Abstract
Fine-leaved Festuca valesiaca Schleich. ex Gaudin (2n = 2x–4x) is native to heavily-grazed, cold, semi-arid, Asian rangelands. However, its potential for low-maintenance turf applications in the semi-arid western United States and its relatedness to other agriculturally important Festuca species have not been investigated. Therefore, a project was designed to identify F. valesiaca accessions that possess horticultural potential when grown under semi-arid growing conditions and to characterize their relatedness to other Festuca species. In 2008, 12 F. valesiaca accessions originating from Kyrgyzstan and eight US. Festuca and one Lolium cultivar were transplanted as replicated, spaced plants to a field nursery at Blue Creek, Utah. Relative vigor, height, width, total biomass (aboveground dry matter yield), seed weight, and seed number were evaluated between 2009 and 2011. Plant height, width, and total biomass of the F. valesiaca accessions examined were approximately equal to the commercial control, ‘Cascade’ (F. rubra L. subsp. commutata Gaudin; 6x; chewings fescue). Plant vigor and seed weight of F. valesiaca accessions PI 659923, PI 659932, W6 30575, and W6 30588 under semi-arid conditions (~300 mm annual precipitation) were significantly (P < 0.05) greater than ‘Cascade’. Moreover, principal component analysis based on all traits as loading factors indicated that these 12 F. valesiaca accessions were distinct from a majority of the other Festuca accessions examined. These F. valesiaca accessions produced abundant amounts of small seed, and this seed yield was significantly correlated with total aboveground biomass (dry weight; r 2 = 0.84, P < 0.001), plant height (r 2 = 0.58, P < 0.05), and plant vigor (r 2 = 0.83, P < 0.001). Amplified fragment length polymorphism (AFLP) analysis (1,454 polymorphic bands) was used to characterize F. valesiaca relatedness to other economically important Festuca species. The AFLP-based, neighbor-joining analysis differentiated F. valesiaca accessions from US Festuca cultivars examined, except for ‘Durar’ (F. ovina L.; 6x; sheep fescue), to which they had strong genetic affinities. Given their morphological attributes, F. valesiaca PI 659923, W6 30575, PI 659932, and W6 30588 should be considered for use in low-maintenance, semi-arid turf improvement programs in the western US.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
About 300 perennial, turfgrass species are members of the genus Festuca L. (Ruemmele et al. 2003). Festuca is a genetically diverse taxon that is comprised of both wide- and narrow-leaved phenotypes that have various commercial uses. There have been several taxonomic classifications of fine-leaved Festuca species that are congruent in many regards (e.g., Hultén and Fries 1986; Tutin et al. 1993; Barkworth et al. 2007). The taxonomic treatment of Barkworth et al. (2007) is used herein because of its historical context to North American flora and its recognition of introduced Festuca species worldwide to the North American continent. Where possible, other species designations are also cited to provide continuity with other taxonomic treatments.
Some Festuca species are used worldwide as turfgrasses {e.g., F. rubra L.; F. ovina L.; F. arundinacea Schreb. [synonym Schedonorus arundinaceus (Schreb.) Dumort.; Soreng et al. 2001]}, and others as roadside plantings [e.g., F. ovina; F. trachyphylla (Hack.) Krajina], and rangeland reclamation [e.g., F. ovina; F. scabrella Torr. ex Hooker (synonym F. altaica Trin.); Pavlick and Looman 1984] (Johnson 2003; Ruemmele et al. 1995). Turfgrass is the most widely planted irrigated crop in the United States (Milesi et al. 2005) and has multi-billion dollar economic impacts (Haydu et al. 2006). Both native and introduced fine-leaved fescues (such as F. rubra, F. ovina, and F. trachyphylla) are important species used in low-maintenance turf applications throughout much of North America, including parts of the western US (Bertin et al. 2009; Ruemmele et al. 2003) because of their drought and shade tolerance (Beard 1997; Ruemmele et al. 1995).
Arndt (2008) has classified diploid F. valesiaca Schleich. ex Gaudin subspecies [subsp. valesiaca (Tzvelev), parviflora (Hack.) Tracey, and hypsophila (St.-Yves) Tzvelev] into a polyploid series [2n = 14 (diploid) − 70 (decaploid)] consisting of at least 20 Festuca species, which are distributed in the sub-continental and continental regions of the meridional and temperate zones in Eurasia, from Germany and France eastwards to northwest China. These and other North American Festuca and closely related species form a polyploid aggregate often called the Ovina Complex with a basic chromosome number of 2n = 2x = 14 (Darbyshire and Warwick 1992; Jones et al. 2008; Ma 2012; Pavlick 1984). The Ovina Complex is composed of F. ovina (sheep fescue; 2x), F. valesiaca, F. filiformis Pourr. [hair fescue; 2x], F. idahoensis Elmer (Idaho fescue; 4x), F. roemeri (Pavlick) E. B. Alexeev (Roemer’s fescue; 4x–6x), F. trachyphylla (hard fescue; 6x), and F. viviparoidea Krajina ex Pavlick subsp. viviparoidea (4x–6x)]. While fine-leaved F. rubra [2n = 42 (hexaploid) or 56 (heptaploid)] is not considered part of the Ovina Complex, morphological comparisons suggest that an Asian fine-leaved fescue, F. valesiaca (Volga fescue; 2x–4x), resembles members of the Ovina Complex (Arndt 2008; Ma 2012; Pils 1984; Pavlick 1984). The distribution of F. ovina (North America, Scotland, Europe, and Asia), F. filiformis (North, Central, and South America, and Western Europe), F. idahoensis (western North America with the exception of Mexico), and F. trachyphylla (North America with the exception of Mexico and Western Europe) is geographically diverse and includes a wide range of growing environments (Jones et al. 2008; Pavlick 1984). Likewise, F. valesiaca has a broad distribution and extends from near sea level in central Europe, up to about 2,000 m in the European Alps and 3,000 m in Xinjiang, Northwestern China (Arndt 2008).
Historically, Festuca germplasm has been collected from their areas of origin to provide needed genetic resources for germplasm preservation and use in plant improvement programs. Several plant exploration expeditions have been made to Eurasia since 1898 by the U.S. Department of Agriculture (USDA; initial collections by Frank N. Meyer and David Fairchild), which collected various Festuca species. Information about these collections can be accessed on the National Plant Germplasm System (NPGS) website (www.ars-grin.gov/npgs). Most recently, collections of F. valesiaca have been made in Kyrgyzstan (2006), Inner Mongolia (2006), and Russia (2010) where annual precipitation ranges from 250 to 1,000 mm and elevation ranges from 132 to 3,100 m above sea level (Johnson 2006, 2010).
Thirty of these F. valesiaca collections had not been evaluated for their horticultural or agronomic potential for low-maintenance applications (www.ars-grin.gov/npgs). Moreover, the genetic relationships among these accessions and other important North American Festuca turfgrass species of the Ovina Complex had not been defined. Adaptations of F. valesiaca to cold and drought conditions on heavily-grazed, barren arid Asian rangelands (Fırıncıoglu et al. 2007, 2009, 2010) may be useful for the development of low-maintenance turfgrasses to increase water conservation in the western US. Therefore, a project was designed to evaluate the phenotypic variation of F. valesiaca accessions under semi-arid conditions of the western US. Great Basin (125–2,032 mm annual precipitation on landscapes ranging from 881 to 2,063 m; Behnke 2011; Comstock and Ehleringer 1992) and to characterize genotypic variation among these accessions and some closely related North American Festuca species. Since some F. valesiaca accessions may possess commercially acceptable turf characteristics for low-maintenance applications (Sărăţeanu and Moisuc 2009), this initial identification and characterization may provide information for their effective use as parental stocks in plant improvement programs.
Materials and methods
Germplasm
Seeds of 12 F. valesiaca (2n = 2x = 14; Ma 2012) accessions (Table 1), chosen based on their native adaptation to overgrazed arid sites from Naryn (41°43′59′′N 75°27′2′′E; W6 30537, PI 659932, PI 659944, W6 30506, W6 30513, W6 30563), Ysyk-Kol (42°10′58′′N 78°0′28′′E; W6 30438, PI 659913, PI 659923), and Chu (42°11′34″N 73°45′22″E; W6 30575, W6 30588, W6 30595) Kyrgyzstan were received from the NPGS (Johnson 2006). All F. valesiaca accessions were diploid, except for W6 30537 and PI 659944, which were tetraploid (Ma 2012). Commercial varieties of several turf species were used as controls for field comparisons. These included ‘Manhattan 4’ [4x; Lolium perenne L.; perennial ryegrass, pasture; Granite Seed Company (GSC), Lehi, Utah], ‘Black sheep’ (6x; F. ovina; sheep fescue, rangeland; GSC), ‘Coronado’ (8x; F. arundinacea; tall fescue, pasture, GSC), ‘Durar’ (6x; F. ovina; sheep fescue, rangeland; GSC), ‘Cascade’ [6x; F. rubra subsp. commutata Gaudin; chewings fescue, turf; US National Turfgrass Evaluation Program (NTEP)], ‘Scaldis’ (6x; F. trachyphylla; hard fescue, turf; NTEP), ‘Shademaster’ (8x; F. rubra L. subsp. rubra; creeping red fescue, turf; NTEP), and ‘Dawson E’ [6x; F. rubra var. littoralis Vasey ex Beal; creeping red fescue, turf; NTEP]. Although ‘Durar’ is sometimes classified under several species names (e.g., F. trachyphylla, F. lemanii, F. ovina, F. brevipila), herein it will be referred to as F. ovina sensu lato (sheep fescue in the broad sense) following Jones et al. (2008) since it is closely related to F. ovina (Ma 2012). Since no commercial varieties of F. valesiaca were available, these other fine-leaved fescue varieties and one ryegrass accession were used for comparison. Given the consistent and exceptional multi-location performance of ‘Cascade’ in the US National Turfgrass Evaluation Program (NTEP; www.ntep.org/contents2.shtml) trial (2004–2010), this turfgrass cultivar was used herein as a standard for comparison. All ploidy levels were confirmed by flow cytometry and/or karyotype analysis (Ma 2012).
The taxa chosen for study were based on morphological and genomic differences. Taxa of the fine-leaved Red Fescue Complex [e.g., F. rubra subspecies arctica (Hack.) Govor., arenaria (Osbeck) F. Aresch., aucta (V. I. Krecz. and Bobrov) Hultén, fallax (Thuill.) Nyman, littoralis, mediana (Pavlick) Pavlick, pruinosa (Hack.) Piper, rubra, secunda (J. Presl) Pavlick, vallicola (Rydb.) Pavlick] are closely related but distinct from those of the Ovina Complex (Catalán et al. 2004; Torrecilla et al. 2004; Jones et al. 2008). Moreover, the fine-leaved fescues, including taxa of the Ovina and Red Fescue Complexes, are genetically distinct from broad-leaved fescues, including tall fescue and other well-known turfgrasses such as perennial ryegrass (Catalán et al. 2004; Gaut et al. 2000; Torrecilla and Catalán 2002; Torrecilla et al. 2004). Based on these relationships, the choice of commercial varieties used allowed for comparisons to define the genetic relationships of the F. valesiaca accessions examined.
Plot establishment and maintenance
In January 2008, seeds of each entry were germinated on blotter paper, and then seedlings were planted in nursery containers (“conetainer”, 164 ml, Stuewe and Sons Inc., Tangent, OR) containing a mixture of 3:1 pumice and peat moss (V/V) in a greenhouse in Logan, UT. Seedlings were grown at 21 °C (daylight conditions)/15 °C (dark conditions) with supplemental light supplied by high-pressure sodium lights [average irradiance = 400 watts (1,800 μmol/m2/s); Sun System III, Sunlight Supply, Inc. Vancouver, WA], at a relative humidity (RH) between 50 and 70 %. Seedlings were fertigated daily with 20 mg/ml of Peters Professional water soluble 20-20-20 fertilizer (NPK) to provide 4,000 ppm N, 1,760 ppm P, and 3,280 ppm K (Scotts Horticultural Company, Marysville, OH).
Seedlings of each accession were transplanted in May 2008 to a field nursery at the Utah State University Blue Creek Experimental Farm in Box Elder County, UT (41°56′3.14′′N 112°26′20.01′′W) approximately 80 km northwest of Logan, UT (elevation = 1,433 m), where the average annual precipitation during the experiment (2009–2011) was about 388 mm (average 20-year precipitation is 307 mm). The soil type was a Parley’s deep silt loam (fine-silty, mixed, mesic, Calcic, Argrixerolls) having a neutral to slightly acidic pH (http://websoilsurvey.nrcs.usda.gov/). Plants were arranged in a randomized complete block design (RCBD) with five plants per plot in six replications spaced at 0.5 m within the rows and 1 m between rows (~20,000 plants/ha) with plants of PI 659984 (F. rubra; Qinghai, China) used as end- and side-borders. Although plants were given water at transplanting, no water or fertilizer was applied during the experiment, and plots were hand-weeded from May to August. Broadleaf weeds were also controlled with herbicide [Mecamine-D; dimethylamine salt of 2,4-D (30.56 %), dimethylamine salt of R-2 propionic acid (8.17 %), and dimethylamine salt of dicamba (2.77 %); EPA Reg. No. 34704-239] application once in April or May of each year at a rate of 4.7 l/ha.
Phenotypic trait evaluation
On April 26, 2010 and June 7, 2011, the relative plant vigor of all accessions in the field plot was assessed using a 0–5 visual rating scale, where plant spring green-up (size, color, and transition from winter to spring growth) was defined as 0 = plant dead, 3 = plants possessing moderate biomass or leaf blade length with green (light to dark) foliage (tussock evident), and 5 = green plants having comparatively abundant above ground biomass and/or long leaf blade length. Because of variation in leaf color among entries, light and dark green were given equal value, and color hue (intensity) was considered in the determination of relative plant vigor. On June 30, 2010, and June 23, 2011, the height (cm) of each plant was measured as the distance from the plant base (soil surface) to the top of the highest panicle at full anthesis. The width (cm) of each plant was measured at the harvest (cutting) height (~10 cm above ground) at the same time. Leaves and seed stalks were harvested at seed maturity and oven dried at 60 °C to estimate dry weight (g). Dried florets of each plant were mechanically threshed to separate seeds and chaff (poorly developed or aborted seeds), and seeds were weighed. One hundred seeds were counted and weighed in at least three replicates for each entry to convert seed weights to an estimate of the number of seeds per plant.
Phenotypic trait analysis
Data for all traits were tested for normality (F test) and homogeneity of variance with the UNIVARIATE procedure of SAS software (version 9.3 for Windows, SAS Institute Inc., Cary, NC, USA). Deviations from normality for data of plant dry matter, seed weight, and seed number were corrected by standard data transformation procedures using square root (dry weight) or natural log functions (seed weight and seed number) to comply with the assumptions necessary for analysis of variance (ANOVA) (Sokal and Rohlf 1995). Nevertheless, data are reported herein in their original scales for biological relevance and interpretation (Draper and Cox 1969). Data distributions for all other traits approached normality and could be used directly for ANOVA. Pearson product-moment correlation was used to measure the strength of linear associations between the traits, a test that assesses a “best fit” line between any two traits (Székely et al. 2007). Thus, tests of putative linear relationships provided assessment of the strength of associations among the traits examined herein.
Morphological trait data (over 3 years) were analyzed as a repeated measure by using a split-plot in time approach, in which year was treated as the whole-plot factor and accession was treated as the split-plot factor in a RCBD. Year and accession main effects, and the year-by-accession interaction effects were tested by using PROC MIXED in SAS software (Oehlert 2000). Accession means were separated using Fisher’s protected least-significant difference (LSD) test by applying the lsmeans statement in SAS. Multivariate principal component analyses (PCA) were performed on lsmean values of three traits in 2009 and six traits in 2010 and 2011 using PROC FACTOR in SAS to clarify accession relationships and to identify those traits that led most to accession discrimination (Kutner et al. 2004). Principal components were visualized using NTsys software (Rohlf 1998), where the least number of principal components that adequately avoided clustering of accessions and explained a high percentage of accession variance (>85 %) were used in visualization of accession relationships (Jolliffe 2005).
DNA extraction and PCR amplification
Leaf samples of each accession were collected, lyophilized, and then ground into fine powder with zinc beads inside extraction tubes by using a Retsch model MM 300 mixer mill (F. Kurt Retsch GmbH and Co., Haan, Germany). Total cellular DNA was extracted using a DNeasy Plant Mini Kit (QIAGEN Inc., Valencia, CA, USA) according to the manufacturer’s instructions, and quantified with a Nanodrop Spectrophotometer (ND-1000, NanoDrop Technologies, Inc., Wilmington, DE, USA). The amplified fragment length polymorphism (AFLP) PCR amplification procedure was performed according to Vos et al. (1995) with fluorescently labeled primers, size fractionating, and fragment size analysis according to Jones et al. (2008). The same E.AC/M.CT primers defined by Jones et al. (2008) were used for the pre-amplification, while the primer combinations of E-ACAC/M-CTAC, E-ACAG/M-CTCA, E-ACCA/M-CTAG, E-ACCA/M-CTTC, E-ACCT/M-CTCT, E-ACTC/M-CTTG, E-ACT/M-CTA, E-ACT/M-CTG, E-ATA/M-CAA, and E-AGG/M-CGC were used for selective amplification. About 4 % of the samples were replicated to identify reproducible marker bands and determine their marker error rates. Only the most distinct (i.e., bright) and reproducible (i.e., consistent during repeated PCR amplicon analysis) bands were used for analysis.
AFLP-based cluster analyses
Data matrices [AFLP band present (1) or absent (0)] were constructed and used to create a rooted neighbor-joining dendrogram with PAUP computer software version 4.0b10 (Swofford 2003). The dendrogram was based on band absence and presence frequency by using the Nei and Li distance method (Nei and Li 1979) which estimates average pair-wise genetic distance (GD) values between each pair of accessions. Internal support for cluster groupings was assessed by using 1,000 bootstrap replications (Felsenstein 1985).
Results
For the phenotypic evaluation, significant (P < 0.001) main effects of year and accession and the interaction of accession × year (3 years) were detected for all traits, except for vigor rating (data not shown; Ma 2012). Therefore, results are presented herein by trait for each year.
Total aboveground dry weight
Average dry weight (DW; aboveground) of all accessions was greater in 2011 than in 2009 or 2010 (Table 1). In 2009, DW ranged from 10.13 (W6 30537) to 241.89 (‘Coronado’) g/plant, with an average of 72.82 g/plant over all accessions. The DW of F. valesiaca accessions averaged 49.96 g/plant, and ranged from 10.13 (W6 30537) to 70.67 (W6 30595) g/plant. Commercial cultivars ‘Coronado’ (tall fescue) and ‘Manhattan’ (perennial ryegrass) produced significantly (P < 0.05) more DW than the fine-leaved commercial control ‘Cascade’ and F. valesiaca accessions, which were similar. In 2010, the DW production of all entries averaged 42.26 g/plant, and ranged from 0.11 (W6 30537) to 117.53 (‘Coronado’) g/plant. The mean DW production of W6 30588 (73.88 g/plant), PI 659923 (64.57), and W6 30575 (62.65) was higher than ‘Cascade’ (36.47), but the differences between ‘Cascade’ and the latter two accessions were not significant. In 2011, the average DW of all entries was 84.60 g/plant, and values ranged from 14.40 (W6 30537) to 154.49 (‘Shademaster’) g/plant. The F. valesiaca accessions produced an average of 85.62 g/plant, with a range from 14.40 (W6 30537) to 126.92 g/plant (PI 659923). Dry weight production in accessions PI 659923 (126.92 g/plant) and W6 30575 (123.11) were higher than ‘Cascade’ (91.23), but not significantly so. The DW mean of the F. valesiaca accessions was lower than the overall mean of the accessions in 2009, but this was not the case in 2010 and 2011.
Height
As already noted for aboveground dry weight, plant height was greatest, on average, in 2011 (Table 1). The average plant height among all accessions in 2010 was 48.4 cm, and values ranged from 8.1 (W6 30537) to 75.8 (‘Coronado’) cm. The height of F. valesiaca accessions was 49.3 cm on average, and ranged from 8.1 (W6 30537) to 58.0 (W6 30575) cm. Although the mean plant height of F. valesiaca accessions W6 30575 (58.0 cm), W6 30506 (56.3 cm), W6 30588 (56.1 cm), and PI 659944 (55.0 cm) were similar to ‘Cascade’ (54.5 cm), they were significantly (P < 0.05) shorter than ‘Coronado’ (75.8 cm). In 2011, the mean plant height of all accessions was 60.8 cm, and values ranged from 46.6 (W6 30537) to 78.8 (‘Durar’) cm. The mean height of F. valesiaca accessions was 59.9 cm, and ranged from 46.6 (W6 30537) to 66.1 (PI 659944) cm.
Width
In 2010, the mean plant width of all accessions was 32.8 cm, and values ranged from 7.7 (W6 30537) to 47.5 (‘Coronado’) cm (Table 1). The mean width of the F. valesiaca accessions was 32.1 cm, and values ranged from 7.7 (W6 30537) to 37.7 (W6 30588) cm. The width of F. valesiaca accessions W6 30588 (37.7 cm), W6 30506 (36.1), PI 659944 (36.6), W6 30563 (35.5), W6 30513 (35.4), PI 659923 (34.5), and W6 30575 (35.7 cm) were similar to ‘Cascade’ (36.6). In 2011, the average plant width of all accessions was 46.6 cm, and values ranged from 27.0 (W6 30537) to 61.3 (‘Shademaster’) cm. The width of ‘Coronado’ (56.1 cm) and ‘Shademaster’ (61.3) was significantly (P < 0.05) greater than ‘Cascade’ (49.3). The mean width of the F. valesiaca accessions was 45.1 cm, ranging from 27.0 (W6 30537) to 52.7 (PI 659944) cm. As a group, the F. valesiaca accessions examined were similar in width to ‘Cascade’.
Vigor rating
The average plant vigor rating among accessions in each year, 2010 and 2011, was 2.1, and values ranged from 0.3 (poor vigor; ‘Manhattan’ in 2010) to 3.5 (most vigorous; ‘Shademaster’ in 2011) (Table 1). While the vigor rating of the F. valesiaca accessions was near 2.2 in both years, values ranged from 0.4 (W6 30537 in 2011) to 3.1 (W6 30588 in 2010). Although the vigor ratings of all F. valesiaca accessions, except PI 659913, W6 30438, W6 30513, and W6 30537, were significantly (P < 0.05) higher than ‘Cascade’ in 2010, the vigor of ‘Cascade’ and all F valesiaca accessions was similar in 2011. In contrast, ‘Durar’ and ‘Shademaster’ were more vigorous than ‘Cascade’ in 2011.
Seed number
Differences in average seed number among all entries from 2009 to 2010 (12,780–10,074 seeds/plant) were not significant, while seed number was significantly (P < 0.001) greater in 2011 (15,676; Table 1). In 2009, the mean number of seeds per plant of all accessions taken collectively was 12,780, and values ranged from 1,254 (W6 30537) to 24,731 (W6 30588) seeds/plant. For F. valesiaca accessions, the average number of seeds per plant was within the same range at 14,800. Festuca valesiaca accessions PI 659913 (21,649 seeds/plant), PI 659923 (19,255), PI 659932 (15,733), W6 30563 (16,372), W6 30575 (22,388), W6 30588 (24,731), and W6 30595 (24,263) produced significantly (P < 0.001) more seeds than did ‘Cascade’ (6,905), but these amounts were similar to those of ‘Coronado’ (18,352). In 2010, the mean seed number of all accessions taken collectively was 10,074 seeds/plant, with a range of 2 (‘Manhattan’) to 33,606 (W6 30588) seeds/plant. The mean seed number of F. valesiaca accessions PI 659913 (18,474 seeds/plant), PI 659923 (27,222), PI 659932 (20,204), W6 30563 (16,744), W6 30575 (16,856), W6 30588 (33,606), and W6 30595 (16,445) was significantly (P < 0.001) higher than those of ‘Cascade’ (3,212) or ‘Coronado’ (6,031). In 2011, the mean seed number of all accessions was 15,676 seeds/plant, and values ranged from 68 (W6 30537) to 47,963 (W6 30595) seeds/plant. For F. valesiaca accessions, the mean seed number was within the same range at 22,208 seeds/plant. The mean seed number of F. valesiaca accessions PI 659913 (28,344 seeds/plant), PI 659932 (32,308), W6 30575 (42,276), W6 30588 (42,539), and W6 30595 (47,963) was significantly (P < 0.001) higher than those of ‘Cascade’ (12,875) or ‘Coronado’ (7,209).
Weight per seed
Based on number of seeds (above) and their weight (three replicates of 100 seeds per entry), F. valesiaca accessions examined possessed abundant amounts of small seeds. While mean seed weight of all entries was similar in 2009 and 2011, production in these years was significantly higher (P < 0.001) than in 2010 (Table 1). For the F. valesiaca accessions, the reduction in 2010 was not as large, and seed production was high in 2011, although this trend was not significantly different from the overall means. In 2009, mean seed weight of all accessions was 10.42 g/plant, and values ranged from 0.79 (W6 30537) to 32.43 (‘Coronado’) g/plant. Mean seed weight of the F. valesiaca accessions was 8.99 g/plant, and values ranged from 0.79 (W6 30537) to 15.34 (W6 30595) g/plant. The mean seed weight of the F. valesiaca accessions PI 659913 (13.49 g/plant), PI 659923 (10.9), PI 659932 (9.84), W6 30563 (8.87), W6 30575 (14.34), W6 30588 (15.10), and W6 30595 (15.34) were significantly greater than ‘Cascade’ (7.87), but significantly lower (P < 0.001) than ‘Coronado’ (32.43) and ‘Manhattan’ (27.48). In 2010, the mean seed weight per plant of the all germplasm examined was 6.12 g/plant, and weights ranged from 0.00 (W6 30537) to 18.38 (W6 30588) g/plant. The mean seed weight of F. valesiaca accessions was 8.28 g/plant, within the same range. The mean seed weight of PI 659913 (10.90 g/plant), PI 659923 (14.68), PI 659932 (10.52), W6 30563 (8.72), W6 30575 (13.40), W6 30588 (18.38), and W6 30595 (7.8) was significantly (P < 0.001) greater than ‘Cascade’ (2.21). In 2011, the mean seed weight of all accessions was 10.06 g/plant, and values ranged from 0.29 (W6 30537) to 22.42 (W6 30595) g/plant, while the F. valesiaca accessions produced an average of 12.22 g of seed per plant within the same range. The mean seed weight per plant of the F. valesiaca accessions PI 659913 (15.19 g/plant), PI 659932 (18.33), W6 30575 (22.38), W6 30588 (21.49), and W6 30595 (22.42) were significantly (P < 0.01) higher than ‘Cascade’ (13.06).
Trait correlations
The F. valesiaca accessions examined possessed abundant amounts of small seed, the weight of which was significantly correlated with the total DW (r 2 = 0.84, P < 0.001), plant height (r 2 = 0.58, P < 0.05), and plant vigor (r 2 = 0.83, P < 0.001) in 2010 (Table 2). Likewise, total DW was significantly correlated with plant height [r 2 = 0.59 (2010) and 0.61 (2011), P < 0.05], vigor rating [r 2 = 0.71 (2010) and 0.88 (2011), P < 0.01], seed number [r 2 = 0.74 (2010), P < 0.001], and width [r 2 = 0.63 (2011), P < 0.05] depending on year.
Principal component analysis
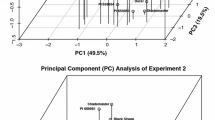
Morphological relationships among 17 accessions as analyzed by PCA are shown in Fig. 1. Trait values for some of the species examined were disproportionately high [‘Manhattan’ (L. perenne), ‘Coronado’ (F. arundinacea)] or low [W6 30537 (F. valesiaca)] when compared to the rest of the accessions (Table 1) and were thus excluded from this analysis. The first two principal components explained large portions of the observed variation (88 %) facilitating a determination of accession relationships (Fig. 1). Commercial cultivars ‘Dawson E’ (F. rubra) and ‘Scaldis’ (F. trachyphylla) and some accessions of F. valesiaca (e.g., PI 659944, W6 30506, W6 30438, and W6 30513) contributed comparatively low values for all traits examined and were positioned in Quadrants I or II. In contrast, commercial cultivars ‘Shademaster’ (F. rubra), ‘Durar’ (F. ovina) and ‘Cascade’ (F. rubra) contributed similar and often comparatively high values for the traits examined and were positioned in Quadrant III. F. valesiaca accessions PI 659923, W6 30563, W6 30575, PI 659932, W6 30595, and W6 30588 were similar in trait values and positioned in Quadrant IV, which was largely due to high seed number and weight values.
Plot of the first two principal components from principal component analysis assessing F. valesiaca accessions and Festuca commercial cultivars (controls) based on three traits in 2009 and six traits in 2010 and 2011 at Blue Creek, UT. Percentage after axis titles indicates the percentage of variation explained for each principal component. The quadrants (I, II, III, IV) illustrate performance over all traits where entries above and to the right of the horizontal and vertical lines, respectively, performed agronomically better than their counterparts
AFLP-based neighbor-joining tree analysis
Ten AFLP selective amplification primer combinations produced 1,530 consistent, bright bands, 95 % (1,454; Ma 2012) of which were polymorphic among the 20 accessions (Table 1) examined. The average number of polymorphic markers recovered per primer combination was 153. A mid-point rooted neighbor-joining tree constructed from GD estimates resulted in the partitioning of all but one (W6 30537, 4x) of the F. valesiaca accessions, and the F. ovina cultivar ‘Durar’, into Cluster 5 and hierarchical Cluster 3 (Fig. 2). Hierarchical Cluster 3, Cluster 4, and Cluster 5 include the taxa F. ovina, F. trachyphylla, and F. valesiaca of the Ovina Complex, and distinguish the fine-leaved fescues from other Festulolium turfgrasses. The Ovina Complex is genetically distinct from Cluster 2 (F. rubra entries) and the F. valesiaca offtype W6 30537. These results indicate that W6 30537 is either misidentified and/or possibly of hybrid origin. As expected, the outgroup taxa, F. arundinacea and L. perenne, were differentiated from all other taxa (Cluster 1).
Rooted neighbor-joining tree illustrating genetic relationships among F. valesiaca accessions (va), commercial cultivars, and outgroups based on Nei and Li’s (1979) GD among AFLP profiles after 1,000 bootstrap permutations (Swofford 2003). Other two-letter species indicators are pe = Lolium perenne, ar = F. arundinacea, ru = F. rubra, tr = F. trachyphylla, and ov = F. ovina. The numbers in parentheses at each cluster indicate bootstrap values after 1,000 permutations
Discussion
Festuca valesiaca is a dominant, erect, sod-forming grass on the intensively-grazed, arid rangeland plateaus of Turkmenistan (1,200–1,900 m) (Habibulla et al. 1999) and Central Anatolian Turkey (1,000–2,000 m) (Fırıncıoglu et al. 2007), and on Asian montane grasslands (e.g., the Emin Valley along the China-Kazakhstan border, 2,000 m) (Taft et al. 2011), which resemble some Great Basin ecosystems of North America (Comstock and Ehleringer 1992). This species is an important, abundant, and persistent rangeland forage grass that shapes vegetation patterns because of its heat and drought tolerance (Fırıncıoglu et al. 2009). We provide herein the first report of the agronomic potential of F. valesiaca in the arid western US, and preliminary evidence of its relationship to North American Festuca species of the Ovina Complex. Substantial genotypic (Fig. 2) and phenotypic (Table 1; Fig. 1) variation exists among the F. valesiaca accessions examined. Our AFLP-based, genetic assessment indicates that these F. valesiaca accessions share considerable genetic affinities with the F. ovina (‘Durar’ and ‘Blacksheep’) and F. trachyphylla (‘Scaldis’) accessions examined. However, given the relatively small number of accessions examined, a broader assessment of these and other Ovina Complex Festuca taxa (e.g., F. filiformis) is needed for a more rigorous determination of such putative relationships.
Plant characteristics associated with plant vigor (e.g., seedling vigor, persistence, sustained green color) and competitiveness (e.g., plant height and width, seed yield) are important prerequisites for turf applications (Ruemmele et al. 2003). For urban uses, plant height and width are important associates of mowing frequency (McKernan et al. 2001) and plant competitiveness (Afolayan 1979; Grime 1977). Although plant height was measured at flowering, which is taller than the typical vegetative growth, the height of F. valesiaca entries was in the main shorter than those of ‘Cascade’, ‘Durar’, and ‘Shademaster’, but generally resembled ‘Black Sheep’ and ‘Scaldis’ in several traits (Table 1). These results suggest that these F. valesiaca accessions (i.e., W6 30438, PI 659923, and PI 659932) may have utility as parental stocks in low-maintenance turf improvement programs. Nevertheless, even though these F. valesiaca accessions are attractive candidates for use in plant improvement, some accessions (e.g., W6 30537) may require further taxonomic clarification because of their genetic dissimilarities to a majority of the accessions examined.
Under minimum management, acceptable aesthetic visual attributes, such as greenness and plant uniformity are essential characteristics of low-maintenance turfgrass (Diesburg et al. 1997; Wang et al. 2011). To the same end, substantial plant vigor in the early spring (i.e., transition from winter dormancy) is also desirable (Bertin et al. 2009). The visual rating (color and aboveground biomass) reported herein indicates that F. valesiaca accessions PI 659923 (2.65), PI 659932 (2.45), W6 30575 (2.65), and W6 30588 (2.95) were significantly (P < 0.001) superior to the commercial control ‘Cascade’ (2.10) during the 2010 and 2011 growing seasons. Moreover, spring vigor ratings were consistent over years, and mean vigor rating (2010 and 2011) was correlated with plant height (P < 0.01; r 2 = 0.64–0.75) and total dry weight (P < 0.01; r 2 = 0.71–0.88) (Table 2).
Traits, such as seed production, biomass production, and plant vigor, in many plants are under quantitative genetic control and can be influenced dramatically by growing environment (Kotecha 1981; van der Heijden and Roulund 2010; Zhang et al. 2006; Brown et al. 2010). Still, such traits can be genetically manipulated during plant improvement (Petr and Frey 1966). In the case of F. valesiaca, the level of phenotypic variation (e.g., seed number; Table 1) suggests that potentially useful cross-progeny might be recovered from matings between F. valesiaca and cross-compatible elite adapted material. However, even if such hybrids were recovered, they would require extensive field evaluation given the large environmental effects on the traits (e.g., seed weight and number, and DW) evaluated (Table 1; large coefficients of variation). Such testing could be facilitated by employing rigorous experimental designs (e.g., lattice) with extensive replication (more than six) and multi-location testing (more than two).
Vigor-related traits, such as leaf color, can also be dramatically influenced by growing environment (Sărăţeanu and Moisuc 2009). The F. valesiaca accessions examined herein possess green to bluish-green coloration and could be selected and developed for specific ornamental and golf course applications after rigorous, mutli-location testing. Because of the moderate to high correlations between seed weight and total aboveground dry weight, plant height, and plant vigor (Table 2; r 2 = 0.58–0.84), it is likely that recurrent selection for these traits among intraspecific F. valesiaca hybrids could result in vigorous, green, tall-statured progeny with abundant seed yield (Fang et al. 2004; Griffiths 1965). This is important given the commercial importance placed on both vegetative (e.g., plant color and size) and seed production (e.g., seed number and weight) characteristics in grasses (Fang et al. 2004).
Under low-precipitation conditions in the Great Basin, the performance of the Kyrgyz F. valesiaca accessions PI 659923 (Naryn), PI 659932 (Naryn), W6 30575 (Chu), and W6 30588 (Chu) for plant vigor [i.e., sustained plant growth (biomass) and green color throughout the growing season] and comparatively high seed production was consistent over 2 years of evaluation. These attributes are important for low-maintenance turfgrass applications, including urban landscapes, golf course fairway fringes/roughs, roadsides, and degraded landscape restoration.
References
Afolayan TA (1979) Change in percentage ground cover of perennial grasses under different burning regimes. Vegetation 39:35–41
Arndt S (2008) Novelties in the Festuca valesiaca group (Poaceae) from the central Alps. Plant Syst Evol 271:129–142
Barkworth ME, Capels KM, Long S, Andeton LK, Piep MB (eds) (2007) Flora of North America. Oxford University Press, New York
Beard JB (1997) Shade stresses and adaptation mechanisms of turf grasses. Intl Turfgrass Soc Res J 8:1186–1195
Behnke RJ (2011) The role of elevation on temperature trends in the western United States: a comparison of two statistical methods. Ph.D. Dissertation of atmospheric and oceanic science, University of Wisconsin, Madison, WI
Bertin C, Senesac AF, Rossi FS, Ditommaso A, Weston LA (2009) Evaluation of selected fine-leaf fescue cultivars for their turfgrass quality and weed suppressive ability in field settings. HortTechnology 19:660–668
Brown RN, Barker RE, Warnke SE, Cooper LD, Brilman LA, Mian MAR, Jung G, Sim SC (2010) Identification of quantitative trait loci for seed traits and floral morphology in a field-grown Lolium perenne x Lolium multiflorum mapping population. Plant Breed 129:29–34
Catalán P, Torrecilla P, López-Rodríguez J-A, Olmstead RG (2004) Phylogeny of the festucoid grasses of subtribe Loliinae and allies (Poeae, Pooideae) inferred from ITS and trnL-F sequences. Mol Phylogenet Evol 31:517–541
Comstock JP, Ehleringer JR (1992) Plant adaptation in the Great Basin and Colorado plateau. Great Basin Nat 52:195–215
Darbyshire SJ, Warwick SI (1992) Phylogeny of North American Festuca (Poaceae) and related genera using chloroplast DNA restriction site variation. Can J Bot 70:2415–2429
Diesburg KL, Christians NE, Moore R, Branham B, Banneberger TK, Reicher ZJ, Voight T, Minner D, Newman R (1997) Species for low-input sustainable turf in the US. Up Midwest Agron J 89:690–694
Draper NR, Cox DR (1969) On distributions and their transformation to normality. J R Stat Soc Series B (Methodological), 31:472–476
Fang C, Aamlid TS, Jorgensen O, Rognli OA (2004) Phenotypic and genotypic variation in seed production traits within a full-sib family of meadow fescue. Plant Breed 123:241–246
Felsenstein J (1985) Confidence-limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Fırıncıoglu HK, Seefeldt SS, Sahin B (2007) The effects of long-term grazing enclosures on range plants in the Central Anatolian Region of Turkey. Environ Manag 39:326–337
Fırıncıoglu HK, Seefeldt SS, Sahin B, Vural M (2009) Assessment of grazing effect on sheep fescue (Festuca valesiaca) dominated steppe rangelands, in the semi-arid Central Anatolian region of Turkey. J Arid Environ 73:1149–1157
Fırıncıoglu HK, Adıguzel N, Bani B, Sahin B (2010) Assessment of grazing effect on two sub-shrubs (Astragalus schottianus and Thymus sipyleus) in dominated mountain Bozoglan Grasslands in the semi-arid Central-Southern Anatolian Region of Turkey. Arid Land Res Manag 24:282–300
Gaut BS, Tredway LP, Kubik C, Gaut RL, Meyer W (2000) Phylogenetic relationships and genetic diversity among members of the Festuca-Lolium complex (Poaceae) based on ITS sequence data. Plant Syst Evol 224:33–53
Griffiths DJ (1965) Breeding for higher seed yields from herbage varieties. J Nat Inst Agric Bot 10:320–331
Grime JP (1977) Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am Nat 111:1169–1194
Habibulla IA, Fet NG, Fet V, Valdez R, Feldman RW (1999) Biodiversity, genetic diversity, and protected areas in Turkmenistan. J Sustain For 9:73–88
Haydu JJ, Hodges AW, Hall CR (2006) Economic impacts of the turfgrass and lawncare industry in the United States. University of Florida Extension publication FE632, Gainesville, FL. [online: http://edis.ifas.ufl.edu/pdffiles/FE/FE63200.pdf] Accessed May 2013
Hultén E, Fries M (eds) (1986) Atlas of North European vascular plants (North of the Tropic of Cancer) Vols. I-III. Koeltz Scientific Books, Königstein
Johnson PG (2003) Mixtures of buffalograss and fine fesuce or streambank wheatgrass as a low-maintenance turf. HortScience 38:1214–1217
Johnson DA (2006) Plant Germplasm Collection Report. Collection of low-maintenance turf germplasm in Kyrgystan and Inner Mongolia. United States Department of Agriculture, Agricultural Research Service, Forage and Range Research Laboratory, Logan, UT. [http://www.ars.usda.gov/SP2UserFiles/Place/54281000/kyrgyzstan06.pdf (or china06.pdf)] Accessed May 2013
Johnson DA (2010) Plant Germplasm Collection Report. Collection of reduced-input turf and forage germplasm in the northern Caucasus mountains of Russia. United States Department of Agriculture, Agricultural Research Service, Forage and Range Research Laboratory, Logan, UT. (http://www.ars.usda.gov/SP2UserFiles/Place/54281000/russia10.pdf) Accessed May 2013
Jolliffe IT (2005) Principal component analysis. Wiley, New York
Jones TA, Larson SR, Wilson BL (2008) Genetic differentiation and admixture among Festuca idahoensis, F. roemeri, and F. ovina detected in AFLP, ITS, and chloroplast DNA. Botany 86:422–434
Kotecha A (1981) Inheritance of seed yield and its components in safflower. Can J Gen Cytol 23:111–117
Kutner MH, Nachtsheim CJ, Neter J (2004) Applied linear regression models, 4th edn. McGraw-Hill/Irwin, NewYork, pp 176–212
Ma YM (2012) Genetic characterization of fine-leaved Festuca valesiaca germplasm and evaluation of their relationship to the F. ovina complex. Master Thesis, Utah State University, Logan, UT. Thesis No. 1352. http://digitalcommons.usu.edu/etd/1352 Accessed May 2013
McKernan DK, Ross JB, Tompkins DK (2001) Evaluation of grasses grown under low maintenance conditions. Int Turfgrass Soc Res J 9:25–33
Milesi C, Running SW, Elvidge CD, Dietz JB, Tuttle BT, Nemani RR (2005) Mapping and modeling the biogeochemical cycling of turf grasses in the United States. Environ Manag 36:426–438
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76:5269–5273
Oehlert GW (2000) A first course in design and analysis of experiments. W. H. Freeman, New York
Pavlick LE (1984) Studies on the Festuca ovina complex in the Canadian Cordillera. Can J Bot 62:2448–2462
Pavlick LE, Looman J (1984) Taxonomy and nomenclature of rough fescues, Festuca altaica, F. campestris (F. scabrella var. major), and F. hallii, in Canada and the adjacent part of United States. Can J Bot 62:1739–1749
Petr FC, Frey KJ (1966) Genotypic correlations, dominance, and heritability of quantitative characters in oats. Crop Sci 6:259–262
Pils G (1984) Systematik, Karyologie und Verbreitung der Festuca valesiaca-Gruppe (Poaceae) in Österreich und Südtirol. Phyton Ann Rei Bot 24:35–77
Rohlf FJ (1998) NTSYS-Pc v2.0 numerical taxonomy and multivariable analysis system. Applied Biostatistics, New York
Ruemmele BA, Brilman LA, Huff DR (1995) Fine fescue germplasm diversity and vulnerability. Crop Sci 35:313–316
Ruemmele BA, Wipff J, Brilman L, Hignight K (2003) Fine-leaved Festuca species. In: Casler MD, Duncan RR (eds) Turf-grass biology, genetics, and breeding. Wiley, Hoboken, NJ, pp 129–174
Sărăţeanu V, Moisuc A (2009) Perennial grasses spontaneous in the grasslands from Western Romania with potential for their use for turf research. Res J Agric Sci 41:197–200
Sokal RR, Rohlf FJ (1995) Biometry: The principles and practice of statistics in biological research, 3rd edn. W. H. Freeman, New York
Soreng RJ, Terrell EE, Wiersema J, Darbyshire SJ (2001) (1488) Proposal to conserve the name Schedonorus arundinaceus (Schreb.) Dumort. against Schedonorus arundinaceus Roem. et Schult. (Poaceae: Poeae. Taxon 50:915–917
Swofford DL (2003) PAUP*: phylogenetic analysis using parsimony (and other methods). Version 4.0. Sinauer Associates, Sunderland, Massachusetts (prerelease test version)
Székely GJ, Rizzo ML, Bakirov NK (2007) Measuring and testing independence by correlation of distances. Annal Stat 35:2769–2794
Taft JB, Phillippe LR, Dietrich CH, Robertson KR (2011) Grassland composition, structure, and diversity patterns along major environmental gradients in the Central Tien Shan. Plant Ecol 212:1349–1361
Torrecilla P, Catalán P (2002) Phylogeny of broad-leaved and fine-leaved Festuca lineages (Poaceae) based on nuclear ITS sequences. Syst Bot 27:241–251
Torrecilla P, López-Rodríguez J-A, Catalán P (2004) Phylogenetic relationships of Vulpia and related genera (Poeae, Poaceae) based on analysis of ITS and trnL-F sequences. Ann Missouri Bot Gard 91:124–158
Tutin TG, Heywood VH, Burges NA, Moore DM, Valentine DH, Walters SM, Webb DA (eds) (1993) Flora Europaea, 2nd edn. Cambridge University Press, Cambridge
van der Heijden SA, Roulund N (2010) Genetic gain in agronomic value of forage crops and turf: a review. In: Huyghe C (ed) Sustainable use of genetic diversity in forage and turf breeding. Springer, The Netherlands pp 247–260
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23:4407–4414
Wang S, Zhang Q, Watkins E (2011) Evaluation of salinity tolerance of prairie junegrass: a potential low-maintenance turfgrass species. HortScience 46:1038–1043
Zhang Y, Mian MAR, Bouton JH (2006) Recent molecular and genomic studies on stress tolerance of forage and turf grasses. Crop Sci 46:497–511
Acknowledgments
Financial support for a graduate assistantship was given through the US. Federal Joint Research and Extension Programs in China (Grant No. 2009-34613-20214) administered by Utah State University, and the U.S. Department of Agriculture, Agricultural Research Service, Forage and Range Research Laboratory gave research support.
Author information
Authors and Affiliations
Corresponding author
Additional information
The cost of publishing this paper was defrayed in part by the payment of page charges. Under postal regulations, this paper therefore must be hereby marked advertisement solely to indicate this fact. Mention of a trade name, proprietary product, or specific equipment does not constitute a guarantee or warranty by the U.S. Department of Agriculture and does not imply its approval to exclusion of other products that may be suitable.
Rights and permissions
About this article
Cite this article
Ma, Y., Staub, J.E., Robbins, M.D. et al. Phenotypic and genetic characterization of Kyrgyz fine-leaved Festuca valesiaca germplasm for use in semi-arid, low-maintenance turf applications. Genet Resour Crop Evol 61, 185–197 (2014). https://doi.org/10.1007/s10722-013-0025-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-013-0025-x