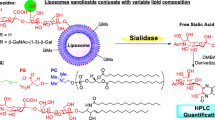
Abstract
GM3 ganglioside interacts specifically with complex-type N-linked glycans having multivalent GlcNAc termini, as shown for (1) and (2) below. (1) Oligosaccharides (OS) isolated from ConA-non-binding N-linked glycans of ovalbumin, whose structures were identified as penta-antennary complex-type with bisecting GlcNAc, having five or six GlcNAc termini (OS B1, B2), or bi-antennary complex-type having two GlcNAc termini (OS I). OS I is a structure not previously described. (2) Multi-antennary complex-type N-linked OS isolated from fetuin, treated by sialidase followed by β-galactosidase, having three or four GlcNAc termini exposed. These OS, conjugated to phosphatidylethanolamine (PE), showed clear interaction with 3H-labeled liposomes containing GM3, when various doses of OS-PE conjugate were adhered by drying to multi-well polystyrene plates. Interaction was clearly observed only with liposomes containing GM3, but not LacCer, Gb4, or GalNAcα1-3Gb4 (Forssman antigen). GM3 interaction with PE conjugate of OS B1 or B2 was stronger than that with PE conjugate of OS I. GM3 interacted clearly with PE conjugate of N-linked OS from desialylated and degalactosylated fetuin, but not native fetuin. No binding was observed to cellobiose-PE conjugate, or to OS-PE conjugate lacking GlcNAc terminus. Thus, GM3, but not other GSL liposomes, interacts with various N-linked OS having multiple GlcNAc termini, in general. These findings suggest that the concept of carbohydrate-to-carbohydrate interaction can be extended to interaction of specific types of N-linked glycans with specific GSLs. Natural occurrence of such interaction to define cell biological phenomena is under investigation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Cell recognition/adhesion based on carbohydrate-to-carbohydrate interaction (CCI) has been studied with two types of glycoconjugate system. One system is based on glycosphingolipid (GSL)-to-GSL interaction in which the same type or different type of cell displays adhesion through the same type (“homotypic interaction”) [1] or different type (“heterotypic interaction”) [2] of GSL. The other system is based on specific, multivalent glycosyl epitopes organized in proteoglycans of sponge cells, which provide the basis for species-specific cell aggregation [3–5]. These two CCI systems were initially studied for self-recognition of (1) Lex by Lex, assumed to occur in autoaggregation of teratocarcinoma F9 cells, as a model of the “compaction process” of preimplantation embryo [1], and (2) GlcNAcβ1-3Fucα1-O-Ser (with or without 3-O-sulfate at terminal GlcNAc) by the same structure [6], as a model of species-specific sponge cell autoaggregation [3–5]. Processes (1) and (2) are observed in the presence of bivalent cation (Ca2+).
Examples of heterotypic interaction between cells expressing two different types of GSLs are: (1) mouse melanoma B16 (high GM3 expressor) with natural killer (NK)-susceptible mouse lymphoma (high Gg3 expressor) [2, 7], (2) melanoma B16 cells (GM3 expressor) with endothelial cells (LacCer expressor) [8, 9], and (3) blastocyst (Ley expressor) with endometrium (H expressor) during the implantation process [10]. Results of these early CCI studies, showing either homotypic or heterotypic interaction, were supported and strengthened by more recent studies with quantitative analysis of CCI using various physical or biophysical methods such as surface plasmon resonance spectroscopy (SPR), atomic force microscopy (AFM), nuclear magnetic resonance spectroscopy, and electrospray mass spectrometry (see Discussion).
In the early studies, GSLs were used for experimental convenience, because they form microdomains by clustering. However, in reality, the process of cell-to-cell interaction may also occur through CCI between glycosyl epitopes in glycoprotein and those in GSLs. To address this possibility, we studied interaction of N-linked glycans with a few types of GSLs. Results reported here indicate that GM3, but not other GSLs, interacts with N-linked glycans having multivalent GlcNAc termini, such as those with 5 or 6 GlcNAc termini [11]. The interaction was lower with a novel structure having 2 GlcNAc termini.
2 Materials and methods
2.1 Materials and reagents
2.1.1 Glycoproteins, GSLs, and other lipids
Chicken ovalbumin (grade V), fetuin from fetal calf serum,1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine (PE), 1,2-dimyristoyl-sn-glycero-3-phosphocholine (PC), cholesterol, and cellobiose (CB) were purchased from Sigma Chemical Co. GM3 was prepared from dog erythrocytes. Forssman GSL antigen was prepared from sheep or goat erythrocytes. Globoside (Gb4) was prepared from human erythrocytes. Gg3 was prepared from guinea pig erythrocytes. LacCer was from Matreya (Pleasant Gap, PA).
2.1.2 Antibodies
mAb directed to GlcNAc termini of glycoproteins or GSLs was prepared from hybridoma J1 [12]. mAb DH2 directed to GM3 [13], and mAb 1D4 directed to Gg3 [14] were prepared in our lab. Anti-mouse IgM-HRP was from Southern Biotech (Birmingham, AL).
2.1.3 Other reagents
Anhydrous hydrazine, sialidase from Clostridium perfringens, concanavalin A conjugated to Sepharose 4B from Canavalia ensiformis (jackbean) (15-mg lectin per ml packed gel), cellulose powder (20-μm mesh size), and tetrabutyl ammonium (TBA) cyanoborohydride were from Sigma. [3H]cholesterol (0.25 mCi/0.25 ml) was from PerkinElmer. Float-A-Lyzer (MWCO 500) and dialysis membrane (MWCO 12,000–14,000) were from Spectrum Labs (Rancho Dominguez, CA). Solvents (HPLC grade) and Rexyn I-300 mixture resin were from Fisher. Distilled water was obtained from a Milli-Q purification system (Millipore, Bedford, MA).
2.2 Oligosaccharides (OS) of N-linked glycans from fetuin and ovalbumin
Fetuin (1 g) or ovalbumin (6 g), dissolved in water, were respectively dialyzed in distilled water for 3 days at 4°C, lyophilized, and subjected to hydrazinolysis as described previously [15]. Hydrazinolysates were N-acetylated and subjected to separation of OS by cellulose-column chromatography [16]. Cellulose-column-bound fraction from 1-g fetuin was 62.8 mg (6.3% yield), and was subjected directly to PE conjugation (see below). Cellulose-column-bound fraction from 6-g ovalbumin was 664 mg (11.1% yield), and was subjected to ConA-chromatography, whereby ConA-non-binding fraction (containing complex-type and hybrid-type OS) was separated from ConA-binding fraction (high-mannose OS) (see below).
2.3 Separation of ConA-non-binding OS from ovalbumin by ConA-Sepharose 4B chromatography
Concanavalin A (ConA) lectin affinity chromatography was performed as described previously [17, 18]. Adaptation of the method for the present study is as follows. ConA-Sepharose 4B column (2.5 × 25, Vb = 10 ml) was equilibrated with buffer containing 10-mM Tris–HCl, 150-mM NaCl, 1-mM CaCl2, and 1-mM MnCl2, pH 7.4 (ConA buffer). OS prepared by cellulose chromatography (664 mg) was dissolved in ConA buffer and applied to the column. Column unbound effluent, containing various salts as in ConA buffer, was evaporated under rotary vacuum and desalted by mixture of anionic and cationic resin (Rexyn I-300, H-OH, Fisher) and limiting dialysis through Float-A-Lyzer dialysis membrane (molecular weight cut-off 500, Spectrum Labs) for 3 days in distilled water at 4°C. The non-dialyzable OS portion was centrifuged (3,000 rpm, 15 min), and the supernatant was lyophilized. The ConA-non-binding fraction was 65.7 mg (9.9% yield).
2.4 Further fractionation of ConA-non-binding OS by high-performance liquid chromatography (HPLC)
ConA-non-binding OS (65.7 mg), after desalting (see above), was further separated by HPLC through normal-phase Microsorb 100 NH2 column (Varian, Lake Forest, CA; 0.46 × 25 cm) using a Varian ProStar chromatography system. Separations were performed at room temperature. Mobile phase consisted of Sol A (90%, v/v, 0.2 M acetic acid/ triethylamine, pH 7.2: acetonitrile = 3:7) and Sol B (10%, v/v, 0.2-M acetic acid/ triethylamine, pH 7.2: acetonitrile = 7:3) to start, followed by a linear gradient from 90 to 80% over 50 min. During the next 10 min, the percentage of Sol B was maintained at 100%. The flow rate was 1 ml/min, fraction size was 1 ml, and the effluent was monitored using a Waters 484 tuneable absorbance UV detector at λ = 215 nm. Yields of major homogeneous OS components thus separated by HPLC were: OS I 3.9 mg, OS VI 7.6 mg (11.6%), OS B 17.4 mg (26.5%). These OS components were subjected to structural analysis (see below).
2.5 Structural determination of OS from ovalbumin
The OS separated by HPLC as above were reductively aminated with 2-aminopyridine and sodium cyanoborohydride [19]. Pyridylaminated (PA)-OS were purified by gel filtration on Sephadex G-15 column, and their structures were analyzed using the HPLC mapping method. This powerful method identifies OS structures based on elution times on two different HPLC columns: a Shim-pack HRC-octadecyl silica (ODS) column (Shimadzu, Kyoto, Japan), and a TSK-gel Amide-80 column (Tosoh, Tokyo, Japan). All experimental procedures used, including chromatographic conditions, have been described previously [20–22]. The PA-OS were identified by comparison with HPLC data of approximately 500 reference PA-OS in a home-made web application, GALAXY (http://www.glycoanalysis.info/) [23]. The PA-glycans were confirmed by co-chromatography with reference to PA-glycans on the columns, and by matrix-assisted laser desorption/ionization time-of-flight mass spectrometric (MALDI-TOF-MS) analyses [24]. MALDI-TOF-MS data were acquired in positive and negative modes using AXIMA-CFR (Shimadzu) operated in linear mode. In preliminary analysis of OS Fr. 1 as PA derivative, the structure appeared to be novel, since the profile of OS Fr. 1-PA did not match the position in GALAXY. Therefore, OS Fr. 1 was β-galactosylated by galactosyltransferase, and the product was analyzed by MALDI-TOF-MS. Galactosylation of OS Fr. 1-PA was carried out in 20 μl of 50-mM Tris–HCl buffer, pH 7.4, containing 10-mM MnCl2, 50 milliU of β1,4-galactosyltransferase (Sigma), with incubation in the presence of 4-mM UDP-Gal overnight at 37°C.
2.6 Conjugation of N-linked OS from fetuin and ovalbumin to PE
Various OS fractions were conjugated to PE [25–27]. PE conjugation was slightly modified for performance under argon atmosphere as follows. Briefly, 1-mg aliquot of OS in each vial was completely dried in a desiccator overnight under vacuum, and dissolved in 100-μl distilled water. After addition of 50-μl boric acid (2 M, in DMSO), 1 ml of PE (4 mg/ml, v/v, in chloroform: ethanol 2:1) and 50 μl tetrabutylammonium-cyanoborohydride (423 mg/ml, in chloroform), the atmosphere of the vial was exchanged with argon gas, the vial was tightly sealed with teflon-lined cap, and heated at 70°C for about 1 week.
2.7 Purification of OS-PE conjugate
2.7.1 Preliminary separation by C18-cartridge
The reaction mixture as above was dried under N2, dissolved in methanol: water 6:4 and applied to a small pre-packed C18-alkylated silica gel (“C18-cartridge”) (Varian) equilibrated with methanol:water 6:4. The column was washed with ten column volumes methanol:water 6:4 to eliminate all water-soluble contaminants. The adsorbed OS-PE conjugate was eluted with chloroform/methanol/water (C/M/W) 2:1:0.1. The PE conjugate and non-reacted PE were eluted from C18 cartridge. OS-PE eluate from the cartridge was dried and dissolved in isopropanol/hexane/water 55:40:5. After centrifugation at 13,000 rpm for 5 min, the pellet, dissolved in C/M/W 25:25:8, showed OS-PE as the major component in preliminary TLC.
2.7.2 Purification of OS-PE conjugate by HPLC with Iatrobeads chromatography
Further purification of OS-PE was performed by HPLC with Iatrobeads 6RS-8010 column (Column Resolution, San Jose, CA, 0.46 × 60 cm) using a Varian ProStar chromatography system. Separations were performed at room temperature. HPLC was initially started with isocratic 100% Sol A (C/M/W 65:30:6) alone for 60 min. Subsequently, a linear gradient elution was performed from 100% Sol A to 100% Sol B (C/M/W 50:55:18) for 60 min. The flow rate was 1 ml/min and fraction size was 1 ml. Aliquots of all eluate fractions from Iatrobeads HPLC were monitored by HPTLC analysis, developed with C/M/W 65:30:6. Spots were revealed by primulin spray [28] and orcinol/sulfuric acid reaction. Eluates containing the same OS-PE conjugates, showing positive reaction with both primulin and orcinol/ sulfuric acid, were combined and concentrated under N2 stream. The yield of OS B-PE conjugate from OS B was ∼27.8%. The dried residue was dissolved in a defined volume of C/M/W 25:25:8. The concentration of OS-PE conjugate was measured based on orcinol/sulfuric acid reactivity, by TLC with Scion image analysis, and aliquots of suitable quantity were used for binding assays with GM3 and other GSLs.
2.8 Preparation of modified OS-PE conjugates from fetuin by treatment with sialidase, or with sialidase followed by β-galactosidase
OS-PE (4 mg) from fetuin was incubated in sodium acetate buffer (170 mM, pH 5.0) containing sialidase (2.5 U) for 88 h at 37°C. The reactant was subjected to C18 cartridge, and desialylated OS-PE conjugate was adsorbed on C18 cartridge in methanol: water 6:4. Non-reacted OS and water-soluble impurities were eluted, and desialylated OS-PE conjugate, eluted from C18 by C:M 2:1, was incubated in Tris–HCl (1 mM, pH 7.5) with β-galactosidase (∼2.5 U) for 39 h at 37°C, after drying. The reaction mixture was purified by C18 cartridge. Thus, OS-PE conjugate from fetuin, or the conjugate without terminal sialic acid (but exposing terminal β-Gal), or the conjugate without terminal sialic acid or penultimate β-Gal (but exposing terminal GlcNAc), were separated, and subjected to GM3 binding assay, using 3H-labeled liposome with GM3.
2.9 Determination of structure of N-linked OS-PE conjugates by ESI mass spectrometry
Structures of oligosaccharides with PE conjugates were confirmed by mass spectrometry using an ion trap mass spectrometer Esquire LC (Bruker Daltonics, Billerica, MA, USA) with electrospray ionization source. To confirm the presence of structures of oligosaccharides after PE-coupling reaction, 25 μg of OS B-PE conjugate dissolved in C/M/W 25:25:8 was diluted tenfold in methanol/ 1% formic acid/10% ammonium acetate. This solution was directly infused into the ion source at a flow rate of 1 µl/min. Spectra were collected in both positive and negative ionization mode. The fragmentation spectra of the analytes were collected with an isolation width of 4 amu, fragmentation amplitude set to 1 V and SmartFrag On (amplitude automatically varied to 30–200% of the set fragmentation amplitude of 1 V).
2.10 GSL-liposome interaction with OS-PE conjugates
Various quantities (0.2–10 μg per well, calculated based on equimolar quantities) of various OS-PE conjugates in 50% aqueous ethanol were added to each well of 96-well flat-bottom polystyrene plates, Costar # 9017 (Corning, Acton, MA), dried for 5 h at 37°C, blocked with 1% BSA for 1 h, and washed with TBS solution (see below). Binding of dried solid phase of OS-PE conjugate to liposomes labeled with [3H]cholesterol containing GSL was determined as follows. 3H-labeled liposomes containing GM3 and other GSLs were prepared as described previously [1, 2], with slight modification. Briefly, 50 μg 1,2 dimyristoyl-sn-glycero-3-phosphocholine, 30 μg non-labeled cholesterol, 2 μl 3H-labeled cholesterol (0.25 mCi per 0.25 ml), and 10-μg GSL were dissolved in 200-μl C/M 2:1, and the solution was evaporated to dryness in a rotary evaporator. The residue was mixed with 2-ml TBS solution (10-mM Tris–HCl (pH 8.0)/0.9-mM CaCl2/0.5-mM MgSO4/0.1-mM MnCl2), vigorously mixed with a Vortex mixer, and sonicated (Bransonic, model 5510R-MT, Branson Ultrasonics, Danbury, CT) at ∼20°C for 2 h. Temperature increase of the sonication bath was prevented by occasional addition of ice. In all experiments, freshly prepared liposomes in TBS solution were used. To each well containing OS-PE conjugate, 100-μl liposome preparation as above was added, the plate covered by lid was wrapped with polyvinyl film, and incubated for 16 h at room temperature with shaking (Red Rocker PR50). Wells were washed 3× with TBS. For effective washing, 200-μl TBS was added to each well, the tip of a thin pipette was placed at the edge, and washing medium was repeatedly sucked out. The last washing was tested for radioactivity. Bound liposomes were extracted twice with isopropanol/hexane/water 55:25:20, and radioactivity of the extract was counted.
3 Results
3.1 Properties and structures of N-linked OS fractions from ConA-non-binding ovalbumin
ConA-non-binding OS fractions from ovalbumin were separated by HPLC. Major fractions separated, containing OS, were termed Fr. 1 (containing OS I), Fr. 2 (containing OS II), Fr. 3 (containing OS III), Fr. 4 (containing OS IV), Fr. 5 (containing OS V), Fr. 6 (containing OS VI), and Fr. B (containing OS B). TLC patterns of these fractions are shown in Fig. 1a, and HPLC profiles on ODS column of Fr. 1, Fr. 6, and Fr. B are shown in Fig. 1b. Fr. 1 and 6 were each indicated mainly as a single peak on the ODS column, whereas Fr. B gave two peaks (Fr. B1 and Fr. B2). Identification of the major OS in Fr. 6, B1, and B2 was based on inspection of their HPLC data, and was confirmed by co-chromatography on ODS column of sample PA-glycans with corresponding reference compounds (Code No. 501.1, 501.1, and 501.5, respectively, in GALAXY). The structure for Fr. B1 and B2 (501.1 and 501.5 in GALAXY) was previously shown as a component of ovalbumin [11].
TLC and HPLC patterns of oligosaccharides (OS) from ConA-non-binding N-linked glycans of ovalbumin. Panel a: TLC patterns of HPLC-separated OS fractions from ConA-non-binding N-linked glycans of ovalbumin. HPLC fractions (Fr. 1-6, and Fr. B) were separated as described in M&M. TLC was developed in a mixture of 1-propanol/N-butyl acetate/ solvent B, 8:1:10. Solvent B is a mixture of acetonitrile and 0.2-M acetic acid/ triethylamine, pH 7.2, in a ratio of 3:7. Glc Glucose, CB cellobiose. Panel b: HPLC patterns of three major OS (Fr. 1, Fr. 6, Fr. B). Each OS peak was recorded as pyridylaminated (PA) derivative and separated on ODS column. Number shown on abscissa for each peak is relative retention time in comparison to glucose (glucose-unit). Detailed conditions of HPLC are described under M&M/ “Structural determination of OS from ovalbumin”
The elution time on ODS and amide columns of PA-glycan in Fr. 1 was not found in the previously-reported HPLC data. The molecular mass of this glycan determined by MALDI-TOF-MS analysis was 1395 Da, which corresponds to (Hex)3(HexNAc)4PA. By β1,4-galactosyltransferase treatment, this glycan gave rise to an oligosaccharide possessing two galactose residues, whose HPLC data and molecular mass values are all in good agreement with those of known reference PA-glycan, Galβ1-4GlcNAcβ1-2Manα1-6(Galβ1-4GlcNAcβ1-4Manα1-3)Manβ1-4GlcNAcβ1-4GlcNAc-PA (Code No. 200.15 in GALAXY). By co-chromatography on the ODS column, we confirmed the identity between the galactosylated glycan and the reference PA-glycan (code No. 200.15). Taking into account the substrate specificity of β-galactosyltransferase, we estimated the glycan structure in Fr. 1 as GlcNAcβ1-2Manα1-6(GlcNAcβ1-4Manα1-3)Manβ1-4GlcNAcβ1-4GlcNAc-PA.
The structures of major OS present in Fr. 1, Fr. 6/ Fr. B1, and Fr. B2 are shown in Fig. 2a, b, and c, respectively. Each glycan has multivalent GlcNAc residues at the non-reducing end.
3.2 Properties and structures of phosphatidylethanolamine (PE) conjugates of major OS fractions
PE conjugates of major OS fractions were prepared as described in M&M. PE conjugates were characterized by strong primulin staining and orcinol/ sulfuric acid reactivity, and were clearly separated by HPTLC, as shown in Fig. 3.
TLC patterns of OS-PE derivatives. Panel a: Fluorescent spot by primulin spray. Panel b: Spot detected by orcinol/ sulfuric acid reaction. CB, cellobiose. CB-PE, cellobiose-PE conjugate. Fr. 1-PE, OS Fr. 1-PE conjugate. Fr. 6-PE, OS Fr. 6-PE conjugate. Fr. B-PE, PE conjugate of a mixture of OS Fr. B-1 and -2. Or, origin. Solvent, C/M/W 60:35:8
Structures of OS B-PE conjugates were determined by ESI-MS with MS-MS, as described in M&M.
Three major ions at m/z 1,393.3, 1,413.8, and 1,494.9 were detected in a positive spectrum from OS B-PE conjugate fraction. These corresponded to doubly charged (M+H+Na)2+ major ions. They were, respectively, Fr. B2-PE minus GlcNAc (Gal, 7 GlcNAc, 3 Man, PE), Fr. B1-PE (8 GlcNAc, 3 Man, PE) and Fr. B2-PE (Gal, 8 GlcNAc, 3 Man, PE) (Fig. 4a). The difference between the three ions suggests the presence of three major molecular species.
Positive-ion ESI-MS spectra of PE conjugates of OS B fractions, and MS-MS data for each major mass. Panel a: PE conjugates of OS B. Note the presence of three major ions, with mass number 1,393.3, 1,413.8, and 1,494.9, corresponding respectively to doubly charged (M+H+Na)2+ major ions with (1 Gal, 7 GlcNAc, 3 Man, PE), (8 GlcNAc, 3 Man, PE) and (1 Gal, 8 GlcNAc, 3 Man, PE). Other minor ions are well assigned by elimination of GlcNAc or Gal from the three major ions. Panel b: MS/MS spectrum of m/z 1413.8 corresponding to Fr. B1-PE. Panel c: MS/MS spectrum of m/z 1494.9 (Fr. B2-PE). Panel d: MS/MS spectrum of m/z 1393.3 (product ion of m/z 1291.3, based on loss of one HexNAc from m/z 1393.3. Panel e: Possible assigned structures of Fr. B-PE conjugates corresponding to expected structure, based on results of MS/MS
The MS/MS spectrum of Fr. B1-PE at m/z 1,413.8 is shown in Fig. 4b. A major product ion of m/z 1,312.2 was assigned to loss of one HexNAc from Fr. B1-PE. The MS/MS of m/z 1,312.2 showed one major ion at m/z 1,210.7, corresponding to loss of one more HexNac (data not shown).
The MS/MS spectrum of Fr. B2-PE at m/z 1,494.9, and assignment of the product ions, are shown in Fig. 4c. A major product ion of m/z 1,393.3 corresponded to loss of one HexNAc from B2-PE. The major ion m/z 1,291.3 in MS/MS of m/z 1,393.3 corresponded to loss of one HexNAc from m/z 1,393.3 (data not shown).
In the positive MS/MS spectrum of m/z 1,393.3 (Fig. 4d), a product ion of m/z 1,291.3 was assigned to loss of two HexNAc from m/z 1,393.3.
Product ions corresponding to dipalmitoyl-glycerol (m/z 551) were detected in all MS/MS (Fig. 4b, c, d), which means that the major three ions originated from PE-conjugate.
Possible structures of Fr. B-PE conjugates corresponding to expected structure, based on results of MS/MS, are shown in Fig. 4e.
Analysis of MS/MS showed that the original structure of oligosaccharide Fr. B is coupled to PE and retained in OS-PE conjugate, without any breakdown of the structure.
3.3 Binding of Fr. OS I-PE, Fr. OS VI-PE, and Fr. OS B-PE conjugates to liposomes containing 3H-labeled GM3 or other GSLs
Various defined equimolar quantities (0.2 through 10 μg/well, as shown on Fig. 5 abscissa) of two OS-PE conjugates (Fr. OS I-PE, Fr. OS B-PE), and cellobiose-PE conjugate, were placed on each well of polystyrene multi-well plates, dried, and blocked by 1% BSA as described in M&M. To each well was added 3H-labeled liposomes containing GM3, LacCer, Gb4, or Forssman antigen. Labeling of liposomes was based on 3H-cholesterol, non-labeled PC, and GSL, as described in M&M. Binding of GM3 to OS B-PE conjugate was clearly higher than binding to OS I-PE conjugate. GM3 did not bind to cellobiose-PE conjugate (Fig. 5a). OS B-PE conjugate did not bind to labeled liposomes containing LacCer, Gb4, or Forssman GSL (Fig. 5b). In some experiments, LacCer showed minimal, variable binding at certain concentrations, but this was insignificant (see Discussion).
Binding of 3H-labeled liposome containing GM3, or other GSLs, to various OS-PE derivatives coated and affixed on multi-well polystyrene plates. Panel a: Various quantities (0.2–10 μg/ well) of various OS-PE derivatives as shown on abscissa were dried at 37°C, blocked with 1% BSA for 1 h, and washed. Each well was added with 100 μl of 3H-labeled GM3 liposome, incubated for 16 h, and degree of binding was determined, as described in M&M. Clear GM3 liposome binding was observed for Fr. B-PE. Binding of Fr. 1-PE was weak. No binding was observed for CB-PE. Data shown are typical results from a single triplicate experiment. Similar results were obtained in two other triplicate experiments. Bars indicate standard deviation. GM3 liposome binding to Gg3-coated plate, indicated as “Gg3” (open squares at left side of figure), is positive control (see Discussion). Panel b: Various quantities (0.1–7 μg/well) of Fr. B-PE were coated on multi-well plates as above. To each well was added 100 μl of 3H-labeled liposome containing LacCer, Forssman antigen, or Gb4. Degree of binding was determined as described in M&M. Only GM3-liposomes, but not other GSL-liposomes, bound to Fr. B-PE coated plates. Data shown are typical results from a single triplicate experiment. Similar results were obtained in two other triplicate experiments. Bars indicate standard deviation
3.4 Binding of multi-antennary OS-PE from fetuin to GM3 when GlcNAc is exposed at terminus
Under experimental conditions similar to those for GM3 binding to OS B-PE conjugate as above, PE conjugate of multi-antennary OS from fetuin did not bind to GM3. However, following treatment with sialidase followed by β-galactosidase as described under M&M, the modified PE conjugate showed strong binding to GM3 (Fig. 6).
Binding of 3H-labeled liposome containing GM3 to N-linked oligosaccharides from fetuin conjugated to PE. Three types of PE-conjugates of fetuin N-linked OS mixtures were prepared: (1) non-treated PE-conjugate; (2) sialidase-treated PE-conjugate (marked “deNeuAc”); (3) sialidase-treated followed by β-galactosidase-treated PE-conjugate (marked “deNeuAc deGal”). 0.1 to 6 μg/well of these PE-conjugates, with calculated equal molar quantities, were adhered to each well of multi-well plates, and binding of 3H-labeled liposome containing GM3 was determined as described in M&M and Fig. 5 legend. “CB-PE” indicates binding of GM3-liposome to cellobiose-PE conjugate coated plate (negative control). “Gg3” (left corner) indicates GM3-liposome binding to Gg3-coated plate (positive control; see Fig. 5 legend, and Discussion)
4 Discussion
The process of CCI was originally studied based on adhesion or interaction of glycosyl vesicles, liposomes, or specific glycosyl cluster assembly (“cyclophane”) [1, 2, 29, 30]. The early findings were later supported by studies using NMR spectroscopy [30, 31], surface plasmon resonance spectroscopy (SPR) [6, 32, 33], atomic force microscopy (AFM) [5, 34, 35], electrospray ionization mass spectrometry [36, 37], and transmission electron microscopy [38] (for review see [39–41].
A few specific conditions are required for the process of CCI: (1) multiple binding sites within a molecule, or clustering of multiple molecules involved in CCI; (2) bivalent cation such as Ca2+; (3) proper orientation of carbohydrate binding sites, interfacing with other carbohydrates involved in CCI. Condition (1) has been often discussed, and demonstrated in many quantitative studies as cited above. Condition (2) has been discussed since the beginning of the CCI concept [1], and was supported by studies based on change of NMR [31], striking change of enthalpy [35], and electrospray ionization MS [36, 37]. The mechanism of bivalent cation dependent effect of CCI remains unclear. In the case of Lex-to-Lex interaction, Ca2+ may mediate binding of Lex to counterpart Lex through O4-Fuc, O5-Fuc, and O2-Gal or O6-GlcNAc, as indicated by study with conformational molecular model [42]. On the other hand, some cases of CCI were claimed not to require bivalent cation [43, 44]. Condition (3) has been revealed clearly, based on the observation that binding ability of Lex epitope carried by ceramide is much stronger than that of the same epitope carried by acyl glyceride, since the orientation of Lex differs greatly between these two carrier lipids [45].
The present findings suggest that OS B having 5 or 6 GlcNAc termini binds strongly to GM3. Binding of OS I, having bivalent GlcNAc, was lower than binding of OS B to GM3. The structures OS B1 (as in Fig. 2b) and OS B2 (as in Fig. 2c) were previously isolated and characterized from ovalbumin [11], and were also identified clearly in the present study. In contrast, OS 1 (Fr. 1 PA-glycan) has a novel structure, clearly characterized for the first time in the present study, as detailed under “Results”. GM3 binding to fetuin OS was observed only when fetuin, having multi-antennary structure, was treated with sialidase followed by β-galactosidase to expose GlcNAc. These findings clearly indicate that multiple GlcNAc termini favor specific binding to GM3. Since GM3 is known to bind to Gg3 [7, 33], binding of GM3 to Gg3-coated plate was used as positive control (see Figs. 5 and 6 legends). GM3 binding efficiency to Gg3-coated plate was much higher than that to plates coated with OS-PE conjugates. GM3-to-Gg3 interaction is enhanced by strong clustering of both GSLs. Inclusion of PC-cholesterol to OS-PE conjugates did not increase the degree of interaction (data not shown).
None of the OS-PE conjugates used in this study showed binding to liposomes containing Gb4 or Forssman antigen. Their binding to LacCer was essentially negligible; however, some variation was observed at certain concentrations (3-6 μg/ well). Interaction of LacCer with GM3 based on CCI was studied previously. Consistent binding was observed only when lactosylsphingosine was presented in micellar form [46].
The occurrence of CCI in vitro has been clearly demonstrated for a limited number of carbohydrate epitopes, such as Lex/Lex, GM3/Gg3, GM3/LacCer, and clustered OS in sponge cells. This is probably due to the limited number of studies, rather than limited occurrence of the CCI phenomenon. Results of the present study extend the concept to in vitro CCI occurring between specific N-linked glycans having multiple GlcNAc termini, and GM3. The major question remains regarding applicability of in vitro CCI results to in vivo processes, i.e., cellular interaction/ adhesion, and signal transduction.
N-linked complex-type glycans having multiple GlcNAc termini are known to be expressed at the early stage of spermatogenesis, as probed by mAb J1 [12]. Differentiation and maturation of sperm cells requires their adhesion to Sertoli cells. N-linked glycans with GlcNAc termini were implicated in mediation of sperm-to-Sertoli adhesion [47], and of annexin V-to-heat shock protein 47 (Hsp47) adhesion [48], although the receptor was not clarified. An interesting possibility is opened that such adhesion could also be mediated by CCI as described in this paper. CCI has been implicated as a basis for cell-to-cell interaction (trans-CCI). On the other hand, modulation of growth factor receptor or integrin receptor by surrounding gangliosides in membrane microdomain is well known [49], although the exact mechanism of the ganglioside effect is unclear. Further extensive study is essential to determine whether such ganglioside effect on receptor occurs through CCI of N-linked glycan of the receptor with surrounding gangliosides (cis-CCI) [40].
Abbreviations
- CB:
-
cellobiose
- CCI:
-
carbohydrate-to-carbohydrate interaction
- C/M/W:
-
chloroform/methanol/water
- ConA:
-
Concanavalin A
- GlcNAc:
-
N-acetylglucosamine
- Gb4:
-
globoside (GalNAcβ3Galα4Galβ4Glcβ1Cer)
- Gg3:
-
GalNAcβ4Galβ4Glcβ1Cer
- GM3:
-
NeuAcα3Galβ4Glcβ1Cer
- GSL:
-
glycosphingolipid
- OS:
-
oligosaccharides
- PC:
-
1,2-dimyristoyl-sn-glycero-3-phosphocholine
- PE:
-
phosphatidylethanolamine (1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine)
References
Eggens, I., Fenderson, B.A., Toyokuni, T., Dean, B., Stroud, M.R., Hakomori, S.: Specific interaction between Lex and Lex determinants: A possible basis for cell recognition in preimplantation embryos and in embryonal carcinoma cells. J. Biol. Chem. 264, 9476–9484 (1989)
Kojima, N., Hakomori, S.: Specific interaction between gangliotriaosylceramide (Gg3) and sialosyllactosylceramide (GM3) as a basis for specific cellular recognition between lymphoma and melanoma cells. J. Biol. Chem. 264, 20159–20162 (1989)
Misevic, G.N., Burger, M.M.: Carbohydrate-carbohydrate interactions of a novel acidic glycan can mediate sponge cell adhesion. J. Biol. Chem. 268, 4922–4929 (1993)
Spillmann, D., Thomas-Oates, J.E., van Kuik, J.A., Vliegenthart, J.F.G., Misevic, G., Burger, M.M., Finne, J.: Characterization of a novel sulfated carbohydrate unit implicated in the carbohydrate-carbohydrate-mediated cell aggregation of the marine sponge Microciona prolifera. J. Biol. Chem. 270, 5089–5097 (1995)
Bucior, I., Scheuring, S., Engel, A., Burger, M.M.: Carbohydrate-carbohydrate interaction provides adhesion force and specificity for cellular recognition. J. Cell Biol. 165, 529–537 (2004)
Haseley, S.R., Vermeer, H.J., Kamerling, J.P., Vliegenthart, J.F.G.: Carbohydrate self-recognition mediates marine sponge cellular adhesion. Proc. Natl. Acad. Sci. USA 98, 9419–9424 (2001)
Kojima, N., Hakomori, S.: Cell adhesion, spreading, and motility of GM3-expressing cells based on glycolipid–glycolipid interaction. J. Biol. Chem. 266, 17552–17558 (1991)
Kojima, N., Shiota, M., Sadahira, Y., Handa, K., Hakomori, S.: Cell adhesion in a dynamic flow system as compared to static system: Glycosphingolipid–glycosphingolipid interaction in the dynamic system predominates over lectin- or integrin-based mechanisms in adhesion of B16 melanoma cells to non-activated endothelial cells. J. Biol. Chem. 267, 17264–17270 (1992)
Rojo, J., Diaz, V., de la Fuente, J.M., Segura, I., Barrientos, A.G., Riese, H.H., Bernad, A., Penades, S.: Gold glyconanoparticles as new tools in antiadhesive therapy. Chembiochem. 5, 291–297 (2004)
Zhu, Z., Kojima, N., Stroud, M.R., Hakomori, S., Fenderson, B.A.: Monoclonal antibody directed to Le(y) oligosaccharide inhibits implantation in the mouse. Biol. Repro. 52, 903–912 (1995)
Yamashita, K., Kamerling, J.P., Kobata, A.: Structural study of the carbohydrate moiety of hen ovomucoid: occurrence of a series of pentaantennary complex-type asparagine-linked sugar chains. J. Biol. Chem. 257, 12809–12814 (1982)
Symington, F.W., Fenderson, B.A., Hakomori, S.: Fine specificity of a monoclonal anti-testicular cell antibody for glycolipids with terminal N-acetyl-d-glucosamine structure. Molec. Immun. 21 (1984)
Dohi, T., Nores, G., Hakomori, S.: An IgG3 monoclonal antibody established after immunization with GM3 lactone: Immunochemical specificity and inhibition of melanoma cell growth in vitro and in vivo. Cancer Res. 48, 5680–5685 (1988)
Young, W.W.J., MacDonald, E.M.S., Nowinski, R.C., Hakomori, S.: Production of monoclonal antibodies specific for distinct portions of the glycolipid asialo GM2 (gangliotriosylceramide). J. Exp. Med. 150, 1008–1019 (1979)
Mizuochi, T.: Microscale sequencing of N-linked oligosaccharides of glycoproteins using hydrazinolysis, Bio-GelP-4, and sequential exoglycosidase digestion. In: Hounsell, E.F. (ed.) In Methods in Molecular Biology: Glycoprotein Analysis in Biomedicine, Vol. 14, pp. 55–68. Humana, Totawa, NJ (1993)
Shimizu, Y., Nakata, M., Kuroda, Y., Tsutsumi, F., Kojima, N., Mizuochi, T.: Rapid and simple preparation of N-linked oligosaccharides by cellulose-column chromatography. Carbohydr. Res. 332, 381–388 (2001)
Merkle, R.K., Cummings, R.D.: Lectin affinity chromatography of glycopeptides. Meth. Enzymol. 138, 232–259 (1987)
Ohyama, Y., Kasai, K., Nomoto, H., Inoue, Y.: Frontal affinity chromatography of ovalbumin glycoasparagines on a concanavalin A-sepharose column. A quantitative study of the binding specificity of the lectin. J. Biol. Chem. 260, 6882–6887 (1985)
Hase, S., Ibuki, T., Ikenaka, T.: Reexamination of the pyridylamination used for fluorescence labeling of oligosaccharides and its application to glycoproteins. J. Biochem. (Tokyo) 95, 197–203 (1984)
Tomiya, N., Awaya, J., Kurono, M., Endo, S., Arata, Y., Takahashi, N.: Analyses of N-linked oligosaccharides using a two-dimensional mapping technique. Anal. Biochem. 171, 73–90 (1988)
Takahashi, N., Nakagawa, H., Fujikawa, K., Kawamura, Y., Tomiya, N.: Three-dimensional elution mapping of pyridylaminated N-linked neutral and sialyl oligosaccharides. Anal. Biochem. 226, 139–146 (1995)
Takahashi, N., Matsuda, T., Shikami, K., Shimada, I., Arata, Y., Nakamura, R.: A structural study of the asparagine-linked oligosaccharide moiety of duck ovomucoid. Glycoconj. J. 10, 425–434 (1993)
Takahashi, N., Kato, K.: GALAXY (Glycoanalysis by the Three Axes of MS and Chromatography): A web application that assists structural analyses of N-glycans. TIGG (Trends in Glycoscience and Glycotechnology) 15, 235–251 (2003)
Yagi, H., Takahashi, N., Yamaguchi, Y., Kimura, N., Uchimura, K., Kannagi, R., Kato, K.: Development of structural analysis of sulfated N-glycans by multi-dimensional HPLC mapping methods. Glycobiology 15, 1051–1060 (2005)
Tang, P.W., Feizi, T.: Neoglycolipid micro-immunoassays applied to the oligosaccharides of human milk galactosyltransferase detect blood-group related antigens on both O- and N-linked chains. Carbohydr. Res. 161, 133–143 (1987)
Feizi, T., Stoll, M.S., Yuen, C.-T., Chai, W., Lawson, A.M.: Neoglycolipids: Probes of oligosaccharide structure, antigenicity, and function. Meth. Enzymol. 230, 484–519 (1994)
Stoll, M.S., Feizi, T.: Preparation of neoglycolipids for structure and function assignments of oligosaccharides. BioMethods 9, 329–348 (1997)
Skipski, V.P.: Thin layer chromatography of neutral glycosphingolipids. Meth. Enzymol. 35, 396–425 (1975)
Stewart, R.J., Boggs, J.M.: A carbohydrate-carbohydrate interaction between galactosylceramide-containing liposomes and cerebroside sulfate-containing liposomes: Dependence on the glycolipid ceramide composition. Biochemistry 32, 10666–10674 (1993)
Coteron, J.M., Vicent, C., Bosso, C., Penades, S.: Glycophanes, cyclodextrin–cyclophane hybrid receptors for apolar binding in aqueous solutions: A stereoselective carbohydrate-carbohydrate interaction in water. J. Am. Chem. Soc. 115, 10066–10076 (1993)
Henry, B., Desvaux, H., Pristchepa, M., Berthault, P., Zhang, Y.-M., Mallet, J.-M., Esnault, J., Sinay, P.: NMR study of a Lewisx pentasaccharide derivative: Solution structure and interaction with cations. Carbohydr. Res. 315, 48–62 (1999)
Hernaiz, M.J., de la Fuente, J.M., Barrientos, A.G., Penades, S.: A model system mimicking glycosphingolipid clusters to quantify carbohydrate self-interactions by surface plasmon resonance. Angew. Chem. Intl. Ed. 41, 1554–1557 (2002)
Matsuura, K., Kitakouji, H., Sawada, N., Ishida, H., Kiso, M., Kitajima, K., Kobayashi, K.: A quantitative estimation of carbohydrate–carbohydrate interaction using clustered oligosaccharides of glycolipid monolayers and of artificial glycoconjugate polymers by surface plasmon resonance. J. Am. Chem. Soc. 122, 7406–7407 (2000)
Tromas, C., Rojo, J., de la Fuente, J.M., Barrientos, A.G., Garcia, R., Penades, S.: Adhesion forces between LewisX determinant antigens as measured by atomic force microscopy. Angew. Chem. Intl. Ed. 40, 3052–3055 (2001)
de la Fuente, J.M., Eaton, P., Barrientos, A.G., Menendez, M., Penades, S.: Thermodynamic evidence for Ca2+-mediated self-aggregation of Lewis X gold glyconanoparticles. A model for cell adhesion via carbohydrate–carbohydrate interaction. J. Am. Chem. Soc. 127, 6192–6197 (2005)
Siuzdak, G., Ichikawa, Y., Caulfield, T.J., Munoz, B., Wong, C.-H., Nicolaou, K.C.: Evidence of Ca2+-dependent carbohydrate association through ion spray mass spectrometry. J. Am. Chem. Soc. 115, 2877–2881 (1993)
Koshy, K.M., Boggs, J.M.: Investigation of the calcium-mediated association between the carbohydrate head groups of galactosylceramide and galactosylceramide I3 sulfate by electrospray ionization mass spectrometry. J. Biol. Chem. 271, 3496–3499 (1996)
Carvalho de Souza, A., Halkes, K.M., Meeldijk, J.D., Verkleij, A.J., Vliegenthart, J.F., Kamerling, J.P.: Gold glyconanoparticles as probes to explore the carbohydrate-mediated self-recognition of marine sponge cells. Chembiochem. 6, 828–831 (2005)
Bovin, N.V.: Carbohydrate–carbohydrate interaction. In: Gabius, H.J., Gabius, S. (eds.) In Glycosciences: Status and Perspectives, pp. 277–289. Chapman & Hall, London (1997)
Hakomori, S.: Carbohydrate-to-carbohydrate interaction in basic cell biology: A brief overview. Arch. Biochem. Biophys. 426, 173–181 (2004)
Rojo, J., Morales, J.C., Penades, S.: Carbohydrate-carbohydrate interactions in biological and model systems. Topics Curr. Chem. 218, 45–92 (2002)
Kojima, N., Fenderson, B.A., Stroud, M.R., Goldberg, R.I., Habermann, R., Toyokuni, T., Hakomori, S.: Further studies on cell adhesion based on Le(x)–Le(x) interaction, with new approaches: embryoglycan aggregation of F9 teratocarcinoma cells, and adhesion of various tumour cells based on Le(x) expression. Glycoconj. J. 11, 238–248 (1994)
Yu, S., Withers, D.A., Hakomori, S.: Globoside-dependent adhesion of human embryonal carcinoma cells, based on carbohydrate-carbohydrate interaction, initiates signal transduction and induces enhanced activity of transcription factors AP1 and CREB. J. Biol. Chem. 273, 2517–2525 (1998)
Yu, S., Kojima, N., Hakomori, S., Kudo, S., Inoue, S., Inoue, Y.: Binding of rainbow trout sperm to egg is mediated by strong carbohydrate-to-carbohydrate interaction between (KDN)GM3 (deaminated neuraminyl ganglioside) and Gg3-like epitope. Proc. Natl. Acad. Sci. USA 99, 2854–2859 (2002)
Gourier, C., Pincet, F., Perez, E., Zhang, Y., Zhu, Z., Mallet, J.M., Sinay, P.: The natural LewisX-bearing lipids promote membrane adhesion: influence of ceramide on carbohydrate–carbohydrate recognition. Angew. Chem. Int. Ed. Engl. 44, 1683–1687 (2005)
Santacroce, P.V., Basu, A.: Probing specificity in carbohydrate–carbohydrate interactions with micelles and Langmuir monolayers. Angew. Chem. Intl. Ed. 42,95–98 (2003)
Akama, T.O., Nakagawa, H., Sugihara, K., Narisawa, S., Ohyama, C., Nishimura, S., O'Brien, D.A., Moremen, K.W., Millan, J.L., Fukuda, M.N.: Germ cell survival through carbohydrate-mediated interaction with Sertoli cells. Science 295, 124–127 (2002)
Gao, C.X., Miyoshi, E., Uozumi, N., Takamiya, R., Wang, X., Noda, K., Gu, J., Honke, K., Wada, Y., Taniguchi, N.: Bisecting GlcNAc mediates the binding of annexin V to Hsp47. Glycobiology 15, 1067–1075 (2005)
Hakomori, S., Igarashi, Y.: Functional role of glycosphingolipids in cell recognition and signaling. J. Biochem. (Tokyo) 118, 1091–1103 (1995)
Acknowledgments
This work was supported by NIH/ National Institute of General Medical Science (R01 GM070593) by NIH/ National Cancer Institute (R01 CA080054), to SH, by Core Research for Evolutionary Science and Technology, Japan Science and Technology Agency, and by a Grant-in-Aid for Scientific Research on Priority Areas (17046017) from the Ministry of Education, Culture, Sports, Science and Technology of Japan. The Esquire LC mass spectrometer was purchased with support by National Science Foundation under Grant No. 9807748. We thank Dr. Kimie Murayama (Pacific Northwest Research Institute) for advice on ESIMS analysis of OS-PE conjugates, and Dr. Stephen D. Anderson for preparation of the MS.
Author information
Authors and Affiliations
Corresponding author
Additional information
All solvent ratios are by volume.
An erratum to this article can be found at http://dx.doi.org/10.1007/s10719-006-9027-7
Rights and permissions
About this article
Cite this article
Yoon, SJ., Nakayama, Ki., Takahashi, N. et al. Interaction of N-linked glycans, having multivalent GlcNAc termini, with GM3 ganglioside. Glycoconj J 23, 639–649 (2006). https://doi.org/10.1007/s10719-006-9001-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-006-9001-4