Abstract
This study tested the hypothesis that abscisic acid (ABA) and ethylene may mediate the effect of post-anthesis soil drying on grain filling of rice (Oryza sativa L.) through regulating its anti-oxidative systems. Two rice cultivars were pot-grown and exposed, from 9 days post anthesis until maturity, to three soil drying treatments: well-watered (WW), moderate drying (MD), and severe drying (SD). The MD treatment, compared with the WW treatment, increased grain filling rate and the activities of CAT, POD, and SOD in superior and inferior spikelets, whereas the SD treatment decreased them. Both MD and SD treatments enhanced the grain contents of ABA, superoxide anion, and MDA, with a higher rise in the SD than in the MD. The MD treatment decreased ethylene evolution rate and 1-aminocycopropane-1-carboxylic acid (ACC) content, whereas the SD treatment increased them. The ratio of ABA–ACC (ABA/ACC) was the highest under the MD and the lowest under the SD treatment. There was a significant positive correlation between the ABA content, the activities of CAT, POD, SOD, and ABA/ACC with the grain filling rate, while the levels of ethylene and ACC and the contents of superoxide anion and MDA correlated negatively with the parameter. Exogenous chemical application verified the roles of ABA and ethylene in grain filling. The results suggest that an increase in ABA and decrease in ethylene in the grains under post-anthesis moderate soil drying could enhance the anti-oxidative enzyme activities, thus reducing free radicals in grains and accelerating grain filling rate of rice.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Grain filling is the final growth stage in cereals when fertilized ovaries develop into caryopses. The filling degree and rate of rice (Oryza sativa L.) spikelets differ largely with their positions on a panicle. In general, the earlier-flowering superior spikelets, usually located on apical primary branches, fill fast and produce larger and heavier grains. Conversely, the later-flowering inferior spikelets, commonly located on proximal secondary branches, are either sterile or fill slowly and poorly (Mohapatra et al. 1993; Yang et al. 2000a). There are many explanations for the physiological mechanism differences between the superior and inferior spikelets attributed mainly to the assimilate supply (Murty and Murty 1982; Fu et al. 2011), hormone balance (Naik and Mohapatra, 1999; Yang et al. 2006a; Zhang et al. 2009), enzyme activities (Nakamura et al. 1989; Kato et al. 2007), and gene expressions (Dai et al. 2009; Jiang et al. 2003; Ishimaru et al. 2003). Grain filling is generally believed to be a process, regulated either genetically or environmentally (Yoshida 1972; Saini and Westgate 2000). Water or drought stress is one of the most important factors limiting crop yields worldwide (Passioura 1996, 2007; Tian et al. 2013). Water stress during the early grain development curtails the kernel sink potential by reducing the number of endosperm cells and amyloplasts formed, thus diminishing grain weight as a result of the decrease in the capacity of endosperm to accumulate starch, both in rate and duration (Bouman and Tuong 2001; Samejima et al. 2004). However, there are reports showing that a moderate soil drying during the grain filling stage could markedly enhance grain growth in both rice and wheat (Triticum aestivum), and consequently rise grain weight, especially in the inferior spikelets (Yang and Zhang 2006; Zhang et al. 2012; Chen et al. 2013). The mechanism underlying the decrease or increase in grain filling under soil drying is yet to be well understood.
Abscisic acid (ABA) and ethylene are the two major phytohormones generally suggested to be involved in linking plant responses to stress (Cheng and Lur 1996; Santner and Estelle 2009). A significant ABA accumulation was observed in aborted maize (Zea mays L.) kernels under drought stress (Ober et al. 1991), and a reduction in the grain set and kernel weight under water stress in wheat was associated with the elevated levels of ABA (Morgan 1980; Ahmadi and Baker 1999). However, there are reports that the inferior spikelets have a lower ABA content than superior spikelets at the early grain filling stage, and the cell division and grain filling rates are significantly positively correlated with ABA contents in grains in both wheat and rice (Yang and Zhang 2006; Yang et al. 2006a). A high ethylene evolution rate, induced by drought, has frequently been related to the abortion in cotton (Gossypium hirsutum L.) and maize (Guinn 1976; Cheng and Lur 1996) and the reduction in wheat grain weight (Beltrano et al. 1999).
It has been reported that many adversities could induce membrane damage, lipid permeability increase, and biological accumulation of free radicals, such as malondialdehyde (MDA), one of the lipid peroxidation products (Rajinder et al. 1981; Huang et al. 2013). Moreover, drought, like other abiotic stress factors, results in oxidative stress through an increase in the reactive oxygen species (ROS), such as the superoxide radical (O2 −). ROS could destroy the normal cellular metabolism through the oxidative damage to lipids, proteins, and nucleic acids, and cause growth impairment in plants. To eliminate these ROS, plants have developed a complex anti-oxidative enzymes system (AES), including catalase (CAT), peroxidase (POD), and superoxide dismutase (SOD) (Nunez et al. 2003). It has been reported that the drought damage degree was negatively correlated with CAT, POD, and SOD activities, but corresponded positively with MDA accumulation (Liu et al. 2011). Although the AES and ROS in rice under water stress are well documented, little is known about the relationships of ABA, ethylene, AES, and ROS with the grain filling of the superior and inferior spikelets of rice under water stress.
The objective of this study was to test the hypothesis that the interaction between ABA and ethylene is involved in grain filling through mediating AES and ROS. The changes of ABA, ethylene, AES, and ROS levels in the superior and inferior spikelets of rice, subjected to post-anthesis soil drying and their relationships with grain filling, were investigated. The effects of the chemical regulators in the grains were also studied to verify the role of the two hormones, ABA and ethylene.
Materials and methods
Plant materials and growth conditions
The experiment was conducted at a farm of Yangzhou University, Yangzhou, China (32° 30′N, 119° 25′E) during the rice growing season (May–October) of 2010, and repeated in 2011. Two rice cultivars, Wuyunjing 8 (japonica) and Yangdao 6 (indica) were grown in pots. The seeds were sown in the paddy field on 8–10 May. Thirty-day-old seedlings were then transplanted to porcelain pots. Each porcelain pot (30 cm in height and 25 cm in diameter, 14.72 l in volume) was filled with 20 kg of sandy loam soil (Typic fluvaquents, Entisols, US classification) that contained 2.42 g kg−1 organic matter and 110 mg kg−1 alkali-hydrolysable N, 34.6 mg kg−1 Olsen-P, and 66.6 mg kg−1 exchangeable K. Each pot was planted with three hills with two seedlings per hill. On the day of transplanting (8–10 June), 1.0 g N as urea, 0.3 g P as single superphosphate, and 0.5 g K as KCl were mixed into the soil in each pot. N as urea was also applied at mid-tillering (0.5 g per pot) and panicle initiation (0.8 g per pot) stages. Both cultivars headed on 24–26 August (50 % of plants), flowered on 26–28 August, and were harvested on 19–20 October. The water level in the pot was kept at 1–2 cm until 9 days post anthesis (DPA) when soil drying treatments were initiated. The total precipitation during the growing season was 496.4 mm, 67.2 % of which was in June and July. The mean solar radiation was 17.8 MJ m−2 day−1. The temperatures, averaged over the 10-day period from anthesis to harvest, were 27.8, 27.2, 24.4, 21.6, 21.1, and 20.2 °C, respectively.
Soil drying treatments
The experiment was a 2 × 3 (two cultivars and three levels of soil moisture) factorial design with six treatments. Each treatment consisted of 60 pots as replicates. From 9 DPA until maturity, three levels of soil water (ψ soil) potentials were imposed by controlling the water application. The well-watered (WW) treatment was flooded with 1–2 cm water depth in the pot (ψ soil = 0 kPa) by manually applying tap water; the moderate soil drying (MD) treatment was maintained at −20 ± 5 kPa; the severe soil drying (SD) treatment was maintained at −40 ± 5 kPa. The soil water potentials in the soil drying treatments were monitored in the 15–20 cm soil depth. A tension meter (Soil Science Research Institute, Nanjing, China) consisting of a sensor of 5 cm length was installed in each pot to monitor the water potential. The tension meter readings were recorded every 3 h from 0600 to 1800 h. When the readings dropped to the desired value, 0.4 and 0.2 l of tap water per pot was added to the MD and SD treatments, respectively. The pots were placed in a field and sheltered from rain by a removable polyethylene shelter, which was placed over them during rain.
Sampling
Six hundred panicles that headed on the same day were chosen and tagged from each treatment. Thirty tagged panicles from each treatment were sampled at 3 days intervals from anthesis to maturity. The sampled panicles were divided into three groups (10 panicles each) as subsamples. Both superior and inferior grains that developed from the fertilized spikelets were removed. One half of the sampled grains were used for measurements of ABA, 1-aminocyclopropane-1-carboxylic acid (ACC), O2 −, MDA contents, and the activities of the anti-oxidative enzymes. Sixty to eighty sampled grains were used for the measurement of ethylene production. About 80-100 grains were dried at 70 °C to constant weight, dehulled, and weighed. The superior spikelets that flowered on the first 2 days (located on apical primary branches) within a panicle and the inferior ones (located on the proximal secondary branches) that flowered on the last 2 days within a panicle were separated from the sampled panicles. The difference in the flowering date between the superior and inferior spikelets within a panicle was 3–4 days. The grain filling processes of both superior and inferior spikelets were fitted by the Richards’s growth equation (Richards 1959), as described by Zhu et al. (1988):
Grain filling rate (R) was calculated as the derivative of the Eq. (1)
where W is the grain weight, A is the maximum grain weight, t is the time after anthesis (d), and B, k, and N are coefficients determined by regression. The active grain filling period was defined as that the time interval when W was from 5 % (t 1) to 95 % (t 2) of A. The average grain filling rate during this period was therefore calculated from t 1 to t 2.
ABA extraction, purification, and quantification
The methods for extraction and purification of ABA were modified from those described by Bollmark et al. (1988) and He (1993). Samples of 0.8–1.0 g spikelets were ground in a mortar (at 0 °C) in 10 ml 80 % (v/v) methanol extraction medium containing 1 mM butylated hydroxytoluene as an antioxidant. The extract was incubated at 4 °C for 4 h and centrifuged at 4,800×g for 15 min at the same temperature. The supernatants were passed through Chromosep C18 columns (C18 Sep-Pak Cartridge, Waters Corp, Millford, MA, USA), prewashed with 10 ml 100 % and 5 ml 80 % methanol, respectively. The hormone fractions were dried under N2 and dissolved in 2 ml phosphate buffered saline (PBS) containing 0.1 % (v/v) Tween 20 and 0.1 % (w/v) gelatin (pH 7.5) for analysis by enzyme-linked immunosorbent assay (ELISA).
The mouse monoclonal antigen and antibody against ABA and immunoglobulin G-horse radish peroxidase (IgG-HRP), used in ELISA, were produced at the Phytohormones Research Institute, China Agricultural University, China (see He 1993). The methods for quantification of ABA by ELISA and the recovery test were described previously (Yang et al. 2001b). The recovery percentage of ABA in spikelets was 85.0 ± 4.9. The specificity of the monoclonal antibody and other possible nonspecific immunoreactive interferences were checked previously and proved reliable (Xie et al. 2003).
Ethylene and ACC determination
The ethylene that evolved from spikelets was determined according to Beltrano et al. (1994) with modifications. Briefly, the sampled spikelets or sheaths were placed between two sheets of moist paper for 1 h at 27 °C in darkness to allow wound ethylene to subside. Each sample contained 0.5–0.8 g spikelets or 1–2 g sheaths. Spikelet/sheaths were then transferred into 10 ml glass vials containing moist filter paper and immediately sealed with airtight stoppers and incubated in the dark for 24 h at 27 °C. A 1 ml gas sample was withdrawn through the airtight stoppers with a gas-tight syringe, and ethylene was assayed by a gas chromatograph (HP5890 Series II, Hewlett Packard, Palo Alto, CA, USA) equipped with a Porapak Q column (0.3 cm × 200 cm, 50–80 mesh) and flame ionization detector (FID). The temperatures of the injection port, column, and detector were kept constant at 140, 100, and 200 °C, respectively. Nitrogen was used as the carrier gas at a flow rate of 30 ml min−1, and hydrogen and air were utilized for FID at the rate of 30 and 300 ml min−1, respectively.
To examine the time-course changes in ethylene production by the spikelets after sampling, the spikelets sampled at 12 day before heading were incubated for 6, 12, 18, 24, and 30 h, respectively, for ethylene measurement through the same method described above.
ACC in the spikelets and sheaths was determined according to the method suggested by Cheng and Lur (1996). The ethylene evolved from ACC was assayed by using gas chromatography, as described above. The transformation rate as a percentage from ACC to ethylene was 90.3 ± 5.42 for spikelets and 88.5 ± 6.4 for sheaths, on average. ABA and ACC concentrations and the rate of ethylene evolution were expressed on a dry weight basis.
Measurements of CAT, POD, and SOD activities
CAT activity was determined by following the consumption of H2O2 (extinction coefficient 39.4 mM−1 cm−1) at 240 nm for 3 min (Aebi 1984). POD activity was assayed by the method described by Cakmak and Marschner (1992). SOD activity was determined through measuring its ability to inhibit the photochemical reduction of nitroblue tetrazolium (NBT), according to the method of Giannopolitis and Ries (1977). The protein content was determined according to the method of Bradford (1976).
Production rate of O2 − and MDA content determination
The production rate of O2 − was determined according to Elstner and Heupel (1976) by monitoring the nitrite formation from hydroxylamine in the presence of O2 −. The absorbance was read at 530 nm. MDA content was assayed via the thiobarbituric acid (TBA) reaction (Heath and Packer 1968).
Chemical applications
The cultivar Yangdao 6 was used for the chemical application. The experimental plants were grown in porcelain pots under the same conditions as described above. From 9 DPA till maturity, either WW or SD treatments were imposed to the plants. The treatment details, water control, and rain prevention were described previously. Synthetic ABA, ethephon (an ethylene-releasing agent), cobaltous nitrate [Co(NO3)2, an inhibitor of ethylene synthesis] (all from Sigma, St Louis, MO, USA), and fluridone (Fluka, Riedel-de Haën, Germany), an inhibitor of ABA synthesis, were applied to the plants for both WW and SD treatments. The preparation of the chemical solutions was described elsewhere (Ober and Sharp 1994; Cheng and Lur 1996; Sharp et al. 2000). Starting at 9 DPA, either 20 × 10−6 M ABA, or 20 × 10−6 M fluridone, or 50 × 10−3 M ethephon, or 5 × 10−5 M Co(NO3)2, were sprayed at the rate of 100 ml per pot on the top of the plants (panicles) daily, for 5 day. The time of application of the exogenous hormones was from 16.00 to 18.00 h. The relative humidity (%RH) during the every day spraying, was 68.6, 73.9, 73.2, 61.9, and 65.9 %, respectively. All the solutions contained ethanol and Tween 20 at final concentrations of 0.1 % (v/v) and 0.01 % (v/v), respectively. The control plants were sprayed with the same volume of deionized water, containing the same concentrations of ethanol and Tween 20. Each chemical treatment had 20 pots. The concentrations of ABA, ethylene evolution rate, and CAT, POD, and SOD activities as well as the O2 − production rate and MDA contents in the grains were determined 3 and 7 day after the chemical treatments (16 and 20 DPA). Twenty panicles for each treatment were harvested at maturity for the determination of yield and its components.
Statistical analysis
The results were analyzed for variance by using the SAS/STAT statistical analysis package (version 6.12, SAS Institute, Cary, NC, USA). The data from each sampling date were analyzed separately and the means were tested by the least significant difference at the P 0.05 level (LSD 0.05). Linear regression was used to evaluate the relationships of grain filling rate, levels of ABA, ethylene, ACC, and the ratio of ABA to ACC with AES and ROS. The differences in data across the two study years and in the interaction between the year and cultivar were not significant (F < 1). Therefore, the data from both years were averaged.
Results
Grain weight and grain filling rate
The grain weight and filling rate varied with the grain type and soil drying treatment (Fig. 1). The grain filling procession of the superior spikelets showed no significant difference among the three soil drying treatments. However, the grain weight and filling rate of the inferior spikelets markedly increased in the MD treatment, whereas the values of these indices significantly decreased in the SD treatment, when compared with the WW treatment. There was no difference in these parameters caused by the cultivars.
Effect of soil drying on grain weight (a, c) and grain filling rate (b, d) of the superior and inferior spikelets of rice. Both japonica cultivar Wuyunjing 8 (a, b) and indica cultivar Yangdao 6 (c, d) were grown in pots. WW, MD, and SD indicate well-watered, moderate soil drying, and severe soil drying, respectively, during the grain filling period (from 9 DPA until maturity). S and I represent the superior grain and inferior grain, respectively. Vertical bars represent ± SE of the mean (n = 6) where these exceed the size of the symbol
Activities of CAT, SOD and POD in grains
The activity levels of CAT, SOD, and POD were increased continuously after the anthesis, reached a maximum, and declined thereafter (Fig. 2). Very similar to the grain filling rate, the highest value of CAT,POD, and SOD activities appeared at the maximum grain filling rate stage. At the early grain filling stage, these activities were with lower magnitude in the inferior than in the superior spikelets, whereas the results were reversed at the middle and late grain filling stages. The activities of the enzymes reaching a maximum appeared later and were affected by the soil drying treatments to a higher degree in the inferior spikelets than in the superior ones (Fig. 2). The MD treatment stimulated the actions of the three enzymes. Under the SD treatment, the enzymatic operations were significantly increased at the early stage of the treatment and decreased at its late stage when compared with those under the WW (Fig. 2).
Effect of soil drying on CAT (a, b), POD (c, d), and SOD (e, f) activity in the superior and inferior spikelets of rice. Both japonica cultivar Wuyunjing 8 (a, c, e) and indica cultivar Yangdao 6 (b, d, f) were grown in pots. WW, MD, and SD indicate well-watered, moderate soil drying, and severe soil drying, respectively, during the grain filling period (from 9 DPA until maturity). S and I represent the superior grains and inferior grains, respectively. Arrows indicate the start of the period in which water was withheld. Vertical bars represent ± SE of the mean (n = 6) where these exceed the size of the symbol
Production rate of O2 − and MDA content in grains
The production rate of O2 − showed no considerable difference between the superior and inferior spikelets at the early grain filling stage, and at the end of grain filling it was greater in the superior than in the inferior ones (Fig. 3). There was no substantial variation in the production rate of O2 − between WW and MD. The SD treatment significantly increased the production rate of O2 − in the spikelets. The effect of soil drying on O2 − production was more pronounced in the inferior than in the superior spikelets (Fig. 3a, b). The tendency of MDA content was very similar to that of the production rate of O2 − (Fig. 3c, d).
Effect of soil drying on production rate of O2 − (a, b) and MDA (c, d) content in the superior and inferior spikelets of rice. Both japonica cultivar Wuyunjing 8 (a, c) and indica cultivar Yangdao 6 (b, d) were grown in pots. WW, MD, and SD indicate well-watered, moderate soil drying, and severe soil drying, respectively, during grain filling (from 9 DPA until maturity). S and I represent the superior grains and inferior grains, respectively. Arrows indicate the start of the period in which water was withheld. Vertical bars represent ± SE of the mean (n = 6) where these exceed the size of the symbol
Levels of ABA, ethylene, ACC, and ABA/ACC in grains
The ABA concentration in both the superior and inferior spikelets was exceedingly low at the early grain filling stage, but increased quickly and reached a peak at 12 DPA in the superior spikelets, and at 18 DPA in the inferior spikelets, and then sharply decreased (Fig. 4a, b). At the early and middle grain filling stage, the superior spikelets had a much higher ABA concentration than the inferior, whereas the trend was reversed at the late grain filling stage. There was no significant difference in the ABA content of the superior spikelets among the three soil moisture treatments. However, both the MD and SD treatments markedly enhanced the ABA concentration of the inferior spikelets when compared to WW, and this rise was more apparent in the SD than in the MD spikelets (Fig. 4a, b).
Effect of soil drying on ABA concentration (a, b), ethylene evolution rate (c, d), ACC concentration (e, f) and the ratio of ABA to ACC (g, h) in the superior and inferior spikelets of rice. Both japonica cultivar Wuyunjing 8 (a, c, e, g) and indica cultivar Yangdao 6 (b, d, f, h) were grown in pots. WW, MD, and SD indicate well-watered, moderate soil drying, and severe soil drying, respectively, during the grain filling period (from 9 DPA until maturity). S and I represent the superior grains and inferior grains, respectively. Arrows indicate the start of the period in which water was withheld. Vertical bars represent ± SE of the mean (n = 6) where these exceed the size of the symbol
In sharp contrast to the instance of ABA content, the ethylene evolution from both the superior and the inferior spikelets was very high at the early grain filling stage, followed by a slight reduction until 9 DPA; then it declined quickly from 9 DPA until 24 DPA, increased thereafter, and decreased again from 30 DPA (Fig. 4c, d). During the grain filling period, the inferior spikelets showed a much higher ethylene evolution rate than the superior spikelets among the three soil moisture treatments. The ethylene release from both superior and inferior spikelets was reduced in the MD treatment, whereas it was substantially increased in the SD treatment, A similar change pattern was observed in the ACC concentration in both the superior and the inferior spikelets (Fig. 4e, f). The ACC concentration was significantly correlated with the ethylene evolution rate (r = 0.99**, P < 0.01).
As shown in Fig. 4, the ratio of ABA to ACC in both superior and inferior spikelets was the highest in the MD treatment and the lowest in the SD treatment, suggesting that the increase in the ACC concentration, relative to the increase in ABA concentration, was greater under the SD treatment. The two cultivars have the same changing tendency (Fig. 4g, h).
Correlations of grain filling rate, levels of ABA, ethylene, ACC, and the ratio of ABA to ACC with the anti-oxidative enzymes activities, and MDA and O2 − contents. As shown in Table 1, the ABA content and the ratio ABA to ACC were significantly or very significantly positively correlated with the CAT, POD, and SOD activities, and very significantly negatively correlated with the MDA content and the production rate of O2 −. The ABA content, ABA/ACC, and the CAT, POD, and SOD activities were correlated very significantly positively with the grain filling rate, while the levels of ethylene and ACC and the contents of superoxide anion and MDA correlated very significantly and negatively with the grain filling intensity.
Effects of chemical applications
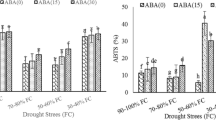
The application of ABA or Co(NO3)2, an inhibitor of ethylene synthesis, considerably increased CAT, POD, and SOD activities, whereas it decreased the production rate of O2 − and the MDA content in the inferior spikelets under both WW and SD treatments at 16 and 20 DPA. Conversely, the application of ethephon, an ethylene-releasing substance, or fluridone, an inhibitor of ABA biosynthesis, showed an opposite effect. There were no significant differences in the CAT, POD, and SOD activities, and the production rate of O2 − and MDA content of the superior spikelets when chemicals were applied to panicles under both WW and SD treatments at 16 and 20 DPA (Supplementary Table 1). When fluridone was applied, the ABA concentration of the superior and inferior spikelets was significantly reduced under both WW and SD treatments. The application of ABA produced the opposite effects in the inferior spikelets under both WW and SD treatments. No influence was observed on ABA contents in the superior spikelets when ABA was employed in the panicles under the SD treatment. When Co(NO3)2 was utilized, the ethylene evolution rate was substantially diminished. The application of ethephon to WW spikes increased the ethylene evolution level (Supplementary Table 1).
Under the WW and SD treatment, when Co(NO3)2 or ABA was applied to the panicles, the quantity of the filled grains and grain weight were enhanced, and the grain yield was augmented. The opposite results were observed when ethephon or fluridone was used (Supplementary Table 2).
Discussion
Previous studies have described that soil drying during grain filling decreased grain filling rate, shortened the grain filling stage, and thereafter reduced the number of filled grains and their weight (Rahman and Yoshida 1985; Wardlaw and Willenbrink 2000). Our study also showed similar results in SD (Fig. 1). We observed, however, that MD increased the grain weight and filling rate, especially in the inferior spikelets, when compared to WW. That indicates that if a water deficit was controlled properly during grain filling, similarly to the conditions in the MD treatment, the moderate soil drying maintained at −20 ± 5 kPa after the fertilization sensitive period, it would not seriously affect plant status and could enhance assimilates remobilization from vegetative tissues to grains, especially to those in the inferior spikelets, thus accelerating grain filling and improving grain weight (Fig. 1). This result is consistent with our previous observations on rice (Zhang et al. 2010, 2012). Yang et al. (2000b, 2001a, 2006b) also reported that moderate soil drying increased the sucrose phosphate synthase and α-/β-amylase activity in stems and sheaths, enhanced the grain sink activity, thus promoted the operation of carbohydrates in stem and sheath to grain. At the same time, the moderate soil drying also improved the key enzymes activity in the sucrose-to-starch pathway in grains, favored the grain filling rate, thus augmented the quantity of filled grains and enhanced grain weight. This suggests that the gain from the accelerated grain filling rate could outweigh the possible loss of photosynthesis as a result of a shortened grain filling period when subjected to water stress during grain filling.
Hormones play important roles in the adaptation of plant growth and development to water stress (Sharp et al. 2004). We established that both MD and SD treatments markedly increased the ABA concentration of the inferior spikelets when compared to WW, with a higher elevation under the SD than under the MD. There was no significant difference in the ABA concentration of the superior spikelets among the three soil moisture treatments (Fig. 4a, b). Numerous observations exist that a reduction in grain set and kernel growth in wheat (Morgan 1980; Ahmadi and Baker 1999), and a decreased rate of endosperm cell division rate in maize (Ober et al. 1991) under soil drying are associated with elevated levels of ABA. However, our results showed that the change in ABA concentration in the grains followed a pattern similar to that of the grain filling rate (Figs. 1b, d, 4a, b). The latter was very significantly positively correlated with the ABA concentration (Table 1). Under the MD treatment, an enhanced ABA accumulation in the grains was closely associated with an increased grain filling rate. The application of ABA to WW panicles considerably fostered ABA concentration at 16 and 20 DPA, whereas the application of fluridone, an indirect inhibitor of ABA synthesis, had the opposite effect (Supplementary Table 1, 2). These effects indicate that the slow grain filling may be attributed to a low ABA concentration in the grains, and the higher ABA content enhances grain filling rate.
Ethylene is an endogenous plant growth regulator mediating many physiological processes, such as cell division, embryogenesis, fruit set and growth, senescence, and responses to environmental stress factors (Davies 2004; Yang et al. 2006a, b). Our results showed that MD reduced the ethylene evolution from both superior and inferior spikelets, whereas the SD treatment stimulated it significantly (Fig. 4c, d). We speculate that the production of ethylene in water-stressed rice grain may depend on the severity and duration of soil drying. A very similar change pattern was observed in the ACC concentration in both superior and inferior spikelets (Fig. 4e, f). Our data demonstrated that the rise in ethylene production is attributed to the enhanced level of ACC, a precursor of ethylene. There are reports indicating that higher levels of ethylene and ACC have frequently been related to abortion in maize (Zea mays) (Cheng and Lur 1996) and a reduction in grain weight of wheat (Triticum aestivum) (Beltrano et al. 1994; Yang et al. 2006a) and rice (Yang et al. 2006b). We observed that the inferior spikelets showed a much higher evolution rate of ethylene and ACC concentrations than the superior spikelets under the three soil moisture treatments during grain filling (Fig. 4c–f). There was a significant negative correlation between the levels of ethylene and ACC with the grain filling rate. The application of ethephon to WW panicles substantially increased the ethylene evolution at 16 and 20 DPA, but decreased the grain filling rate, grain weight, and yield, whereas the application of Co(NO3)2, an inhibitor of the ethylene synthesis, had the opposite effect (Supplementary Table 1, 2).
The facilitating mechanism of ABA and ethylene on grain filling is not completely understood. The interaction between ABA and ethylene in the superior and inferior spikelets of rice has been described and the relationship between this interaction and the rate of grain filling from the assimilates supply (Mohapatra et al. 1993), the enzymatic activity (Kato et al. 2007; Zhang et al. 2012), and the mechanism at a molecular level (Ishimaru et al. 2005; Wang et al. 2008) has been represented. Plant hormones can act either synergistically or antagonistically, and it is the balance between promoting and inhibiting agents that ultimately determines plant growth and development (Davies 2004). Our results demonstrated that the changes in the ratio of ABA to ACC in the grains were also associated with the grain filling rate (Figs. 1b, d, 4g, h). The rate of grain filling correlated not only with the levels of ABA and ethylene, but also with the ratio of ABA to ACC (Table 1). These findings suggest that antagonistic interactions between ABA and ethylene may mediate and influence grain filling in rice when subjected to soil drying during meiosis.
Previous studies have described that the lipid permeability of the plant membrane increased under adversities, contributing to the biological accumulation of free radicals, such as O2 − and MDA (Liu et al. 2011). In order to cope with the harsh environment, plants themselves could generate a complex AES, such as CAT, POD, and SOD to counteract and eliminate these hazardous substances. It was reported that under moderate water stress the CAT, POD, and SOD activities in rice seedlings were stimulated constantly over time, while the SOD and POD effects increased under severe water stress conditions (Jiang et al. 1991). Wang et al. (2007) found that the operation level of CAT, POD, and SOD in the flag leaves of rice grew constantly under moderate water stress, and the activities of the three enzymes decreased over time and along with the aggravation of the stress degree. Our results showed that compared to WW, MD increased the production rate of O2 − and MDA content in spikelets, but there was no significant difference between WW and MD (Fig. 3). At the same time, the MD treatment increased the activities of the three enzymes, mentioned above (Fig. 2), indicating that AES could eliminate the harmful effects caused by the MD treatment. However, AES could not deal with the accumulation of hazardous substances in the SD treatment (Figs. 2, 3). Meanwhile, we noticed that there were no substantial variations in the production rate of O2 − and MDA content between the superior and inferior spikelets at the early grain filling stage, while they were considerably greater in the superior than in the inferior spikelets at the end of grain filling (Fig. 3). Moreover, the activity of the three enzymes in the superior spikelets was higher than that in the inferior at the early grain filling stage, whereas the results were reversed at the late grain filling stage (Fig. 2). The evidence infers that the effect of the soil drying on the production rate of O2 −, MDA content, and AES was remarkably dissimilar because of the variations of the soil drying status, the measurement time, and the differences between the inferior and superior spikelets.
Agarwal et al. (2005) proposed that ABA enhanced the CAT and SOD activities, reduced the H2O2 and membrane lipid peroxidation contents, and the responses of SOD to ABA was most obvious. Our results showed that the ABA level was very significantly positively correlated with the CAT, POD, and SOD activities, and very significantly, but negatively correlated with MDA content and the production rate of O2 − (Table 1). When ABA was applied to the spikes at the early grain filling stage (9–13 DPA), the ABA concentration in the spikelets was elevated or substantially exceeded that of the control (Supplementary Table 1). The CAT, POD, and SOD activities were greatly intensified, while the O2 − production rate and MDA content were considerably lower in the inferior spikelets, leading to the improvement of grain filling and the promotion of grain weight and yield (Supplementary Table 1, 2). Our data clearly demonstrated that ABA could enhance the effects of the enzymes protecting the membrane from lipid peroxidation. We also observed that after the application of ethephon to the spikes at the early grain filling stage (9-13 DPA), the activities of CAT, POD, and SOD decreased significantly, and the O2 − production rate and MDA content increased greatly in the inferior spikelets, causing a decline in the grain filling rate and grain weight (Supplementary Table 1, 2). Basing on the above results, we speculated that ethylene could enhance the active oxygen system and stimulate the free radicals production.
It is noteworthy that the ratio of ABA to ACC level was significantly or very significantly positively correlated with the activities of CAT, POD, and SOD, and very significantly negatively correlated with MDA content and the production rate of O2 −. The grain filling rate was very significantly positively correlated with CAT, POD, and SOD activities, but very significantly negatively correlated with MDA content and production rate of O2 − (Table 1). The results indicate that ABA could increase CAT, POD, and SOD activities and enhance the ability of the plant to eliminate free radicals, while ethylene could promote the formation of active oxygen system and increase the content of free radicals. The higher ratio ABA to ACC may enhance the endosperm cell physiological activity, restrain or delay the endosperm cell senescence, enhance the grain filling rate, finally leading to an increase in the grain weight.
In conclusion, the effects of post-anthesis soil drying on grain filling were mediated by the balance between ABA and ethylene through regulating anti-oxidative systems. The moderate soil drying during grain filling could enhance ABA and reduce ethylene levels; moreover, the increase in the ratio of ABA to ethylene could stimulate the activities of the anti-oxidative systems and the ability of the plant to counteract the free radical in grains, and consequently accelerate the grain filling during the grain filling stage of rice.
Abbreviations
- ABA:
-
Abscisic acid
- AES:
-
Anti-oxidative enzymes system
- ACC:
-
1-Aminocycopropane-1-carboxylic acid
- CAT:
-
Catalase
- MD:
-
Moderate soil-drying
- DPA:
-
Days post anthesis
- O2 − :
-
Superoxide anion
- POD:
-
Peroxidase
- SD:
-
Severe soil-drying
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- WW:
-
Well-watered
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Agarwal S, Sairam RK, Srivastava GC, Meena RC (2005) Changes in antioxidant enzymes activity and oxidative stress by abscisic acid and salicylic acid in wheat genotypes. Biol Plantarum 4:541–550
Ahmadi A, Baker DA (1999) Effects of abscisic acid (ABA) filling period on grain filling processes in wheat. Plant Growth Regul 28:187–197
Beltrano J, Carbone A, Montaldi ER, Guiamet JJ (1994) Ethylene as promoter of wheat grain maturation and ear senescence. Plant Growth Regul 15:107–112
Beltrano J, Ronco MG, Montaldi ER (1999) Drought stress syndrome in wheat is provoked ethylene evolution imbalance and reversed by rewatering, aminoethoxyvinylglycine, or sodium benzoate. Plant Growth Regul 18:59–64
Bollmark M, Kubat B, Eliasson L (1988) Variations in endogenous cytokinin content during adventitious root formation in pea cuttings. J Plant Physiol 132:262–265
Bouman BAM, Tuong TP (2001) Field water management to save water and increase its productivity in irrigated lowland rice. Agr Water Manage 49:11–30
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein using the principal of protein-dye binding. Anal Biochem 72:248–254
Cakmak I, Marschner H (1992) Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol 98:1222–1227
Chen T, Xu Y, Wang J, Wang Z, Yang J, Zhang J (2013) Polyamines and ethylene interact in rice grains in response to soil drying during grain filling. J Exp Bot 64:2523–2538
Cheng CY, Lur HS (1996) Ethylene may be involved in abortion of the maize caryopsis. Physiol Plantarum 98:245–252
Dai Z, Yin Y, Wang Z (2009) Comparison of starch accumulation and enzyme activity in grains of wheat cultivars differing in kernel type. Plant Growth Regul 57:153–162
Davies PJ (2004) The plant hormones: their nature, occurrence and function. In: Davies PJ (ed) Plant Hormones, biosynthesis, signal transduction, action!. Kluwer Academic Publishers, Dordrecht, pp 1–15
Elstner FF, Heupel C (1976) Inhibition of nitrite formation from hydroxylammoniumchloride: a simple assay for superoxide dismutase. Anal Biochem 2:616–620
Fu J, Huang Z, Wang Z, Yang J, Zhang J (2011) Pre-anthesis nonstructural carbohydrate reserve in the stem enhances the sink strength of inferior spikelets during grain filling of rice. Field Crop Res 123:170–182
Giannopolitis GN, Ries SK (1977) Superoxide dismutase I. Occurrence in higher plants. Plant Physiol 59:309–314
Guinn G (1976) Water deficit and ethylene evolution by young cotton bolls. Plant Physiol 57:403–405
He Z (1993) Enzyme linked immunosorbent assay for endogenous plant hormones. In: He Z (ed) Guidance to experiment on chemical control in crop plants. Agricultural University Publishers, Beijing, China, pp 60–68 (in Chinese)
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Huang C, Zhao S, Wang L, Shakeel AA, Chen M, Zhou H, Zou C (2013) Alteration in chlorophyll fluorescence, lipid peroxidation and antioxidant enzymes activities in hybrid ramie (Boehmeria nivea L.) under drought stress. Aust J Crop Sci 5:594–599
Ishimaru T, Ohsugi R, Matsuda T, Yamagishi T (2003) Morphological development of rice caryopses located at the different positions in a panicle from early to middle stage of grain filling. Funct Plant Biol 30:1139–1149
Ishimaru T, Hirose T, Matsuda T, Goto A, Takahashi K, Sasaki H, Terao T, Ishii R, Ohsugi R, Yamagishi T (2005) Expression patterns of genes encoding carbohydrate-metabolizing enzymes and their relationship to grain-filling in rice (Oryza sativa L.): comparison of caryopses located at different positions in a panicle. Plant Cell Physiol 46:620–628
Jiang M, Jing J, Wang S (1991) Effect of osmotic stress on membrane-lipid peroxidation and endogenous protective systems in rice seedlings. Acta Phytophysiol Sinica 1:80–84 (in Chinese)
Jiang D, Cao W, Dai T, Jing Q (2003) Activities of key enzymes for starch synthesis in relation to growth of superior and inferior grains on winter wheat (Triticum aestivum L.) spike. Plant Growth Regul 41:247–257
Kato T, Shinmura D, Taniguchi A (2007) Activities of enzymes for sucrose-starch conversion in developing endosperm of rice and their association with grain filling in extra-heavy panicle types. Plant Prod Sci 10:442–450
Liu S, Chen G, Yin J, Lu C (2011) Response of the flag leaves of a super-hybrid rice variety to drought stress during grain filling period. J Agro Crop Sci 4:322–328
Mohapatra PK, Patel R, Sahu SK (1993) Time of owering affects grain quality and spikelet partitioning with in the rice panicle. Aust J Plant Physiol 20:231–242
Morgan JM (1980) Possible role of abscisic acid in reducing seed set in water-stressed wheat plants. Nature 285:655–657
Murty PSS, Murty KS (1982) Spikelet sterility in relation to nitrogen and carbohydrate contents in rice. Indian J Plant Physiol 25:40–48
Naik PK, Mohapatra PK (1999) Ethylene inhibitors promote male gametophyte survival in rice. Plant Growth Regul 28:29–39
Nakamura Y, Yuki K, Park SY (1989) Carbohydrate metabolism in the developing endosperm of rice grains. Plant Cell Physiol 30:833–839
Nunez M, Mazzafera P, Mazorra LM, Siqueira WJ, Zullo MAT (2003) Influence of a brassinosteroid analogue on antioxidant enzymes in rice grown in culture medium with NaCl. Biol Plant 47:67–70
Ober ES, Sharp RE (1994) Proline accumulation in maize (Zea mays L.) primary roots at low water potentials. I. Requirement for increased levels of abscisic acid. Plant Physiol 105:981–987
Ober ES, Setter TL, Madison JT, Thompson JF, Shapiro PS (1991) Influence of water deficit on maize endosperm development. Enzyme activities and RNA transcripts of starch and zein synthesis, abscisic acid, and cell division. Plant Physiol 97:154–164
Passioura JB (1996) Drought and drought tolerance. Plant Growth Regul 2:79–83
Passioura JB (2007) The drought environment: physical, biological and agricultural perspectives. J Exp Bot 2:113–117
Rahman MS, Yoshida S (1985) Effect of water stress on grain filling in rice. Soil Sci Plant Nutr 31:497–511
Rajinder SD, Pamela P, Trevor AT (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 1:93–101
Richards FJ (1959) A exible growth function for empirical use. J Exp Bot 10:290–300
Saini HS, Westgate ME (2000) Reproductive development in grain crops during drought. Adv Agron 68:59–95
Samejima H, Kondo M, Ito O, Nozoe T, Shinano T, Osaki M (2004) Root-shoot interaction as a limiting factor of biomass productivity in new tropical rice lines. Soil Sci Plant Nutr 50:545–554
Santner A, Estelle M (2009) Recent advances and emerging trends in plant hormone signaling. Nature 459:1071–1078
Sharp RE, LeNoble ME, Else MA, Thorne ET, Gherardi F (2000) Endogenous ABA maintains shoot growth in tomato independently of effects on plant water balance: evidence for an interaction with ethylene. J Exp Bot 51:1575–1584
Sharp RE, Poroyko V, Hejlek LG, Spollen WG, Springer GK, Bohnert HJ, Nguyen HT (2004) Root growth maintenance during water deficits: Physiology to functional genomics. J Exp Bot 55:2343–2351
Tian F, Gong J, Zhang J, Zhang M, Wang G, Li A, Wang W (2013) Enhanced stability of thylakoid membrane proteins and antioxidant competence contribute to drought stress resistance in the tasg1 wheat stay-green mutant. J Exp Bot 6:1509–1520
Wang H, Ma J, Li X, Li Y, Zhang R, Wang R (2007) Effects of water stress on active oxygen generation and protection system in rice during grain filling stage. Sci Agric Sin 7:1379–1387 (in Chinese)
Wang E, Wang J, Zhu X, Hao W, Wang L, Li Q, Zhang L, He W, Lu B, Lin H, Ma H, Zhang G, He Z (2008) Control of rice grain-filling and yield by a gene with a potential signature of domestication. Nat Genet 40:1370–1374
Wardlaw IF, Willenbrink J (2000) Mobilization of fructan reserves and changes in enzymes activities in wheat stems correlate with water stress during kernel filling. New Phytol 148:413–422
Xie Z, Jiang D, Cao W, Dai T, Jing Q (2003) Relationships of endogenous plant hormones to accumulation of grain protein and starch in winter wheat under different post-anthesis soil water statuses. Plant Growth Regul 41:117–127
Yang J, Zhang J (2006) Grain filling of cereals under soil drying. New Phytol 169:223–236
Yang J, Peng S, Visperas RM, Sanico AL, Zhu Q, Gu S (2000a) Grain filling pattern and cytokinin content in the grains and roots of rice plants. Plant Growth Regul 30:261–270
Yang J, Zhang J, Huang Z, Zhu Q, Wang L (2000b) Remobilization of carbon reserves is improved by controlled soil drying during grain filling of wheat. Crop Sci 6:1645–1655
Yang J, Zhang J, Wang Z, Zhu Q, Liu L (2001a) Water deficit-induced senescence and its relationship to remobilization of pre-stored carbon in wheat during grain filling. Agron J 93:196–206
Yang J, Zhang J, Wang Z, Zhu Q, Wang W (2001b) Hormonal changes in the grains of rice subjected to water stress during grain filling. Plant Physiol 127:315–323
Yang J, Zhang J, Liu K, Wang Z, Liu L (2006a) Abscisic acid and ethylene interact in wheat grains in response to soil drying during grain filling. New Phytol 271:293–303
Yang J, Zhang J, Wang Z, Liu K, Wang P (2006b) Post-anthesis development of inferior and superior spikelets in rice in relation to abscisicacid and ethylene. J Exp Bot 57:149–160
Yoshida S (1972) Physiological aspects of grain yield. Annu Rev Plant Physiol 23:437–464
Zhang H, Tan G, Yang L, Yang J, Zhang J, Zhao B (2009) Hormones in the grains and roots in relation to post-anthesis development of inferior and superior spikelets in japonica/indica hybrid rice. Plant Physiol Biochem 47:195–204
Zhang H, Chen T, Wang Z, Yang J, Zhang J (2010) Involvement of cytokinins in the grain filling of rice under alternate wetting and drying irrigation. J Exp Bot 13:3719–3733
Zhang H, Li H, Yuan L, Wang Z, Yang J, Zhang J (2012) Post-anthesis alternate wetting and moderate soil drying enhances activities of key enzymes in sucrose-to-starch conversion in inferior spikelets of rice. J Exp Bot 63:215–227
Zhu Q, Cao X, Luo Y (1988) Growth analysis in the process of grain filling in rice. Acta Agro Sinica 14:182–192 (in Chinese)
Acknowledgments
We are grateful for grants from the National Basic Research Program (2015CB150404, 2012CB114306), the National Natural Science Foundation of China (NSFC-IRRI Joint Research Project 31061140457; General Program 31271641, 31171481; Young Program 31201155), China National Public Welfare Industry (Agriculture) Plan (201203031), the Ph.D. Programs Foundation of Ministry of Education of China (20123250120001), the National Key Technology Support Program of China (2011BAD16B14), the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, H., Liu, K., Wang, Z. et al. Abscisic acid, ethylene and antioxidative systems in rice grains in relation with grain filling subjected to postanthesis soil-drying. Plant Growth Regul 76, 135–146 (2015). https://doi.org/10.1007/s10725-014-9983-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-014-9983-z