Abstract
Enzyme activity was evaluated in the intestine of juvenile pacu, Piaractus mesopotamicus, fed diets containing 0, 10 or 20 % of lyophilized bovine colostrum (LBC) inclusion for either 30 or 60 days. The enzymes intestinal acid and alkaline phosphatase (ACP and ALP, respectively), nonspecific esterase (NSE), lipase (LIP), dipeptidyl aminopeptidase IV (DAP IV) and leucine aminopeptidase (LAP) were studied using histochemistry in four intestinal segments (S1, S2, S3 and rectum). Moderate activity of the DAP IV was detected in the three last intestinal segments, but no differences among the treatments were detected. Enzymes LAP, NSE and LIP were weakly stained in all intestinal segments and the inclusion of 10 or 20 % of LBC in the diet commanded a moderate reaction to NSE in the S3 segment at day 60. ACP activity was detected only in the brush border of the S1 segment of fish fed 0 % LBC for either 30 or 60 days. The activity of ALP was very strong in the first intestinal segment, but a weak reaction was seen in the last segments. The inclusion of 20 % of LBC changed the pattern of staining to the ALP, eliciting moderate staining in S2 at day 30 and S1 at day 60. The consumption of diets containing LBC by juvenile pacu did not have significant implications in intestinal enzymatic activity, which still was not fully stimulated.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bovine colostrum is a dietary source of lipids, carbohydrates, proteins, vitamins and minerals, having also high concentrations of hormones and growth factors, such as the insulin-like growth factor type I and the epidermal growth factor, both associated with calf gastrointestinal growth and development (Pandey et al. 2011). Studies have been carried out on immunological and nutritional properties of bovine colostrum as feed supplement to sheep, goats, pigs, rodents, and more recently by our group, fish (Huguet et al. 2006; Lima et al. 2009; Rodrigues et al. 2009; Moretti et al. 2010, 2012; Bodammer et al. 2011).
The administration of biologically active substances, as present in colostrum, is an important area to be considered in aquaculture. This method can be implemented due to a unique characteristic of teleost, the ability of macromolecules internalization by the enterocytes in the posterior intestine for a prolonged period (Schep et al. 1999). However, enzyme activity can hamper intestinal absorption and make this process less efficient (Schep et al. 1999; Barbieri and Hernández-Blazquez 2002). In the omnivorous Nile tilapia (Oreochromis niloticus), the greater enzyme activity in the first four segments of the intestine indicates that the initial portions of the tract are the most important for digestion and absorption of nutrients (Tengjaroenkul et al. 2000).
The pacu (Piaractus mesopotamicus, Holmberg 1887), an intensively farmed Neotropical characin, forages on leaves, fruits, crabs, small fish and insects in the wild (Pelli et al. 1997; Urbinati and Gonçalves 2005). The intestinal digestive physiology of pacu is ostensibly complex and has not been completely characterized. This study investigated the distribution of selected intestinal enzymes of juvenile pacu fed diets with lyophilized bovine colostrum (LBC).
Materials and methods
Farm-raised, feed-conditioned juvenile pacu (8.5 ± 0.7 g and 7.8 ± 0.3 cm) were stocked in 300 L, covered plastic cages, in a closed loop, continuous water circulation and aeration system (temperature 26.8 ± 1.5 °C; pH 7.9 ± 0.3, dissolved oxygen 5.8 ± 1.0 mg L−1; dissolved ammonia: <0.05 mg L−1). Fish were assigned to experimental groups in a totally randomized design (n = 3; 18 fish per cage), 3 × 2 factorial scheme with three diets (0, 10 and 20 % of LBC) and two feeding periods (30 and 60 days) Fish were adapted to the experimental diets for 10 days and then hand-fed pelleted experimental diets to apparent satiety twice a day (08h30 and 16h30). Fish were kept, maintained and treated according to accepted standards for the humane treatment of animals (authorized by the ESALQ/USP ethics committee).
The bovine colostrum was collected from multiparous Holstein cows of commercial dairy farms. Animals were milked manually and lacteal secretions were stored in sterile containers. Next, the colostrum was homogenized to form a unique pool of bovine colostrum and stored at −20 °C. The frozen pool was lyophilized and resulting powder homogenized and stored in sealed containers at −20 °C until addition in the diet.
Formulation of experimental pellet diets, isonitrogenous and isoenergetic, and their centesimal composition are presented in Table 1. Experimental diets and LBC were submitted to chemical analysis according to AOAC (2000) methods.
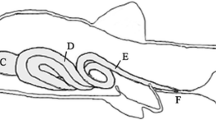
Seven juveniles per treatment were sampled at each mentioned date for tissue collection. Sampling routine included 24 h of fasting and anesthesia with a benzocaine medium (0.1 g L−1). After slaughter by cervical separation, the abdominal cavity was opened, and the gastrointestinal tract removed within 5–10 min. Intestinal tracts (pyloric sphincter to anus) were then isolated, separated into middle intestine and rectum by identification of the ileum-rectal valve (Bértin 1958), and the former divided into three segments: S1 (anterior section), S2 (middle section) and S3 (posterior section), Fig. 1. Intestinal samples were collected for histochemical analysis of cellular enzyme activity. The tissues were fixed in 4 % phosphate buffered paraformaldehyde solution, dehydrated with solutions of ethanol (30, 50, 70, 90 and 100 %) and, thereafter, embedded in glycol methacrylate (JB-4, Polysciences, Inc., Warrington, PA, USA). For each fish and segment, four sections of 5 µm thickness were taken.
The activity of dipeptidil aminopeptidase IV (DAP IV) was evaluated in sections incubated for 50 min at room temperature in a solution containing 3 mg of Gly-Pro-4-methoxy-2-naphthylamine dispersed in 500 µL of N,N-dimethylformamide added to 10 mg of fast blue B in 10 mL of sodium phosphate buffer (0.1 M, pH 7.4). The slides were then washed with distilled water, immersed in 2 % copper sulfate for 5 min, washed again and post-stained (Lodja 1979).
The activity of leucine aminopeptidase (LAP) was evaluated in sections incubated for 120 min at room temperature in a solution containing 5 mL of acetate buffer (0.1 M, pH 6.5), 4 mL of 0.85 % sodium chloride, 0.5 mL of enzyme substrate solution (0.08 % l-leucyl-4-methoxy-β-naphthylamide diluted in 2 % ethyl alcohol solution), 0.5 mL of distilled water and 5 mg of fast blue B salt (Bancroft 1996). The slides were then washed with sodium chloride solution, immerged in copper sulfate (0.1 M) for 2 min, washed again and post-stained.
Nonspecific esterase (NSE) activity was evaluated by the naphthyl acetate method (Bancroft 1996). Sections were incubated for 10 min at 37 °C in a solution containing 0.25 mL of 4 % sodium nitrite, disodium hydrogen phosphate buffer (0.2 M), pararosanilin-HCl solution (4 % pararosanilin hydrochloric in hydrochloric acid 2 M), and enzyme substrate (1 % α-naphthyl acetate in acetone), washed with distilled water and post-stained.
The activity of lipase (LIP) was evaluated by the tween method (Bancroft 1996). Sections were incubated for 90 min at 37 °C in a medium solution containing 9 mL of Tris buffer (0.05 M, pH 7.2), 0.6 mL of tween 60 (5 % in Tris buffer) and 0.3 mL of 2 % calcium chloride. The slides were then washed with distilled water, incubated with 2 % lead nitrate, washed again, immersed in 1 % ammonium sulfate for 3 min, washed and post-stained.
The activity of acid phosphatase (ACP) was evaluated by the Gomori lead method (Bancroft 1996). Sections were incubated for 90 min at 37 °C in Gomori medium solution containing 10 mL of sodium acetate buffer (0.05 M, pH 5), 20 mg of lead nitrate and 32 mg of enzyme substrate (β-sodium glycerophosphate). The slides were then washed with distilled water, incubated with 1 % ammonium sulfate for 2 min, washed again and post-stained.
The activity of alkaline phosphatase (ALP) was evaluated by the Gomori calcium method (Bancroft 1996) in sections incubated for 90 min at 37 °C in a medium solution containing 2.5 mL of glycine buffer (0.2 M, pH 9.4), 5.0 mL of 2 % calcium nitrate, 0.25 mL of 1 % magnesium chloride and 2.5 mL of enzyme substrate (2 % β-sodium glycerophosphate). The slides were then washed with distilled water, incubated with 2 % cobalt nitrate, washed again, immersed in 1 % ammonium sulfate for 2 min, washed and post-stained.
Post-staining was performed with toluidine blue O (Bancroft 1996). Slides were then examined under light microscopy (JANAMED 2 Zeiss). Enzyme activities were assessed as strong (+++), moderate (++), weak (+) or absent (−) depending on the staining intensity of the azo dye in the tissue (Hirji and Courtney 1982; Gawlicka et al. 1995; Tengjaroenkul et al. 2000). Intestinal sections incubated without substrate were used as negative controls.
Results
Enzyme activity was detected along the brush border of the intestinal segments (Fig. 2). Weak reaction of dipeptidyl peptidase IV and nonspecific esterases was detected in the cytoplasm of the enterocytes.
Intestinal sections showing enzyme staining in the brush border of epithelial cells; a moderate dipeptidil aminopeptidase IV staining in the S2 of juvenile pacu fed diets containing 20 % lyophilized bovine colostrum (LBC) after 60 experimental days; b weak leucine aminopeptidase staining in the rectum of juvenile pacu fed diet containing 10 % LBC after 60 experimental days; c weak nonspecific esterase staining in the S1 of juvenile pacu fed diet 20 % LBC after 30 experimental days; d weak lipase staining in the S1 of juvenile pacu fed diet containing 10 % LBC after 30 experimental days; e weak acid phosphatase staining in the S1 of juvenile pacu fed diet containing no LBC after 30 experimental days; f strong alkaline phosphatase staining in the S1 of a juvenile pacu fed diet containing 20 % LBC after 30 experimental days; arrow enzyme activity; bar 50 µm
Moderate activity of the DAP IV was detected in the last three intestinal segments, but no differences among the treatments were detected. Enzymes LAP, NSE and LIP were weakly stained (azo dye) in all intestinal segments and the diets with 10 or 20 % of LBC commanded a moderate reaction to NSE in the S3 segment at day 60. Acid phosphatase activity was detected only in the brush border of the S1 segment of fish fed 0 % LBC for either 30 or 60 days. In the other groups and segments, the activity of ACP was not detected or was very weak to be considered as positive. The activity of ALP was very strong in the first intestinal segment, but a weak reaction was seen in the last segments. The inclusion of 20 % of LBC changed the pattern of staining to the ALP, eliciting moderate staining in S2 at day 30 and S1 at day 60 (Table 2).
Discussion
According to Tengjaroenkul et al. (2000), the DAP IV and LAP, enzymes responsible for breaking down long peptides chains into smaller chains and amino acids, exhibit greater activity in the first intestinal segments of Nile tilapia, a herbivorous species. The authors, including Bowen (1981) who worked with the T. mossambica, suggest that the proteins are completely digested and absorbed in the cranial half of the intestinal tract of these species. In this study, the LAP enzyme activity remained weak in the four intestinal segments, while DAP IV activity was moderate in the last three segments. These results indicate a possible change in the distribution pattern of proteolytic activity, a condition that could be related to the developmental stage and feeding habits of the juvenile pacu. Kuz’mina and Gelman (1997) reported that results about the localization of maximum proteolytic activity in intestinal segments are contradictory, with greater recorded activity in the proximal, medial and distal intestine both in equal and different species. The presence of proteases in the intestinal segments may be a barrier to the absorption of intact macromolecules in the distal regions of the intestine, as proposed by Schep et al. (1999). Unlike what happens with higher vertebrates, teleost have extensive uptake of molecules by the enterocytes in the posterior intestine (Schep et al. 1999; Barbieri and Hernández-Blazquez 2002). Although this absorptive capacity can be commercially exploited in aquaculture, the present study revealed that the presence of enzymatic activity can interfere with this process.
Regarding lipid metabolism, enzyme activity was also observed weak for enzymes NSE and LIP in the four intestinal segments. Hussain and Channa (2010) found that LIP activity in the intestinal tract of adult snow trout (Schizothorax curvifrons) is located in the brush border and cytoplasm of enterocytes decreased toward the posterior intestinal tract, and suggesting, as Kuz’mina and Gelman (1997) did, lipolysis activity only in the anterior intestine. Only juveniles who ingested diets containing bovine colostrum showed moderate reaction of the NSE in the middle intestine (S3 segment) after 60 days, indicating an influence of diet on lipase distribution. Studies of herbivores (Opuszynski and Shireman 1995; Tengjaroenkul et al. 2000; Hussain and Channa 2010), showed a restricted distribution of lipase, which is explained by the lower amount of fat present in plants. Thus, lipase activity in the intestinal tract seems to be related to the percentage of fat in the diet of fish, and considering the wide range of it in the pacu diet, we can also expect some variation in the activity of this enzyme. In this work, as Deimling and Bocking (1976), Wassmer et al. (1988), Van Lith et al. (1992) and Tengjaroenkul et al. (2000) found, NSE activity was also observed in the cytoplasm of enterocytes, especially in the S1, stating a route of intracellular lipid metabolism.
The enzyme acid phosphatase, present mainly in lysosomes, has been related to intracellular digestive activity and maturation of the intestinal epithelium and constitutes an auxiliary element of the digestion process (Baintner 1994). In newborn mammals, the intracellular activity of the acid phosphatase in epithelial cells is very low during the first hours of life (Brown and Moon 1979; Wilson et al. 1991; Bessi et al. 2002a, b; Moretti et al. 2012). In pacu, we found that the enzymatic activity of ACP was not significant in the brush border and absent in lysosomes, indicating that in the juvenile the intracellular degradation is not fully stimulated. The activity of ACP was present in the S1 segment of juveniles that did not consume bovine colostrum, 0 % LBC group, suggesting a possible inhibition of this enzyme by the bovine lacteal secretion. Contradictorily, colostrum deprivation may decrease or delay the enzymatic activity in the neonatal period of mammals (Kelly and Coutts 2000). Playford et al. (2000), Sauter et al. (2004) and Zhang et al. (1997) suggest that the increase in enzyme activity after ingestion of colostrum by newborn mammals may be a response to nutritional components present in this lacteal secretion, such as proteins, or to non-nutrition factors, such as growth factors and hormones.
Mahmood et al. (1994), Tengjaroenkul et al. (2000) and Lallès (2010) stated that the ALP is related to nutrient absorption by the enterocytes. The higher activity of this enzyme in the cranial region of the intestine suggests lower nutrient absorption in the posterior region (Tengjaroenkul et al. 2000). In the present work, in agreement with the above authors, we observed a strong reaction of the ALP in the first intestinal segment. Although the group that consumed a diet containing 20 % of LBC showed a moderate reaction in the S2 segment after 30 days and the S1 segment after 60 days, the nutrient absorption probably was not affected overall.
Considering the juveniles that did not consumed colostrum that is, in a normal condition, weak activity of LAP, NSE and LIP was observed in all four intestinal segments and the presence of ACP only in the S1 segment. DAP IV activity was moderate in the last three segments and the ALP activity was strong in the first segment and weak in others. These results suggest that intestinal enzyme activity was not fully active in juvenile pacu and that the consumption of diets containing bovine colostrum, a rich source of proteins, did not cause significant alteration in activity of the studied enzymes.
References
Association of Official Analytical Chemists—AOAC (2000) Methods of analysis. AOAC, Washington
Baintner K (1994) Demonstration of acidity intestinal vacuoles of the suckling rat and pig. J Histochem Cytochem 42:231–238
Bancroft JD (1996) Enzyme histochemistry. In: Bancroft JD, Stevens A (eds) Theory and practice of histological techniques, 4th edn. Churchill Livingstone, New York, pp 391–410
Barbieri RL, Hernández-Blazquez FJ (2002) Análise ultra-estrutural da absorção intestinal de macromolécula protéica com o uso de peixe como modelo experimental. ConSci Saúde 1:21–30
Bértin L (1958) Appareil digestif. In: Grassé PP (ed) Traite´ de Zoologie, vol 13. Masson, Paris, pp 1249–1301
Bessi R, Pauletti P, D’Arce RD, Machado Neto R (2002a) Colostral antibodies absorption in calves. II. Distal small intestine study. Braz J Anim Sci 31:2325–2331
Bessi R, Pauletti P, D’Arce RD, Machado Neto R (2002b) Colostral antibodies absorption in dairy calves. I: proximal small intestine study. Braz J Anim Sci 31:2314–2324
Bodammer P, Maletzki C, Waitz G, Emmrich J (2011) Prophylatic application of bovine colostrum ameliorates murine colitis via induction of immunoregulatory cells. J Nutr 141:1056–1061
Bowen SH (1981) Digestion and assimilation of periphytic detrital aggregate by Tilapia mossambica. Trans Am Fish Soc 110:239–245
Brown HH, Moon HW (1979) Localization and activities of lysosomal enzymes in jejunal and ileal ephitelial cells of the young pig. Am J Vet Res 40:1573–1577
Deimling OV, Bocking A (1976) Esterases in histochemistry and ultrahistochemistry. J Histochem 8:215–252
Gawlicka A, Teh S, Hung SSO, Hinton E, de Li Noüe J (1995) Histological and histochemical changes in the digestive tract of white sturgeon larvae during ontogeny. Fish Physiol Biochem 14:357–371
Hirji KN, Courtney WAM (1982) Leucine aminopeptidase activity in the digestive tract of perch, Perca fluviatilis L. J Fish Biol 21:615–622
Holmberg EL (1887) Viaje á Misiones. Bol Acad Nac Ci 10:5–391
Huguet A, Sève B, Le Dividich J, Le Huërou-Luron I (2006) Effects of a bovine colostrum-supplemented diet on some gut parameters in weaned piglets. Reprod Nutr Dev 46:167–178
Hussain I, Channa A (2010) Histochemical distribution of lipase and acid phosphatase in the intestinal tract of the snow trout, Schizothorax curvifrons Heckel. J Biol Sci 10:643–647
Kelly D, Coutts AGP (2000) Development of digestive and immunological function in neonates: role of early nutrition. Liv Prod Sci 66: 161–167
Kuz’mina VV, Gelman AG (1997) Membrane-linked digestion in fish. Rev Fish Sci 5:99–129
Lallès J-P (2010) Intestinal alkaline phosphatase: multiple biological roles in maintenance of intestinal homeostasis and modulation by diet. Nutr Rev 68:323–332
Lima AL, Pauletti P, Susin I, Machado-Neto R (2009) Fluctuation of serum variables in goats and comparative study of antibody absorption in new-born kids using cattle and goat colostrum. Braz J Anim Sci 38:2211–2217
Lodja Z (1979) Studies on dipeptidyl(amino)peptidase IV (glycyl-proline naphthylamidase). Histochemistry 59:153–166
Mahmood A, Yamagishi F, Eliakim R, DeSchryver-Kecskemeti K, Gramlich TL, Alpers DH (1994) A possible role for rat intestinal surfactant-like particles in transepithelial triacylglycerol transport. J Clin Invest 93:70–80
Moretti DB, Kindlein L, Pauletti P, Machado-Neto R (2010) IgG absorption by Santa Ines lambs fed Holstein bovine colostrum or Santa Ines ovine colostrum. Animal 4:933–937
Moretti DB, Nordi WM, Lima AL, Pauletti P, Susin I, Machado-Neto R (2012) Enzyme activity in the small intestine of goat kids during the period of passive immunity acquisition. Small Rumin Res 105:321–328
Opuszynski K, Shireman JV (1995) Digestive mechanisms. In: Opuszynski K, Shireman JV (eds) Herbivorous fishes: culture and use for weed management. CRC, Boca Raton, pp 21–31
Pandey NN, Dar AA, Mondal DB, Nagaraja L (2011) Bovine colostrum: a veterinary nutraceutical. J Vet Med Anim Health 3:31–35
Pelli A, Dumont Neto R, Silva JD da, Ramos SM, Souza DS, Barbosa NDC (1997) Ingestion of ration by “pacu” (Piaractus mesopotamicus Holmberg, 1887), “curimba” (Prochilodus scrofa Steindachner, 1881) and “piau” (Leporinus friderici Bloch, 1794) in condition of semi-intensive rearing. B Inst Pesca 24:119–123
Playford RJ, MacDonald CE, Johnson WS (2000) Colostrum and milk-derived peptide growth factors for the treatment of gastrointestinal disorders. Am J Clin Nutr 72:5–14
Rodrigues APO, Pauletti P, Kindlein L, Cyrino JEP, Delgado EF, Machado-Neto R (2009) Intestinal morphology and histology of the striped catfish Pseudoplatystoma fasciatum (Linnaeus, 1766) fed dry diets. Aquac Nutr 15:55–563
Sauter SN, Roffler B, Philipona C, Morel C, Rome V, Guilloteau P, Blum JW, Hammon HM (2004) Intestinal development in neonatal calves: Effects of glucocorticoids and dependence on colostrum feeding. Biol Neon 85: 94–104
Schep LJ, Tucker IG, Young G, Ledger R, Butt AG (1999) Controlled release opportunities for oral peptide delivery in aquaculture. J Control Release 59:1–14
Tengjaroenkul B, Smith BJ, Caceci T, Smith SA (2000) Distribution of intestinal enzyme activities along the intestinal tract of cultured Nile tilapia, Oreochromis niloticus L. Aquaculture 182:317–327
Urbinati EC, Gonçalves FD (2005) Pacu (Piaractus mesopotamicus). In: Baldisseroto B, Gomes LC (eds) Espécies nativas para piscicultura no Brasil. UFSM, Santa Maria, pp 225–246
Van Lith HA, Meijer GW, Van Der Wouw MJA, Den Bieman M, Van Tintelen G, Van Zutphen LFM, Beynen AC (1992) Influence of amount of dietary fat and protein on esterase-1 (ES-1) activities of plasma and small intestine in rats. Br J Nutr 67: 379–390
Wassmer B, Augenstein U, Ronai A, De Looze S, Von Deimling O (1988) Lymph esterases of the house mouse (Mus musculus): II. The role of esterase-2 in fat resorption. Comp Biochem Physiol B 91:179–185
Wilson JM, Whitney JA, Neutra MR (1991) Biogenesis of the apical endosome-lysosome complex during differentiation of absorptive epithelial cells in the rat ileum. J Cell Sci 100:133–143
Zhang H, Malo C, Buddington RK (1997) Suckling induces rapid intestinal growth and changes in brush border digestive functions of newborn pigs. J Nutr 127:418–426
Acknowledgments
Authors are indebted to Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the funding and support to the research project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moretti, D.B., Nordi, W.M., Cruz, T.M.P. et al. Histochemical distribution of intestinal enzymes of juvenile pacu (Piaractus mesopotamicus) fed lyophilized bovine colostrum. Fish Physiol Biochem 40, 1487–1493 (2014). https://doi.org/10.1007/s10695-014-9942-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-014-9942-7