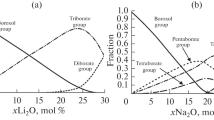
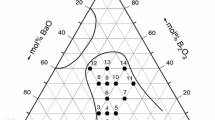
Abstract
The dependences of the dilatometric properties on the hypothetical structure for a number of borate, aluminoborate, and lead-containing glasses are analyzed over a wide range of compositions. Special attention is focused on the specific features observed in the behavior of the structural temperature coefficient of linear expansion. It is demonstrated that the revealed regularities allow the justified inferences regarding the structural transformations of glasses with variations in their composition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
Lindig, O., Die Zahigkeit von Glasern bei Transformationstemperatur, in Beitrage zur angewandte Glasforschung, Stuttgart, 1959, pp. 209–220.
Mazurin, O.V., Steklovanie (Glass Transition), Leningrad: Nauka, 1986.
Mazurin, O.V., Mazurina, E.K., Klyuev, V.P., and Dorofeeva, N.P., Specific Features of Composition Dependences of Properties for Silicate Glasses in the Range of High Concentrations of Modifier Oxides, Fiz. Khim. Stekla, 1977, vol. 3, no.3, pp. 261–265.
Klyuev, V.P. and Bulaeva, A.V., Viscosity and Thermal Expansion of Lead Borate Glasses in the Glass Transition Range, Fiz. Khim. Stekla, 1980, vol. 6, no.6, pp. 674–678.
Klyuev, V.P. and Bulaeva, A.V., Viscosity and Thermal Expansion of Lead Silicate Glasses in the Glass Transition Range, Fiz. Khim. Stekla, 1983, vol. 9, no.5, pp. 635–638.
Klyuev, V.P. and Pevzner, B.Z., Structural Interpretation of the Glass Transition Temperature and Thermal Expansion of Glasses in the System BaO-Al2O3-B2O3, Phys. Chem. Glasses, 2000, vol. 41, no.6, pp. 380–383.
Mazurin, O.V., Otzhig spaev stekla s metallom (Annealing of Glass-Metal Seals), Leningrad: Energiya, 1980.
Klyuev, V.P. and Pevzner, B.Z., Glass Transition Temperature, Instantaneous and Structural Thermal Expansion in the Systems R 2O-Al2O3-B2O3 (R = Li, Na) and RO-Al2O3-B2O3 (R = Ca, Ba), Phys. Chem. Glasses, 2003, vol. 44, no.2, pp. 121–124.
Mazurin, O.V. and Klyuev, V.P., Dilatometric Investigation into the Structural Hysteresis in Some Multicomponent Glasses, Fiz. Khim. Stekla, 1975, vol. 1, no.3, pp. 245–251.
Bray, P.J. and O'Keefe, J.G., Nuclear Magnetic Resonance Investigations of the Structure of Alkali Borate Glasses, Phys. Chem. Glasses, 1963, vol. 4, no.2, pp. 37–46.
Bray, P.J., Leventhal, M., and Hooper, H.O., Nuclear Magnetic Resonance Investigations of the Structure of Lead Borate Glasses, Phys. Chem. Glasses, 1963, vol. 4, no.2, pp. 47–66.
Greenblatt, S. and Bray, P.J., Nuclear Magnetic Resonance Investigations of the System BaO-B2O3, Phys. Chem. Glasses, 1967, vol. 8, no.5, pp. 190–193.
Bishop, S.G. and Bray, P.J., Nuclear Magnetic Resonance of Calcium Boroaluminate Glasses, Phys. Chem. Glasses, 1966, vol. 7, no.3, pp. 73–81.
Klyuev, V.P. and Pevzner, B.Z., Influence of Aluminum and Gallium Oxides on the Thermal Expansion and Viscosity of Barium Borate Glasses, Fiz. Khim. Stekla, 1998, vol. 24, no.4, pp. 532–545 [Glass Phys. Chem. (Engl. transl.), 1998, vol. 24, no. 4, pp. 372–381].
Klyuev, V.P. and Pevzner, B.Z., Composition Dependence of Thermal Expansion and Glass Transition Temperature for Barium Aluminoborate Glasses, Fiz. Khim. Stekla, 2001, vol. 27, no.1, pp. 79–90 [Glass Phys. Chem. (Engl. transl.), 2001, vol. 27, no. 1, pp. 54–61].
Klyuev, V.P. and Pevzner, B.Z., The Influence of Aluminum Oxide on the Thermal Expansion, Glass Transition Temperature, and Viscosity of Lithium and Sodium Aluminoborate Glasses, Fiz. Khim. Stekla, 2002, vol. 28, no.4, pp. 295–314 [Glass Phys. Chem. (Engl. transl.), 2002, vol. 28, no. 4, pp. 207–220].
Klyuev, V.P. and Pevzner, B.Z., Thermal Expansion and Glass Transition Temperature of Calcium Borate and Calcium Aluminoborate Glasses, Fiz. Khim. Stekla, 2003, vol. 29, no.2, pp. 191–204 [Glass Phys. Chem. (Engl. transl.), 2003, vol. 29, no. 2, pp. 127–136].
Park, M.J. and Bray, P.J., B11 NMR Studies of Strontium Borate Glasses and Compounds, Phys. Chem. Glasses, 1972, vol. 13, no.2, pp. 50–62.
Yiannopoulos, Y.D., Chryssikos, G.D., and Kamitsos, E.I., Structure and Properties of Alkaline Earth Borate Glasses, Phys. Chem. Glassed, 2001, vol. 42, no.3, pp. 164–172.
Zhong, J. and Bray, P.J., Change in Boron Coordination in Alkali Borate Glasses, and Mixed Alkali Effects, as Elucidated by NMR, J. Non-Cryst. Solids, 1989, vol. 111, pp. 67–76.
Clarida, W.J., Berryman, J.R., Affatigato, M., Feller, S.A., Kroeker, S.C., Ash, J., Zwanziger, J.W., Meyer, B., Borsa, F., and Martin, S.W., Dependence of N 4 upon Alkali Modifier in Binary Borate Glasses, Phys. Chem. Glasses, 2003, vol. 44, no.3, pp. 215–217.
Griscom, D.L., Borate Glass Structure, in Borate Glasses: Structure, Properties, Applications, Pye, L.D., Frechette, V.D., and Kreidl, N.J., Eds., N.Y.: London: Plenum, 1978, pp. 11–50.
Dietzel, A., Zusammenhang zwischen Ausdehnungkoeffizient und Struktur von Glasern, Glastech. Ber., 1941, vol. 19, no.10, pp. 319–325.
Appen, A.A., Khimiya stekla (Chemistry of Glass), Leningrad: Khimiya, 1970.
Takaishi, T., Jisun, Jin., Takahashi, M., Uchino, T., and Yoko, T., The Local Structure of Pb2+ in Borate and Silicate Glasses, Proceedings of the International Congress on Glass, Edinburg, 2001, vol. 2, pp. 10–11.
Nemilov, S.V., Viscosity and Structure of Binary Germanate Glasses in the Range of Softening Temperatures, Zh. Prikl. Khim. (Leningrad), 1970, vol. 43, no.12, pp. 2602–2610.
Ivanov, A.O. and Evstrop'ev, K.S., On the Problem of Structure of the Simplest Germanate Glasses, Dokl. Akad. Nauk SSSR, 1962, vol. 145, no.4, pp. 797–800.
Mazurin, O.V. and Klyuev, V.P., Structural Temperature Coefficients of Density, Enthalpy, and Viscosity of Lead Borate and Lead Silicate Melts in the Glass Transition Range, Fiz. Khim. Stekla, 1983, vol. 9, no.5, pp. 600–607.
Author information
Authors and Affiliations
Additional information
Original Russian Text Copyright © 2005 by Fizika i Khimiya Stekla, Klyuev.
Rights and permissions
About this article
Cite this article
Klyuev, V.P. Dependence of the Dilatometric Properties of Glasses on Their Structure: I. Borate, Aluminoborate, and Lead-Containing Glasses. Glass Phys Chem 31, 749–759 (2005). https://doi.org/10.1007/s10720-005-0123-8
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10720-005-0123-8