Abstract
A hallmark symptom of heart failure (HF) is exercise intolerance, typically evidenced by excessive shortness of breath, and/or fatigue with exertion. In recent years, the physiologic response to progressive exercise using direct measures of ventilation and gas exchange, commonly termed the cardiopulmonary exercise test (CPX), has evolved into an important clinical tool in the management of patients with HF. There is currently debate regarding the optimal CPX response to apply when stratifying risk for mortality, hospitalization, or other outcomes in patients with HF. Early studies in this area focused on the application of peak VO2 in predicting outcomes in patients considered for transplantation. More recently, the focus of these studies has shifted to an emphasis on ventilatory inefficiency, in lieu of or in combination with peak VO2, in estimating risk. The most widely studied index of ventilatory inefficiency has been the minute ventilation/carbon dioxide production (VE/VCO2) slope. A growing body of studies over the last decade has demonstrated that among patients with HF, the VE/VCO2 slope more powerfully predicts mortality, hospitalization, or both, than peak VO2. A number of investigations have also simultaneously examined the diagnostic importance of peak VO2 and the VE/VCO2 slope as well as their respective response to various interventions. This review examines the body of evidence which has used aerobic capacity and ventilatory efficiency as prognostic and diagnostic markers as well as endpoints in interventional trials. Based on this evidence, recommendations for future clinical and research applications of these CPX variables are provided.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The risk of the eventual development of heart failure (HF) has increased as recent treatment advances have decreased the age-adjusted death rates for most other cardiovascular diseases. It is therefore not surprising that the prevalence of HF has risen dramatically in the last two decades [1]. Therefore, a great deal of effort has been directed toward diagnostic tools and interventions designed to optimally stratify risk in these patients. A hallmark symptom of HF is exercise intolerance, typically evidenced by excessive shortness of breath, and/or fatigue with exertion. In recent years, the physiologic response to progressive exercise using direct measures of ventilation and gas exchange, commonly termed the cardiopulmonary exercise test (CPX), has evolved into an important clinical tool in the management of patients with HF. This technology is useful in terms of quantifying responses to therapy, evaluating disability, assessing the mechanism of exercise intolerance, making activity recommendations, and estimating prognosis. The latter application has received a particular amount of attention in recent years; numerous studies have been published over the last decade documenting the prognostic utility of the CPX in predicting outcomes in patients with HF [2, 3].
There is currently debate regarding the optimal CPX variable(s) to apply when stratifying risk for mortality, hospitalization, or other outcomes in patients with HF. Early studies in this area focused on the application of peak oxygen consumption (VO2) in predicting outcomes in patients considered for transplantation. It is logical that peak VO2 would be associated with mortality risk in HF since it is widely considered a global marker of cardiopulmonary health. Peak VO2 reflects the degree of impairment in ventricular function (pumping capacity), vascular function (O2 delivery), and skeletal muscle metabolic capacity (O2 utilization). In a landmark 1991 study by Mancini et al. [4], patients who achieved a peak VO2 >14 ml O2 kg−1 min−1 had a survival rate that was similar to those who received a cardiac transplantation ( >90% at 1 year). Conversely, those who achieved a peak VO2 value ≤ 14 ml O2 kg−1 min−1 had a 1-year survival rate of only 47%. The enduring implication of this finding is that scarce donor hearts should be reserved for patients whose one year prognosis (judged by peak VO2) is significantly worse than the one year prognosis following transplant. Numerous subsequent studies have confirmed the value of peak VO2 stratifying risk in patients with HF [2, 3].
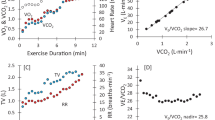
More recently, the focus of these studies has shifted to an emphasis on ventilatory inefficiency, in lieu of or in combination with peak VO2, in estimating risk [2]. The underlying concept behind the use of ventilatory inefficiency is the fact that patients with HF exhibit excessive ventilation in accordance with the degree of HF severity. This response is reflected by an excessive rise in minute ventilation relative to work rate, VO2, or CO2 production (VCO2). The most widely studied index of ventilatory inefficiency has been the VE/VCO2 slope, defined as the slope of the linear relation between minute ventilation (VE) and VCO2. Examples of different VE/VCO2 slope responses in three patients with HF undergoing symptom-limited CPX are illustrated in Fig. 1. A VE/VCO2 slope <30 is widely accepted as a normal response. Increased ventilation–perfusion mismatching [5, 6] and an abnormally heightened chemosensitivity and ergoreflex response [7–9] all appear to be linked to the elevated VE/VCO2 slope observed in HF.
A growing body of studies over the last decade has demonstrated that among patients with HF, the VE/VCO2 slope more powerfully predicts mortality, hospitalization, or both, than peak VO2. Although data are sparse, there has been recent interest in other markers of ventilatory efficiency, including the oxygen uptake efficiency slope (OUES, derived by the slope of a semi-log plot of minute ventilation versus VO2) [10], and exercise oscillatory ventilation (EOV, commonly defined as oscillatory fluctuations in ventilation for greater than 60% of the exercise test at an amplitude greater than 15% of the resting oscillatory fluctuations) [11–13]. However, the body of available literature at this time does not allow for meaningful comparisons of these responses to the VE/VCO2 slope. Because ventilatory efficiency provides important information in both the clinical and research settings, there exists a need to better define its calculation and application in relation to aerobic capacity, which is presently the most commonly assessed CPX variable. While a recent American Heart Association Scientific Statement [14] briefly addressed the prognostic value of both aerobic capacity and ventilatory efficiency in HF, a comprehensive review of the literature, which has compared these two CPX markers does not exist. Therefore, in the following, the body of evidence which has used aerobic capacity and ventilatory efficiency as prognostic and diagnostic markers as well as endpoints in interventional trials in patients with HF is reviewed. Based on this review, recommendations for clinical and research applications of these CPX responses are provided.
Prognostic characteristics of aerobic capacity and ventilatory efficiency
Summary of prognostic investigations
The landmark investigation by Mancini et al. [4] in 1991 initially demonstrated the prognostic value of peak VO2 in patients with HF. While the value of peak VO2 has been confirmed by numerous subsequent studies, indices of ventilatory efficiency were not assessed until the late 1990’s. In 1997, MacGowan et al. [14] reported peak VO2 was a significant predictor of mortality in a cohort of HF patients, and this study appeared to be the first to include the VE–VCO2 relationship in a univariate prognostic analysis. Although a multivariate regression was not performed, the ratio of VE to VCO2 at the ventilatory threshold was a stronger prognostic marker when compared to peak VO2. Over the past 10 years, over 20 peer-reviewed publications have included both aerobic capacity and ventilatory efficiency in prognostic analyses of patients diagnosed with HF. Details from these investigations are outlined in Table 1.
On average, investigations describing the VE–VCO2 relationship dichotomously used a threshold value ≥34 to define an abnormal response. Four level classification systems for the VE–VCO2 relationship have a general range of <30 for the most favorable class, from 30 to the low 40s for the middle classes and from the low 40s and above for the least favorable class. Twenty-four of the 26 investigations reported the VE–VCO2 relationship (reported as the slope in 22 investigations and as a ratio in 4) was superior to peak VO2 as a prognostic marker. Three investigations only reported a univariate analysis while the remaining studies performed a multivariate regression. Ten investigations using multivariate analyses found peak VO2 added significant prognostic value to the VE–VCO2 relationship and was retained in the regression. Eleven investigations reported peak VO2 did not add prognostic value to the VE–VCO2 relationship and was removed from the regression.
Areas requiring additional study regarding the prognostic characteristics of CPX
The pharmacologic and surgical treatment of patients with HF has changed dramatically since the initial prognostic analyses of CPX in the early 1990s. These changes in HF care have raised additional questions regarding the prognostic applications of CPX that require clarification. Beta-blocker therapy has become a standard of care in patients with HF [39]. This drug class has been shown to significantly reduce the VE/VCO2 slope without significantly altering peak VO2 [40–42]. Most early analyses assessing the prognostic characteristics of these variables did not report beta-blocker use, attributable to the fact that these agents were not considered a standard at the time. Later investigations began to report beta-blockade use, ranging between 12% and 60% of the overall patient cohorts. A limited number of investigations have specifically examined the prognostic impact of beta-blockade use on aerobic capacity and ventilatory efficiency with mixed results. Corra et al. [27] found that peak VO2, but not the VE/VCO2 slope, significantly predicted mortality risk in a subgroup of HF patients prescribed a beta-blocking agent. Arena et al. [37] however, found the VE/VCO2 slope was prognostically superior to peak VO2 irrespective of beta-blocker use. A key difference between these two investigations was the method employed to calculate the VE/VCO2 slope. The former investigation only utilized data to the point of the anaerobic threshold while the latter investigation incorporated all exercise data. Several investigations have now demonstrated that calculation of the VE/VCO2 slope with all exercise data more powerfully predicts risk [25, 34, 43, 44]. A more thorough discussion of the calculation of the VE/VCO2 slope is provided below.
A growing number of patients with HF are undergoing implantation of resynchronization devices as well as implantable cardioverter defibrillators. Cardiac resynchronization therapy has been shown to improve both aerobic capacity and ventilatory efficiency [45, 46]. In addition, these devices have been shown to favorably impact prognosis in patients with HF [47]. We are unaware of any investigation that has examined the impact cardiac resynchronization devices and/or implantable cardioverter defibrillators on the prognostic characteristics of CPX. This issue warrants further analysis given the growing prevalence of these devices in patients with HF.
It has been estimated that approximately 30–40% of the HF cases are attributable to diastolic dysfunction [48–50]. Patients with diastolic HF have a unique pathophysiology and different prognostic trajectory [51] as compared to individuals with systolic HF. Moreover, it appears that ventilatory efficiency and aerobic capacity characteristics differ between patients with systolic and diastolic HF [28, 52]. The majority of studies listed in Table 1 have assessed the prognostic characteristics of CPX in cohorts exclusively with a diagnosis of systolic HF. Presently, only one investigation has reported on the prognostic characteristics of CPX in patients with diastolic HF. In a small group of subjects with diastolic HF, Guazzi et al. [28] found that both the VE/VCO2 slope and peak VO2 were significant univariate predictors of mortality, hospitalization, or both. Multivariately however, the VE/VCO2 slope was the superior prognostic marker while peak VO2 did not add value and was removed from the regression. It should be noted that this analysis included a small number of patients with diastolic HF (<50 subjects with an ejection fraction >50%). The findings of the study by Guazzi et al. [28] should therefore only be viewed with caution at this time. Significant further study is required before any definitive conclusions are reached regarding the prognostic utility of CPX in patients with diastolic HF.
Although the prevalence of HF is similar between genders [53], all of the studies assessing the prognostic value of both aerobic capacity and ventilatory efficiency listed in Table 1 examined predominantly male cohorts. Notably, several investigations have found peak VO2 to be a significant prognostic marker in females with HF [54–56]. However, these investigations did not include ventilatory efficiency in their analyses. Guazzi et al. [31] appears to be the only investigation to date that has assessed the prognostic characteristics of both aerobic capacity and ventilatory efficiency separately in male and female patients with HF. Peak VO2 was significantly lower while the VE/VCO2 slope was significantly higher in females, suggesting that gender needs to be considered when applying the CPX to assess prognosis. In a multivariate analysis, the VE/VCO2 slope was the strongest prognostic marker while peak VO2 added significant prognostic value in both males and females. It should be noted that this analysis was conducted in a small number of females diagnosed with HF (n = 75). Therefore, while these initial findings indicate that both the VE/VCO2 slope and peak VO2 possess prognostic value in females with HF, additional research is needed in this area.
In recent years, prognostic scoring systems, such as the Seattle HF Model [57] and the Heart Failure Survival Score [56, 58] have been shown to be prognostically valuable. These models include a host of baseline variables such age, medications, HF etiology, and ejection fraction. The Heart Failure Survival Score also includes peak VO2 in its predictive model. We are not aware of any investigation that has compared the prognostic value of either scoring system to ventilatory efficiency or assess the value of adding ventilatory efficiency to the scoring model. Given, the continued interest in both CPX and the implementation of scoring systems in the HF population, future research should be directed toward assessing the combined prognostic value of these evaluation techniques.
Mode of exercise and protocol considerations
There is no consensus as to whether testing using the treadmill, cycle ergometer, or a particular protocol optimally predicts risk in patients with HF. This is potentially important since both the exercise mode and protocol influence the ventilatory gas exchange response to exercise [14]. Witte and Clark [59] reported that both peak VO2 and the VE/VCO2 slope were significantly lower during CPX utilizing a cycle ergometer compared to a treadmill in patients with HF. Nevertheless, Arena et al. [60] reported the prognostic characteristics of the VE/VCO2 slope and peak VO2 were similar in two separate HF cohorts, one group utilizing a treadmill while the other utilized a cycle ergometer for CPX. As indicated in Table 1, 12 investigations utilized a treadmill, 9 investigations utilized a cycle ergometer, and 5 utilized both for CPX. In addition, while some investigations listed in Table 1 employed more aggressive protocols (e.g., Bruce or modified Bruce), most opted for more conservative ramping protocols. Even with differences in mode of exercise and protocol selection, the prognostic value of ventilatory efficiency and aerobic capacity remained consistent, indicating ventilatory expired gas data possesses universally applicable characteristics across exercise testing laboratories with differing procedures.
Differences in endpoints used for prognostic investigations
The investigations listed in Table 1 used widely differing endpoints to assess the prognostic value of CPX. Thirteen investigations only considered mortality as an endpoint, eight considered mortality or heart transplantation/left ventricular assist device implantation, and five considered mortality or hospitalization as endpoints. Mortality is considered the only hard endpoint, resistant to selection bias. Of note, in the 13 investigations only considering mortality as an endpoint, the VE/VCO2 slope was prognostically superior to peak VO2 in each instance. Notably, of these 13 investigations, only two addressed the impact of beta-blocker therapy on the prognostic value of CPX.
Defining optimal prognostic thresholds for aerobic capacity and ventilatory efficiency
The optimal prognostic thresholds for aerobic capacity and ventilatory efficiency require further clarification, but depend upon the characteristics of the population studied. Initially, a peak VO2 threshold of </≥14 ml O2 kg−1 min−1 was proposed for transplant consideration [4] and this cutpoint is still the most frequently cited value in clinical practice. However, this threshold was proposed prior to the standard use of beta-blocker therapy which has been shown to improve survival without increasing peak VO2 in HF. As a result, it has been suggested that the peak VO2 threshold for prognostic purposes be reduced to </≥10 ml O2 kg−1 min−1 in patients prescribed a beta-blocking agent [61]. The most commonly cited dichotomous threshold for the VE/VCO2 slope is </≥34 [15, 22, 26]. Other investigations have assessed the prognostic characteristics of the VE/VCO2 slope using a four-level classification [18, 38]. Both of these latter studies found that mortality risk increases progressively as the VE/VCO2 slope increases from <30 to >40. Furthermore, in a subgroup analysis by Arena et al. [38], prognosis likewise became progressively worse as the VE/VCO2 slope increased from <30 to >40 in subjects prescribed a beta-blocking agent. Given the body of evidence presently available, clinicians should consider patients with a peak VO2 <10 ml O2 kg−1 min−1 or a VE/VCO2 slope >40 to be in the highest risk category. Patients with HF who present both of these characteristics have a particularly poor prognosis. However, in patients with a preserved aerobic capacity, a VE/VCO2 slope >40 should still be considered a strong indicator of poor prognosis given the independent prognostic value of ventilatory inefficiency.
Optimal expression of ventilatory efficiency
While the expression of peak VO2 is relatively straightforward and has been standardized for many years, this is not the case for ventilatory efficiency. The VE–VCO2 relationship has been expressed as both as slope and ratio. In addition, the VE/VCO2 slope has been calculated using data from the onset of exercise to both the point of the ventilatory threshold and maximal exertion. The VE/VCO2 ratio has likewise been calculated at both the ventilatory threshold and peak exercise. Four investigations have compared the prognostic value of the VE/VCO2 slope using submaximal exercise data to that using all exercise data during a symptom-limited test [25, 34, 43, 44]. In all instances, while both were significant predictors of prognosis, the VE/VCO2 slope calculated using all exercise data was superior to submaximal expressions of ventilatory efficiency in terms of predicting risk. Investigations supporting the exclusion of data past the ventilatory threshold in the calculation of the VE/VCO2 slope suggest this eliminates the influence of increasing lactic acidosis, which increases the steepness of the slope and creates a degree of nonlinearity. Arena et al. [43] found the change in steepness of the VE/VCO2 slope from the ventilatory threshold to maximal exercise varied considerably in a group of patients with HF. In addition, this investigation reported prognosis significantly worsened as the VE/VCO2 slope steepened beyond the ventilatory threshold. This would not be expected if lactic acidosis was the only factor driving the increase in steepness of the VE/VCO2 slope when maximal exercise data were incorporated. These investigators hypothesized that a greater increase in the VE/VCO2 slope during the final stages of a symptom-limited exercise test may reflect a further impairment in cardiopulmonary function, a response with important prognostic implications not captured by submaximal expressions of ventilatory efficiency. To date, no investigation has examined the relationship between changes in cardiopulmonary performance and changes in the VE/VCO2 slope during an exercise test. This type of diagnostic investigation is needed to provide physiologic support for studies finding the VE/VCO2 slope calculated with all exercise data is prognostically superior.
While both the VE/VCO2 slope and ratio provide significant prognostic information, the former expression incorporates a far greater amount of exercise data. The VE/VCO2 slope should therefore be considered more resistant to variability in CPX data not reflective of a true physiologic response. Given the fact that presently available metabolic exercise testing systems commonly provide both these markers of ventilatory efficiency, opting for the slope for clinical/research purposes does not entail additional time or inconvenience for the individual interpreting the exercise test.
Diagnostic characteristics of aerobic capacity and ventilatory efficiency
A number of cardiac, pulmonary, neurohormonal, and autonomic physiologic abnormalities underlie heart failure. These abnormalities are identified by several different diagnostic testing techniques including invasive hemodynamic measurements, echocardiography, neurohormonal blood analysis, electrocardiography, sleep studies, and pulmonary function. Investigations assessing the relationship between these diagnostic techniques and both aerobic capacity and ventilatory efficiency are listed in Table 2.
Both peak VO2 and the VE/VCO2 slope/ratio are to be significantly related to resting and exercise cardiac output as well as resting pressures in the pulmonary vasculature. It appears however, that the relationships between invasive hemodynamics and ventilatory efficiency are somewhat stronger than those for peak VO2. Several variables obtained from echocardiography, such as the E wave, deceleration time, and left ventricular ejection fraction, have been shown to be significantly related to both peak VO2 and the VE/VCO2 slope. Certain echocardiographic variables are more strongly associated with peak VO2 while others have demonstrated a stronger correlation with the VE/VCO2 slope. The relationship between neurohormonal markers assessed in the resting state and both aerobic capacity and ventilatory efficiency appear to be mixed. Peak VO2 has demonstrated a significant correlation with norepinephrine and epinephrine. The VE/VCO2 slope was not significantly related to either norepinephrine or epinephrine in one investigation while there was a significant correlation with norepinephrine in another. Several investigations have reported a significant correlation between b-type natriuretic peptide and both peak VO2 and the VE/VCO2 slope. In two instances, the correlation between this neurohormonal marker and the VE/VCO2 slope was stronger than that for peak VO2. In a third investigation the relationship between b-type natriuretic peptide and peak VO2 was greater. One study examining the relationship between heart rate variability (via holter monitoring) and both peak VO2 and the VE/VCO2 slope found several measures reflecting autonomic dysfunction were significantly related to both CPX variables. The correlation between the markers of autonomic function and the VE/VCO2 slope was found to be stronger than that for peak VO2. One study examining the relationship between central sleep apnea and CPX responses reported the VE/VCO2 slope was significantly related with the apnea–hypopnea index. Moreover, an elevated VE/VCO2 slope effectively discriminated between subjects with and without central sleep apnea. Peak VO2 was not significantly correlated with the apnea–hypopnea index and did not discriminate between patients with and without central sleep apnea. Lastly, one investigation examined the relationship between alveolar–capillary membrane conductance and both peak VO2 and the VE/VCO2 slope, finding that the correlation was significant for both variables. The correlation between alveolar–capillary membrane conductance and the VE/VCO2 slope was however, stronger than that for peak VO2.
The impact of heart failure interventions on aerobic capacity and ventilatory efficiency
Numerous HF intervention trials have included CPX as an endpoint. Surgical, pharmacological, aerobic exercise training, inspiratory muscle training, and central sleep apnea interventions that reported their respective impact on both aerobic capacity and ventilatory efficiency are listed in Table 3.
One left ventricular assist device implantation trial reported both a significant reduction in the VE/VCO2 ratio at peak exercise and a significant improvement in peak VO2. All four cardiac resynchronization trials reported a significant reduction in the VE/VCO2 slope following device implantation. Three of the four trials also reported a significant increase in peak VO2 while the fouth reported no significant change following cardiac resynchronization. Pharmacologic investigations examining the impact of angiotensin converting enzyme inhibition, insulin infusion (in diabetic HF patients), and Sildenafil therapy have all reported a significant reduction in the VE/VCO2 slope and a significant increase in peak VO2 following treatment. The two investigations examining the impact of angiotensin II receptor blocker treatment were mixed, with one reporting a significant increase in peak VO2 and no change in the VE/VCO2 slope while the other reported a significant decrease in the VE/VCO2 slope and no change in peak VO2. Trials examining the impact of beta-blockade have consistently reported a significant reduction in the VE/VCO2 slope with no change in peak VO2. Aerobic exercise training studies have consistently reported both a significant increase in peak VO2 and a significant decrease in the VE/VCO2 slope following 2–6 months of training. The impact of inspiratory muscle training and continuous positive airway pressure (in patients with central sleep apnea) on aerobic capacity and ventilatory efficiency have been described in two separate investigations. In both instances, the VE/VCO2 slope was significantly reduced while no change in peak VO2 was noted.
Summary
Aerobic capacity and ventilatory efficiency provide important prognostic and diagnostic insights and are responsive to a multitude of accepted HF interventions. This body of evidence clearly supports the application of CPX in clinical management and research investigations involving patients with HF. Peak VO2 continues to be the most commonly assessed variable in clinical practice as well as in the research arena. Given the investigations cited in the present review, we propose the following broad paradigm shifts for present day clinical and research settings: (1) Peak VO2 and the VE/VCO2 slope provide independent and complementary information for the study of interventions in HF. Both variables should be considered for prognostic studies. Use of the VE/VCO2 slope as the primary variable obtained from CPX should be considered for prognostic studies; (2) All exercise data, from the initiation of exercise to maximal exertion should be used to calculate the VE/VCO2 slope; (3) For diagnostic purposes, both the VE/VCO2 slope and peak VO2 should be assessed although the former variable may better reflect the overall, multi-system pathophysiology associated with HF; and (4) Both the VE/VCO2 slope and peak VO2 should be considered endpoints for intervention trials. It should be noted, however, that certain interventions may impact one CPX variable while having little influence on the other. Finally, it is recognized that additional areas of research must be addressed, particularly in terms of utilizing the CPX for prognostic purposes. Research directions that may warrant priority are: (1) The prognostic assessment of CPX in HF cohorts receiving beta-blocker therapy; (2) The prognostic assessment of CPX in HF cohorts undergoing cardiac resynchronization therapy and/or automated implantable cardioverter defibrillation procedures; (3) The prognostic assessment of CPX in female cohorts with HF; and (4) The prognostic assessment of CPX in HF cohorts with diastolic HF.
References
Ansari M, Massie BM (2003) Heart failure: how big is the problem? Who are the patients? What does the future hold? Am Heart J 146:1–4
Arena R, Myers J, Guazzi M (2007) Ventilatory abnormalities during exercise in heart failure: a mini review. Curr Resp Med Rev 3:179–187
Myers J (2005) Applications of cardiopulmonary exercise testing in the management of cardiovascular and pulmonary disease. Int J Sports Med 26(Suppl 1):S49–S55
Mancini DM, Eisen H, Kussmaul W, Mull R, Edmunds LH Jr, Wilson JR (1991) Value of peak exercise oxygen consumption for optimal timing of cardiac transplantation in ambulatory patients with heart failure. Circulation 83:778–786
Wada O, Asanoi H, Miyagi K, Ishizaka S, Kameyama T, Seto H, Sasayama S (1993) Importance of abnormal lung perfusion in excessive exercise ventilation in chronic heart failure. Am Heart J 125:790–798
Uren NG, Davies SW, Agnew JE, Irwin AG, Jordan SL, Hilson AJ, Lipkin DP (1993) Reduction of mismatch of global ventilation and perfusion on exercise is related to exercise capacity in chronic heart failure. Br Heart J 70:241–246
Ponikowski P, Francis DP, Piepoli MF, Davies LC, Chua TP, Davos CH, Florea V, Banasiak W, Poole-Wilson PA, Coats AJ, Anker SD (2001) Enhanced ventilatory response to exercise in patients with chronic heart failure and preserved exercise tolerance: marker of abnormal cardiorespiratory reflex control and predictor of poor prognosis. Circulation 103:967–972
Chua TP, Clark AI, Amadi AA, Coats AJS (1996) Relation between chemosensitivity and the ventilatory response to exercise in chronic heart failure. J Am Coll Cardiol 27:650–657
Piepoli M, Clark AL, Volterrani M (1996) Contribution of muscle affarents to the hemodynamic, autonomic, and ventilatory responses to exercise in patients with chronic heart failure. Circulation 93:940–952
Davies LC, Wensel R, Georgiadou P, Cicoira M, Coats AJ, Piepoli MF, Francis DP (2006) Enhanced prognostic value from cardiopulmonary exercise testing in chronic heart failure by non-linear analysis: oxygen uptake efficiency slope. Eur Heart J 27:684–690
Corra U, Giordano A, Bosimini E, Mezzani A, Piepoli M, Coats AJ, Giannuzzi P (2002) Oscillatory ventilation during exercise in patients with chronic heart failure: clinical correlates and prognostic implications. Chest 121:1572–1580
Corra U, Pistono M, Mezzani A, Braghiroli A, Giordano A, Lanfranchi P, Bosimini E, Gnemmi M, Giannuzzi P (2006) Sleep and exertional periodic breathing in chronic heart failure: prognostic importance and interdependence. Circulation 113:44–50
Guazzi M, Arena R, Ascione A, Piepoli M, Guazzi MD (2007) Exercise oscillatory breathing and increased ventilation to carbon dioxide production slope in heart failure: an unfavorable combination with high prognostic value. Am Heart J 153:859–867
MacGowan GA, Janosko K, Cecchetti A, Murali S (1997) Exercise-related ventilatory abnormalities and survival in congestive heart failure. Am J Cardiol 79:1264–1266
Chua TP, Ponikowski P, Harrington D, Anker SD, Webb-Peploe K, Clark AL, Poole-Wilson PA, Coats AJ (1997) Clinical correlates and prognostic significance of the ventilatory response to exercise in chronic heart failure. J Am Coll Cardiol 29:1585–1590
Robbins M, Francis G, Pashkow FJ, Snader CE, Hoercher K, Young JB, Lauer MS (1999) Ventilatory and heart rate responses to exercise: better predictors of heart failure mortality than peak oxygen consumption. Circulation 100:2411–2417
Kleber FX, Vietzke G, Wernecke KD, Bauer U, Opitz C, Wensel R, Sperfeld A, Glaser S (2000) Impairment of ventilatory efficiency in heart failure: prognostic impact. Circulation 101:2803–2809
Francis DP, Shamim W, Davies LC, Piepoli MF, Ponikowski P, Anker SD, Coats AJ (2000) Cardiopulmonary exercise testing for prognosis in chronic heart failure: continuous and independent prognostic value from VE/VCO(2)slope and peak VO(2). Eur Heart J 21:154–161
Cicoira M, Davos CH, Florea V, Shamim W, Doehner W, Coats AJ, Anker SD (2001) Chronic heart failure in the very elderly: clinical status, survival, and prognostic factors in 188 patients more than 70 years old. Am Heart J 142:174–180
Cohen-Solal A, Tabet JY, Logeart D, Bourgoin P, Tokmakova M, Dahan M (2002) A non-invasively determined surrogate of cardiac power (’circulatory power’) at peak exercise is a powerful prognostic factor in chronic heart failure. Eur Heart J 23:806–814
Scharf C, Merz T, Kiowski W, Oechslin E, Schalcher C, Brunner-La Rocca HP (2002) Noninvasive assessment of cardiac pumping capacity during exercise predicts prognosis in patients with congestive heart failure. Chest 122:1333–1339
Corra U, Mezzani A, Bosimini E, Scapellato F, Imparato A, Giannuzzi P (2002) Ventilatory response to exercise improves risk stratification in patients with chronic heart failure and intermediate functional capacity. Am Heart J 143:418–426
Mejhert M, Linder-Klingsell E, Edner M, Kahan T, Persson H (2002) Ventilatory variables are strong prognostic markers in elderly patients with heart failure. Heart 88:239–243
Gitt AK, Wasserman K, Kilkowski C, Kleemann T, Kilkowski A, Bangert M, Schneider S, Schwarz A, Senges J (2002) Exercise anaerobic threshold and ventilatory efficiency identify heart failure patients for high risk of early death. Circulation 106:3079–3084
Tabet JY, Beauvais F, Thabut G, Tartiere JM, Logeart D, Cohen-Solal A (2003) A critical appraisal of the prognostic value of the VE/VCO2 slope in chronic heart failure. J Cardiovasc Risk 10:267–272
Arena R, Myers J, Aslam SS, Varughese EB, Peberdy MA (2004) Peak VO2 and VE/VCO2 slope in patients with heart failure: a prognostic comparison. Am Heart J 147:354–360
Corra U, Mezzani A, Bosimini E, Scapellato F, Temporelli PL, Eleuteri E, Giannuzzi P (2004) Limited predictive value of cardiopulmonary exercise indices in patients with moderate chronic heart failure treated with carvedilol. Am Heart J 147:553–560
Guazzi M, Myers J, Arena R (2005) Cardiopulmonary exercise testing in the clinical and prognostic assessment of diastolic heart failure. J Am Coll Cardiol 46:1883–1890
Guazzi M, Reina G, Tumminello G, Guazzi MD (2005) Exercise ventilation inefficiency and cardiovascular mortality in heart failure: the critical independent prognostic value of the arterial CO2 partial pressure. Eur Heart J 26:472–480
Arena R, Myers J, Abella J, Peberdy MA (2005) Influence of heart failure etiology on the prognostic value of peak oxygen consumption and minute ventilation/carbon dioxide production slope. Chest 128:2812–2817
Guazzi M, Arena R, Myers J (2006) Comparison of the prognostic value of cardiopulmonary exercise testing between male and female patients with heart failure. Int J Cardiol 113:395–400
Tsurugaya H, Adachi H, Kurabayashi M, Ohshima S, Taniguchi K (2006) Prognostic impact of ventilatory efficiency in heart disease patients with preserved exercise tolerance. Circ J 70:1332–1336
Tabet JY, Metra M, Thabut G, Logeart D, Cohen-Solal A (2006) Prognostic value of cardiopulmonary exercise variables in chronic heart failure patients with or without beta-blocker therapy. Am J Cardiol 98:500–503
Bard RL, Gillespie BW, Clarke NS, Egan TG, Nicklas JM (2006) Determining the best ventilatory efficiency measure to predict mortality in patients with heart failure. J Heart Lung Transpl 25:589–595
Nanas SN, Nanas JN, Sakellariou DC, Dimopoulos SK, Drakos SG, Kapsimalakou SG, Mpatziou CA, Papazachou OG, Dalianis AS, Nastasiou-Nana MI, Roussos C (2006) VE/VCO2 slope is associated with abnormal resting haemodynamics and is a predictor of long-term survival in chronic heart failure. Eur J Heart Fail 8:420–427
Dimopoulos K, Okonko DO, Diller GP, Broberg CS, Salukhe TV, Babu-Narayan SV, Li W, Uebing A, Bayne S, Wensel R, Piepoli MF, Poole-Wilson PA, Francis DP, Gatzoulis MA (2006) Abnormal ventilatory response to exercise in adults with congenital heart disease relates to cyanosis and predicts survival. Circulation 113:2796–2802
Arena RA, Guazzi M, Myers J, Abella J (2007) The prognostic value of ventilatory efficiency with beta-blocker therapy in heart failure. Med Sci Sports Exerc 39:213–219
Arena R, Myers J, Abella J, Peberdy MA, Bensimhon D, Chase P, Guazzi M (2007) Development of a ventilatory classification system in patients with heart failure. Circulation 115:2410–2417
Hunt SA (2005) ACC/AHA 2005 guideline update for the diagnosis and management of chronic heart failure in the adult: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (Writing committee to update the 2001 guidelines for the evaluation and management of heart failure). J Am Coll Cardiol 46:e1–e82
Agostoni P, Guazzi M, Bussotti M, De Vita S, Palermo P (2002) Carvedilol reduces the inappropriate increase of ventilation during exercise in heart failure patients. Chest 122:2062–2067
Agostoni P, Contini M, Magini A, Apostolo A, Cattadori G, Bussotti M, Veglia F, Andreini D, Palermo P (2006) Carvedilol reduces exercise-induced hyperventilation: A benefit in normoxia and a problem with hypoxia. Eur J Heart Fail 8:729–735
Wolk R, Johnson BD, Somers VK, Allison TG, Squires RW, Gau GT, Olson LJ (2005) Effects of [beta]-blocker therapy on ventilatory responses to exercise in patients with heart failure. J Cardiac Fail 11:333–339
Arena R, Myers J, Aslam S, Varughese EB, Peberdy MA (2003) Technical considerations related to the minute ventialtion/carbon dioxide output slope in patients with heart failure. Chest 124:720–727
Ingle L, Goode K, Carroll S, Sloan R, Boyes C, Cleland JGF, Clark AL (2007) Prognostic value of the VE/VCO2 slope calculated from different time intervals in patients with suspected heart failure. Int J Cardiol 118:350–355
Auricchio A, Stellbrink C, Sack S, Block M, Vogt J, Bakker P, Huth C, Schondube F, Wolfhard U, Bocker D (2002) Long-term clinical effect of hemodynamically optimized cardiac resynchronization therapy in patients with heart failure and ventricular conduction delay. J Am Coll Cardiol 39:2026–2033
Varma C, Sharma S, Firoozi S, McKenna WJ, Daubert JC (2003) Atriobiventricular pacing improves exercise capacity in patients with heart failure and intraventricular conduction delay. J Am Coll Cardiol 41:582–588
Lindenfeld J, Feldman AM, Saxon L, Boehmer J, Carson P, Ghali JK, Anand I, Singh S, Steinberg JS, Jaski B, DeMarco T, Mann D, Yong P, Galle E, Ecklund F, Bristow M (2007) Effects of cardiac resynchronization therapy with or without a defibrillator on survival and hospitalizations in patients with new york heart association class IV heart failure. Circulation 115:204–212
Bursi F, Weston SA, Redfield MM, Jacobsen SJ, Pakhomov S, Nkomo VT, Meverden RA, Roger VL (2006) Systolic and diastolic heart failure in the community. JAMA 296:2209–2216
Redfield MM, Jacobsen SJ, Burnett JC Jr, Mahoney DW, Bailey KR, Rodeheffer RJ (2003) Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic. JAMA 289:194–202
Zile MR, Brutsaert DL (2002) New concepts in diastolic dysfunction and diastolic heart failure: part I: diagnosis, prognosis, and measurements of diastolic function. Circulation 105:1387–1393
Ahmed A, Perry GJ, Fleg JL, Love TE, Goff J, Kitzman DW (2006) Outcomes in ambulatory chronic systolic and diastolic heart failure: a propensity score analysis. Am Heart J 152:956–966
Moore B, Brubaker PH, Stewart KP, Kitzman DW (2007) VE/VCO2 slope in older heart failure patients with normal versus reduced ejection fraction compared with age-matched healthy controls. J Cardiac Fail 13:259–262
American Heart Association (2007) 2007 Heart and stroke statistical update. Dallas, Texas. Ref Type: Pamphlet
Elmariah S, Goldberg LR, Allen MT, Kao A (2006) Effects of gender on peak oxygen consumption and the timing of cardiac transplantation. J Am Coll Cardiol 47:2237–2242
Richards DR, Mehra MR, Ventura HO, Lavie CJ, Smart FW, Stapleton DD, Milani RV (1997) Usefulness of peak oxygen consumption in predicting outcome of heart failure in women versus men. Am J Cardiol 80:1236–1238
Green P, Lund LH, Mancini D (2007) Comparison of peak exercise oxygen consumption and the heart failure survival score for predicting prognosis in women versus men. Am J Cardiol 99:399–403
Levy WC, Mozaffarian D, Linker DT, Sutradhar SC, Anker SD, Cropp AB, Anand I, Maggioni A, Burton P, Sullivan MD, Pitt B, Poole-Wilson PA, Mann DL, Packer M (2006) The Seattle heart failure model: prediction of survival in heart failure. Circulation 113:1424–1433
Koelling TM, Joseph S, Aaronson KD (2004) Heart failure survival score continues to predict clinical outcomes in patients with heart failure receiving [beta]-blockers. J Heart Lung Transpl 23:1414–1422
Witte KKA, Clark AL (2005) Cycle exercise causes a lower ventilatory response to exercise in chronic heart failure. Heart 91:225–226
Arena R, Guazzi M, Myers J, Peberdy MA (2005) Prognostic characteristics of cardiopulmonary exercise testing in heart failure: comparing american and european models. Eur J Cardiovasc Prev Rehabil 12:562–567
O’Neill JO, Young JB, Pothier CE, Lauer MS (2005) Peak oxygen consumption as a predictor of death in patients with heart failure receiving {beta}-blockers. Circulation 111:2313–2318
Sullivan MJ, Higginbotham MB, Cobb FR (1988) Increased exercise ventilation in patients with chronic heart failure: intact ventilatory control despite hemodynamic and pulmonary abnormalities. Circulation 77:552–559
Reindl I, Wernecke KD, Opitz C, Wensel R, Konig D, Dengler T, Schimke I, Kleber FX (1998) Impaired ventilatory efficiency in chronic heart failure: possible role of pulmonary vasoconstriction. Am Heart J 136:778–785
Myers J, Dziekan G, Goebbels U, Dubach P (1999) Influence of high-intensity exercise training on the ventilatory response to exercise in patients with reduced ventricular function. Med Sci Sports Exerc 31:929–937
De Feo S, Franceschini L, Brighetti G, Cicoira M, Zanolla L, Rossi A, Golia G, Zardini P (2005) Ischemic etiology of heart failure identifies patients with more severely impaired exercise capacity. Int J Cardiol 104:292–297
Kruger S, Graf J, Kunz D, Stickel T, Hanrath P, Janssens U (2002) Brain natriuretic peptide levels predict functional capacity in patients with chronic heart failure. J Am Coll Cardiol 40:718–722
Passino C, Poletti R, Bramanti F, Prontera C, Clerico A, Emdin M (2006) Neuro-hormonal activation predicts ventilatory response to exercise and functional capacity in patients with heart failure. Eur J Heart Fail 8:46–53
Scardovi AB, De MR, Coletta C, Aspromonte N, Perna S, Infusino T, D’Errigo P, Rosato S, Greggi M, Di GT, Ricci R, Ceci V (2006) Brain natriuretic peptide is a reliable indicator of ventilatory abnormalities during cardiopulmonary exercise test in heart failure patients. Med Sci Monit 12:CR191–CR195
Ponikowski P, Chua TP, Piepoli M, Banasiak W, Anker SD, Szelemej R, Molenda W, Wrabec K, Capucci A, Coats AJS (1998) Ventilatory response to exercise correlates with impaired heart rate variability in patients with chronic congestive heart failure. Am J Cardiol 82:338–344
Arzt M, Harth M, Luchner A, Muders F, Holmer SR, Blumberg FC, Riegger GAJ, Pfeifer M (2003) Enhanced ventilatory response to exercise in patients with chronic heart failure and central sleep apnea. Circulation 107:1998–2003
Guazzi M, Reina G, Tumminello G, Guazzi MD (2005) Alveolar–capillary membrane conductance is the best pulmonary function correlate of exercise ventilation efficiency in heart failure patients. Eur J Heart Fail 7:1017–1022
de Jonge N, Kirkels H, Lahpor JR, Klopping C, Hulzebos EJ, de la Riviere AB, Robles de Medina EO (2001) Exercise performance in patients with end-stage heart failure after implantation of a left ventricular assist device and after heart transplantation: an outlook for permanent assisting? J Am Coll Cardiol 37:1794–1799
Abraham WT, Young JB, Leon AR, Adler S, Bank AJ, Hall SA, Lieberman R, Liem LB, O’Connell JB, Schroeder JS, Wheelan KR, on behalf of the Multicenter InSync ICD II Study Group (2004) Effects of cardiac resynchronization on disease progression in patients with left ventricular systolic dysfunction, an indication for an implantable cardioverter-defibrillator, and mildly symptomatic chronic heart failure. Circulation 110:2864–2868
Wasserman K, Sun XG, Hansen JE (2007) Effect of biventricular pacing on the exercise pathophysiology of heart failure. Chest 132(1):250–261
Guazzi M, Marenzi G, Alimento M, Contini M, Agostoni P (1997) Improvement of alveolar–capillary membrane diffusing capacity with enalapril in chronic heart failure and counteracting effect of aspirin. Circulation 95:1930–1936
McConnell TR, Menapace FJ Jr, Hartley LH, Pfeffer MA (1998) Captopril reduces the VE/VCO2 ratio in myocardial infarction patients with low ejection fraction. Chest 114:1289–1294
Guazzi M, Palermo P, Pontone G, Susini F, Agostoni P (1999) Synergistic efficacy of enalapril and losartan on exercise performance and oxygen consumption at peak exercise in congestive heart failure. Am J Cardiol 84:1038–1043
Guazzi M, Tumminello G, Matturri M, Guazzi MD (2003) Insulin ameliorates exercise ventilatory efficiency and oxygen uptake in patients with heart failure-type 2 diabetes comorbidity. J Am Coll Cardiol 42:1044–1050
Kinugawa T, Kato M, Ogino K, Osaki S, Igawa O, Hisatome I, Shigemasa C (2004) Effects of angiotensin II type 1 receptor antagonist, losartan, on ventilatory response to exercise and neurohormonal profiles in patients with chronic heart failure. Jpn J Physiol 54:15–21
Lewis GD, Lachmann J, Camuso J, Lepore JJ, Shin J, Martinovic ME, Systrom DM, Bloch KD, Semigran MJ (2007) Sildenafil improves exercise hemodynamics and oxygen uptake in patients with systolic heart failure. Circulation 115:59–66
Coats AJ, Adamopoulos S, Radaelli A, McCance A, Meyer TE, Bernardi L, Solda PL, Davey P, Ormerod O, Forfar C, et al (1992) Controlled trial of physical training in chronic heart failure. Exercise performance, hemodynamics, ventilation, and autonomic function. Circulation 85:2119–2131
Kiilavuori K, Sovijarvi A, Naveri H, Ikonen T, Leinonen H (1996) Effect of physical training on exercise capacity and gas exchange in patients with chronic heart failure. Chest 110:985–991
Guazzi M, Reina G, Tumminello G, Guazzi MD (2004) Improvement of alveolar–capillary membrane diffusing capacity with exercise training in chronic heart failure. J Appl Physiol 97:1866–1873
Van Laethem C, Van De Veire N, Backer GD, Bihija S, Seghers T, Cambier D, Vanderheyden M, Sutter JD (2007) Response of the oxygen uptake efficiency slope to exercise training in patients with chronic heart failure. Eur J Heart Fail 9:625–629
Dall’Ago P, Chiappa GR, Guths H, Stein R, Ribeiro JP (2006) Inspiratory muscle training in patients with heart failure and inspiratory muscle weakness: a randomized trial. J Am Coll Cardiol 47:757–763
Arzt M, Schulz M, Wensel R, Montalvan S, Blumberg FC, Riegger GAJ, Pfeifer M (2005) Nocturnal continuous positive airway pressure improves ventilatory efficiency during exercise in patients with chronic heart failure. Chest 127:794–802
Author information
Authors and Affiliations
Corresponding author
Appendix 1: Commonly used terms in cardiopulmonary exercise testing
Appendix 1: Commonly used terms in cardiopulmonary exercise testing
-
CPX or CPET: Cardiopulmonary exercise testing
-
EOV: Exercise oscillatory ventilation
-
May also be referred to as EOB (exercise oscillatory breathing)
-
-
MET: Metabolic equivalent
-
One MET = 3.5 ml O2 kg−1 min−1
-
-
ml O2 kg−1 min−1: milliliters of oxygen/kilogram of body weight/minute
-
OUES: Oxygen uptake efficiency slope
-
Peak VO2: Peak oxygen consumption
-
RER: Respiratory exchange ratio
-
VCO2: Carbon dioxide production/output
-
VE: Minute ventilation
-
VE/VCO2: Minute ventilation/carbon dioxide production
-
Expressed as a slope or ratio
-
May be referred to as “ventilatory efficiency”
-
-
VT: Ventilatory threshold
-
Non-invasive representation of anaerobic threshold
-
-
W: Watts
Rights and permissions
About this article
Cite this article
Arena, R., Myers, J. & Guazzi, M. The clinical and research applications of aerobic capacity and ventilatory efficiency in heart failure: an evidence-based review. Heart Fail Rev 13, 245–269 (2008). https://doi.org/10.1007/s10741-007-9067-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-007-9067-5