Abstract
The objective is to investigate the relationship between arterial stiffness measured by arterial tonometry and echocardiographic indices of diastolic dysfunction—a basis for the diagnosis of heart failure with preserved ejection fraction (HFpEF). OvidSP Medline, Embase and PubMed were systematically searched. Eligible articles correlated arterial stiffness measured by brachial-ankle pulse wave velocity (baPWV), carotid-femoral pulse wave velocity (cfPWV), augmentation index (AIx) or cardio-ankle vascular index (CAVI) with indices of diastolic dysfunction, E/A ratio, peak early mitral annular velocity (e′) and E/e′ ratio. Correlation coefficients were determined using a random-effects model. Twenty-seven studies with 6,626 patients were included. baPWV was significantly correlated with E/A ratio (r = −0.434, 95 % CI −0.387 to −0.479), e′ (r = −0.499, 95 % CI −0.448 to −0.548) and E/e′ ratio (r = 0.372, 95 % CI 0.251–0.481). cfPWV was significantly correlated with E/A ratio (r = −0.391, 95 % CI −0.304 to −0.470) and E/e′ ratio (r = 0.210, 95 % CI 0.300–0.116), but not e′. AIx was significantly correlated with E/A ratio (r = −0.356, 95 % CI −0.255 to −0.450), e′ (r = −0.313, 95 % CI −0.195 to −0.423) and E/e′ ratio (r = 0.321, 95 % CI 0.250–0.388). CAVI was significantly correlated with E/A ratio (r = −0.405, CI −0.324 to −0.481), e′ (r = −0.449, 95 % CI −0.340 to −0.630), but not E/e′. baPWV showed significantly greater correlation with diastolic dysfunction compared to most other tonometric techniques. Arterial stiffness measured by arterial tonometry is an indicator of diastolic dysfunction with baPWV demonstrated the most consistent and strongest association. These data suggest a refocusing on the impact of arterial stiffness on the left ventricle as a potential causative factor leading to HFpEF.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Heart failure (HFpEF) with preserved ejection fraction is currently a major health challenge because of its high prevalence, high morbidity and mortality, poorly understood pathophysiology and paucity of specific therapy [1, 2]. The pathophysiology of HFpEF is complex and multifactorial [2], but its definition maybe condensed to the presence of normal systolic left ventricular performance in the presence of diastolic left ventricular dysfunction [1]. Indeed, left ventricular diastolic function identifies patients at high risk of the development of heart failure as well as serving as a predictor of an increased mortality from heart failure [3, 4]. The clinical and radiographic differences between diastolic and systolic heart failure are often subtle and can be indistinguishable, prompting the need for additional diagnostic tools. Although cardiac catheterization is considered the most accurate method for diagnosing diastolic dysfunction, it is an invasive procedure that cannot be easily used in the general population. The primary imaging modality currently used to assess diastolic dysfunction is Doppler echocardiography, although there are controversies over the accuracy of its assessment [5–8].
Conventional Doppler echocardiography measures intracardiac blood flow velocities, providing values of early diastolic mitral flow velocity (E), late diastolic mitral flow velocity (A) and the resulting ratio (E/A ratio) [9]. With diastolic dysfunction, ventricular relaxation is impaired, which decreases E velocity, and there is a greater atrial contraction, which increases A velocity [6]. These changes in diastolic function are reflected in a decreased E/A ratio [10]. The E/A ratio is an important echocardiography index for characterizing the severity and staging diastolic dysfunction [9]. Conventional flow Doppler echocardiography assessment of diastolic function has been critiqued for its dependence on preload [11, 12]. Myocardial velocities are measured throughout the cardiac cycle, including the peak early diastolic mitral annular velocity (e′) [12]. The ratio of E mitral flow velocity to e′ (E/e′ ratio) has been reported to be comparable to invasive measures for assessment of diastolic function [13]. Furthermore, e′ and E/e′ are more preload independent and have utility in evaluating diastolic dysfunction [13–15].
Arterial stiffness has been proposed to play a role in the prediction of left ventricular diastolic dysfunction [16–18]. There are invasive and noninvasive methods of evaluating arterial stiffness, including vascular catheterization, ultrasound, magnetic resonant imaging [19] and arterial tonometry [20]. Arterial tonometry is an accessible, noninvasive assessment of arterial stiffness that can be done at the bedside [21]. Brachial-ankle pulse wave velocity (baPWV), carotid-femoral pulse wave velocity (cfPWV), augmentation index (AIx) and cardio-ankle vascular index (CAVI) are tonometric techniques to measure arterial stiffness [22–24]. baPWV is calculated from the distance between the arms and ankles determined by anthropomorphic data based on a person’s height, divided by the propagation time of the pulse wave between the two points, measured with occlusion cuffs [25]. cfPWV is determined by manually measuring the distance between the carotid and femoral arteries, and dividing by the time for pulse wave propagation between the two points [26]. Augmentation index is defined as the percentage of total pulse pressure attributed to the reflected pulse wave, calculated from the two pressure peaks of a pulse waveform [27]. CAVI is measured similarly to baPWV, but is calculated using an equation involving systolic and diastolic blood pressures, heart-ankle pulse wave velocity, pulse pressure and blood density [28].
Observational studies suggest that the different tonometric techniques correlate with markers of diastolic dysfunction. However, most studies have had relatively small sample sizes, and only a few studies have compared the different techniques [18, 24, 29]. Meta-analysis has the advantage of combining the study populations to come to meaningful conclusions. There has not been a comprehensive analysis of arterial stiffness and diastolic dysfunction. We performed a systematic review of original studies that compared the four different tonometric measures of arterial stiffness with echocardiographic indices of diastolic dysfunction. This meta-analysis tested the hypothesis that arterial stiffness measured by arterial tonometry correlates significantly with echocardiographic parameters of diastolic dysfunction.
Methods
Search strategy
A systematic search was conducted by the two authors to identify studies that examined the relationship between arterial stiffness and left ventricular diastolic dysfunction. We searched the Medline and EMbase databases using the PubMed and OvidSP platforms. The full electronic search strategy used in PubMed and Medline was (“pulse wave velocity” OR “pulse wave analysis” OR “arterial stiffness” OR “vascular stiffness” OR “aortic stiffness” OR “CAVI” OR “augmentation index”) AND (“left ventricular diastolic function” OR “left ventricular diastolic dysfunction” OR “heart failure, diastolic” OR “heart failure with preserved ejection fraction”) limited to humans. Similar search terms were used when searching OvidSP Medline and OvidSP EMbase. The last date of search was August 30, 2014.
Eligibility criteria
Studies that met the following criteria were included: (1) was an original study published in a peer-review journal, (2) used a tonometric assessment of arterial stiffness (i.e., baPWV, cfPWV, AIx or CAVI), (3) assessed diastolic dysfunction with Doppler echocardiographic parameters (E/A ratio, e′ or E/e′ ratio), (4) presented a univariate correlation between arterial stiffness and diastolic dysfunction, (5) subjects were mainly adults (over 18 years of age) and (6) did not consists of persons with uncommon diseases or conditions (e.g., pseudoxanthoma elasticum). Duplicate studies, non-English studies, abstracts from unpublished studies, reviews, case reports and letters were excluded. In a few cases, we could not exclude some studies from the same author [30–34], and because we could not confirm that they were duplicate studies of the same patient population, the studies were included in our analysis.
Data extraction
From each eligible study, patient characteristics, method of arterial stiffness assessment and findings were systematically recorded. Patient characteristics included average age, sex, presence of hypertension and diabetes. Studies were grouped by method of assessing arterial stiffness—as baPWV, cfPWV, AIx or CAVI. Studies that used more than one technique were placed in multiple groups. The principal summary measure was the univariate correlation between one of the four tonometric assessments of arterial stiffness and one of the three measures of diastolic dysfunction (E/A ratio, e′ or E/e′), equating to twelve sets of correlation coefficients. For each eligible finding, the correlation coefficient, direction of correlation and significance were recorded.
Statistical analysis
Meta-analyses of the aggregate patient data were conducted with the Comprehensive Meta-Analysis version 2 (Biostat, Englewood, New Jersey, USA). From each entry, the study name, sample size, correlation coefficient and direction of correlation were entered. The random-effects model was chosen to generate a summary correlation coefficient due to the variation in participant characteristics and operators between studies. Fischer’s z transformation was used to compare the summary correlation coefficients of the different techniques [35]. To assess heterogeneity, the Cochrane Q statistic, I 2 statistic and Tau-squared statistic were calculated. Publication bias was assessed with funnel plots of standard error versus Fisher’s Z-scores. Statistical significance was set as p < 0.05 unless otherwise specified.
To determine whether any one paper had a disproportionate effect on the summary correlation coefficient, sensitivity analyses were conducted (Comprehensive Meta-Analysis version 2). Each study in every group was sequentially removed, and the analysis was repeated. The point estimates of correlation coefficients were manually compared to the primary result to identify any changes in significance or direction of correlation.
Results
Study selection
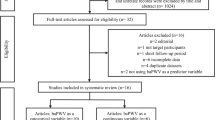
The systematic search is summarized in Fig. 1, following the Preferred Reporting Items for Systematic reviews and Meta-Analyses guidelines [36]. The systematic search presented 328 unique records. After screening the abstracts, 70 articles were removed because they were reviews, case reports, editorials or letters. Ninety-two abstracts from unpublished studies were excluded. The remaining 160 full-text articles were reviewed. We removed 87 studies that did not measure arterial stiffness using tonometric techniques and 46 studies that did not examine the correlation between arterial stiffness and diastolic dysfunction. Twenty-seven articles fulfilled the eligibility criteria [18, 24, 28–34, 37–54].
A total of 6,626 subjects were enrolled across the 27 studies. Thirteen studies measured baPWV, 8 studies measured cfPWV, 8 studies measured AIx and 7 studies measured CAVI. Five studies used two measures of arterial stiffness, and one study used three measures. Characteristics of the study populations are described in Table 1. baPWV was measured in 4,035 subjects, cfPWV was measured in 1,249 subjects, AIx was measured in 1,764 subjects and CAVI was measured in 548 subjects. The majority of studies showed a high proportion of males and persons with hypertension. Most studies consecutively enrolled eligible subjects. Other methods of enrollment included patient invitation [43, 49] and random selection [30, 31].
Outcomes
The summary correlation coefficients of baPWV, cfPWV, AIx and CAVI were all significantly associated with E/A ratio (Figs. 2, 3). baPWV had the strongest correlation with E/A ratio (r = −0.434, 95 % CI −0.387 to −0.479). Table 2 outlines the summary correlation coefficients for each meta-analysis. Sensitivity analysis showed that exclusion of any single study from the analysis did not significantly alter the overall findings. Heterogeneity analysis indicated statistical heterogeneity for baPWV versus E/A (Q = 17.5, I 2 = 48.5 %), cfPWV versus E/A (Q = 14, I 2 = 57.3 %) and AIx versus E/A (Q = 13.42, I 2 = 62.8 %) but not CAVI versus E/A (Q = 5, I 2 = 0.5 %).
Comparison of arterial stiffness with diastolic dysfunction measured by E/A ratio for a brachial-ankle pulse wave velocity (baPWV) and b cardio-ankle vascular index (CAVI). Sample size of each study is listed under the “Total” heading. The size of the squares is proportional to the weight of each study. The summary correlation coefficient and 95 % CIs determined by the random-effects models are shown at the bottom of each column. Heterogeneity statistics are also shown
Comparison of arterial stiffness with diastolic dysfunction measured by E/A ratio for a carotid-femoral pulse wave velocity (cfPWV) and b augmentation index (AIx). Sample size of each study is listed under the “Total” heading. The size of the squares is proportional to the weight of each study. The summary correlation coefficient and 95 % CIs determined by the random-effects models are shown at the bottom of each column. Heterogeneity statistics are also shown
baPWV, AIx and CAVI were significantly correlated with e′, but cfPWV did not correlate (Figs. 4, 5). baPWV and CAVI were equally the strongest correlations (r = −0.499, 95 % CI −0.448 to −0.548 and r = −0.449, 95 % CI −0.340 to −0.630, respectively). baPWV and CAVI were both significantly stronger correlated than cfPWV (z = −6.54, p < 0.001; z = −4.21, p < 0.001) and AIx (z = −4.23, p < 0.001; z = −2.77, p < 0.01). Sensitivity analysis indicated that exclusion of Roos et al. [52] from the meta-analysis of cfPWV versus e′ changed the nonsignificant summary correlation coefficient (r = −0.208, p = 0.140) to a significant point estimate of the correlation coefficient (r = −0.323, p < 0.001). This change is not unexpected given Roos et al. reported a positive correlation between cfPWV versus e′ (r = 0.320, p < 0.01), while the other four studies found negative correlation. This discrepancy is reflected in the Cochrane Q statistic, which indicated statistical heterogeneity for cfPWV versus e′ (Q = 52, I 2 = 92.3 %). The Cochrane Q statistic did not indicate statistical heterogeneity for baPWV versus e′ (Q = 2.7, I 2 = 0 %), AIx versus e′ (Q = 4.6, I 2 = 56.8 %) or CAVI versus e′ (Q = 5.4, I 2 = 44.9 %).
Comparison of arterial stiffness with diastolic dysfunction measured by peak early diastolic mitral annular velocity (e′) for a brachial-ankle pulse wave velocity (baPWV) and b cardio-ankle vascular index (CAVI). Sample size of each study is listed under the “Total” heading. The size of the squares is proportional to the weight of each study. The summary correlation coefficient and 95 % CIs determined by the random-effects models are shown at the bottom of each column. Heterogeneity statistics are also shown
Comparison of arterial stiffness with diastolic dysfunction measured by peak early diastolic mitral annular velocity (e′) for a carotid-femoral pulse wave velocity (cfPWV) and b augmentation index (AIx). Sample size of each study is listed under the “Total” heading. The size of the squares is proportional to the weight of each study. The summary correlation coefficient and 95 % CIs determined by the random-effects models are shown at the bottom of each column. Heterogeneity statistics are also shown
baPWV, cfPWV and AIx were significantly correlated with E/e′ ratio, but CAVI did not correlate (Figs. 6, 7). baPWV had the strongest correlation with E/e′ ratio (r = 0.372, 95 % CI 0.251–0.481) and significantly better than cfPWV (z = 2.92, p < 0.01) and CAVI (z = 3.56, p < 0.01), but not AIx (z = 1.09, p = 0.435). AIx (r = 0.321, 95 % CI 0.250–0.388) was significantly better than CAVI (z = 2.77, p < 0.01), but not cfPWV (z = 1.4, p = 0.0561). Sensitivity analysis showed that exclusion of any single study from the analysis did not significantly alter the overall findings. The Cochrane Q statistic indicated statistical heterogeneity for baPWV versus E/e′ (Q = 9.2, I 2 = 67.4 %) and CAVI versus E/e′ (Q = 11, I 2 = 72.8 %), but not cfPWV versus E/e′ (Q = 0.5, I 2 = 0 %) or AIx versus E/e′ (Q = 1.1, I 2 = 0 %).
Comparison of arterial stiffness with diastolic dysfunction measured by E/e′ ratio for a brachial-ankle pulse wave velocity (baPWV) and b cardio-ankle vascular index (CAVI). Sample size of each study is listed under the “Total” heading. The size of the squares is proportional to the weight of each study. The summary correlation coefficient and 95 % CIs determined by the random-effects models are shown at the bottom of each column. Heterogeneity statistics are also shown
Comparison of arterial stiffness with diastolic dysfunction measured by E/e′ ratio for a carotid-femoral pulse wave velocity (cfPWV) and b augmentation index (AIx). Sample size of each study is listed under the “Total” heading. The size of the squares is proportional to the weight of each study. The summary correlation coefficient and 95 % CIs determined by the random-effects models are shown at the bottom of each column. Heterogeneity statistics are also shown
Funnel plots for the meta-analysis of baPWV versus E/A ratio, e′ and E/e′ ratio were reviewed visually and appeared symmetric, indicating no any publication bias (Fig. 8). The funnel plots the other meta-analyses were similarly reviewed and did not reveal any publication bias.
Discussion
To our knowledge, this study is the first meta-analysis of arterial tonometry in assessment of left ventricular diastolic dysfunction. Reviewing 27 studies, we compared four different tonometric measures of arterial stiffness with three different indicators of diastolic dysfunction determined by echocardiography. The primary finding of our meta-analysis is that increased arterial stiffness is an indicator of diastolic dysfunction. Second, we found that of the four techniques, baPWV demonstrated the highest correlative strength and was significantly associated with all three indicators of diastolic dysfunction.
Our study showed the different strengths of the correlation of each tonometric method with diastolic dysfunction. Each method of tonometry correlated significantly with at least one index of diastolic dysfunction. We found that only baPWV and AIx, however, were significantly correlated with all three accepted indices of diastolic dysfunction. Of note, baPWV was more strongly correlated with E/A ratio and e′ than AIx. The ability of baPWV to outperform AIx, as a marker of arterial stiffness indicative of diastolic dysfunction, was demonstrated [18, 22, 53]. Augmentation index has been reported to be more influenced by antihypertensive medications, vasoactive drugs, gender and height when compared to pulse wave velocity [55–57].
When compared to the other three techniques, we found that baPWV consistently had the highest correlation with diastolic dysfunction. When compared with cfPWV, baPWV was more strongly correlated with e′ and E/e′ indices. baPWV and cfPWV are closely correlated as indicators of arterial stiffness [18, 58, 59]. Another study which did not provide a correlation coefficient to be used in this analysis found a significant correlation between baPWV and e′ [60]. baPWV, however, is consistently 17–20 % higher than cfPWV [18, 58], indicating baPWV measures additional aspects of arterial stiffness. Unlike cfPWV, which only involves the central arterial tree, baPWV includes both the central and peripheral arterial trees, which may be more representative of the afterload effects leading to diastolic dysfunction [18, 59]. Other potential explanations for the differences between baPWV and cfPWV in this study include the fact that cfPWV is more operator dependent than baPWV with necessary measurement of the pulse waves at the neck and groin with a handheld pressure transducer [61]. In addition, there are different methods proposed for measurement of the carotid-femoral arterial path [26]. In the eight studies that measured cfPWV, three defined carotid-femoral length as the direct measurement from the carotid artery to the femoral artery [18, 46, 52], one study subtracted the distance between the suprasternal notch to the carotid artery from the distance between the suprasternal notch and the femoral artery [43], and one study subtracted the distance between the manubrium to the carotid artery from the distance between the manubrium and the femoral artery [49]. Three studies did not provide a definition for the carotid-femoral length [29, 39, 48]. baPWV avoids these issues by measuring the pulse waves with occlusion cuffs on the arms and ankles and estimating the arterial path from the patient’s height [58]. It is not the purpose of this study to compare the relative merits of baPWV and cfPWV in overall cardiovascular assessment, but rather to focus on the relationship with diastolic dysfunction.
There was between-study variability in the methods of reporting peak early diastolic mitral annular velocities (e′). Five studies measured e′ at the septal site [33, 47, 48, 51, 53]; three studies measured e′ at the lateral; and one measured e′ at the posterior site [40]. Five studies calculated an average velocity from various combinations of lateral, septal, anterior, inferior, anteroseptal and posterior wall velocities [29, 34, 39, 44, 52]. One study did not report the sampling site [49].
There was no significant difference between the correlations of CAVI and baPWV with E/A ratio and e′. However, CAVI did not significantly correlate with E/e′. Some investigators have suggested measurement of CAVI may be superior to baPWV because it is less affected by systolic blood pressure and has better reproducibility [62, 63]. The populations in most of the studies included in this meta-analysis consisted of high percentage of subjects with hypertension. If blood pressure were to influence baPWV more than CAVI, we would have expected to see a difference in their correlations. However, the similar correlative ability of CAVI and baPWV suggests that the effect of blood pressure may not be a significant factor when assessing the relationship of arterial stiffness and diastolic dysfunction. Our meta-analyses of baPWV and CAVI showed similar ranges of heterogeneity. Significant heterogeneity was present in the analysis of baPWV versus E/A and E/e′. The heterogeneity can be partially attributed to the different patient populations examined, specifically in the proportion of subject characteristics such as gender, hypertension and diabetes mellitus, factors that are independent predictors of diastolic function and arterial stiffness [64]. Our review of studies examining baPWV versus E/A populations showed a range from 0 to 100 % for the prevalence hypertension or diabetes mellitus (Table 1). The heterogeneity statistic is reflective of the variation in these patient characteristics across the studies. The E/e′ ratio was the index where CAVI showed the most heterogeneity (I 2 = 72.8 %). Two of four studies included in that meta-analysis reported nonsignificant negative correlation coefficients [40, 47], while the other two studies reported significant positive correlation coefficients [33, 34]. This heterogeneity coupled with the inability to correlate with E/e′ suggests that CAVI does not offer any advantages over baPWV in evaluating diastolic dysfunction and may in fact be less sensitive.
Arterial stiffness is a predictor of cardiovascular events and all-cause mortality [65–67]. This applies to both baPWV and cfPWV [65–67]. Considering that left ventricular diastolic function is also a predictor of an increased mortality from heart failure [3, 4], it is possible that part of the adverse effects of increased arterial stiffness on cardiovascular events is operative through producing diastolic dysfunction. The mechanism by which increased arterial stiffness produces diastolic dysfunction remains uncertain. Increased arterial stiffness, by increasing systolic load on the ventricles and decreasing aortic pressures during diastole, amplifies the pulse pressure [68]. The increased pulse pressure increases myocardial oxygen demands during systole, is associated with left ventricular hypertrophy and also compromises coronary perfusion during diastole [69, 70]. These changes can lead to impaired relaxation and eventual diastolic dysfunction and failure [69]. The relationship of arterial stiffness to diastolic dysfunction, however, cannot be simply ascribed to production of left ventricular hypertrophy because arterial stiffness is independently associated with diastolic dysfunction in the presence of ventricular hypertrophy [29, 53].
Study limitations
There are several potential limitations of our study that warrant consideration. In this meta-analysis, aggregate patient data reported as correlation coefficients were collected rather than individual patient data. If we would have had individual patient data from each study that measured arterial stiffness and diastolic dysfunction, we would have had a larger patient population, allowing for a more robust meta-analysis. However, the resources, time and international cooperation required for meta-analysis of individual patient data have made it impractical for most systematic reviews, as in our case [71]. Arterial stiffness can be more prominent in women, who are also more susceptible to diastolic dysfunction [72]. The current meta-analysis is limited in its ability to examine the relationship of arterial stiffness to diastolic dysfunction in each sex because individual patient data were unavailable. The large majority of studies presented data on the combined patient population of both males and females. The one study that did separate the findings by sex examined the relationships between AIx, cfPWV, e′ and E/e′ [48]. Shim et al. [48] found that AIx and cfPWV were both significantly correlated with e′ and E/e′ when males and females were combined (n = 158). In women (n = 79), AIx was significantly correlated with e′, but not cfPWV but there was no significant correlation between E/e′ and AIx or cfPWV. In men (n = 79), none of the relationships were significant [48]. A third concern is that we excluded studies published in non-English languages. There is controversy over the prevalence of over-estimation of effect size in non-English studies versus English studies [73, 74]. However, exclusion of non-English studies in our meta-analysis is reasonable because excluding non-English studies does not affect the outcome of most meta-analysis [73, 75, 76]. Fourthly, although our systematic search was able to gather a similar number of studies for each tonometric technique, there was a noticeable regional preference for certain techniques. Studies measuring baPWV were mainly from Asian countries, and CAVI was exclusively studied in Japan. AIx and cfPWV had more global usage. Additionally, there were multiple studies from the same author with similar patient characteristics [30–32]. These studies measured the same tonometric techniques and were included because duplicate studies could not be confirmed. Fifthly, the population size attributed to each technique was not uniform. baPWV had the largest study size, followed by cfPWV, AIx and CAVI. The bias toward certain techniques may be compounded by the variation due to operator dependency of arterial tonometry and Doppler echocardiography [77]. By employing the random-effects model, we aim to account for this variation [78].
This meta-analysis focused on arterial tonometry studies to address vascular stiffness. There are other approaches to assess vascular stiffness such as those based on Windkessel models of arterial compliance. These are challenged by the need to measure changes in total arterial volume because while volume enters the aorta in systole, volume simultaneously leaves the arterial circulation into the venous system so that even more complex models have been proposed [79] but have not yet been readily adapted for patient application. Furthermore, there are no gold standards against which to compare the different methods for measurement of total aortic compliance, a necessary component for calculation of arterial stiffness, using Windkessel models [80]. In addition, there are only a limited number of studies that utilized this approach and related it to diastolic function so that their inclusion in this meta-analysis was not possible. The relationship of left ventricular diastolic function to the measurement of vascular stiffness using devices employing modified Windkessel models to separate the capacitance and oscillatory components of arterial compliance [81] need to be performed.
Another consideration is that the indices of vascular stiffness examined in this study are reflective of different aspects of the arterial circulation. PWV is considered to be a direct representation of the arterial stiffness over the measured vascular segment (e.g., carotid-femoral, brachial-ankle) and largely omits the contribution of the peripheral circulation not involved in its assessment [59]. AIx is considered to be a complex surrogate marker for arterial stiffness, indicative mainly of proximal aortic stiffness and is dependent on the arterial stiffness gradient between central aorta and the periphery [82]. CAVI is thought to represent both the state of smooth muscle contraction and the sclerotic change in the arterial wall [83].
Overall, this meta-analysis found that there is a significant correlation between arterial stiffness and diastolic dysfunction ranging from r = 0.210 to r = 0.498. The modest absolute values of the summary correlation coefficients indicate that arterial stiffness does not account for all the variability in diastolic dysfunction and that arterial stiffness is one factor among several that determine diastolic function. Other factors such as age, sex, blood pressure and left ventricular hypertrophy are associated with diastolic dysfunction [6, 64]. Our meta-analysis examined the relationship between arterial stiffness and diastolic dysfunction at the univariate level. The relationship between arterial stiffness and diastolic function was analyzed on a multivariate level by 18 of the included papers. After multivariate analyses, three studies found a significant relationship between AIx and E/A [42, 52, 53], three studies found a significant relationship between baPWV and E/A [18, 37, 53], one study found a significant relationship between CAVI and E/A [40], and one study found a significant relationship between cfPWV and E/A [46]. Two studies found a significant relationship between AIx and e′ [44, 52]; two studies found a significant relationship between baPWV and e′ [29, 32]; two studies found a significant relationship between CAVI and e′ [33, 47]; and no study found a significant relationship between cfPWV and e′. One study found a significant relationship between AIx and E/e′ [44], and one study found a significant relationship between baPWV and E/e′ [53]. Common covariates such as age, sex, blood pressure and left ventricular hypertrophy were included in some studies, but each study varied in the number and types of covariates. Individual participant data would be required to evaluate the correlation between arterial stiffness, diastolic dysfunction and a third covariate (e.g., age) to conduct a proper pooled multivariate analysis. A single large well-designed study including all covariates would contribute robust data recognizing that any one study has a risk of being wrong by chance alone and pooling data from a number of studies has the strength of multiple observations.
Another consideration is that our measure of diastolic dysfunction was strictly limited to Doppler echocardiography. We cannot with certainty extrapolate our results to other modalities of evaluating diastolic dysfunction. There still exists some controversy over the studied indices of diastolic dysfunction. The changes in E/A ratio with diastolic dysfunction are complex, with an initial negative trend that may later increase when a restrictive pattern of diastolic dysfunction arises [9]. The parameters, e′ and E/e′, have been suggested to be influenced by age and presence of cardiovascular disease, prompting cautionary recommendation of using these indices alone in evaluation diastolic dysfunction [6]. A single index from Doppler echocardiography may not be sufficient to evaluate diastolic dysfunction leading us to evaluate several Doppler indices. Evaluation of the relationship of arterial tonometry with other modalities of diagnosing diastolic dysfunction may be a topic of future research to further establish its predictive ability.
Conclusion
This study suggests that increased arterial stiffness, measured by tonometry, is indicative of a potential mechanistic link between vascular changes and the development of HFpEF. The different tonometric measures (baPWV, cfPWV, AIx and CAVI) are not the same in their correlations with the different indices of diastolic dysfunction. baPWV was consistently a strong predictor of diastolic dysfunction when compared to the other techniques. These data suggest a refocusing on the impact of arterial stiffness on the left ventricle leading to HFpEF, and efforts to improve arterial stiffness merit investigation in the prevention and treatment of HFpEF. Further research investigations by prospectively integrating data on age, gender and ethnicity data along with newer measures of vascular stiffness should provide information with regard to the value of vascular stiffness over and above age changes for the effect on diastolic function, and whether gender and ethnicity influence these relationships.
References
Borlaug BA, Paulus WJ (2011) Heart failure with preserved ejection fraction: pathophysiology, diagnosis, and treatment. Eur Heart J 32:670–679
Butler J, Fonarow GC, Zile MR et al (2014) Developing therapies for heart failure with preserved ejection fractioncurrent state and future directions. JACC Heart Fail 2:97–112
Kane GC, Karon BL, Mahoney DW et al (2011) Progression of left ventricular diastolic dysfunction and risk of heart failure. JAMA 306:856–863. doi:10.1001/jama.2011.1201
Lam C, Lyass A, Kraigher-Krainer E et al (2011) Cardiac dysfunction and noncardiac dysfunction as precursors of heart failure with reduced and preserved ejection fraction in the community. Circulation 124:24–30. doi:10.1161/CIRCULATIONAHA.110.979203
Tschope C, Paulus WJ (2009) Is echocardiographic evaluation of diastolic function useful in determining clinical care? Doppler echocardiography yields dubious estimates of left ventricular diastolic pressures. Circulation 120:810–820
Nagueh SF, Appleton CP, Gillebert TC et al (2009) Recommendations for the evaluation of left ventricular diastolic function by echocardiography. J Am Soc Echocardiogr 22:107–133
Little WC, Oh JK (2009) Echocardiographic evaluation of diastolic function can be used to guide clinical care. Circulation 120:802–809
Ogunyankin KO (2011) Assessment of left ventricular diastolic function: the power, possibilities, and pitfalls of echocardiographic imaging techniques. Can J Cardiol 27:311–318
Garcia MJ, Thomas JD, Klein AL (1998) New Doppler echocardiographic applications for the study of diastolic function. J Am Coll Cardiol 32:865–875
Nishimura RA, Tajik AJ (1997) Evaluation of diastolic filling of left ventricle in health and disease: Doppler echocardiography is the clinician’s Rosetta Stone. J Am Coll Cardiol 30:8–18
Farias CA, Rodriguez L, Garcia MJ et al (1999) Assessment of diastolic function by tissue doppler echocardiography: comparison with standard transmitral and pulmonary venous flow. J Am Soc Echocardiogr 12:609–617. doi:10.1053/je.1999.v12.a99249
Wang M, Yip GWK, Wang AYM et al (2003) Peak early diastolic mitral annulus velocity by tissue Doppler imaging adds independent and incremental prognostic value. J Am Coll Cardiol 41:820–826
Ommen SR, Nishimura RA, Appleton CP et al (2000) Clinical utility of Doppler echocardiography and tissue doppler imaging in the estimation of left ventricular filling pressures: a comparative simultaneous Doppler-catheterization study. Circulation 102:1788–1794. doi:10.1161/01.CIR.102.15.1788
Sohn D-W, Chai I-H, Lee D-J et al (1997) Assessment of mitral annulus velocity by Doppler tissue imaging in the evaluation of left ventricular diastolic function. J Am Coll Cardiol 30:474–480
Kasner M, Westermann D, Steendijk P et al (2007) Utility of Doppler echocardiography and tissue Doppler imaging in the estimation of diastolic function in heart failure with normal ejection fraction: a comparative Doppler-conductance catheterization study. Circulation 116:637–647. doi:10.1161/CIRCULATIONAHA.106.661983
Chung C-MM, Chu C-MM, Chang S-TT et al (2010) Quantification of aortic stiffness to predict the degree of left ventricular diastolic function. Am J Med Sci 340:468–473
Desai AS, Mitchell GF, Fang JC et al (2009) Central aortic stiffness is increased in patients with heart failure and preserved ejection fraction. J Card Fail 15:658–664
Chow B, Rabkin SW (2013) Brachial-ankle pulse wave velocity is the only index of arterial stiffness that correlates with a mitral valve indices of diastolic dysfunction, but no index correlates with left atrial size. Cardiol Res Pract 2013:986847
Groenink M, de Roos A, Mulder BJM et al (1998) Changes in aortic distensibility and pulse wave velocity assessed with magnetic resonance imaging following beta-blocker therapy in the marfan syndrome. Am J Cardiol 82:203–208. doi:10.1016/S0002-9149(98)00315-4
Mackenzie IS, Wilkinson IB, Cockcroft JR (2002) Assessment of arterial stiffness in clinical practice. QJM 95:67–74. doi:10.1093/qjmed/95.2.67
DeLoach SS, Townsend RR (2008) Vascular stiffness: its measurement and significance for epidemiologic and outcome studies. Clin J Am Soc Nephrol 3:184–192. doi:10.2215/CJN.03340807
Nakae I, Matsuo S, Matsumoto T et al (2008) Augmentation index and pulse wave velocity as indicators of cardiovascular stiffness. Angiology 59:421–426
Asmar R, Topouchian J, Pannier B et al (2001) Pulse wave velocity as endpoint in large-scale intervention trial. The Complior(R) study. J Hypertens 19:813–818
Takaki A, Ogawa H, Wakeyama T et al (2008) Cardio-ankle vascular index is superior to brachial-ankle pulse wave velocity as an index of arterial stiffness. Hypertens Res–Clin Exp 31:1347–1355
Munakata M, Nunokawa T, Yoshinaga K, Toyota T (2005) The brachial-ankle pulse wave velocity is a better predictor for pulse pressure than augmentation index in older hypertensives. Jpn Med Assoc J 48:224–233
Weber T, Ammer M, Rammer M et al (2009) Noninvasive determination of carotid-femoral pulse wave velocity depends critically on assessment of travel distance: a comparison with invasive measurement. J Hypertens 27:1624–1630
Nürnberger J, Keflioglu-Scheiber A, Opazo Saez AM et al (2002) Augmentation index is associated with cardiovascular risk. J Hypertens 20:2407–2414. doi:10.1097/01.hjh.0000045501.82010.fa
Sakane K, Miyoshi T, Doi M et al (2008) Association of new arterial stiffness parameter, the cardio-ankle vascular index, with left ventricular diastolic function. J Atheroscler Thromb 15:261–268
Wang CP, Hung WC, Yu TH et al (2009) Brachial-ankle pulse wave velocity as an early indicator of left ventricular diastolic function among hypertensive subjects. Clin Exp Hypertens (New York) 31:31–43
Hsu P-CC, Lin T-HH, Lee C-SS et al (2010) Mismatch between arterial stiffness increase and left ventricular diastolic dysfunction. Heart Vessel 25:485–492
Hsu P-C, Lin T-H, Lee C-S et al (2011) Impact of a systolic parameter, defined as the ratio of right brachial pre-ejection period to ejection time, on the relationship between brachial-ankle pulse wave velocity and left ventricular diastolic function. Hypertens Res 34:462–467
Hsu P-C, Tsai W-C, Lin T-H et al (2012) Association of arterial stiffness and electrocardiography-determined left ventricular hypertrophy with left ventricular diastolic dysfunction. PLoS One 7:e49100
Masugata H, Senda S, Goda F et al (2009) Tissue Doppler echocardiography for predicting arterial stiffness assessed by cardio-ankle vascular index. Tohoku J Exp Med 217:139–146
Masugata H, Senda S, Okuyama H et al (2010) Aortic annular velocity assessed by tissue Doppler echocardiography as a potential parameter of arterial stiffness. Tohoku J Exp Med 221:169–174
Field AP (2001) Meta-analysis of correlation coefficients: a Monte Carlo comparison of fixed- and random-effects methods. Psychol Methods 6:161–180
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62:1006–1012
Yambe M, Tomiyama H, Hirayama Y et al (2001) Arterial stiffening as a possible risk factor for both atherosclerosis and diastolic heart failure. Hypertens Res 27:625–631
Masugata H, Senda S, Yoshikawa K et al (2005) Relationships between echocardiographic findings, pulse wave velocity, and carotid atherosclerosis in type 2 diabetic patients. Hypertens Res–Clin Exp 28:965–971
Tsioufis C, Chatzis D, Dimitriadis K et al (2005) Left ventricular diastolic dysfunction is accompanied by increased aortic stiffness in the early stages of essential hypertension: a TDI approach. J Hypertens 23:1745–1750
Mizuguchi Y, Oishi Y, Tanaka H et al (2007) Arterial stiffness is associated with left ventricular diastolic function in patients with cardiovascular risk factors: early detection with the use of cardio-ankle vascular index and ultrasonic strain imaging. J Card Fail 13:744–751
Ikonomidis I, Tzortzis S, Papaioannou T et al (2008) Incremental value of arterial wave reflections in the determination of left ventricular diastolic dysfunction in untreated patients with essential hypertension. J Hum Hypertens 22:687–698
Kaji Y, Miyoshi T, Doi M et al (2009) Augmentation index is associated with B-type natriuretic peptide in patients with paroxysmal atrial fibrillation. Hypertens Res 32:611–616
Libhaber CD, Norton GR, Majane OH et al (2009) Contribution of central and general adiposity to abnormal left ventricular diastolic function in a community sample with a high prevalence of obesity. Am J Cardiol 104:1527–1533
Fukuta H, Ohte N, Wakami K et al (2010) Impact of arterial load on left ventricular diastolic function in patients undergoing cardiac catheterization for coronary artery disease. Circ J 74:1900–1905
Kang S, Fan H-M, Li J et al (2010) Relationship of arterial stiffness and early mild diastolic heart failure in general middle and aged population. Eur Heart J 31:2799–2807
Triantafyllidi H, Tzortzis S, Lekakis J et al (2010) Association of target organ damage with three arterial stiffness indexes according to blood pressure dipping status in untreated hypertensive patients. Am J Hypertens 23:1265–1272
Noguchi S, Masugata H, Senda S et al (2011) Correlation of arterial stiffness to left ventricular function in patients with reduced ejection fraction. Tohoku J Exp Med 225:145–151
Shim CY, Park S, Choi D et al (2011) Sex differences in central hemodynamics and their relationship to left ventricular diastolic function. J Am Coll Cardiol 57:1226–1233
Soldatos G, Jandeleit-Dahm K, Thomson H et al (2011) Large artery biomechanics and diastolic dysfunction in patients with Type 2 diabetes. Diabet Med 28:54–60
Usui Y, Takata Y, Inoue Y et al (2011) Severe obstructive sleep apnea impairs left ventricular diastolic function in non-obese men. Sleep Med 4:4
Han J-Y, Choi D-H, Choi S-W et al (2012) Predictive value of brachial-ankle pulse wave velocity for cardiovascular events. Am J Med Sci 346:92–97
Roos CJ, Auger D, Djaberi R et al (2013) Relationship between left ventricular diastolic function and arterial stiffness in asymptomatic patients with diabetes mellitus. Int J Cardiovasc Imaging 29:609–616
Takami T, Saito Y (2013) Azelnidipine plus olmesartan versus amlodipine plus olmesartan on arterial stiffness and cardiac function in hypertensive patients: a randomized trial. Drug Des Dev Ther 7:175–183
Canepa M, Alghatrif M, Strait JB et al (2014) Early contribution of arterial wave reflection to left ventricular relaxation abnormalities in a community-dwelling population of normotensive and untreated hypertensive men and women. J Hum Hypertens 28:85–91
Matsui Y, Kario K, Ishikawa J et al (2005) Smoking and antihypertensive medication: interaction between blood pressure reduction and arterial stiffness. Hypertens Res 28:631–638
Kelly RP, Millasseau SC, Ritter JM, Chowienczyk PJ (2001) Vasoactive drugs influence aortic augmentation index independently of pulse-wave velocity in healthy men. Hypertension 37:1429–1433. doi:10.1161/01.HYP.37.6.1429
Yasmin Brown MJ (1999) Similarities and differences between augmentation index and pulse wave velocity in the assessment of arterial stiffness. QJM 92:595–600. doi:10.1093/qjmed/92.10.595
Tanaka H, Munakata M, Kawano Y et al (2009) Comparison between carotid-femoral and brachial-ankle pulse wave velocity as measures of arterial stiffness. J Hypertens 27:2022–2027
Yu WC, Chuang SY, Lin YP, Chen CH (2008) Brachial-ankle vs carotid-femoral pulse wave velocity as a determinant of cardiovascular structure and function. J Hum Hypertens 22:24–31
Tsai WC, Lee KT, Kuo HF et al (2013) Association of increased arterial stiffness and p wave dispersion with left ventricular diastolic dysfunction. Int J Med Sci 10:1437–1444
Chuang S-Y, Chen C-H, Cheng C-M, Chou P (2005) Combined use of brachial-ankle pulse wave velocity and ankle-brachial index for fast assessment of arteriosclerosis and atherosclerosis in a community. Int J Cardiol 98:99–105. doi:10.1016/j.ijcard.2004.01.019
Shirai K, Utino J, Otsuka K, Takata M (2006) A novel blood pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI). J Atheroscler Thromb 13:101–107
Kubozono T, Miyata M, Ueyama K et al (2007) Clinical significance and reproducibility of new arterial distensibility index. Circ J 71:89–94
Zile MR, Brutsaert DL (2002) New concepts in diastolic dysfunction and diastolic heart failure: part II: causal mechanisms and treatment. Circulation 105:1503–1508
Laurent S, Cockcroft J, Van Bortel L et al (2006) Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 27:2588–2605
Vlachopoulos C, Aznaouridis K, Stefanadis C (2010) Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol 55:1318–1327
Vlachopoulos C (2012) Progress towards identifying biomarkers of vascular aging for total cardiovascular risk prediction. J Hypertens 30:S19–S26
Safar ME, Levy BI, Struijker-Boudier H (2003) Current perspectives on arterial stiffness and pulse pressure in hypertension and cardiovascular diseases. Circulation 107:2864–2869
O’Rourke MF (2001) Diastolic heart failure, diastolic left ventricular dysfunction and exercise intolerance. J Am Coll Cardiol 38:803–805
Rabkin SW, Chan SH (2012) Correlation of pulse wave velocity with left ventricular mass in patients with hypertension once blood pressure has been normalized. Heart Int 7:27–31
Lyman GH, Kuderer NM (2005) The strengths and limitations of meta-analyses based on aggregate data. BMC Med Res Methodol 5:14
Coutinho T, Borlaug BA, Pellikka PA et al (2013) Sex differences in arterial stiffness and ventricular–arterial interactions. J Am Coll Cardiol 61:96–103
Jüni P, Holenstein F, Sterne J et al (2002) Direction and impact of language bias in meta-analyses of controlled trials: empirical study. Int J Epidemiol 31:115–123
Egger M, Zellweger-Zähner T, Schneider M et al (1997) Language bias in randomised controlled trials published in English and German. Lancet 350:326–329. doi:10.1016/S0140-6736(97)02419-7
Grégoire G, Derderian F, Le Lorier J (1995) Selecting the language of the publications included in a meta-analysis: is there a tower of babel bias? J Clin Epidemiol 48:159–163. doi:10.1016/0895-4356(94)00098-B
Moher D, Pham Klassen TP et al (2000) What contributions do languages other than English make on the results of meta-analyses? J Clin Epidemiol 53:964–972. doi:10.1016/S0895-4356(00)00188-8
Ehler D, Carney DK, Dempsey AL et al (2001) Guidelines for cardiac sonographer education: recommendations of the american society of echocardiography sonographer training and education committee. J Am Soc Echocardiogr 14:77–84. doi:10.1067/mje.2001.109922
Deeks JJ, Higgins JPT, Altman DG (2011) Analysing data and undertaking meta-analyses. Cochrane Handb. Syst. Rev. Interv. Version 5.1.0
Chung CS, Strunc A, Oliver R, Kovacs SJ (2006) Diastolic ventricular-vascular stiffness and relaxation relation: elucidation of coupling via pressure phase plane-derived indexes. Am J Physiol–Heart Circ Physiol 291:H2415–H2423
Haluska BA, Jeffriess L, Brown J et al (2010) A comparison of methods for assessing total arterial compliance. J Hum Hypertens 24:254–262
Cohn JN, Finkelstein S, McVeigh G et al (1995) Noninvasive pulse wave analysis for the early detection of vascular disease. Hypertension 26:503–508
Mitchell GF, Lacourciere Y, Arnold JM et al (2005) Changes in aortic stiffness and augmentation index after acute converting enzyme or vasopeptidase inhibition. Hypertension 46:1111–1117
Sun CK (2013) Cardio-ankle vascular index (CAVI) as an indicator of arterial stiffness. Integr Blood Press Control 6:27–38
Conflict of interest
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chow, B., Rabkin, S.W. The relationship between arterial stiffness and heart failure with preserved ejection fraction: a systemic meta-analysis. Heart Fail Rev 20, 291–303 (2015). https://doi.org/10.1007/s10741-015-9471-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-015-9471-1