Abstract
Drought stress is a common issue that affects agriculture production. The soybean plant is one of the drought-sensitive crops that could lose the yield of up to 40% under severe drought years. Improvements in drought tolerance have been one of the main objectives of breeding programs in soybean. In this study, 62 soybeans [Glycine max (L.) Merri.] germplasms including landraces and elite cultivars were evaluated by analyzing canopy and root morphological characteristics at seedling stage under polyethylene glycol (2.5 M PEG 8000, − 0.54 MPa) simulated drought stress. The results showed that the wilting index of sixty-two soybean germplasms showed significant polymorphism differences after 4 days PEG treatment. The lowest wilting index was 2.75 in Tiefeng 31, while the largest reached 9 in Fengdou 93 and Songzidou. The wilting index was positively correlated with plant height, electrolyte leakage, and negatively correlated with a dry weight of the above-ground part, stomatal conductance, and transpiration rate by regression analysis. The root system architecture (RSA) study showed root-canopy ratio, root length, number of the lateral roots, root surface area, and root volume were significantly negatively correlated with the wilting index and positively correlated with the root volume. Based on these results, sixty-two soybean germplasms were comprehensively evaluated for drought-tolerant cultivar by the principal component analysis (PCA). The results showed that Tiefeng 31 was the most drought-tolerant elite cultivar. However, Fengdou 93 was the most drought-sensitive elite variety. The drought tolerance screening results were consistent with the wilting index and RSA analysis, especially the root length. Both droughts tolerant and sensitive elite cultivar could be further used to breed drought-tolerant germplasms and to clarify the drought tolerance mechanism in soybean.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Global warming affects not only temperature but also rainfall size, intensity, frequency, and distribution. Additionally, climate change will also reduce the predictability of rainfall and lead to increased drought and flooding stress (Douglas et al. 2008). Drought stress is a major global issue that adversely affects plant growth and limits crop productivity. Yield losses of significant crops due to the fluctuating weather will be challenging to meet the food requirements of the world’s growing population in 2050 (Foley et al. 2011). Soybean is one of the most abundant oil and plant protein crops in the world (Hasanah et al. 2015). It needs 450–700 mm of water during the growing season and considers as a drought-sensitive crop (Dogan et al. 2007). Drought stress exposure during flowering and pod stages could severely limit the yield and may reduce yield by approximately 20–40% in soybean (Thao and Tran 2012). Obtaining more water resources, absorbing more water, and reducing water loss are useful measures to improve drought stress in crops (Passioura 1983). Therefore, tremendous efforts have been placed on planting drought-tolerant soybean cultivars are an effective strategy for enhancing seed yield under drought stress (Tester and Langridge 2010). The acceptable policy is selecting the drought tolerance trait from soybean germplasms and using traditional breeding, which combines the desirable feature into elite cultivar via crossing and backcrossing process.
It is essential to assess the drought tolerance in soybean germplasms. Experiments have been conducted to evaluate the soybean drought tolerance by seed yield reduction, drought tolerance index, and wilting index (Bouslama and Schapaugh 1984; Liu 1986). The first visible symptom of soil water deficit is canopy wilting in soybean (Charlson et al. 2009). The sign of canopy wilting included leaves fading, upper canopy curling, leaf angles movement, and petioles shrinking (Oosterhuis et al. 1985). However, the time of onset and severity of canopy wilting in response to water deficit were different in soybean germplasms (Sloane et al. 1990). The slow-wilting genotype PI 416937 from Japan and PI 471938 from Nepal had been identified among soybean germplasms (King et al. 2009), and the progeny of these genotypes had shown to have high yield under water deficit conditions (Bagherzadi et al. 2017). Slow wilting was the main factor for drought-tolerant soybean to increase the yield (Sinclair et al. 2010). The slow-wilting genotypes reportedly had a high capacity allowing high CO2 assimilations even with partial stomata closure (Sadok et al. 2012), increased transpiration rate under air vapor pressure deficit (Fletcher et al. 2007; Pathan et al. 2014; Bagherzadi et al. 2017). Compared to the fast-wilting genotypes, the slow-wilting genotypes generally had less radiation use efficiency and improved water use efficiency. Still, the stomatal conductance or canopy temperature had no difference (Ries et al. 2012). Soybean plants could withstand drought by root length elongation and prolonged nitrogen fixation besides slow canopy wilting (Sinclair et al. 2008).
Plant roots are the first tissues to percept the moisture change in the soil. The root system architecture (RSA) plays a crucial role in determining crop productivity in water-limited environments (Bengough et al. 2011). Root system architecture (RSA) refers to the shape and physical space of the roots. More deep-seated taproot and more lateral roots could absorb more water from deep soil layer and avoid drought stress in most of a dicotyledonous root system (Gilbert et al. 2011; Battisti and Sentelhas 2017; Ye et al. 2018). The drought-tolerant landrace (PI 416937) had a slow canopy wilting index and more fibrous root morphology (Hudak and Patterson 1996). Pantalone et al. (1996) also found the slow-wilting genotype showed a highly prolific root system and increased lateral root growth. Besides, the soybean plants can adapt to drought by developing a longer taproot under water-deficit environment (Manavalan et al. 2009). Root morphology parameters were used to classify the different cultivars in conferring drought tolerance traits in soybean. The drought-sensitive variety had a shallow root and root angle < 40°, and the drought-escaping cultivar had a deep root and root angle > 60° (Fenta et al. 2014). Some researchers had been studied the influence of drought on the growth of soybean crops, especially on leaves. However, fewer studies are focusing on combination above-ground part and root system architecture for screening drought-tolerant cultivars in soybean. Therefore, the objectives in this study will be (i) to evaluate differential wilting index and other physiological characteristics among soybean genotypes after polyethylene glycol (PEG) -simulated drought stress at the seedling stage; (ii) to determine the root morphological characteristics among soybean genotypes after PEG-simulated drought stress; (iii) to explore the relationship between the wilting index and root morphological characteristics; (iv) to identify some varieties more tolerant or more sensitive to the drought stress and select the drought-tolerant varieties for further soybean drought tolerance breeding program.
Materials and methods
Plant materials
62 soybean (Glycine max L. Merri.) germplasms including landraces and elite cultivars, comprising of 29 spring-type, 11 summer-type, and 22 autumn-type, which were obtained from Oil crops Research Institute, Chinese Academy of Agricultural Sciences (Wuhan, China), were evaluated in this research. The full list of germplasms is given in Table 1.
Drought stress trial
Drought stress trial was conducted at the walk-in growth chamber in the Institute of Crop Science, Zhejiang University, Zijinggang campus, Hangzhou, China (30° 17′ 55″ N, 120° 5′ 12″ E) in 2017 and 2018. The day/night temperature was about 25 °C/22 °C, the light length was 14 h/10 h (day/night), and the relative air humidity was about 57%. Sixteen seeds of each germplasm were sowed on the 19 × 13 × 12 cm germinating box containing 1.3 kg sterilized river sand with a 130 ml half-length Hoagland nutrient solution (Hoagland and Arnon 1938). Plants were watered 100 ml ddH2O daily until the beginning of PEG simulated drought stress. Nine days after sowing, the seedlings were thinned to ten. Fourteen days after sowing when the first trifoliolate leave was coming out, PEG simulated drought stress was imposed by watering plants with 100 ml 2.5 M PEG 8000 (ψ-0.54 MPa) solution and watering with 100 ml ddH2O as control. Each treatment for each germplasm was repeated three times at least 30 plants.
Physiological traits of above-ground part assessment
Four days after PEG-simulated drought stress treatment, the treated and control plants were analyzed for wilting canopy index, stomatal conductance, transpiration rate, electrolyte leakage, and root morphological characteristics.
Canopy wilting index
Four days after PEG treatment, the leaf wilting was recorded according to Pathan et al. (2014). The leaf wilting was rated visually on a scale of 0 to 4. The wilting scale of 0 represented no wilting. 1 represented the cotyledon wilting. 2 represented the wilting of both the cotyledonary and unifoliolate leave. 3 represented the cotyledonary, unifoliolate, and the first trifoliolate leave wilting together. 4 represented the whole plant wilting (Fig. 1). The wilting index was calculated by formula as follows:
Grade represented the degree of wilting scale, e.g., 0, 1, 2, 3, 4.
Stomatal conductance, transpiration rate, and electrolyte leakage
The stomatal conductance (Sc) and transpiration rate (Tr) were measured with Li-6400 portable photosynthesis system (Li-Cor Inc. USA) at constant CO2 of 380 µmol and light intensity of 1200 μmol m−2 s−1 at the time of 09:00–11:00 when the first trifoliolate leave fully expanded. Measurement was conducted from 10 plants of each genotype exposed under drought and control treatments. The highest and lowest values were removed during analyses, and the remaining data were averaged.
The electrolyte leakage of leaves was measured according to the manufacturer’s protocol using the Lei-magnetic conductivity meter (DDS-307, China). Three biological replicates of each germplasm were measured.
Root system architecture analysis
Four days after PEG-simulated drought stress, the above-ground part from five plants, which were selected randomly was used to determine the dry weight, and the roots were washed thoroughly using tap water and making the root flat in the plexiglass tray for scanning the root system architecture by EPSON Perfection V850 Pro. Root system architecture parameters such as root length (mm), number of lateral roots, root diameter (mm), root surface area (mm2), and root volume (mm3) were obtained through WinRHIZO software (Reagent Instrument, Quebc, Canada).
A comprehensive evaluation of drought resistance
The method of Yue et al. (2003) was adopted for evaluating drought-tolerance cultivars by Principal component analysis (PCA). Those indexes which are easy to measure and relevant to drought-tolerance, such as wilt index, root length, number of the lateral root, root diameter, root surface area and root volume, root-canopy ratio, were used as components of PCA. The component score of PCA is calculated by formula as follows:
Principal component score \(\left({\mathrm{F}}_{\mathrm{n}}\right)=\mathrm{factor} \, \mathrm{n} \, \mathrm{score} \times {\uplambda }_{\mathrm{n}}\)
The principal component synthesis score \(\left(\mathrm{F}\right)=\frac{{\uplambda }_{1}}{\left({\uplambda }_{1}+\cdots +{\uplambda }_{\mathrm{n}}\right)}\times {\mathrm{F}}_{1}+\frac{{\uplambda }_{2}}{\left({\uplambda }_{1}+\cdots +{\uplambda }_{\mathrm{n}}\right)}\times {\mathrm{F}}_{2}+\cdots +\frac{{\uplambda }_{\mathrm{n}}}{\left({\uplambda }_{1}+\cdots +{\uplambda }_{\mathrm{n}}\right)}\times {\mathrm{F}}_{\mathrm{n}}\)
The λ represents the square root of the eigenvalue corresponding to the principal component.
Data analysis
All data were calculated as the relative reduction percentage, i.e., the data under non-drought stress minus the data under drought stress then divided by the data under non-drought stress. The data were analyzed by using SPSS 20.0 statistic software package (Chicago, USA), and the linear fitting was analyzed by OriginPro 9.0.0 (OriginLab Corporation, USA).
Results
PEG-simulated drought stress influence on the wilting index
We observed that the seedling wilting was more severe with the prolongation of PEG-induced drought stress (Fig. 2). The leaves were healthy before PEG simulated drought stress (Fig. 2a). The cotyledons and the unifoliolate leave started wilting after 1d PEG simulated drought stress (Fig. 2b). The cotyledons, the unifoliolate leave, and the first trifoliolate leave became wilting after 3d PEG simulated drought stress (Fig. 2c). The whole plants showed complete wilting 5 d after PEG simulated drought stress (Fig. 2d). The wilting index of 62 soybean genotypes showed polymorphism (Fig. 3). From the Fig. 3, we found the lower wilting index were from Tiefeng 31, Pengshan Huangkezi-3, Jiyu 403, Jihei 3 and Dongnong 43 and the corresponding wilting index was 2.75, 3.25, 3.5, 3.5, and 4.25, respectively. The medium wilting indexes were 5, 5, 6, 6, and 7 respectively from Yannong 12, Zhonghuang 203, Shaxindou, Yangyan dou, and Lvhuangdou. The highest wilting indexes were 8, 9, 9, 9, and 9, respectively, which were from Madai Qingdou-2, Ludou 8, Fendou 93, Pine Bean, and Dahuangdou-2.
PEG-simulated drought stress influence on above-ground growth
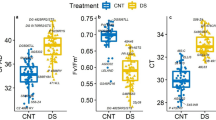
Drought stress can inhibit the growth of the above-ground part. Also, plant height, dry weight of the above-ground, stomatal conductance, and transpiration rate showed a significant down-regulation after PEG-simulated drought stress (Table 2). The wilting indexes were associated with plant height, above-ground part dry weight, electrolyte leakage, stomatal conductance and transpiration rate. The results of the linear analysis showed that the wilting index was positively correlated with plant height, and electrolyte leakage (Fig. 4a, c), and the correlation coefficients were 0.63 and 0.63, respectively. However, the wilting indexes were negatively correlated with a dry weight of the above-ground part, stomatal conductance, and transpiration rate (Fig. 4b–e), and the correlation coefficients were 0.72, 0.82, and 0.85, respectively.
PEG-simulated drought stress influenced on root system architecture
The PEG-simulated drought stress altered the root morphology of soybean plants (Fig. 5 and Table 3). From Fig. 5 and Table 3, we found the more extensive the wilting index, the shorter the root length, the fewer the number of lateral roots, and the smaller the root surface area and root volume. Furthermore, linear analysis between root morphological characteristics such as root length, lateral root number, root mean diameter, root surface area, root volume, and wilting index in sixty-two soybean cultivars was evaluated. The results indicated that the root length, lateral root number, root surface area, and root volume were negatively correlated with the wilting index, and the correlation coefficients were 0.80, 0.82, 0.85, and 0.81, respectively. Meanwhile, the average root diameter was positively correlated with the wilting index, and the correlation coefficient was 0.86 (Fig. 6a–f).
A comprehensive evaluation of drought tolerance among soybean genotypes
PCA comprehensively evaluated the 62 soybean germplasm for drought tolerance. The wilting index was positively correlated with the principal component, and R2 was 0.997 (Fig. 7). Root length, lateral root number, root surface area, root volume, plant height, and shoot growth contributed more to drought tolerance in principal component 1. In contrast, stomatal conductance and transpiration rate contributed more to drought tolerance in principal component 2, and the root-canopy ratio contributed more to drought tolerance in principal component 3 (Table 4). The linear correlation analysis between the wilting index and 11 drought tolerance related factors showed that stomatal conductance, transpiration rate, root length, lateral root number, root surface area, root volume, and average root diameter were strongly correlated with the wilting index under PEG simulated drought stress.
Meanwhile, the results of the principal component analysis showed that the principal component scores of Tiefeng 31, Wuyanwo, and Jilin 33 were higher than Tonghua Pingdingxiang, Yudou 20, and Shaxindou. The scores of Fendou 93, Liuyuehuang and Suinong 10 were the lowest. Therefore, Tiefeng 31, Wuyanwo and Jilin 33 can be considered as drought-tolerant soybean germplasms. Besides, Fendou 93, Liuyuehuang and Suinong 10 can be considered as drought-sensitive germplasms.
Discussion
Soybean productivity is lower compared to cereal crops. How to increase the global soybean productivity to satisfy the demand for soybean products is a concern of many researchers and farmers. Given the importance of soybean to the world economy, it is imperative to select drought-tolerant germplasm to improve soybean yield under drought conditions. The results showed that the plant height, shoot growth, stomatal conductance, and transpiration rate of soybean varieties decreased after PEG-simulated drought stress. Generally, drought tolerance soybean germplasm has strong water retention and is slow-wilting than that of drought-sensitive germplasm. The wilting index is the most intuitive index that can be seen by naked eyes after drought stress. Researchers use the wilting index as an indicator of drought tolerance directly (Sloane et al. 1990). The wilting of different genotypes may be associated with soil water conservation. The slow wilting genotype PI 416937 had higher soil water conservation by limiting the transpiration rate and decreasing stomatal conductance (King et al. 2009). Our result was coincidence with this, in which the wilting index was strongly negatively correlated with transpiration rate and stomatal conductance. Our results found the slow-wilting was negatively correlated with a dry weight of the above part, and this was following slow-wilting germplasms resulting in yield increases in many locations in 70% or more (Sinclair et al. 2010).
Drought tolerance is a very complex trait, which involves the morphological, physiological, and biochemical characteristics of above-ground and below-ground parts. So far, there is no valid, reasonable protocol and criteria for screening drought tolerance germplasms. Root system architecture, including root morphology and development, had a significant influence on plant survival and productivity, especially in water deficit stress or excess water (Manavalan et al. 2010; Jogaiah et al. 2013). Battisti and Sentelhas (2017) found the deeper root depth was an important characteristic to make soybean crops more elastic to droughts, and that was an important strategy to be considered for adapting the crop to climate change. The root is the first organ that could experience drought stress, and soybean taproots are the first roots that formed. Therefore, screening germplasms with long taproots under non-stress conditions by a cone system to evaluate the root system architecture in soybean germplasms might determine the profound rooting ability, which is helpful to drought tolerance (Manavalan et al. 2010). Our results showed that root length, lateral root number, root surface area, root volume, and root-canopy ratio increased, and the root diameter decreased with the PEG-simulated drought stress. The wilting index was strongly negatively correlated with root traits such as root length, the number of lateral roots, root surface area, and root volume, strongly positively correlated with root mean diameter and moderate negatively correlated with root-canopy ratio. Soybean plants that experienced drought stress before flowering had a higher yield than those stressed after flowering since the root system architecture had been developed well before flowering (Hirasawa et al. 1994). Liu et al. (2005) were also found significant correlations between drought resistance and various root traits such as dry weight, total length, root volume, and the number of lateral roots in soybean.
Recently, Prince et al. (2019) attempted to identify genes linked to root system architecture using GWAS in soybean landraces and elite cultivars and find four loci were detected in landraces for lateral root number, no major was detected for lateral root number in elite germplasm. The discovery that evaluated for drought tolerance germplasms to integrate wilting index and root system architecture could consider being used in soybean breeding programs in the future.
Conclusion
In summary, this study provided both above‐and below‐ground characteristics of sixty-two soybean germplasms during PEG-simulated drought stress in the seedling stage and evaluated the drought tolerance germplasm by PCA. The results showed the wilting index was positively correlated with plant height, electrolyte leakage and root mean diameter, but negatively correlated with shoot dry weight, stomatal conductance, transpiration rate, root-canopy ratio, root length, number of lateral roots, root surface area and root volume. Six drought-tolerance evaluation indexes were obtained by wilting index, correlation analysis, and PCA, which contain stomatal conductance, transpiration rate, root length, lateral root number, root surface area, and root volume. Furthermore, by comparison, the wilting index and the main component overall score, the drought-tolerant cultivar Tiefeng 31, and the drought-sensitive cultivar Fendou 93 were evaluated. Therefore, the selected drought-tolerant and drought-sensitive soybean cultivars could be used to understand further the mechanism of drought tolerance and breed soybean drought-tolerant cultivars in the future.
References
Bagherzadi L, Sinclair TR, Zwieniecki M, Secchi F, Hoffmann W, Carter TE, Rufty TW (2017) Assessing water-related plant traits to explain slow-wilting in soybean PI 471938. J Crop Improv 31(3):400–417
Battisti R, Sentelhas PC (2017) Improvement of soybean resilience to drought through deep root system in Brazil. Agron J 109:1612–1622
Bengough AG, McKenzie B, Hallett P, Valentine T (2011) Root elongation, water stress, and mechanical impedance: a review of limiting stresses and beneficial root tip traits. J Exp Bot 62:59–68
Bouslama M, Schapaugh WT (1984) Stress tolerance in soybeans. I. Evaluation of three screening techniques for heat and drought tolerance. Crop Sci 24(5):933–937
Charlson DV, Bhatnagar S, King CA, Ray JD, Sneller CH, Carter TE, Purcell LC (2009) Polygenic inheritance of canopy wilting in soybean [Glycine Max (L.) Merr.]. Theor Appl Genet 119:587–594
Dogan E, Kirnak H, Copur O (2007) Deficit irrigations during soybean reproductive stages and CROPGRO-soybean simulations under semi-arid climatic conditions. Field Crop Res 103:154–159
Douglas I, Alam K, Maghenda M, McDonnell Y, McLean L, Campbell J (2008) Unjust waters: climate change, flooding and the urban poor in Africa. Environ Urban 20:187–205
Fenta BA, Beebe SE, Kunert KJ, Burridge JD, Barlow KM, Lynch JP, Foyer CH (2014) Field phenotyping of soybean roots for drought stress tolerance. Agronomy 4:418–435
Fletcher AL, Sinclair TR, Allen LH Jr (2007) Transpiration responses to vapor pressure deficit in well-watered and ‘slow wilting’ and commercial soybean. Environ Exp Bot 61:145–151
Foley JA, Ramankutty N, Brauman KA, Cassidy ES, Gerber JS, Johnston M, Mueller ND, Connell C, Ray DK, West PC, Balzer C, Bennett EM, Carpenter SR, Hill J, Monfreda C, Polasky S, Rockström J, Sheehan J, Siebert S, Tilman D, Zaks DPM (2011) Solutions for a cultivated planet. Nature 478:337–342. https://doi.org/10.1038/nature10452
Gilbert ME, Zwieniecki MA, Holbrook NM (2011) Independent variation in photosynthetic capacity and stomatal conductance leads to differences in intrinsic water use efficiency in 11 soybean genotypes before and during mild drought. J Exp Bot 62(8):2875–2887
Hasanah Y, Nisa T, Armidin H, Hanum H (2015) Isoflavone content of soybean (Glycine max (L.) merr.) cultivars with different nitrogen sources and growing season under dry-and conditions. J Agric Environ Int Dev (JAEID) 109(1):5–17
Hirasawa T, Tanaka K, Miyamoto D, Takei M, Ishihara K (1994) Effects of pre-flowering moisture deficits on dry matter production and ecophysiological characteristics in soybean plants under drought conditions during grain filling. J Crop Sci 63:721–730
Hoagland DR, Arnon DI (1938) The water-culture method for growing plants without soil. University of California Agricultural Research Station, Davis, C.A. Circular 347:1–39
Hudak CM, Patterson RP (1996) Root distribution and soil moisture depletion pattern of a drought-resistant soybean plant introduction. Agro J 88:478–485
Jogaiah S, Govind SR, Phan Tran LS (2013) Systems biology-based approaches toward understanding drought tolerance in food crops. Crit Rev Biotechnol 33(1):23–39
King CA, Purcell LC, Brye KR (2009) Differential wilting among soybean genotypes in response to water deficit. Crop Sci 49(1):290–298
Liu X (1986) Discussing assessment methods of drought resistance of soybean. Chin J Oil Crop Sci 4(2):23–26
Liu F, Anderson MN, Jacobson SE, Jensen CR (2005) Stomatal control and water use efficiency of soybean (Glycine max L. Merr.) during progressive soil drying. Environ Exp Bot 54:33–40
Manavalan LP, Guttikonda SK, Phan Tran LS, Nguyen HT (2009) Physiological and molecular approaches to improve drought resistance in soybean. Plant Cell Physiol 50(7):1260–1276
Manavalan LP, Guttikonda SK, Nguyen VT, Shannon JG, Nguyen HT (2010) Evaluation of diverse soybean germplasm for root growth and architecture. Plant Soil 330:503–514
Oosterhuis DM, Walker S, Eastham J (1985) Soybean leaflet movements as an indicator of crop water stress1. Crop Sci 25:1101–1106
Pantalone VR, Rebetzke GJ, Burton JW, Carter TE (1996) Phenotypic evaluation of root traits in soybean and applicability to plant breeding. Crop Sci 36:456–459
Passioura JB (1983) Root and drought resistance. Agric Water Manag 7:265–280
Pathan SM, Lee JD, Sleper DA, Fritschi FB, Sharp RE, Carter TE, Nelson RL, King CA, Schapaugh WT, Ellersieck MR, Nguyen HT, Shannon JG (2014) Two soybean plant introductions display slow leaf wilting and reduced yield loss under drought. J Agron Crop Sci 200:231–236
Prince SJ, Valliyodan BB, Ye H, Yang M, Tai SS, Hu WS, Murphy M, Durnell LA, Song L, Joshi T, Liu Y, de Velde JV, Vandepoele K, Shannon JG, Nguyen HT (2019) Understanding genetic control of root system architecture in soybean: insights into the genetic basis of lateral root number. Plant Cell Environ 42:212–229
Ries LL, Purcell LC, Carter TE, Edwards JT, King CA (2012) Physiological traits contributing to differential canopy wilting in soybean under drought. Crop Sci 52:272–281
Sadok W, Gilbert ME, Raza MS, Sinclair TR (2012) Basis of slow-wilting phenotype in soybean PI 471938. Crop Sci 52:1261–1269
Sloane RJ, Patterson RP, Carter TE (1990) Field drought tolerance of a soybean plant introduction. Crop Sci 30:118–123
Sinclair TR, Messina CD, Beatty A, Samples M (2010) Assessment across the united states of the benefits of altered soybean drought traits. Agron J 102(2):475–482
Sinclair TR, Zwieniecki MA, Holbrook NM (2008) Low leaf hydraulic conductance associated with drought tolerance in soybean. Physiol Plant 132:446–451
Tester M, Langridge P (2010) Breeding technologies to increase crop production in a changing world. Science 327:818–822
Thao NP, Tran LSP (2012) Potentials toward genetic engineering of drought-tolerant soybean. Crit Rev Biotechnol 32:349–362
Ye H, Roorkiwal M, Valliyodan B, Zhou LJ, Chen PG, Varshney RK, Nguyen HT (2018) Genetic diversity of root system architecture in response to drought stress in grain legumes. J Exp Bot 69:3267–3277
Yue CL, Huang YX, Yan Z (2003) SAS system and economic statistical analysis. China University of Science and Technology Press, Hefei (in Chinese)
Acknowledgements
This work was supported by the National Major Special Project for Transgenic Organisms, Ministry of Agriculture in China (No. 2016ZX08004-004-005).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, G., Zhou, Q., He, M. et al. Wilting index and root morphological characteristics used as drought-tolerance variety selection at the seedling stage in soybean (Glycine max L.). Plant Growth Regul 92, 29–42 (2020). https://doi.org/10.1007/s10725-020-00617-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00617-0