Abstract
The adoption of reduced height (Rht) genes has dramatically enhanced yield potential of cereal crops. The semi-dwarfing gene uzu in barley is widely used in East Asia. Its value as an Rht gene in the warm and dry environments in Australia was evaluated in this study. In addition to plant height, this gene showed significant effects on a wide range of characteristics. It reduced the overall plant height by an average of 33.7 % with the most significant reduction occurred on the internode below the peduncle. The gene reduces spike length by 31.6 % and grain weight by 18.8 %. It increases grain number per spike by 5.6 % and grain density by 51.8 %. Seedling assays suggested that the effects of this gene became more significant with increased temperature. It reduced coleoptile and seedling length by 19.4 % and 15.1 %, respectively at 7 °C. The reduction increased to 61 % and 46.7 %, respectively, at 25 °C. These results indicated that the uzu gene could have limited values in some high temperature and drought prone environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reduced height (Rht) genes enhance crops’ resistance to lodging which could be caused by either weather factors or management practices. Together with some other factors, these genes have made tremendous contributions to a significant increase in cereal production worldwide, which has been dubbed as the ‘Green Revolution’ (Milach and Federizzi 2001). Different Rht genes not only affect height differently under different growth conditions, but also have different effects on many other morphological and agronomic traits. Thus selecting appropriate Rht genes is a major consideration in breeding for different environments (Ellis et al. 2005).
Rht genes have been reported on each of the seven chromosomes in barley (Wang et al. 2010). However, only three of them, including denso, sdw1 and uzu, have been widely used in breeding programs (Hellewell et al. 2000; Zhang and Zhang 2003; Saisho et al. 2004). Surprisingly, these three widely used Rht genes all locate on the long arm of chromosome arm 3H, occupying two different loci. One of these loci contains at least three different alleles: sdw1 (previously known as sdw), denso and another one reported by Barua et al. (1993). This Rht locus has been widely used in breeding programs in the United States, Canada and Europe (Rasmusson 1991; Mickelson and Rasmusson 1994) and contributed to many malting and feeding barley varieties (Mickelson and Rasmusson 1994). Although sdw1 and denso were derived from separate mutation events, they are allelic and have similar effects on agronomic traits. They have been associated with late heading, low 1000 kernel weight, lower yield, high screenings and high β-glucan content (Powell et al. 1985; Foster and Thompson 1987; Thomas et al. 1991; Mickelson and Rasmusson 1994; Hellewell et al. 2000).
Another Rht locus, which locates 23 cM proximally to the sdw1/denso locus, contains the uzu gene (Li et al. 2015). It is reported that this gene reduces plant height to about 80–90 % of its normal counterpart under normal cultivation in Japan (Saisho et al. 2004). It was noted that ‘uzu’ lines showed lower adaptability to deep-seeding than non-uzu lines (Takeda and Kakahashi 1999) and that the gene seems to have inhibitory effects on the elongation of leaf, culm, rachis internode, awn and glume (Saisho et al. 2004). Further, it was also found that the uzu gene suppresses elongation of coleoptiles and first internode under dark conditions (Saisho et al. 2004), possibly for the reason that light is known to reduce brassinosteroids (or BR) synthesis (Fujioka and Yokota 2003).
Previous studies demonstrated that the uzu phenotype is due to a single-nucleotide substitution (A>G) at position 2612 in the sequence encoding kinase domain of HvBRI1 polypeptide. This substitution results in an amino acid change at a highly conserved residue in the kinase domain of the BR -receptor protein (Chono et al. 2003). The marker synteny of rice and barley chromosomes suggests that the uzu gene is likely homologous to D61, a rice homolog of Arabidopsis BR-insensitive 1 (BRI1), encoding a BR-receptor protein (Chono et al. 2003). A report on a new barley semi-dwarf gene allelic with uzu showed that similar effects on seedling length, plant height and awn length were detected by mutating the HvBRI1 gene at a position different from the single-nucleotide substitution characterizing the uzu gene (Gruszka et al. 2011).
Many of the existing Rht genes in barley have negative effects on grain yield and other traits of agronomic importance (Hellewell et al. 2000) thus additional genes are highly desirable. Although the uzu gene has been widely used as a semi-dwarfing gene in East Asia (Chono et al. 2003; Rikiishi et al. 2008), its usefulness in other regions including Australia has not been reported. In the course of fine-mapping a locus controlling several characteristics of spike morphology (Chen et al. 2012), it was found that it co-located with the well-known semi-dwarf gene uzu. When investigating possible effects of plant height on Fusarium crown rot resistance, it was also noticed in experiments conducted in growth cabinet that the uzu gene has more drastic effect on plant height in environments of higher temperatures (Chen et al. 2014). These findings led to the systematic study described here which showed that the so-called semi-dwarfing gene uzu affects many characteristics and that the magnitudes of its effects are temperature-dependent.
Materials and methods
Near isogenic lines (NILs) for the uzu gene
The 15 pairs of NILs used in this study were developed in a study investigating possible effects of plant height on Fusarium crown rot resistance (Chen et al. 2014). They were developed from two F2 populations (TX9425/Franklin and TX9425/Gairdner) based on the heterogeneous inbred family method (Tuinstra et al. 1997). The co-dominant simple sequence repeat (SSR) marker HVM33, which was most closely linked to the peak of the QTL controlling plant height, was used to select F2 individuals that were heterozygous at the marker locus (Chen et al. 2014). Briefly, 15 individual heterozygous F2 plants were identified and self-pollinated. Six F3 plants were grown from each heterozygous F2 plant, and a single heterozygote was again selected and self-pollinated. This process of heterozygous individual selection followed by self-pollination was repeated until the F8 generation. From each of the 15 original F2 plants, two homozygous F8 lines, one with and the other without the ‘TX9425’ allele, were selected and were treated as a pair of NILs. A ‘fast generation’ procedure described by Zheng et al. (2013) was used in developing these NIL pairs.
Field trials in assessing the effects of the uzu gene on plant height and other traits of agronomic importance
The NILs were assessed in two field trials conducted at the CSIRO Research Station at Gatton, Queensland, Australia (27°34′S, 152°20′E) in 2012 and 2013, respectively. Two replicates of the test lines were planted in two separate blocks and a randomized complete block design were applied in each of the trials. Twenty seeds of each NIL were grown in a single 1.5 m row in each replicate, and the space between rows was 25 cm. Measurements of plant height, spike length (excluding awns), seed number per spike, grain weight (100 kernel weight), grain density (number of grains per cm spike) and internode (counted from the spike downward) length were taken on the six tallest tillers in each row, and the average values were used in subsequent statistical analyses.
The NILs and the genotype TX9425 (the donor of the uzu gene to the NILs used in this study) were also assessed in two field trials conducted at University of Tasmania, Launceston, Australia (41°26′S, 147°8′E) in 2013 and 2014. Two replicates of the 15 pairs of NILs were planted in two separate blocks and a randomized complete block design were applied in each of the trials. Twenty seeds of each NIL were grown in a single 1.5 m row in each replicate, the space between rows was 25 cm. Internode lengths were taken on the six tallest tillers in each row, and the average values were used in subsequent statistical analyses.
Effects of the uzu gene on coleoptile and seedling lengths at different temperatures
These experiments were conducted in growth cabinet using four pairs of the NILs. Seeds were surface sterilised in 0.5 % hypochlorite solution for 30 s and then rinsed in sterile de-ionised water. The sterilised seeds were then germinated in Petri dishes on three layers of filter paper saturated with water and newly germinated seedlings were then planted into 56-well seedling trays (Rite Grow Kwik Pots, Garden City Plastics, Australia) containing sterilised University of California mix C (50 % sand and 50 % peat v/v). Seven seedlings per line were used in each replicate and two replicates were used in each trial. Seedling trays were then placed into the growth cabinets pre-set at 7 °C, 15 °C, 20 °C, or 25 °C with a fixed lighting period of 14 h. Measurements of coleoptiles (from soil surface to tip of coleoptile) and seedling lengths (from soil surface to tip of longest leaf) were taken when seedlings of the tall isolines reached 15–20 cm.
Statistical analysis
All statistical analyses were performed using GenStat for Windows in the 11th edition (copy right Lawes Agricultural Trust, Rothamsted Experimental Station, UK). Descriptive statistics and an analysis of variance (ANOVA) were conducted to detect if there were significant genetic effects for uzu in the 15 pairs of tall and dwarf NILs. Within each trial, the following mixed effects model was used: Yij = l + ri + gj + wij, where Yij observation on the jth genotype in the ith replication; l general mean; ri effect due to ith replication; gj effect due to the jth genotype; wij error or genotype by replication interaction, where genotype was treated as a random effect and that of replicates as fixed effect.
Results
Effects of the uzu on plant height and internode lengths
Highly significant reductions in plant height caused by the uzu gene were detected from each of the 15 NIL pairs assessed in both field trials, although the effects of this gene varied between genetic backgrounds and environments. Plant heights of the dwarf isolines ranged from 41.3 to 63.3 cm in 2012 and 43.5 cm to 68.5 cm in 2013, while the heights of the tall isolines ranged from 75.8 cm to 92.6 cm in 2012 and 67.5 cm to 104.0 cm in 2013. Plant heights between the uzu lines and their tall counterparts among the 15 NIL pairs differed between 18.6 and 46.9 % in 2012 and between 10.2 and 50.9 % in 2013, with an average reduction of 37 % and 30.4 %, respectively, in the two seasons (Table 1).
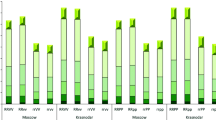
Based on the experiments conducted at Launceston in Australia, the reduction in plant height conferred by the uzu gene was not evenly distributed across the various internodes. The gene had the largest effect on the 2nd internode (the one below the peduncle) with an average reduction of 64.4 % (Table 2). An extreme example is shown in Fig. 1.This internode hardly elongated in some of the uzu lines in the spring sowing trial under Australian environments. The reductions of the gene on the 1st (peduncle) and the 3rd internode were similar, with an average of 49.7 % and 40.8 %, respectively. The gene showed a much small reduction on the 4th internode, with an average of 24.6 % between the two seasons. Unexpectedly, the uzu gene increased the 5th internode by an average of 45.9 % between the two experiments conducted (Table 2). However, this internode was very short in the non-uzu isolines and the low baseline could have contributed to the observed differences between the uzu isolines and their counterparts.
Showing that the reduction in plant height conferred by the uzu gene was not evenly distributed across the various internodes (left panel). The gene had the largest effect on the 2nd internode which was hardly elongated in some of the uzu lines sown in spring under Australian environments (right panel)
Effects of the uzu gene on spike characteristics
Barley yield is the product of the number of spikes, the number of grains per spike and the average kernel weight. Thus a gene affecting any characteristic of spikes could have significant implication in yield potential. Based on the trials conducted at the CSIRO Gatton Research Station, the uzu gene reduced spike length across the 15 pairs of NILs by an average of 32.3 % and 30.9 %, respectively, over the two seasons. The dramatic decrease in spike length was accompanied by a slight increase in grain number per spike. The combined effects resulted in dramatic changes in grain density, which was increased by an average of 48.7 % and 54.8 %, respectively, over the two seasons across the 15 pairs of NILs. Kernel weight of the uzu isolines was also significantly lower than that of their counterparts, with an average difference across the 15 pairs of NILs of 18.1 % and 19.5 %, respectively, over the two seasons (Table 3).
Effects of the uzu gene on coleoptile and seedling lengths at different temperatures
As coleoptile length is strongly associated with stand establishment and grain yield of deep-sown crops (Rebetzke et al. 2007), possible effects of the uzu gene on these characteristics was assessed using four pairs of the NILs at four different temperatures. Data from these experiments showed that the uzu gene had highly significant effects on both of these characteristics and its effects are temperature dependent. The gene reduced coleoptile length by an average of 17.4 % at 7 °C, 23.4 % at 15 °C, 40.1 % at 20 °C, and 61.8 % at 25 °C across the four NIL pairs. The gene reduced total seedling length by an average of 15.1 %, 20.0 %, 33.6 % and 46.5 % at 7 °C, 15 °C, 20 °C, and 25 °C, respectively (Table 4). Clearly, the effects of the uzu gene on coleoptile and seedling lengths increased with increased temperature.
Discussion
Rht genes have played a key role worldwide in increasing yield potential of cereal crops. The barley semi-dwarfing gene uzu has been widely used in East Asia and we assessed its potential for Australian breeding programs. Our results showed that this gene has drastic effects on not only plant height but a wide array of other characteristics. Importantly, the effects of this gene are temperature-dependent, and its effects on these characteristics become more drastic in environments of higher temperatures. The drastic effects of this gene on some of these characteristics could make it difficult to exploit in breeding varieties for high temperature environments. For example, deep-sowing is often necessary to get better seed-zone moisture in high temperature and drought prone environments and it is known that genotypes with long coleoptiles improve stand establishment, vigour and grain yield of deep-sown crops (Rebetzke et al. 2007). Thus the significant reduction in coleoptile length conferred by the uzu gene would restrict the exploitation of this gene in breeding for such environments. Further, the significant reduction on grain weight by this semi-dwarfing gene in high temperature environments is also of concern as it would lead to reduced grain quality due to increased screenings.
Considering that the uzu gene is due to a single nucleotide substitution which results in an amino acid change (Chono et al. 2003), it could be difficult to apprehend the substantial effects of this gene on so many characteristics. In fact, the effects of this gene are not limited to the characteristics described in this paper. We noticed that it also affects many other characteristic including leaf size and flowering time although no measurements were taken on these traits in the study reported here. It has been reported that this gene plays an inhibitory role in the elongation of various tissues including reducing coleoptile length in the dark (Saisho et al. 2004), possibly for the reason that light is known to reduce BR synthesis (Fujioka and Yokota 2003). However, the effect of darkness on the functions of the uzu gene is debatable based on the results from this study. We observed significant inhibitory effect of the gene on coleoptile elongation in the condition of 14 h lighting as well, and this is the case at each of the four temperatures assessed. It has also been reported that genotypes with the uzu gene show dark green leaves and suppressed plant regeneration from calli derived from immature embryos (Rikiishi et al. 2008).
Pleiotropic effects of this gene have also been noted in several other species. For example, it has been reported that BR-deficient mutants show greatly reduced longitudinal cell expansion (Takahashi et al. 1995; Kauschmann et al. 1996; Szekeres et al. 1996; Azpiroz et al. 1998) and exhibit delayed flowering (Chory et al. 1991; Li and Chory 1997; Azpiroz et al. 1998). In Prunus avium, the gene reduces fertility and extends life span and delays senescence (Hewitt et al. 1985). Clearly, although the uzu gene has routinely been referred to as a semi-dwarfing gene in barley (Chono et al. 2003; Saisho et al. 2004; Rikiishi et al. 2008), it significantly affects a wide range of characteristics related to plant development in not only barley but other plant species as well. Results from this study show that the pleiotropic effects of this gene on plant development and kernel quality need to be considered when exploiting it to modulate plant height and this is especially the case in high temperature and drought prone environments.
References
Azpiroz R, Wu Y, LoCascio JC, Feldmann KA (1998) An Arabidopsis brassinosteroid-dependent mutant is blocked in cell elongation. Plant Cell 10:219–230
Barua UM, Chalmers KJ, Thomas WT, Hackett CA, Lea V, Jack P, Forster BP, Waugh R, Powell W (1993) Molecular mapping of genes determining height, time to heading, and growth habit in barley (Hordeum vulgare L.). Genome 36:1080–1087
Chen GD, Li HB, Zheng Z, Wei YM, Zheng YL, McIntyre CL, Zhou MX, Liu CJ (2012) Characterization of a QTL affecting spike morphology on the long arm of chromosome 3H in barley (Hordeum vulgare L.) based on near isogenic lines and a NIL-derived population. Theor Appl Genet 125:1385–1392
Chen GD, Yan W, Liu Y, Wei YM, Zhou MX, Zheng Y-L, Manners JM, Liu CJ (2014) The non-gibberellic acid-responsive semi-dwarfing gene uzu affects Fusarium crown rot resistance in barley. BMC Plant Biol 14:22
Chono M, Honda I, Zeniya H, Yoneyama K, Saisho D, Takeda K, Takatsuto S, Hoshino T, Watanabe Y (2003) A semi-dwarf phenotype of barley uzu results from a nucleotide substitution in the gene encoding a putative brassinosteroid receptor. Plant Physiol 133:1209–1219
Chory J, Nagpal P, Peto CA (1991) Phenotypic and genetic analysis of det2, a new mutant that affects light-regulated seedling development in Arabidopsis. Plant Cell 3:445–459
Ellis MH, Rebetzke GJ, Azanza F, Richards RA, Spielmeyer W (2005) Molecular mapping of gibberellin-responsive dwarfing genes in bread wheat. Theor Appl Genet 111:423–430
Foster AE, Thompson AP (1987) Effects of a semi-dwarf gene from Jotun on agronomic and quality traits of barley. In: Yasuda S, Kanishi T (eds) Proceedings of the 5th international barley genetics symposium, 1986. Sanyo, Okayama, pp 979–982
Fujioka S, Yokota T (2003) Biosynthesis and metabolism of brassinosteroids. Annu Rev Plant Biol 54:137–164
Gruszka D, Szarejko I, Maluszynski M (2011) New allele of HvBR1 gene encoding brassinosteroid receptor in barley. J Appl Genet 52:257–268
Hellewell KB, Rasmusson DC, Gallo-Meagher M (2000) Enhancing yield of semidwarf barley. Crop Sci 40:352–358
Hewitt FR, Hough T, O’Neill P, Sasse JM, Williams EG, Rowan KS (1985) Effect of brassinolide and other growth regulators on the germination and growth of pollen tubes of Prunus avium using a multiple hanging drop assay. Aust J Plant Physiol 1:201–211
Kauschmann A, Jessop A, Koncz C, Szekeres M, Willmitzer L, Altmann T (1996) Genetic evidence for an essential role of brassinosteroids in plant development. Plant J 9:701–713
Li J, Chory J (1997) A putative leucine-rich repeat receptor kinase involved in brassinosteroid signal transduction. Cell 90:929–938
Li HB, Chen GD, Yan W (2015) Molecular characterization of barley 3H semi-dwarf genes. PLoS One 10(3):e0120558
Mickelson HR, Rasmusson DC (1994) Genes for short stature in barley. Crop Sci 34:1180–1183
Milach SCK, Federizzi LC (2001) Dwarfing genes in plant improvement. Adv Agron 73:35–63
Powell W, Caligari PDS, Swanston JS, Jinks JL (1985) Genetic investigations into beta-glucan content in barley. Theor Appl Genet 71:461–466
Rasmusson DC (1991) A plant breeder’s experience with ideotype breeding. Field Crop Res 26:191–200
Rebetzke GJ, Richards RA, Fettell NA, Long M, Condon AG, Forrester RI, Botwright TL (2007) Genotypic increases in coleoptile length improves stand establishment, vigour and grain yield of deep-sown wheat. Field Crop Res 100:10–23
Rikiishi K, Saisho D, Takeda K (2008) Uzu, a barley semi-dwarf gene, suppresses plant regeneration in calli derived from immature embryos. Breed Sci 58:149–155
Saisho D, Tanno K, Chono M, Honda I, Kitano H, Takeda K (2004) Spontaneous brassinolide-insensitive barley mutants ‘uzu’ adapted to East Asia. Breed Sci 54:409–416
Szekeres M, Nemeth K, Koncz-Kalman Z, Mathur J, Kauschmann A, Altmann T, Redei GP, Nagy F, Schell J, Koncz C (1996) Brassinosteroids rescue the deficiency of CYP90, a cytochrome P450, controlling cell elongation and de-etiolation in Arabidopsis. Cell 85:171–182
Takahashi T, Gasch A, Nishizawa N, Chua N (1995) The DIMINUTO gene of Arabidopsis is involved in regulating cell elongation. Genes Dev 9:97–107
Takeda K, Takahashi H (1999) Varietal variation for deep-seeding tolerance in barley and wheat. Breed Res 1:1–8
Thomas WTB, Powell W, Swanston JS (1991) The effects of major genes on quantitatively varying characters in barley. 4. The GPert and denso loci and quality characters. Heredity 66:381–389
Tuinstra MR, Ejeta G, Goldsbrough PB (1997) Heterogeneous inbred family (HIF) analysis: a method for developing near-isogenic lines that differ at quantitative trait loci. Theor Appl Genet 95:1005–1011
Wang JM, Yang JM, McNeil DL, Zhou MX (2010) Identification and molecular mapping of a dwarfing gene in barley (Hordeum vulgare L.) and its correlation with other agronomic traits. Euphytica 175:331–342
Zhang J, Zhang W (2003) Tracing sources of dwarfing genes in barley breeding in China. Euphytica 131:285–292
Zheng Z, Wang HB, Chen GD, Yan GJ, Liu CJ (2013) A procedure allowing up to eight generations of wheat and nine generations of barley per annum. Euphytica 191:311–316
Acknowledgments
This publication is based upon work supported by CSIRO (Project R-04818) and the International Science & Cooperation Program of China (No. 2015DFA30600). GC thanks the China Scholarship Council for funding his visit to CSIRO. The authors are also grateful to Caritta Eliasson for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Guangdeng Chen and Haobing Li have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, G., Li, H., Wei, Y. et al. Pleiotropic effects of the semi-dwarfing gene uzu in barley. Euphytica 209, 749–755 (2016). https://doi.org/10.1007/s10681-016-1668-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-016-1668-4