Abstract
Urochloa spp. grow vigorously in the dry-season of the tropics, and have been used successfully to provide abundant surface residue as cover crop for no-till soybean (Glycine max). Nitrogen (N) fertilizer application could enhance cover crop biomass production and its ground cover and accelerate residue decomposition, but how these cascading factors affect nutrient availability to the subsequent soybean crop is not known. We evaluated nutrient cycling and soybean nutrition and yield components following different timings of N application to living and desiccated Urochloa cover crops. The experiment was conducted in two growing seasons at Botucatu, São Paulo State, Brazil. Treatments consisted of two cover crop grasses (Urochloa brizantha and Urochloa ruziziensis) and six N management systems [control (no N application); N application at soybean sowing (40 kg N ha−1) plus topdressing (60 kg N ha−1 at V5 stage); N application 20 days before desiccation (DBD) of cover crops, N application 10 DBD; N application 5 DBD; and N application 1 day before sowing of soybean, using the rate of 100 kg N ha−1 in the latter four treatments]. Both cover crops produced high amount of shoot biomass (> 9.7 Mg ha−1), but U. brizantha was 48% more productive than U. ruziziensis. Nutrient accumulation in cover crop straw was enhanced due to greater biomass production in treatments with N applied 20 and 10 DBD. Soybean grain yield was 17% greater following U. brizantha than following U. ruziziensis. Nitrogen application at different times did not affect soybean grain yield. These results suggest that Urochloa biomass, macronutrient accumulation, and subsequent release rates can be enhanced with N application, but it had little short-term impact on soybean yield components.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soil degradation is on on-going challenge facing agriculture, and therefore, affects global climate change by altering the cycling of water, carbon (C), nitrogen (N), sulfur (S), and other elements (Lal and Stewart 1990). According to FAO (2015), 33% of soil in the world is degraded due to agriculture, resulting in serious economic and social instability, deforestation, water pollution, enhanced greenhouse gas emissions, intensive use of marginal lands, and accelerated runoff and erosion (Lal and Stewart 1990). Conservation agricultural systems using cover crops and appropriate fertilizer management could be important practices to enhance the sustainability of agricultural systems.
No-till systems (NTS) maintain crop residue on the soil surface, decrease soil disturbance, and often have rotation of crops over time, resulting in one of the most effective strategies to improve agricultural sustainability (Caires et al. 2015), particularly when combined with crop-livestock integration (Crusciol et al. 2015). Urochloa is well-suited for the tropical region with dry winters and low fertility, because these grasses have vigorous and deep roots and high tolerance to water deficit stress (Fisher and Kerridge 1998; Gichangi et al. 2017). These perennial forage grasses have high biomass production potential and efficiency in nutrient cycling. The root system can explore more than 1 m deep, taking advantage of residual fertilization from the previous summer crop, and increasing soil biological activity and organic matter content (Pacheco et al. 2011; Nascente and Crusciol 2012). Urochloa straw quality and quantity are considered ideal for maintaining the integrity of NTS (Pariz et al. 2011a) with significant persistence at the soil surface (Kluthcouski et al. 2003; Nascente and Crusciol 2012), which could lead to improved soil organic matter and regeneration of long-term productivity (Franzluebbers et al. 2014; Crusciol et al. 2015).
To keep the soil surface covered, cover crops should have high potential biomass production with low decomposition (Pacheco et al. 2011). However, residues with high C/N ratio may cause temporary immobilization of N in soil (Calonego et al. 2012). Seasonal changes in soil organic C and N fractions are a result of crop residue inputs (quantity and quality) and soil microbial biomass and activity (Franzluebbers et al. 1995). Fluctuations in soil microbial biomass can cause net N mineralization or immobilization during decomposition of plant residues (Santi et al. 2003). Low quantity of soil and crop residue N could limit subsequent crop growth during decomposition of cover crop straw.
Forage grasses as cover crops may promote constant and more efficient nutrient cycling by changing time of availability and/or release of nutrients (Assmann et al. 2017). A key concept of nutrient cycling in any ecosystem is mass balance. To be sustainable, nutrient outputs should be balanced with nutrient inputs to a system (Wedin and Russelle 2007), and the cycling of nutrients are determined by the soil biotic and abiotic conditions (Ferreira et al. 2011). Therefore, forage grasses as cover crops can maximize benefits to nutrient cycling by supplying sufficient C to keep nutrients in organic forms during non-crop growing periods (Assmann et al. 2017).
Soybean [Glycine max (L.) Merr.] is one of the most important crops cultivated in Brazil, grown on 35 million ha (CONAB 2018). This crop grown in succession or rotation with grasses achieves high grain yield (Moraes et al. 2014). Soybean has high quantity of N, mainly as grain protein (50–60 g N kg−1). To meet the demand of N, soybean relies primarily on biological nitrogen fixation (BNF), which occurs from symbiosis with the bacterial genus Bradyrhizobium supplying 70–200 kg N ha−1 (Herridge et al. 2008). However, some researchers have reported the necessity to fertilize soybean with N in order to avoid yellowing of leaves during initial growth (Nogueira et al. 2010; Pereira et al. 2010), or at the reproductive stage since BNF may be insufficient to provide enough N to soybean (Petter et al. 2012; Bahry et al. 2013).
One alternative to improve soybean N availability might be to supply N to a cover crop, aiming to both carryover sufficient N to soybean via decomposition of the cover crop and enhance the quantity of cover crop straw. Therefore, our objective was to develop an understanding of how N fertilization timing (i.e. applied to cover crop before desiccation or on cover crop residues) affects cover crop biomass production, nutrient accumulation and release, and subsequent soybean growth and yield components.
Materials and methods
Site description
The experiment was conducted during the 2013–2014 and 2014–2015 growing seasons in Botucatu, State of São Paulo, southeastern Brazil (48°26′W; 22°51′S; 740 m asl). The soil is a clayey, kaolinitic, thermic Typic Haplorthox (USDA Taxonomy). Rainfall and temperature during the experimental period are reported in Table 1.
The experiment was carried out in an area cultivated with Urochloa brizantha (Hochst. Ex A. Rich.) R.D. Webster (syn. Brachiaria brizantha) cv. Marandu and U. ruziziensis (R. Germ. and C.M. Evrard) Morrone and Zuloaga (syn. B. ruziziensis). In each growing season, the experiment was conducted in a new area of the field. Management history was NTS for 6 years. Cover crops were sown at a density of 10 kg seed ha−1 (34% viable seed). In both growing seasons, cover crops were not previously fertilized, i.e., they grew only with the residual fertilization from a previous crop. Cover crops were cultivated approximately 8 months (April to November) before soybean seeding, and managed without weed control.
Before the experiment, soil (0–0.2-m depth) was sampled to evaluate its chemical characteristics (Table 2), according to the method of van Raij et al. (2001). Only in the 2013–2014 growing season, ammonifier and nitrifier bacterial populations were determined (Table 3), according to Sarathchandra (1978), Kowalchuck et al. (1997), and Treusch et al. (2005).
Experimental design and treatments
The experimental design was a randomized complete block, arranged in a 2 × 6 factorial scheme, with four replications. Treatment factors consisted of two cover crops (U. brizantha and U. ruziziensis) and six forms of N management: control (no N application), N application at soybean sowing and topdressing [40 kg N ha−1 + 60 kg N ha−1, respectively (S + T)], N application on cover crop at 20 days before desiccation (DBD), N application on cover crop at 10 DBD, N application on cover crop at 5 DBD, and N application after cover crop desiccation at 1 day before sowing (DBS) of soybean, using the rate of 100 kg N ha−1 in the last four treatments. The N rate of 100 kg ha−1 was applied with the intent to increase cover crop biomass production and N uptake, as well as reduce straw C/N ratio when applied before desiccation (20 DBD, 10 DBD, or 5 DBD) as a strategy to minimize N immobilization during early soybean development. Nitrogen application at 1 DBD and at soybean sowing and topdressing was intended to supply N directly to the soybean crop, since in conditions of high grain yield, soybean may demand more N than BNF can supply (Salvagiotti et al. 2008). Each plot consisted of ten 5-m-long rows spaced at 0.45 m, in a total area of 22.5 m2. Samples were collected in four central rows, avoiding 0.5 m from the end of each row.
Prior to N application, forage accumulated as cover crop was cut by mechanical mower at 0.3-m height. Application of N started on 5 October 2013 and 8 October 2014 for the 20 DBD treatment and applications of N at 10 and 5 DBD were made in sequence. Cover crops were desiccated on 25 October 2013 and 28 October 2014 with the herbicide glyphosate (2.8 kg acid-equivalent ha−1). Cover crop desiccation was made 30 DBS of soybean. For the treatment with application of N at 1 DBS, N was applied on the cover crop straw on 26 November 2013 and 30 November 2014. For the treatment with application at time of sowing, 40 kg N ha−1 was applied at 0.05–0.10 m distance from the sowing furrow and 60 kg N ha−1 was topdressed at V5 stage of soybean. The N source for all applications was ammonium nitrate.
Crop management
Soybean cultivar BMX Potencia RR was sown on 27 November 2013 and 1 December 2014 using a no-till drill at a density of 377,000 seeds ha−1 and a depth of 0.03 m. Right before sowing, soybean seeds were inoculated with Bradyrhizobium japonicum. For all treatments, the basic fertilization in the sowing furrows consisted of 90 kg P2O5 ha−1 as triple superphosphate and 45 kg K2O ha−1 as potassium chloride (Mascarenhas and Tanaka 1997).
Soybean was cultivated according to crop needs with the following: fungicide application of azoxystrobin and cyproconazole, pyraclostrobin and epoxiconazole; and insecticide application of acephate, methomyl and thiamethoxam.
Sampling and analyses
Shoot dry matter (DM) of cover crops was evaluated at 0 and 90 days after desiccation (DAD). Biomass was cut at ground level (0.25 m2 each) from three random locations along a diagonal within each plot and composited, according to Crusciol et al. (2005). Biomass was dried by forced-air circulation at 65 °C for 72 h until constant weight and weighed.
Soybean shoot DM was determined by sampling 10 plants at flowering (R2 stage). Additionally, samples were collected for leaf nutrition diagnosis from the upper third leaf from 30 plants per plot (Ambrosano et al. 1997). Leaves were washed, dried in forced air circulation at 65 °C for 72 h, and ground. Concentrations of N, P, K, Ca, Mg, and S in leaves were determined according to Malavolta et al. (1997).
Soybean was harvested at maturity. Plant height, grain yield, and yield components (final population of plants, number of pods per plant, number of grains per pod, and 100-seed weight) were determined at harvest. Final population was determined by counting the number of plants in two central 4-m rows per plot. Plant height, number of pods per plant, and number of grains per pod were evaluated from 15 plants per plot chosen at random. Plants from four central 4-m rows were threshed mechanically, grain weighed, and yield calculated. The 100-grain weight was performed by randomly collecting eight samples per plot.
Statistical analyses
A combined analysis of variance across the two growing seasons was performed using the statistical package SISVAR (Ferreira 2011). Before analysis, data were tested for normality and homogeneity of variance. Blocks and block interactions were considered random effects. Cover crop, N management, growing season, and their interactions were considered fixed effects. For all variables, an F test was performed and means were separated using Fisher’s protected LSD test at 0.05 probability level.
Results
Weather conditions
In the first growing season (October 2013–May 2014), rainfall (683 mm) was 45% lower than the long-term average of 1244 mm and mean temperature (23.0 °C) was higher than the long-term average of 22.2 °C (Table 1). In the second growing season (October 2014–May 2015), rainfall (1361 mm) was 9% greater than the long-term average and mean temperature (21.9 °C) was lower than the long-term average.
Cover crop biomass production and nutrient accumulation
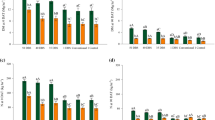
Accumulation of DM and macronutrients in cover crop shoots at 0 and 90 DAD were affected by cover crop species, N management system, growing season, and cover crop × N management system interaction (Table 4). At 0 DAD, U. brizantha had greater DM than U. ruziziensis in all treatments with N (20 DBD, 10 DBD, 5 DBD, 1 DBS, and S + T) and in the control (no N application), when averaged across growing seasons (Fig. 1a). U. brizantha biomass production varied by N management system: 20 DBD > 10 DBD = 5 DBD > 1 DBS = S + T = control. Meanwhile, U. ruziziensis had greatest biomass production in the treatment with N applied 20 DBD and the lowest in the treatment with N applied 1 DBS. At 90 DAD, U. brizantha straw remaining was greater than U. ruziziensis only when N was applied at 20 DBD, at 10 DBD, and at 5 DBD (Fig. 1b). In the S + T and control (no N application) treatments, DM of U. brizantha was lower than that of U. ruziziensis (0.60 and 0.71 Mg ha−1, respectively), whereas at 1 DBS there was no difference between species in cover crop DM remaining.
Shoot biomass (a and b) and N (c and d), P (e and f), and K (g and h) contents of cover crops at 0 (a, c, e, g) and 90 (b, d, f, h) days after desiccation (DAD) as affected by N management system and cover crop species in a field study in Botucatu, State of São Paulo, Brazil. Data are means of two growing seasons. Different lowercase letters denote significant difference between cover crops and different uppercase letters denote significant difference among N management systems (LSD, P < 0.05). Control = no N application; 20 DBD = 100 kg N ha−1 broadcast over cover crop 20 days before desiccation; 10 DBD = 100 kg N ha−1 broadcast over cover crop 10 days before desiccation; 5 DBD = 100 kg N ha−1 broadcast over cover crop 5 days before desiccation; 1 DBS = 100 kg N ha−1 broadcast over desiccated cover crop 1 day before soybean sowing; S + T = 40 kg N ha−1 at soybean sowing plus 60 kg N ha−1 topdressing at V5
For all macronutrients (N, P, K, Ca, Mg, and S) at 0 DAD, U. brizantha had greater accumulation than U. ruziziensis in all N management systems, including the control (Figs. 1c, e, g, 2a, c, e). Nitrogen and K had greatest contents of all elements in both cover crops, leading to large sources of N and K recycled (Fig. 1c, g). For both cover crops, the treatment with N applied at 20 DBD also had greater macronutrient content than all other N management systems (Figs. 1c, e, g, 2a, c, e).
Ca (a and b), Mg (c and d), and S (e and f) contents of cover crops at 0 (a, c, and e) and 90 (b, d, and f) days after desiccation (DAD) as affected by N management system and cover crop species in a field study in Botucatu, State of São Paulo, Brazil. Data are means of two growing seasons. Different lowercase letters denote significant difference between cover crops and different uppercase letters denote significant difference among N management systems (LSD, P < 0.05). Control = no N application; 20 DBD = 100 kg N ha−1 broadcast over cover crop 20 days before desiccation; 10 DBD = 100 kg N ha−1 broadcast over cover crop 10 days before desiccation; 5 DBD = 100 kg N ha−1 broadcast over cover crop 5 days before desiccation; 1 DBS = 100 kg N ha−1 broadcast over desiccated cover crop 1 day before soybean sowing; S + T = 40 kg N ha−1 at soybean sowing plus 60 kg N ha−1 topdressing at V5
At 90 DAD, N, Ca, and S contents were greater in U. brizantha straw than in U. ruziziensis straw when N was applied at 20 DBD, 10 DBD and 5 DBD (Figs. 1d, 2b, f). Likewise, U. brizantha straw had greater P, K, and Mg contents than U. ruziziensis straw at 1 DBS (Figs. 1f, h, 2d). However, N and Ca contents were not different between cover crops when N was applied at 1 DBS (Figs. 1d, 2b). The lowest macronutrient content of U. brizantha was when the N management was S + T and without N (control).
On average across cover crops and N management system, the 2013–2014 cover-crop growing season had greater DM biomass and N, K, Ca, and Mg contents than the 2014–2015 at 0 DAD, but the reverse was true for P and S contents (Table 4). At 90 DAD, the remaining amounts of DM and macronutrients were greater in the 2014–2015 cover-crop growing season than in the 2013–2014 season.
Nutrient concentrations in soybean leaves
Nutrient concentrations in soybean leaves were affected by growing season, but there was no effect of cover crop species, N management system, and interaction among factors (Table 5), except for Mg concentration. The system without N addition had lower Mg concentration than all other treatments with N addition. Greatest N, K, and Ca concentrations in soybean leaves were found in the first growing season and greatest P, Mg, and S concentrations were found in the second growing season (Table 5).
Plant height, shoot dry matter, yield components and grain yield of soybean
Soybean plant height, shoot DM, plant population, number of pods per plant, and grain yield were lower in 2013–2014 than in 2014–2015 (Table 6), likely due to differences in rainfall with significant drought periods in the vegetative stage of soybean in 2013–2014 (Table 1). Plant height was affected by N management system and growing season (Table 6). Plant height was greater with all N-fertilized treatments than the control. Soybean plant height was greater in the wetter 2014–2015 growing season than the drier 2013–2014 growing season. Soybean shoot DM was not affected by cover crop species or N management system (Table 6). Shoot DM was greater in the wetter 2014–2015 growing season than the drier 2013–2014 growing season.
Soybean plant population and number of pods per plant were greater following U. brizantha than following U. ruziziensis (Table 6). Neither response variable was affected by N management system. Both plant population and number of pods were greater in the wetter 2014–2015 growing season than the drier 2013–2014 growing season (Table 1). Number of grains per pod was unaffected by any factor. The 100-grain weight was unaffected by cover crop species and N management system, but was lower in the wetter 2014–2015 growing season than the drier 2013–2014 growing season (Table 6). Soybean grain yield was 17% greater following U. brizantha than following U. ruziziensis (Table 6). Grain yield was unaffected by N management system. Soybean grain yield was 39% greater in the wetter 2014–2015 growing season than the drier 2013–2014 growing season.
Discussion
Cover crop biomass production, accumulation, and remaining amounts of nutrients
In the 2013–2014 cover-crop growing season, rainfall distribution was uniform and favored cover-crop growth and development (Table 1). In the 2014–2015 cover-crop growing season, there were periods of drought that limited biomass production. Rainfall differences explained differences in amount of cover crop biomass production and nutrient accumulation (Figs. 1, 2).
Nitrogen fertilizer application before desiccation stimulated greater growth rate and shoot biomass production (Fig. 1a). Greatest shoot biomass production occurred when N was applied 20 DBD, which could be attributable to longer time between N application and cover crop desiccation. Both cover crops had less biomass production the shorter the time they were supplied with N. Difference in biomass production between species at the same N treatment was likely due to U. brizantha being more N-use efficient than U. ruziziensis (Alvim et al. 1990).
Greater shoot biomass production with U. brizantha than with U. ruziziensis in control (no N application) (Fig. 1a) could be attributable to greater root production and its propensity for high mulching capacity, although self-shading could be an issue in high biomass conditions (EMBRAPA 2002). Pacheco et al. (2011, 2013) observed production of 9.7–11.4 Mg ha−1 of U. brizantha straw and 6.7–7.0 Mg ha−1 of U. ruziziensis straw without N supply. Cover crop shoot biomass production in our study was greater than in previous studies with U. brizantha [6.3 Mg ha−1 (Crusciol and Soratto 2007); 10.3 Mg ha−1 (Simidu et al. 2010)] and with U. ruziziensis [8.6 Mg ha−1 (Menezes et al. 2009); 7.0 Mg ha−1 (Pacheco et al. 2011)]. Although U. ruziziensis produced lower shoot DM than with U. brizantha in the control treatment, the amount of straw was greater than considered necessary to control runoff (4 Mg ha−1) (Lopes et al. 1987). Moreover, the amount of straw with both cover crop species in all N treatments, including the control without N, was sufficient to provide full coverage of soil (> 7 Mg ha−1) (Kluthcouski et al. 2003).
Although U. brizantha produced more biomass than U. ruziziensis (0 DAD) regardless of N management, the remaining straw (90 DAD) of U. ruziziensis was equal to U. brizantha when N was applied 1 DBS, and greater than U. brizantha when N was applied as S + T and without N application (control) (Fig. 1b). These results might be linked to the amount of N in the biomass on 0 DAD (Fig. 1c) since straw decomposition is associated with chemical composition of biomass (Xu and Hirata 2005). Nitrogen application to cover crops before desiccation can also alter the chemical composition of biomass (Costa et al 2016) and its decomposition by microorganisms. Bacterial and fungal communities mineralize and release nutrients (N, P, K, Ca, and Mg) of cover crops via decomposition processes (Christensen 1989; Glassman et al. 2018). In addition, rate of straw decomposition is determined by biotic and abiotic factors at the soil surface (Espindola et al. 2006), e.g., water availability and temperature (Parton et al. 2007), inorganic N supply (Mary et al. 1996), and C/N ratio of plant residues (Carvalho et al. 2008; Torres and Pereira 2008). Due to greater remaining biomass of U. ruziziensis (90 DAD), macronutrient contents in the straw of U. brizantha were lower than in U. ruziziensis in the S + T and control treatments (Figs. 1d, f, h, 2b, d, f).
Application of N on U. brizantha before desiccation increased nutrient accumulation with biomass production (Figs. 1, 2). Macronutrient contents (N, P, K Ca, Mg, and S) at 0 DAD were greater when N was applied 20 DBD in U. brizantha than in U. ruziziensis due to greater biomass production. This result occurred because when plants are well supplied with N, plants have greater development of root system to support shoot development and uptake of other nutrients (Brouwer 1962). Even when N was not supplied to the system, U. brizantha had greater accumulation of nutrients than U. ruziziensis because of differences between species and biomass production. Pacheco et al. (2013) also studied the accumulation of nutrients in U. brizantha and U. ruziziensis and observed no differences in N, P, K, Ca, and Mg accumulation. Authors attributed this to lack of difference in shoot biomass production. Pariz et al. (2011b) also observed greater straw inputs of N, P, and K to the soil surface by U. brizantha than with Megathyrsus maximus (Jacq.) B.K. Simon & S.W.L. Jacobs (syn. Panicum maximum Jacq.) cv. Tanzânia, M. maximum cv. Mombaça and U. ruziziensis. They reported that use of these grasses over the winter-spring have a great potential to cover the soil surface in the Brazilian Savanna. In both cover crops, the most extracted nutrients were N and K, similar to that reported by Torres et al. (2008) and Pacheco et al. (2011, 2013). Additionally, cover crops, such as Urochloa and other grasses, have deep root systems that can reach 1 m deep, and consequently, can take up nutrients otherwise lost by leaching and return them to the soil surface (Crusciol et al. 2015).
When N was applied before desiccation, U. brizantha accumulated greater amounts of N, P, K, Ca, and Mg than U. ruziziensis as a consequence of greater biomass production (Figs. 1, 2). However, at 90 DAD, lower quantities of N, P, K, Ca, Mg, and S remained in U. brizantha straw than in U. ruziziensis straw in the S + T and control treatments. These results can be attributed to the high decomposition rate of substrates, which was dependent on biomass amount, rainfall, temperature, and soil microbial activity (Sinsabaugh et al. 2015; Bell et al. 2018). Nutrient release from cover crop biomass can be altered by growth and decay of environment-specific microbial communities (Christensen 1989; Varela et al. 2017). Production and decomposition of biomass influence the availability of nutrients in soil (N’Dri et al. 2018), and therefore, our results suggest that well-fertilized U. brizantha may release greater quantities of nutrients to the following crop. More detailed studies are needed to determine cover crop-specific decomposition rates and nutrient availability in soil.
Nutrient concentration in soybean leaves
All nutrient concentrations were within or near the ranges considered suitable for soybean production (Table 5), according to Ambrosano et al. (1997). Nutrient concentrations in soybean leaves ranged from medium to high (van Raij et al. 1997) (Table 2). Nutrients appeared to cycle effectively through cover crops via decomposition and leaching (Figs. 1, 2), as well as supplied from fertilizer and soil. Silva et al. (2011) showed that inorganic fertilization of 20 kg N ha−1 reduced nodulation and efficiency of BNF. However, in the present study it is possible that much of the N applied had been temporarily immobilized by soil microorganisms decomposing the large amount of straw on the soil surface, thereby not altering the N supply to soybean. The lack of difference in soybean leaf N concentration among N management systems, even with disparate N timing from 50 DBS to the day of sowing, suggests that inorganic N availability may not have affected BNF. Greater N, K, and Ca concentrations in soybean leaves in the first growing season and greater P, Mg, and S concentrations in the second growing season (Table 5) were related with nutrient contents in cover crop biomass in the respective growing seasons (Figs. 1, 2), except for Mg. Greater Mg concentration with N application may have been due to reduced soil moisture with high biomass yield, C/N ratio, and root-microbial activity, all of which have been shown to increase plant-available Mg release from soils (Mayland and Wilkinson 1989; Senbayram et al. 2015). Soybean leaf Mg concentration without N fertilizer application had a value considered adequate for crop growth, i.e. in the range of 3.0–10.0 g kg−1 for soybean (Ambrosano et al. 1997).
Plant height, dry matter, yield components, and grain yield of soybean
Low rainfall in 2013–2014 adversely affected soybean growth and nutrient uptake (Table 5). Despite lower grain yield in the 2013–2014 growing season (2.5 Mg ha−1), it was close to the national average (2.8 Mg ha−1) (CONAB 2015).
Soybean grain yield was a reflection of the results observed in plant population and number of pods per plant, which had greater values when following U. brizantha than following U. ruziziensis (Table 6). Greater amount of straw left on the soil surface by U. brizantha may have favored the establishment of soybean plants and pod formation under adverse weather conditions during critical development stages (Table 1; Figs. 1, 2). Number of pods per plant is one of the main yield components determining grain yield (Carpentieri-Pípolo et al. 2005). Pacheco et al. (2013) did not find a difference in soybean grain yield when comparing the two forages grasses as cover crops, despite slower nutrient release with U. brizantha than with U. ruziziensis.
Soybean height was the only plant trait affected by N management system, which was 10% lower in the control without N compared with all other N treatments (Table 6). This response reflected N availability to soybean at an early growth stage, and consequently, plant height was not a critical factor for grain yield. Similar results were inferred by Silva et al. (2011) in a fallow area with conventional tillage system, in which N fertilization at sowing (24 kg N ha−1) led to greatest plant height of 0.74 m. Pereira et al. (2010) also reported increased plant height with N application than without.
Lack of N management effects on shoot DM, yield components, and grain yield of soybean (Table 6) corroborate previous results. Aratani et al. (2008) found that N application in soybean did not influence yield components and grain yield. Thus, in soils with efficient nodulation, use of N fertilizer has no effect on grain yield, but may decrease nodulation and efficiency of BNF (EMBRAPA 2011). In contrast, Bahry et al. (2013) demonstrated that N application in the reproductive stage was effective to increase soybean grain yield, since from this stage the efficiency of BNF begins to decrease.
This study showed that the use of either Urochloa species as a cover crop in the winter dry season of the Brazilian Savanna would be a viable option for farmers to diversify their operations for more sustainable soybean production (Crusciol et al. 2015). In addition, N application to the forage grass or to the soybean following them did not improve soybean grain yield (Table 6). However, N application to grass cover crops before desiccation could benefit soil fertility and organic matter accumulation in the long-term to promote even greater cycling of nutrients in the system. Economic impacts of such long-term application strategies would also be needed to make effective recommendations.
Conclusions
Despite large shoot biomass production and nutrient accumulation from both Urochloa cover crops, soybean grain yield was greater following U. brizantha than following U. ruziziensis. Regardless of whether or when N was applied, soybean leaf nutrient concentrations, yield components, and grain yield were unaffected. However, it is noteworthy that application of N to cover crops before desiccation resulted in greater nutrient accumulation in cover crop biomass, and consequently, improved nutrient cycling. However, this greater nutrient accumulation and cycling had little consequence on subsequent soybean grain yield, at least in this short-term evaluation. U. brizantha was considered superior to U. ruziziensis as a cover crop to promote greater nutrient cycling, increase food production, and allowing farmers to produce in a sustainable way.
References
Alvim MJ, Botrel MA, Verneq RS, Salvati JA (1990) Nitrogen application in access to Brachiaria. 1-Dry matter production effect. Past Trop 2:1–5 (In Portuguese)
Ambrosano EJ, Tanaka RT, Mascarenhas AA, van Raij B, Quaggio JA, Cantarella H (1997) Leguminosas e Oleaginosas. In: van Raij B, Cantarela H, Quaggio JA, Furlani AMC (eds) Boletim Técnico 100, Recomendações de adubação e calagem para o Estado de São Paulo, 2nd ed. Instituto Agronômico de Campinas (IAC), Campinas, SP, Brazil, pp 189–195 (In Portuguese)
Aratani RG, Lazarini E, Marques RR, Backes C (2008) Nitrogen Fertilization in soybean in no tillage system introduction. Biosc J 30:31–38 (In Portuguese)
Assmann JM, Martins AP, Anghinoni I, Denardim LGO, Nichel GH, Costa SEVGA, Silva RAP, Balerini F, Carvalho PCF, Franzluebbers AJ (2017) Phosphorus and potassium cycling in a long-term no-till integrated soybean-beef cattle production system under different grazing intensities in subtropics. Nutr Cycl Agroecosyst 108:21–33. https://doi.org/10.1007/s10705-016-9818-6
Bahry CA, Venske E, Nardino M, Fin SS, Zimmer PD, Souza VQ, Caron BO (2013) Morphological traits and soybean yield components subjected to nitrogen fertilization (In Portuguese, with English abstract). Agrarian 21:281–288. https://doi.org/10.30612/agrarian.v6i21.2240
Bell MC, Ritson JP, Verhoef A, Brazier RE, Templeton MR, Graham NJD, Freeman C, Clark JM (2018) Sensitivity of peatland litter decomposition to changes in temperature and rainfall. Geoderma 331:29–37. https://doi.org/10.1016/j.geoderma.2018.06.002
Brouwer R (1962) Nutritive influences on the distribution of dry matter in the plant. Neth J Agri Sci 10:399–408
Caires EF, Haliski A, Bini AR, Scharr DA (2015) Surface liming and nitrogen fertilization for crop grain production under no-till management in Brazil. Eur J Agron 66:41–63. https://doi.org/10.1016/j.eja.2015.02.008
Calonego JC, Gil FC, Rocco VF, dos Santos EA (2012) Persistence and nutriente release from maize, brachiaria and lablab straw. (In Portuguese, with English abstract.). Biosc J 28:770–781
Carpentieri-Pípolo V, Gastaldi LF, Pipolo AE (2005) Phenotypic correlations between quantitative characteristics in soybean. (In Portuguese, with English abstract.). Semina: Cienc Agrar 26:11–16
Carvalho AM, Bustamante MMC, Sousa Junior JGA, Vivaldi LJ (2008) Decomposition of plant residues in latosol under corn crop and cover crops. (In Portuguese, with English abstract.). Rev Bras Ciên Solo 32:2831–2838. https://doi.org/10.1590/S0100-06832008000700029
Christensen M (1989) A view of fungal ecology. Mycologia 81:1–19
CONAB—National Supply Company (2018) 5th Survey—2017–2018 harvest: grains. Harvest surveys. https://www.conab.gov.br/index.php/info-agro/safras/graos/boletim-da-safra-de-graos/item/download/12569_5b3e0e675171f49a5b1e9215edc1064a/. Accessed 03 Feb 2018
CONAB—National Supply Company (2015) 9th Survey—2014–2015 harvest: grains. Harvest surveys. https://www.conab.gov.br/index.php/info-agro/safras/graos/boletim-da-safra-de-graos/item/download/1290_0c593ee0b2d2259f8e84778987f08a51/. Accessed 14 Feb 2017
Costa CHM, Crusciol CAC, Soratto RP, Ferrari Neto J, Moro E (2016) Nitrogen fertilization on palisadegrass: phytomass decomposition and nutrients release. Pesq Agropec Trop 46:159–168. https://doi.org/10.1590/1983-40632016v4639297
Crusciol CAC, Soratto RP (2007) Peanut crop nutrition and yield in no-tillage system in succession to cover crops growth (In Portuguese, with English abstract). Pesq Agrop Bras 42:1553–1560. https://doi.org/10.1590/S0100-204X2007001100006
Crusciol CAC, Cottica RL, Lima EV, Andreotti M, Moro E, Marcon E (2005) Persistence and nutrients release of forage turnip straw utilized as mulching in no-tillage crop system (In Portuguese, with English abstract). Pesq Agropec Bras 40:161–168. https://doi.org/10.1590/S0100-204X2005000200009
Crusciol CAC, Nascente AS, Borghi E, Soratto RP, Martins PO (2015) Improving soil fertility and crop yield in a tropical region with palisadegrass cover crops. Agron J 107:2271–2280. https://doi.org/10.2134/agronj14.0603
EMBRAPA—Empresa Brasileira de Pesquisa Agropecuária (2002) Fixação biológica de nitrogênio associada a pastagens de braquiária e outras gramíneas forrageiras. https://ainfo.cnptia.embrapa.br/digital/bitstream/CPAC-2009/24678/1/doc_52.pdf/. Accessed 10 Feb 2017
EMBRAPA—Empresa Brasileira de Pesquisa Agropecuária (2011) Centro Nacional de Pesquisa de soja. Tecnologias de produção de soja – região central do Brasil 2012 e 2013. Londrina, PR, Brazil (In Portuguese)
Espindola JAA, Guerra JGM, Almeida DL, Teixeira MG, Urquiaga S (2006) Decomposition and nutrient release of perennial herbaceous legumes intercropped with banana. (In Portuguese, with English abstract.). Rev Bras Ciên Solo 30:321–328. https://doi.org/10.1590/S0100-06832006000200012
FAO—Food and Agriculture Organization of the United Nation (2015) Status of the world’s soil resources: main report. http://www.fao.org/3/a-i5199e.pdf/. Accessed 10 Feb 2017
Ferreira DF (2011) Sisvar: a computer statistical analysis system. Cienc Agrotec 35:1039–1042
Ferreira EVO, Anghinini I, Andrighetti M, Martins AP, Carvalho PFC (2011) Potassium cycling and balance and soybean yield in an integrated crop-livestock system under no-till. Rev Bras Cienc Solo 35:161–169. https://doi.org/10.1590/S0100-06832011000100015
Fisher MJ, Kerridge PC (1998) The agronomy and physiology of Brachiaria species. In: Miles JW, Maass BL, do Valle CB (eds) Brachiaria: Biology, agronomy, and improvement. International Center for Tropical Agriculture (CIAT), Cali, Colombia, pp 43–52
Franzluebbers AJ, Hons FM, Zuberer DA (1995) Tillage and crop effects on seasonal soil carbon and nitrogen dynamics. Soil Sci Soc Am J 59:1618–1624
Franzluebbers AJ, Sawchik J, Taboada MA (2014) Agronomic and environmental impacts of pasture-crop rotations in temperate North and South America. Agr Ecosyst Environ 190:18–26. https://doi.org/10.1016/j.agee.2013.09.017
Gichangi EM, Njarui BMG, Gatheru M (2017) Plant shoots and roots biomass of Brachiaria grasses and their effects on soil carbon in the semi-arid tropics of Kenya. Trop Subtrop Agroecosyst 20:65–74
Glassman SI, Weihe C, Li J, Albright MBN, Looby CI, Martiny AC, Treseder KK, Allison SD, Martiny JBH (2018) Decomposition responses to climate depend on microbial community composition. PNAS 115:47. https://doi.org/10.1073/pnas.1811269115
Herridge DF, Peoples MP, Boddey RM (2008) Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 311:1–18. https://doi.org/10.1007/s11104-008-9668-3
Kluthcouski J, Stone LF, Aidar H (2003) Integracao lavoura-pecuaria. http://livraria.sct.embrapa.br/liv_resumos/pdf/00075060.pdf. Accessed 10 Feb 2017
Kowalchuck GA, Stephen JR, De Boer W, Prosser JI, Embley TM, Woldendorp JW (1997) Analysis of β-proteobacteria ammonia-oxidizing bacteria in coastal and sand dunes using denaturing gradient gel electrophoresis and sequencing of PCR amplified 16S rDNA fragments. Appl Environ Microbiol 63:1489–1497
Lal R, Stewart BA (1990) Soil degradation: a global threat. Adv Soil Sci 11:13–17
Lopes PRC, Cogo NP, Levien R (1987) Eficácia relativa de tipo e quantidade de resíduos culturais espalhados uniformemente sobre o solo na redução da erosão hídrica. Rev Bras Ciên Sol. 1:71–75 (In Portuguese)
Malavolta E, Vitti GC, Oliveira SA (1997) Avaliação do estado nutricional das plantas: princípios e aplicações, 2nd edn. POTAFOS, Piracicaba (In Portuguese)
Mary B, Recous S, Darwis D, Robin D (1996) Interaction between decomposition of plant residues and nitrogen and nitrogen cycling in soil. Plant Soil 181:71–82. https://doi.org/10.1007/BF00011294
Mascarenhas HAA, Tanaka RT (1997) Soja. In: van Raij B, Cantarella H, Quaggio JA, Furlani AMC (eds) Boletim Tecnico 100, Recomendações de adubação e calagem para o Estado de São Paulo, 2nd ed. Instituto Agronômico de Campinas (IAC), Campinas, SP, Brazil, pp 202–203 (In Portuguese)
Mayland HF, Wilkinson SR (1989) Soil factors affecting magnesium availability in plant–animal systems: a review. J Anim Sci 67:3437–3444
Menezes LAS, Leandro WM, Oliveira Juniro JP, Ferreira ACB, Santana JG, Barros RG (2009) Yield of plant mass of different species, single and mixed, with potential to coverage of the soil. (In Portuguese, with English abstract.). Biosci J 25:7–12
Moraes A, Carvalho PCF, Anghinoni I, Lustosa SBC, Costa SEVGA, Kunrath TR (2014) Integrated crop–livestock systems in the Brazilian subtropics. Europ J Agron 57:4–9. https://doi.org/10.1016/j.eja.2013.10.004
N’Dri JK, Guei AM, Edoukou EF, Yeo JG, N’Guessan KK, Lagerlof J (2018) Can litter production and litter decomposition improve soil properties in the rubber plantations of different age in Cte d’Ivoire? Nutr Cycl Agroecosys 111:203–215. https://doi.org/10.1007/s10705-018-9923-9
Nascente AS, Crusciol CAC (2012) Cover crops and herbicide timing management on soybean yield under no-tillage system. Pesqui Agropecu Bras 47:187–192. https://doi.org/10.1590/S0100-204X2012000200006
Nogueira PDM, Sena Junior DG, Ragagnin VA (2010) Leaf chlorophyll and nodulation in soybean with sidedress nitrogen fertilization. (In Portuguese, with English abstract.). Gl Sci Technol 3:117–124
Pacheco LP, Leandro WM, Machado PLOA, Assis RL, Cobucci T, Madari BE, Petter FA (2011) Biomass production and nutrient accumulation and release by cover crops in the off-season. (In Portuguese, with English abstract.). Pesq Agropec Bras 46:17–25. https://doi.org/10.1590/S0100-204X2011000100003
Pacheco LP, Monteiro MMS, Silva RF, Soares LS, Fonseca WL, Nobrega JCA, Petter FA, Alcantra Neto F, Osajima JA (2013) Biomass production and nutrient accumulation by cover crops in the Brazilian Cerrado of Piaui State. (In Portuguese, with English abstract.). Bragantia 72:237–246. https://doi.org/10.1590/brag.2013.041
Pariz CM, Andreotti M, Azenha MV, Bergamaschine AF, Mello LMM, Lima RC (2011a) Corn grain yield and dry mass of Brachiaria intercrops in the crop–livestock integration system. Cienc Rural 41:875–882. https://doi.org/10.1590/S0103-84782011000500023
Pariz CM, Andreotti MV, Buzatti S, Bergamaschine AF, Ulian NA, Furlan LC, Meirelles PRL, Cavasano FA (2011b) Straw decomposition of nitrogen-fertilized grasses intercropped with irrigated maize in an integrated crop-livestock system. Rev Bras Cien Solo 35:2029–2037. https://doi.org/10.1590/s0100-06832011000600019
Parton W, Silver WL, Burke IC, Grassens L, Harmon ME, Currie WS, King JY, Adair EC, Brandt LA, Hart SC, Fasth B (2007) Global-scale similarities in nitrogen release patterns during long-term decomposition. Science 315:361–364. https://doi.org/10.1126/science.1134853
Pereira VJ, Rodrigues JF, Gomes RR, Filho JMRR (2010) Performance of soybean (Glycine max (L.) Merrill) submitted to nitrogen of planting. (In Portuguese, with English abstract.). Encic Bios: Centro Científico Conhecer 6:1–5
Petter FA, Pacheco LP, Alcântara Neto F, Santos GG (2012) Responses of soybean cultivars to nitrogen fertilizer late in Cerrado soils. (In Portuguese, with English abstract.). Rev Caatinga 25:67–72
Salvagiotti F, Cassman KG, Specht JE, Walters DT, Weiss A, Dobermann A (2008) Nitrogen uptake, fixation and response to fertilizer N in soybeans: a review. Field Crops Res 108:1–13. https://doi.org/10.1016/j.fcr.2008.03.001
Santi A, Amado TJC, Acosta JAA (2003) Black oat biomass and nutrient cycling as affected by nitrogen fertilization in soil under no-tillage. (In Portuguese, with English abstract.). Rev Bras Ciên Solo 27:1075–1083
Sarathchandra SV (1978) Nitrification activies and the changes in the population of nitrifying bacteria in soil perfused with two different H-ion concentrations. Plant Soil 50:99–111
Senbayram M, Gransee A, Wahle V, Thiel H (2015) Role of magnesium fertilisers in agriculture: plant–soil continuum. Crop Pasture Sci 66:1219–1229. https://doi.org/10.1071/CP15104
Silva AF, Carvalho MAC, Schoninger EL, Monteiro S, Caione G, Santos PA (2011) Doses of inoculant and nitrogen at sowing of soybean in firts cultivation area. Biosc J 27:404–412
Simidu HM, de Sá ME, Souza LCD, Lima Abrantes F, Silva MP, Arf O (2010) Effect of green manure and sowing date on the productivity of bean no-tillage in the Cerrado region. Acta Sci Agron 32:309–315. https://doi.org/10.4025/actasciagron.v32i2.2061
Sinsabaugh RL, Belnap J, Rudgers J, Kuske CR, Martinez N, Sandquist D (2015) Soil microbial responses to nitrogen addition in arid ecosystems. Front Microb 6:819. https://doi.org/10.3389/fmicb.2015.00819
Torres JLR, Pereira MG (2008) Potassium dynamics in crop residues of cover plants in Cerrado. (In Portuguese, with English abstract.). Rev Bras Ci Solo 32:1609–1618. https://doi.org/10.1590/S0100-06832008000400025
Torres JLR, Pereira MG, Fabian AJ (2008) Cover crops biomass production and its residues mineralization in a Brazilian no-till Oxisol. (In Portuguese, with English abstract.). Pesq Agropec Bras 43:421–428
Treusch AH, Leininger S, Kletzin A, Schuster SC, Klent HP, Schleper C (2005) Novel genes for nitrite reductase and AMO related proteins indicate a role of uncultivated mesophilic crenarchaeota in nitrogen cycling. Environ Microbiol 7:1985–1995. https://doi.org/10.1111/j.1462-2920.2005.00906.x
van Raij B, Cantarella H, Quaggio JA, Furlani AMC (1997) Boletim Tecnico 100, Recomendações de adubação e calagem para o Estado de São Paulo. Intituto Agronomico de Campinas (IAC), Campinas (In Portuguese)
van Raij B, Andrade JC, Cantarella H, Quaggio JA (2001) Análise química para avaliação da fertilidade de solos tropicais. IAC, Campinas (In Portuguese)
Varela MF, Barraco M, Gili A, Taboada MA, Rubio G (2017) Biomass decomposition and phosphorus release from residues of cover crops under no-tillage. Agron J 109:317–326. https://doi.org/10.2134/agronj2016.03.0168
Wedin DA, Russelle MP (2007) Nutrient cycling in forage production. In: Barnes RF, Nelson CJ, Moore KJ, Collins M (eds) Forages: the science of grasslands agriculture, 6th edn. Wiley, Ames, pp 137–148
Xu X, Hirata E (2005) Decomposition patterns of leaf litter of seven common canopy species in a subtropical forest: N and P dynamics. Plant Soil 273:279–289. https://doi.org/10.1007/s11104-004-8069-5
Acknowledgements
The authors would like to thank the Coordination of Improvement of Higher Education Personnel (CAPES) for the financial support, as well as the National Council for Scientific and Technological Development (CNPq) for an award for excellence in research to the second, third, and eighth authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tanaka, K.S., Crusciol, C.A.C., Soratto, R.P. et al. Nutrients released by Urochloa cover crops prior to soybean. Nutr Cycl Agroecosyst 113, 267–281 (2019). https://doi.org/10.1007/s10705-019-09980-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10705-019-09980-5