Abstract
The proximate composition of the whole body and the fatty acid composition of the liver, muscle, eye and brain of wild and cultured rohu (Labeo rohita) were analyzed. The cultured species was found to have significantly (P < 0.05) higher lipid contents than its wild counterpart. The saturated (SFA) and monounsaturated (MUFA) fatty acid contents were significantly higher in the cultured species, whereas the n-6 and n-3 polyunsaturated fatty acid (PUFA) levels were higher in the wild species. Fatty acids C16:0 and C18:1 n-9 were the principal fatty acids of the SFAs and MUFAs, respectively, identified in the analyses. Docosahexaenoic acid, eicosapentaenoic acid, and arachidonic acid were the predominant PUFAs in both groups, and all three were found to be present at significantly (P < 0.05) higher levels in the wild species. Erucic acid (C22:1 n-9), which was the predominant fatty acid (30.76%) in the feed, was detected only at low levels in muscle (0.30%), liver (1.04%) and eye (1.28%) of cultured fish tissue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fish are a rich source of polyunsaturated fatty acids (PUFAs), namely, the n-6 and n-3 PUFAs, which are beneficial to human health. Compared to beef and chicken, fish meat contains higher levels of n-3 PUFAs (Calder 2004), which are known to be cardio-protective (Sanderson et al. 2002), anti-atherosclerotic (Givens et al. 2006), antithrombotic (Calder 2004; Harris 2004; Givens et al. 2006), and anti-arrythmitic (Givens et al. 2006). Studies with non-human primates and human newborns indicate that Docosahexaenoic acid (DHA) is essential for the normal functional development of the retina and brain, particularly in premature infants (Montano et al. 2001). Arachidonic acid (AA), a PUFA of the n-6 series, is a precursor of biologically important products, such as epoxides, AA-ethanolamide, anandamide, and iso-prostanes, an isomer of prostaglandins (Galli and Marangoni 1997). Fish meat containing high n-3 to n-6 ratios of PUFAs are considered to be the most beneficial in terms of human health.
Rohu (Labeo rohita) is one of the most preferred fish among Indian consumers and cultured extensively throughout the Indian subcontinent (Chondar 1999; Misra and Samantaray 2004), mostly in ponds. The majority of rohu marketed commercially is the product of fish farming, although a main capture fishery is active on the Ganga river and it tributaries (Chondar 1999). Rohu along with other two carps found extensively in India, namely catla (Catla catla) and mrigal (Cirrhinus mrigala), contribute the bulk of fish farm production, with over 1.8 million tonnes (FAO 2003). The taste and nutritional quality of the fish meat, such as the fatty acid composition, varies with the season (Grun et al. 1999; Ackman et al. 2002) region (Ackman et al. 2002), and diet (Grigorakis et al. 2002). The fatty acid profiles of different fish, such as gilthead sea bream (Grigorakis et al. 2002; Mnari et al. 2007), sea bass (Alasalvar et al. 2002), sharp snout sea bream (Rueda et al. 2001), and Murray cod (De Silva et al. 2004), have been compared. However, to date, there has been no attempt to study the differences in fatty acid profiles between wild and cultured carps of the major commercial carp species in India. The aim of this study was, therefore, to compare the fatty acid profiles of cultured and wild rohu.
Materials and methods
Three wild rohu (n = 3; average weight ± SE 108 ± 5.34 g) were caught in a reservoir in Powerkheda, Madhya Pradesh, India in January 2006, and three cultivated rohu (n = 3; average weight ± SE 105 ± 4.95 g) were collected from a pond in same region and in the same season. The proximate composition of the farmed and wild rohu were then analyzed (AOAC 1995). The muscle, liver, eye and brain were dissected out of the fish, and total lipids were extracted with a chloroform:methanol mixture (2:1, v/v; Folch et al. 1957). The extracted lipids were esterified with BF3 methanol and recovered in heptane (AOAC 1995). The fatty acid methyl ester (FAME) was analyzed by gas chromatography–mass spectrometry (GC-MS; model QP 2010, quadruple mass-spectrometer with ionization energy of 70 eV) equipped with a dB-wax column (25 × 25 mm; film thickness 0.25 μm) with helium gas as the carrier gas. The sample was injected at a split mode injection port with a 1:15 split ratio at 250°C, and the oven temperature was programmed to increase from 50 to 230°C in steps of 10°C/min and then to remain stable for 35 min. The mass spectrometer was adjusted to obtain relative abundance of m/z ranging from 40.00 to 550.00. The values of the fatty acids are presented in area percentage of total identified fatty acids.
Statistical analysis
Data are presented as mean ± standard error of the mean (SEM), and significant differences between the means were determined by an independent t test.
Results and discussion
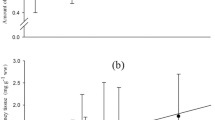
Our analyses of the proximate composition of cultured and wild rohu revealed that cultured fish contain significantly higher lipid levels and a lower moisture content than its wild counterpart (Table 1). The fatty acid profiles of the feed, muscle, liver, eye and brain are given in Tables 2, 3, 4, 5, 6, respectively. The liver, muscle, and eye of cultured rohu contained higher levels of total saturated (SFA) and monounsaturated (MUFA) fatty acids than the wild rohu, but the level of total SFAs was found to be insignificant in the brain. Mnari et al. (2007) reported higher SFA levels in cultured sea bream than in its wild counterpart, but the latter contained higher levels of MUFAs. In contrast, Alasalvar et al. (2002) observed the reverse in sea bass. The principal SFAs identified in both the wild and cultured fish were C16:0 and C18:0; in terms of MUFA, these were C18:1 n-9 and C16:1 n-9. These results are similar to those reported by Grigorakis et al. (2002) in sea bream and Alasalvar et al. (2002) in sea bass. Palmitic acid was identified as the most abundant fatty acid in the muscle, liver, eye, and brain of both cultured and wild rohu; in the muscle and eye, it was significantly (P < 0.05) higher in cultured fish than in wild ones, but in the eye and brain tissue the difference was insignificant. The levels of the SFAs found in the brain in this study remain stable despite dietary influence, which is in contrast to the report of Connor et al. (1990) who found that the fatty profile of brain tissue of rhesus monkeys was altered by dietary supplement. The presence of higher amounts of palmitic acid in the muscle and liver of farmed rohu may be attributed to the use of supplementary feed containing high amounts of palmitic acid.
As observed by Rueda et al. (2001) in red porgy, the total n-6 and n-3 PUFA levels present in the liver, muscle, and brain of wild rohu were significantly (P < 0.05) higher than those in its cultured counterpart. This finding is similar to that of Tocher and Harvie (1988). Pagliarani et al. (1986) reported that fish brain lipid composition seems to be affected by dietary input, which partially supports our finding of similar levels of palmitic acid and total SFA in wild and cultured fish. The brain fatty acid profile was different from that reported by Stoknes et al. (2004) who indicated that the brain fatty acid composition in fish species is dominated by PUFA from the n-3 series; with those in fish being eicosapentaenoic acid (EPA) and DHA in particular. The total n-3 level in the eye of wild rohu was significantly higher than that in the eye of its cultured counterpart, but the n-6 level in the same organ was found to be significant. Rohu is an omnivorous fish, and the fatty acid accumulation in wild fish is influenced by the plankton it consumes (Chakrabarti et al. 1995). Mnari et al. (2007) observed significantly high n-3 PUFA levels in cultured sea bream in the muscle and liver; the same findings were reported by De Silva et al. (2004) in Murray cod. In contrast, Serot et al. (1998) reported a higher proportion of long chain n-3 PUFAs in wild turbot.
The observed higher levels of arachidonic acid (AA), EPA, and DHA in the muscle of both wild and farmed fish compared to those found in the liver indicate that the rohu has a tendency to conserve highly unsaturated fatty acids (HUFAs). These fatty acids were found to be retained in several fish species, even during starvation, suggesting a metabolic priority for their conservation (Rainuzzo et al. 1994). The higher proportion of n-6 PUFAs may be due to a conservation of n-6 PUFAs in preference to n-3 PUFAs (De Silva et al. 2004). Arachidonate is conserved during periods in which there is a deficiency of essential fatty acids in the heart, renal cortex, and liver, when the formation of prostaglandins is also blocked (Lefkowith et al. 1985). The conservation of arachidonate and the depletion of other PUFAs in these tissues is required for maintaining eicosanoid production (Moussa et al. 1996). The major n-3 PUFA in both the cultured and wild rohu was DHA (C22:6 n-3), with significantly higher levels being found in the muscle, liver, eye, and brain of wild rohu; these results are in agreement with those previously reported by Grigorakis et al. (2002) and Serot et al. (1998). Similarly, EPA (C20:5 n-3) was also found to be significantly higher in wild fish than in its cultured counterpart. The level of AA (C20:4 n-6), the principal n-6 fatty acid, was significantly higher—in all of the organs analyzed—in wild rohu than in cultured rohu. Similar results were obtained by Mnari et al. (2007) and Grigorakis et al. (2002) in sea bream, De Silva et al. (2004) in Murray cod, Serot et al. (1998) in turbot, and Alasalvar et al. (2002) in sea bass.
The most important finding of our study was the distinct difference between wild and farmed rohu in terms of AA levels in the muscle and liver tissue: wild rohu had fourfold and twofold higher levels of AA in the liver and muscle, respectively, than farmed rohu. Ackman et al. (2002) reported that polar lipid levels reached 10.6% in Indian major carp feeding on natural food, while Rahman et al. (1995) reported 8.51% in rohu and 14% in big head carp. Tropical freshwater fish have higher levels of AA than marine fish (Ackman 2002). The abundance of linoleic acid (C18:2 n-6), a precursor of n-6 fatty acids, was also found to be higher in wild fish, which may explain the high levels of AA and n-6 fatty acids in these fish. Consequently, the higher levels of AA, EPA and DHA in wild fish may be due to higher levels of these fatty acids and their precursors in their naturally available food. Along similar lines, the lower levels of PUFA in cultured fish may be explained on the basis of the fatty acid profile of the feed (Table 2), which does not contain AA, EPA, and DHA and only limited levels of linoleic (12.41%) and linolenic (5.52%) acids. Wild fish were found to have a significantly higher n-6/n-3 ratio in the muscle and liver, which is in contrast to the finding of Grigorakis et al. (2002) in sea bream and Serot et al. (1998) in turbot. However, Mnari et al. (2007) reported high a n-6/n-3 ratio in wild sea bream. The higher n-6/n-3 ratio in wild fish is due to the significantly greater amounts of n-6 fatty acids than n-3 fatty acids, with better ratios in the eye and brain than in the muscle and liver.
Although our analysis of the fatty acid profile of feed revealed a high amount (30.76%) of erucic acid (C22:1 n-9), erucic acid levels were at very low levels in the muscle (0.30%), liver (1.04%), eye (1.28%), and brain (0.31%) of the cultured fish. These low levels may be due to its poor absorption, as erucic acid has been reported to be linked to the sn-1 and sn-3 of triacylglycerol (Farnworth 1983) and high 1, 3-lipase activities in rohu (Nayak et al. 2003). The poor assimilation of erucic acid that we observed in our study is of some importance as this fatty acid has been reported to be an antinutritional factor and shown to be associated with an increased incidence of myocardial lipidosis in animals (Food Standards Australia New Zealand 2003).
Due to the general trend towards increased standards of living worldwide, human beings are becoming more and more health conscious, preferring to consume nutritious food with some added health benefits. Fish rich in PUFAs have several health benefits, as PUFAs are considered to be cardio-protective, anti-atherosclerotic, antithrombotic, and anti-arrythmitic; they also contribute to normal eye and brain development in humans. Based on the results of our study, we conclude that the difference in the fatty acid profiles of wild and farmed rohu may be attributed primarily to the dietary intake of the fish. The nutritional value of farmed rohu can be improved by incorporating desired fatty acids, such as linoleic acid, linolenic acid, EPA, and DHA, into the feed. The high amounts of DHA and the favorable n-6/n-3 ratios in the eye and brain indicate that the fish head is a very omega-rich edible portion of the fish body. Well-designed comparative studies of lipid and fatty acids of the same species of fish need to be carried out in the future. In addition, regional and seasonal variations in the lipid and fatty acid patterns of different body parts, such as the head, dorsal muscle, ventral muscle, and caudal muscle, need to be analyzed for a better understanding of the nutritive lipid and fatty acid profile of this species. The fatty acid profile of the natural food of rohu needs to be analyzed in all regions and seasons.
References
Ackman RG (2002) Freshwater fish lipids—an overlooked source of beneficial long chain –3 fatty acids. Eur J Lipid Technol 104:253–254. doi:10.1002/1438-9312(200205)104:5<253::AID-EJLT253>3.0.CO;2-B
Ackman RG, Mcleod C, Rakshit S, Misra KK (2002) Lipids and fatty acids of five freshwater food fishes of Indian. J Food Lipids 9:127–145
Alasalvar CK, Taylor DA, Zubcov E, Shahidi F, Alexis M (2002) Differentiation of cultured and wild sea bass (Dicentrarchus labrax): total lipid content, fatty acid and trace mineral composition. Food Chem 79:145–150. doi:10.1016/S0308-8146(02)00122-X
AOAC (1995) Official methods of analysis of AOAC International, vol 1, 16th edn. AOAC International, Arlington
Calder PC (2004) Long-chain fatty acids and cardiovascular disease: further evidence and insights. Nutr Res 24:761–772. doi:10.1016/j.nutres.2004.04.008
Chakrabarti I, Gani MA, Chaki KK, Sur R, Misra KK (1995) Digestive enzymes in eleven freshwater teleost fish species in relation to food habit and niche segregation. Comp Biochem Physiol 11:167–177. doi:10.1016/0300-9629(95)00072-F
Chondar SL (1999) Labeo rohita. Biology of finfish and shellfish. SCSC Publishers, Howrah, West Bengal, India, pp 199–211
Connor WE, Neuringer M, Lin DS (1990) Dietary effects on brain fatty acid composition: the reversibility of n-3 fatty acid deficiency and turnover of docosahexaenoic acid in the brain, erythrocytes, and plasma of rhesus monkeys. J Lipid Res 31:237–247
De Silva SS, Gunasekera RM, Ingram BA (2004) Performance of intensively farmed Murray cod (Maccullochella peelii peelii) (Mitchell) fed newly formulated vs. currently used commercial diets, and a comparison of fillet composition of farmed and wild fish. Aquacult Res 35:1039–1052. doi:10.1111/j.1365-2109.2004.01111.x
FAO (2003) Aquaculture production. Yearbook of fishery statistics, vol 96. Food and Agriculture Organization of the United Nations, Rome, Italy
Farnworth ER (1983) The composition of diets used in rapeseed oil feeding trials. In: Kramer KG, Sauer FD, Pigden WJ (eds) High and low erucic acid rapeseed oils: production, usage, chemistry, and toxicological examination. Academic Press, New York, pp 315–333
Folch J, Lees M, Sloane-Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Food Standards Australia New Zealand (2003) Erucic acid in food: a toxicological review and risk assessment. Technical Report series no. 21. Food Standards Australia New Zealand, Canberra
Galli C, Marangoni F (1997) Recent advances in the biology of n-6 fatty acids. Nutrition 13:978–985. doi:10.1016/S0899-9007(97)00341-9
Givens DI, Kliem KE, Gibbs RA (2006) The role of meat as a source of n-3 polyunsaturated fatty acids in the human diet. Meat Sci 74:209–218. doi:10.1016/j.meatsci.2006.04.008
Grigorakis K, Alexis MN, Taylor A, Hole M (2002) Comparison of wild and cultured gilthead sea bream (Sparus aurata): composition, appearance and seasonal variations. Int J Food Sci Technol 37:1–8. doi:10.1046/j.1365-2621.2002.00604.x
Grun IU, Shi H, Fernando LN, Clarke AD, Ellersieck MR, Beffa DA (1999) Differentiation and identification of cultured and wild Crappie (Promoxix spp.) based on fatty acid composition. Lebensm Wiss Technol 32:305–311
Harris WS (2004) Omega-3 fatty acids, thrombosis and vascular disease. Int Congr Ser 1262:380–383. doi:10.1016/j.ics.2003.12.097
Lefkowith JB, Flippo V, Sprecher H, Needleman P (1985) Paradoxical conservation of cardiac and renal arachidonate content in essential fatty acid deficiency. J Biol Chem 260:15736–15744
Misra K, Samantaray K (2004) Interacting effects of dietary lipid level and temperature on growth, body composition and fatty acid profile of rohu (Labeo rohita). Aquacult Nutr 10:359–369. doi:10.1111/j.1365-2095.2004.00311.x
Mnari A, Bouhlel I, Chraief I, Hammami M, Romdhane MS, Cafs MEI, Chaouch A (2007) Fatty acids in muscles and liver of Tunisian wild and farmed gilthead sea bream (Sparus aurata). Food Chem 100:1393–1397. doi:10.1016/j.foodchem.2005.11.030
Montano N, Gavina G, Gavino VC (2001) Polyunsaturated fatty acids contents of some traditional fish and shrimp paste condiments of the Philippines. Food Chem 75:611–614. doi:10.1016/S0308-8146(01)00126-1
Moussa M, Garcia J, Ghisolfi J, Pariquet B, Thouvehot JP (1996) Dietary essential fatty acid deficiency differentially affects tissues of rats. J Nutr 126:340–345
Nayak J, Nair PG, Viswanathan AK, Mathew S (2003) Lipase activity in different tissues of four species of fish: rohu (Labeo rohita Hamilton), oil sardine (Sardinella longiceps Linnaeus), mullet (Liza subviridis Valenciennes) and Indian mackerel (Rastrelliger kanagurta Cuvier). J Sci Food Agric 83:1139–1142. doi:10.1002/jsfa.1515
Pagliarani A, Pirini M, Trigari G, Ventrella V (1986) Effects of diets containing different oils on brain fatty acids compositions in seabass (Dicentrarchus lebrax L.). Comp Biochem Physiol B 83:277–283. doi:10.1016/0305-0491(86)90366-4
Rahman SA, Huah TS, Hassan O, Daud NM (1995) Fatty acid composition of some Malaysian freshwater fish. Food Chem 54:45–49. doi:10.1016/0308-8146(95)92660-C
Rainuzzo JR, Reitan KI, Jorgensen L, Olsen Y (1994) Lipid composition in turbot larvae fed live feed cultured by emulsions of different lipid classes. Comp Biochem Physiol 107:699–710. doi:10.1016/0300-9629(94)90372-7
Rueda F, Hernandez MD, Egea MA, Aguado F, Garcia B, Martinez FJ (2001) Differences in tissue fatty acid composition between reared and wild sharp snout sea bream, Diplodus puntazzo (Cetti, 1177). Br J Nutr 86:617–622. doi:10.1079/BJN2001438
Sanderson P, Finnegan YE, Williams CM, Calder PC, Burdge GC, Wootton SA, Griffin BA, Millward DJ, Pegge NC, Bemelmans WJE (2002) UK Food standards agency alpha-linolenic acid workshop report. Br J Nutr 88:573–579. doi:10.1079/BJN2002691
Serot T, Gandermer G, Demaimay M (1998) Lipid and fatty acid compositions of muscle from farmed and wild turbot. Aquacult Int 6:331–343. doi:10.1023/A:1009284905854
Stoknes IS, Okland HMW, Falch E, Syness M (2004) Fatty acid and lipid class composition in eyes and brain from teleosts and elasmobranchs. Comp Biochem Physiol 138B:183–191
Tocher DR, Harvie DG (1988) Fatty acid composition of the major phosphoglycerides from fish neural tissue; (n-3) and (n-6) polyunsaturated fatty acids in rainbow trout (Salmo gairdneri) and cod (Gadus morhua) brains and retinas. Fish Physiol Biochem 5:229–239. doi:10.1007/BF01874800
Acknowledgements
The authors are grateful to the Director, Central Institute of Fisheries Education, Mumbai, for providing facilities for carrying out the work. The first author is grateful to the Central Institute of Fisheries Education, Mumbai for awarding the institutional fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, P., Kumar, V., Sinha, A.K. et al. Comparative fatty acid profiles of wild and farmed tropical freshwater fish rohu (Labeo rohita). Fish Physiol Biochem 36, 411–417 (2010). https://doi.org/10.1007/s10695-009-9309-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-009-9309-7