA (0.01 ng), 16th June 2003; lane 4, phytoplankton template DNA (10 ng), 17th June 2003; lane 5, phytoplankton template DNA (0.1 ng), 17th June 2003; lane 6, phytoplankton template DNA (0.01 ng), 17th June 2003. M, size standards; arrow indicates reactions that produced a single species-specific PCR product
Discussion
Without understimating the setbacks to the tourism industry, the ecological impact of A. taylorii blooms may be important considering the high levels of biomass produced, as in the case of the West Bay, Vulcano (up to 3 × 107 cells l−1) and coincident conditions of oxygen supersaturation of the waters, that may threaten marine life. Trophic trends in the Tyrrhenian target site indicate high amounts of nutrients linked to the increased anthropogenic activity in summer (see also Penna et al., 2001), although we observed an apparent shift of the marked eutrophic conditions towards a slighter eutrophy. This could be due to a recent improvement of the sewage treatment. Previous studies in this area suggested that nutrients play a relevant role in the development and maintenance of A. taylorii blooms, the high concentrations in summer of inorganic nitrogen and phosphorus being probably due to the hotel sewage, when the anthropogenic pressure increases in relation to the tourism (Penna et al., 2002). Then, climatic conditions such as high water temperature, calm sea and weak tide, seem to favour the bloom development. Garcés et al. (2002) suggested that under short-term environmental perturbations, for instance storms or swells, A. taylorii encysts to withstand the unfavourable conditions and restores the population after a few days of calm weather, thus functioning as a population stock. In addition, encystment would also reduce the population losses from the area, also through cell aggregation in clusters of cysts. This, together with the physical characteristics (relatively low water renewal) and favourable wind conditions, may favour population growth and persistence (Garcés et al., 1999, Basterretxea et al., 2005).
Many factors, natural or anthropogenic, are known to contribute to the spreading worldwide of HAB species. Such phenomena are amplified by human activities, such as the wrong management of the coastal area leading to increased amounts of nutrients in the waters and eutrophy, lack of seawage treatments, use of fertilizers in agriculture, the sea discharge of ship’s ballast water favouring the introduction of non-indigenous organisms, the so-called “alien species”. Cell dispersion can also be favoured by anthropogenic means, as plastics (Masò et al., 2003) or movement of recreational boats in summer. Therefore, areas previously free by harmful species can later become subject to HABs, if such species find in the new environment conditions favouring cell growth.
In the specific case of A. taylorii, we obtained further evidence for its spreading in the Mediterranean region, from the western side (the Catalan coast of Spain) to the eastern area (Greece).
Aiming at the improvement of the HAB detection tools, species-specific primers for A. taylorii were applied in this study in parallel to conventional techniques. This allowed the detection of the target organism also when it occurred as resting stage and the exact species identity was more difficult to be determined. The application of the PCR analysis to field seawater samples, evidenced the presence of A. taylorii in all mixed phytoplankton population samples collected in different sampling points in the Vulcano West Bay, with exception of the Station 3, where the cell abundance was often extremely low. Further, the species-specific primer application to the PCR analyses resulted also suitable for the Greek and Spanish field samples containing the target species. These results on the species-specific detection of A. taylorii by PCR assay were always in agreement with the microscopic examination.
The phylogenetic analyses of geographically distinct isolates of A. taylorii (Italy, Spain, Greece) indicated the existence of a unique, homogeneous Mediterranean population, constituted by isolates with identical ITS-5.8S rDNA sequences.
In this study, the confirmation of the specific identity through the coupling of microscopic and molecular analyses provided new insights into the biogeography of A. taylorii, showing the extension of its geographic range from the western Mediterranean to the eastern area. Future researches will give priority to the advancement in both control and prevention strategies as safeguard of the coastal marine environment and public health, and development of advanced technologies for the rapid detection and mitigation of HAB events through laboratory experiments and in situ application.
References
Anderson, D. M., 1989. Toxic algal blooms and red tides: a global perspective. In Okaichi T., D. M. Anderson & T. Nemoto (eds), Red Tides: Biology, Environmental Science and Toxicology. Elsevier, New York, 11–16.
Aminot, A. & M. Chaussepied 1983. Manuel des analyses chimiques en milieu marin. Centre National pour l’Exploration des Oceans, 1–1395.
Basterretxea, G., E. Garcés, A. Jordi, M. Masò & J. Tintoré, 2005. Breeze conditions as a favoring mechanism of Alexandrium taylori blooms at a Mediterranean beach. Estuarine, Coastal & Shelf Science 62: 1–12.
Delgado, M., E. Garcés, M. Vila & J. Camp, 1997. Morphological variability in three populations of the dinoflagellate Alexandrium taylori. Journal of Plankton Research 19: 749–757.
Felsenstein, J., 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39: 783–791.
Fritz, L. & R. E. Triemer, 1985. A rapid simple technique utilizing calcofluor white M2R for the visualization of dinoflagellate thecal plates. Journal of Phycology 21: 662–664.
Garcés, E., M. Masò & J. Camp, 1999. A recurrent and localized dinoflagellate bloom in a Mediterranean beach. Journal of Plankton Research 21: 2373–2391.
Garcés, E., M. Masò & J. Camp, 2002. Role of temporary cysts in the population dynamics of Alexandrium taylori (Dinophyceae). Journal of Plankton Research 24: 681–686.
Giacobbe, M. G. & X. Yang, 1999. The life history of Alexandrium taylori (Dinophyceae). Journal of Phycology 35: 331–338.
Masó, M., E. Garcés, F. Pagès & J. Camp, 2003. Drifting plastic debris as a potential vector for dispersing Harmful Algal Blooms (HAB) species. Scientia Marina 67: 107–111.
Penna A., G. Fusco, E. Bertozzini, M. Vila, E. Garcés, M.G. Giacobbe, A. Luglié, M. Masò, L. Galluzzi & M.Magnani, 2004. Monitoring of HAB species in the Mediterranean Sea through a filter system PCR assay detection method. 11th International Conference on Harmful Algae. Cape Town, 15–19 November 2004. Abstract.
Penna, A., M. G. Giacobbe, F. Andreoni, E. Garcés, S. Berluti, R. Cantarini, N. Penna & M. Magnani, 2001. Blooms of Alexandrium taylori (Dinophyceae) in the Mediterranean: a preliminary molecular analysis of different isolates. In Hallegraeff G. M., S. I. Blackburn, C. J. Bolch & R. J. Lewis (eds), Harmful Algal Blooms 2000. UNESCO, Paris, 218–221.
Penna, A., M. G. Giacobbe, N. Penna, F. Andreoni & M. Magnani, 2002. Seasonal blooms of the HAB dinoflagellate Alexandrium taylori Balech in a new Mediterranean area (Vulcano, Aeolian Islands). Marine Ecology 23: 320–328.
Penna, A. & M. Magnani, 1999. Identification of Alexandrium (Dinophyceae) species using PCR and rDNA-targeted probes. Journal of Phycology 35: 615–621.
Saitou, N. & M. Nei, 1987. The Neighbour-Joining method: a new method for reconstructing phylogenetic trees. Molecular Biology and Evolution 4: 406–25.
Strickland, J. D. H. & T. R. Parsons, 1972. A Practical Handbook of Seawater Analysis. Fisheries Research Board of Canada, Ottawa.
Throndsen, J., 1995. Estimating Cell Numbers. In Hallegraeff G. M., D. M. Anderson & A. D. Cembella (eds), Manual on Harmful Marine Microalgae Intergovernamental Oceanographic Commission Manuals and Guides no 33. UNESCO, Paris, 63–80.
Vila, M., M. G. Giacobbe, M. Maso’, E. Gangemi, A. Penna, N. Sampedro, F. Azzaro, J. Camp & L. Galluzzi, 2005. A comparative study on recurrent blooms of Alexandrium minutum in two Mediterranean coastal areas. Harmful Algae 4: 673–695.
Acknowledgements
This study was supported by the EU project STRATEGY (EVK3-2001-00046). Thanks to Mariagrazia Galletta, Antonella Marini, Franco Decembrini, Francesco Raffa for analyses of nutrients, biomass and technical cooperation, Dr. Francesca Andreoni and Dr. Elena Bertozzini for molecular analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Giacobbe, M.G., Penna, A., Gangemi, E. et al. Recurrent high-biomass blooms of Alexandrium taylorii (Dinophyceae), a HAB species expanding in the Mediterranean. Hydrobiologia 580, 125–133 (2007). https://doi.org/10.1007/s10750-006-0459-7
Issue Date:
DOI: https://doi.org/10.1007/s10750-006-0459-7