Abstract
A rapid increase in the application of synthetic pesticides in agricultural practices causes a severe hazard to agro-sustainability and soil microflora. Considering these threatening problems, the present study aimed to investigate the impact of different groups of pesticides on growth, cellular morphostructure, permeability and antioxidant enzyme of a symbiotic Gram-negative bacterium recovered from chickpea nodules. Pesticide-tolerant strain BRM5 was characterized and phylogenetically (accession no. KY013481) identified as Mesorhizobium ciceri. Strain BRM5 tolerated up to the level of 1200, 1600, 1600, 2400, 2400, 1800, 2400, 2400 and 3200 μg mL−1 of glyphosate, quizalofop, atrazine, kitazin, metalaxyl, hexaconazole, fipronil, monocrotophos and imidacloprid, respectively. Among all concentrations, 3X concentrations of each pesticide had most fatal to bacterial cells. The growth pattern of strain BRM5 was decreased linearly with increasing incubation times. Scanning electron micrograph (SEM) images of pesticide-treated bacterial cells revealed the distorted, broken, irregular, misshaped and larger-sized cell as compared to untreated cells. Upon staining with acridine orange (AO)/propidium iodide (PI), active/living cells appeared as green colour, whereas dead cells appeared as red colour under confocal microscope (CLSM). This is the clear indication of loss in cell viability following the pesticide exposure. The generation of oxidative stress (catalase) by strain BRM5 induced under pesticides pressure further revealed the pesticidal toxicity. CAT activity of BRM5 was decreased with the enhancement in pesticide concentrations. For instance, CAT activity of BRM5 was decreased by 45%, 71% and 83% when cultured in liquid medium treated with 3X doses of each glyphosate, kitazin and fipronil, respectively. Overall, the present study visibly indicated that pesticides inhibited the growth, disrupted the morphology and reduced the permeability and antioxidant producing ability of bacteria. So, from this study, it is suggested that before applying the pesticides in legume production strategies, the recommended pesticides be carefully examined to avoid the toxicity problems, if any.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
In agronomic practices, pesticides are habitually applied to combat the damages caused by numerous pests. Extreme usage of these agrochemicals, in turn, has led to serious problems such as loss in soil fertility (Saha et al. 2017), non-target vegetations (Aktar et al. 2009) and serious environmental pollution (Datta et al. 2018). Apart from these, pesticides could be poisonous to beneficial soil microbiota (Wu et al. 2018), useful soil insects (Heard et al. 2017) and food crops (Carvalho 2017) following consumption via food chain (Nollet and Fernandez-Alba 2017). Much information is available regarding the impact of pesticides on soil bacteria, fungi, algae and higher organisms (Asad et al. 2017). Among the several groups of pesticides, herbicidal toxicity could be higher that constrains the chemical and biological processes of both the plants and microorganisms (Rubio-Bellido et al. 2018). In addition, pesticides hamper the growth of soil beneficial bacteria (Shahid and Khan 2017) and nodule-inhabiting microsymbiont such as Rhizobium (Raghavendra et al. 2017) depending on the concentration and their mode of action and hence decline the nodulation process in legumes (Kaur et al. 2016). During nodulation, entry of rhizobia inside the leguminous plants occurs via rhizobial infection of root hairs, and since the bacterial infection period is temporary and restricted to actively growing hairs, pesticides affect the growth of root hair and thus destruct the infection process. In addition, pesticides downregulate the expression of nod genes involved in the process of nodulation and nitrogenase production (Niewiadomska and Klama, 2005). In addition, pesticides also produce a number of free radicals that upsurge the oxidative responses of cellular machinery in microbes (Abdollahi et al. 2004). Furthermore, plant-associated soil microbes designated as plant growth-promoting rhizobacteria (PGPR) possess antioxidant defence enzymes (Sarkar et al. 2018) such as catalases (CAT), peroxidases (POD) and superoxide dismutase (SOD) which help in reducing the oxidative stress exerted by pesticides. However, pesticides also alter the antioxidant defence enzymes in soil bacterial population (Shahid and Khan 2018a, b). Contrarily, development of resistance in soil microbial population reduces the impact of pesticide toxicity (Srivastava et al. 2016). Therefore, the increased collective concern towards the indiscriminate and injudicious use of toxic pesticides has encouraged the agronomists and microbiologists to apply beneficial soil rhizobacteria in combination with integrated pest management (IPM) strategies. If properly and meticulously applied, this strategy of using pesticide-resistant microorganisms in agronomic practices is very likely to significantly reduce the pesticide usage and might be more economic, target-specific and eco-friendly approach.
Despite a few successes achieved so far, there is a critical gap in revealing the fact that how pesticides influence the architecture and survival of root-nodulating rhizobacteria. Therefore, to better explain such mechanisms and to explore the possibility of emerging pesticide resistance in bacterial entities capable of forming symbiotic relationship with leguminous plants, the current study was intended to: (1) identify and characterize the pesticide-tolerant root-nodulating bacterium of Cicer arietinum, (2) evaluate the effect of different groups of pesticides (herbicides, fungicides and insecticides) on growth pattern of bacterial strain, (3) assess the oxidative stress induced by pesticides in strain BRM5 and (4) determine the impact of pesticides on morphological structure and cellular permeability of bacterial strain.
2 Materials and methods
2.1 Recovery of root nodule bacteria and pesticide tolerance
Fresh and undamaged healthy nodules were detached from Cicer arietinum (chickpea) plants raised in soils (sandy clay loam and having organic C 6.2 g kg−1, Kjeldahl N 0.75 g kg−1, Olsen P 16 mg kg−1, pH 7.2 and WHC 0.44 mL g−1, cation exchange capacity 11.7 and 5.1 cmol kg−1 anion exchange capacity) of experimental fields of Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh (27°53′N 78°05′E 27.88°N 78.08°E), western district of Uttar Pradesh, India, which had previous history of pesticide application for the last 18 years. Nodules were surface-sterilized by dipping the nodules in 4% sodium hypochlorite (NaOCl) for 2 min, washed thrice with sterilized double-distilled water (DDW) and crushed gently under an aseptic condition. A 100-μL of freshly extracted nodule suspensions was streaked on plates of yeast extract mannitol (YEM) agar, and the plates were incubated at 28 ± 2 °C for 3–5 days. A total of 15 Mesorhizobial isolates were recovered from chickpea root nodule and morphologically and biochemically characterized (Holt et al. 2009). A plant infection technique was carried out to determine the host specificity (Vincent 1970). Mesorhizobium isolates were exposed to different dose rates of different pesticides tested (Table 1) using minimal salt agar (MSA) (g L−1: KH2PO4 1.0, K2HPO4 1.0, NH4NO3 1.0, MgSO4·7H2O 0.2, CaCl2·2H2O 0.02, FeSO4·7H2O 0.01, pH 6.5) for selection of pesticide-tolerant Mesorhizobium strains. The freshly prepared agar plates were amended individually with concentrations ranging from 0 to 6400 μg mL−1 at twofold dilution intervals for each pesticide. The plates were spot (10 μL) inoculated with 108 cells mL−1 of freshly grown bacterial cells and incubated at 28 ± 2 °C for 3 days. Rhizobial colonies growing and efficiently persisting at the maximum dose rates of pesticides were chosen and referred to as pesticide-tolerant mesorhizobial strains (PTMS). Among all the recovered Mesorhizobium strains, BRM5 strain exhibited the maximum tolerance to pesticides, so this strain was selected for further studies.
2.2 Identification of Mesorhizobial strain and construction of phylogenetic tree
Pesticide-tolerant Mesorhizobium strain BRM5 was characterized at the level of species adopting 16S rDNA gene sequence method by means of universal primers (785F and 907R). The nucleotide sequence data so found were deposited in the GenBank sequence database. Moreover, the sequences were pooled and phylogenetic tree was built with the help of MEGA 6.0 software.
2.3 Growth kinetics of bacterial strain under pesticide stress
The growth response/behaviour of bacterial strain (BRM5) was assessed by growing the log-phase cells (1 × 107 CFU mL−1) into minimal salt (MS) broth medium comprising of one (1X), two (2X) and three (3X) times more of normal rates of pesticides. The normal doses of glyphosate, quizalofop, atrazine, kitazin, hexaconazole, metalaxyl, fipronil, monocrotophos and imidacloprid applied throughout the experimental trial were 300, 400, 300, 500, 600, 400, 500, 600 and 700 μg mL−1, respectively. The MS medium without pesticides but containing only bacterial strain served as control. The inoculated medium was maintained at 28 ± 2 °C on a rotary shaker, and growth response was measured with the help of a spectrophotometer at 620 λ at periodic intermissions (0–24 h), and a curve was plotted.
2.4 Oxidative damage experiment
2.4.1 Catalase (CAT) assay
The CAT activity of bacterial cell (strain BRM5) grown in pesticide-supplemented medium was assessed (Abei 1974). For the assay, 100 μL of bacterial cell lysate from the tissue homogenate was properly mixed with 2 mL of 50 mM PBS (phosphate-buffered saline) (pH 7.0), and 30 mM of 100 μL of hydrogen peroxide (H2O2) was added to it. The decrease in the absorbance was recorded at the wavelength of 240 λ for 3 min using a UV–vis spectrophotometer (Shimadzu 2600, Japan). The results were interpreted as the nanomolar of H2O2 min−1 μg−1 protein.
2.5 Assessment of structural damage and permeability determination
2.5.1 Cellular/structural damage by scanning electron microscopy (SEM)
Pesticide-induced cellular damage/distortion in BRM5 strains was observed under SEM by growing the bacterial strains in NB medium treated with 900 µg mL−1 glyphosate, 1200 µg mL−1 quizalofop, 900 µg mL−1 atrazine, 1500 µg mL−1 kitazin, 1800 µg mL−1 hexaconazole, 1200 µg mL−1 metalaxyl, 1500 µg mL−1 fipronil and 1800 µg mL−1 monocrotophos and incubated at 28 ± 2 °C for 24 h. The culture grown in pesticide-free medium served as control. After incubation, cultures were centrifuged at 12,000 rpm for 10 min, and pellet was suspended in 1 × PBS and the cell pellets were washed again three times with 1 × PBS and pre-fixed with 2.5% glutaraldehyde for overnight at 4 °C. The cells were recovered by centrifugation at 10,000 rpm for 5 min, and pellets were again washed with the same buffer. After three successive washing cycles, the fixed specimens were dehydrated in a graded series (30, 50, 70, 90 and 100%) of ethanol for 5 min each. Thereafter, cell pellets were centrifuged and re-suspended in PBS. Five μL of bacterial suspension was smeared on coverslip and dried. The specimens were mounted and analysed under the SEM to see the changes in bacterial structures, if any, and images were recorded.
2.5.2 Membrane integrity and bacterial mortality under CLSM
Alterations in the membrane integrity and bacterial mortality were assessed by fluorescence microscopy with some modifications (Rizvi et al. 2019). To 200 μL of treated and untreated bacterial suspensions, 10 μL of acridine orange (AO: 15 μg mL−1 prepared in PBS) and 10 μL of propidium iodide (PI: 50 μg mL−1 prepared in PBS) were added. Bacterial suspensions treated with both AO and PI stains were incubated at room temperature for 10 min and centrifuged (at 5000 rpm) for 10 min. The supernatants were discarded to remove the unbound dyes, while cell pellets were re-suspended in 500 μL of PBS. The experimental set-up was maintained in the dark conditions to avoid photobleaching of dyes.
2.6 Statistical analysis
The data obtained throughout the study were analysed statistically by Duncan’s multiple range (DMRT) test and LSD between the treatments calculated by means of Minitab 17 statistical software at 5% probability level.
3 Results and discussion
3.1 Strain identification and characterization
In the present investigation, a total of 15 mesorhizobial isolates varying in colonial morphology on YEMA plates were recovered from root nodules of Cicer arietinum. Among the total mesorhizobial strain, BRM5 was found as Gram negative and rod in shape displaying inconstant biochemical reactions (Table 2). Based on the variable biochemical characteristics, strain BRM5 was presumptively identified as Mesorhizobium ciceri. Additionally, the molecular characterization was done by using 16S rDNA sequencing technology to know the precise species of this strain (BRM5). After molecular sequencing, the 16S rDNA nucleotide sequences (approximately 614 nbp) of BRM5 strain were submitted to GenBank, NCBI (accession number KY013481.1). Kinship of the strain BRM5 was explored by basic local alignment search tool for nucleotide (BLASTn) which suggested that the test strain was phylogenetically related to M. ciceri, and due to the high frequency of relatedness (> 95%), it was authenticated as M. ciceri. Later, a phylogenetic tree was constructed using multiple sequence alignment by ClustalW and neighbour-joining (NJ) method using the FASTA format sequences of similar bacteria (Fig. 1). In recent time, the isolation and molecular characterization of rhizobial population in various regions of the world have received increased attention due to their important characteristic features of biological nitrogen fixation (BNF) and symbiotic association with leguminous crops (Lindström and Mousavi 2019). The sequence analysis of 16S rRNA has emerged as one of the most important methods in phylogenetic analysis of bacteria (Pandey et al. 2019). Mesorhizobium ciceri was isolated from chickpea nodules and very first time identified by Nour et al. (1994). In recent time, various workers have isolated Mesorhizobium strains from root nodules of chickpea nodule plants grown in different agro-climatic regions and molecularly characterized using 16S RNA partial gene sequence analysis (Martinez Condom 2019; Singh et al. 2019; Pandey et al. 2018; Zafar et al. 2017). However, due to the high sequence conservation, 16S RNA partial gene sequence analysis is insufficient to distinguish the strains of one species or even closely related species (Martens et al. 2008). Recently, housekeeping genes located on the bacterial chromosome have been used in combination with 16S rRNA for elucidation of taxonomic relationship (Rong and Huang 2010). In Mesorhizobium spp., symbiotic genes present in their chromosome have also been characterized alongside the housekeeping genes to understand the phylogenetic relationship among the different strains (Chen et al. 2008).
3.2 Pesticide tolerance
In our study, mesorhizobial isolate M. ciceri BRM5 tolerated a significant amount of fungicides (hexaconazole, kitazin and metalaxyl), herbicides (atrazine, quizalofop and glyphosate) and insecticides (imidacloprid, monocrotophos and fipronil) when grown on C- and N-deprived minimal salt agar (MSA) plates added with the ordered doses (0–6400 μg mL−1) of nine pesticides. The tolerance levels of M. ciceri BRM5 against pesticides ranged between 900 and 3200 μg mL−1. M. ciceri when treated with varying rates of pesticides in semi-solid agar and liquid broth MS medium (solid/liquid), displayed tolerance in the following order: imidacloprid (3200:2100 μg mL−1) > monocrotophos/hexaconazole (2400:1800 μg mL−1) > fipronil/kitazin (2400:1500 μg mL−1) > metalaxyl (1800:1200 μg mL−1) > quizalofop (1600:1200 μg mL−1) > atrazine (1600:900 μg mL−1) > glyphosate (1200:900 μg mL−1).
In this study, M. ciceri BRM5 displayed the maximum tolerance against the imidacloprid in broth as well as semi-solid agar media. Usually, pesticides are applied in cultivation practices to negate dreadful pests, for example, phytopathogens, insects and weeds, to safeguard the crop damages and hence to enhance the food productivity. However, haphazard and redundant application and deposition of agricultural pesticides in soils lead to both the decline in plant productivity and development of resistance between the soil microorganisms. In order to better understand the toxicity of pesticides, we identified and cultivated a new and unique pesticide-tolerant nodule-inhabiting bacterium that alleviated the pesticidal toxicity and might concurrently be used to augment the crop production even in soils contaminated with pesticides. M. ciceri strain BRM5 when exposed to variable concentrations of pesticides exhibited exceptional tolerance ability to different pesticides. Since strain BRM5 was grown on MS medium amended with different doses of pesticides, it is supposed that this symbiotic bacterium might have degraded the test pesticides and utilized it as a nutrient source for their growth and existence. This ability of M. ciceri BRM5 expressing higher tolerance and degradation of pesticides among plant beneficial bacteria is a fascinating and intricate phenomenon (Jahn et al. 2017). However, the tolerance to any pesticides could be: (1) temporary: pesticides induce the changes at physiological level that alters microbial metabolism (Wu et al. 2018) so that a new metabolic pathway is generated (Cycon et al. 2013) which helps in degrading the encountered pesticide. As a result, microbial populations protect an essential biochemical reaction which otherwise could be suppressed by pesticide and (2) permanent: they depend upon genetic modifications which are inherited by subsequent generations of microorganisms (Ahemad and Khan 2010; Bellinaso et al. 2003; Johnsen et al. 2001). Microbes that have established either temporary or permanent resistance to pesticides have been found capable of degrading pesticides and hence altering its toxicity (Sang et al. 2016).
3.3 Growth kinetics of bacterial strain under pesticide pressure
Growth behaviour of M. ciceri strain BRM5 to different concentrations of glyphosate (Fig. 2a), quizalofop (Fig. 2b), atrazine (Fig. 2c), kitazin (Fig. 2d), hexaconazole (Fig. 2e), metalaxyl (Fig. 2f), fipronil (Fig. 2g), monocrotophos (Fig. 2h) and imidacloprid (Fig. 2i) noticeably wandered. In the start (lag phase), the growth of bacterial cells was slow, which, further, increased an exponential mode with an enhancement in number of cells and afterwards it dropped sharply. Among the three selected doses, 900, 1200, 900, 1500, 1800, 1200, 1500, 1800 and 2100 μg mL−1 of each glyphosate, quizalofop, atrazine, kitazin, hexaconazole, metalaxyl, fipronil, monocrotophos and imidacloprid were most fatal to the cells of BRM5 and caused most detrimental impact on the growing cells compared to other doses (1X and 2X) of pesticides. The noxious and deadly effect of all the chosen pesticides on growing cells of Mesorhizobial strain was probably just because of the soluble and mobile nature of these agricultural chemicals in liquid medium relative to those assessed on solid agar medium. The subsequent exposure of pesticides to the growing cells of BRM5 and a consistent decline in growth could possibly be due to the interaction of such chemical compounds with the bacterial cytoplasmic membrane enhancing its permeability (Wasi et al. 2011). In the same way, arrest of membrane respiration is a common phenomenon that is responsible for the bacterial mortality (Haroon et al. 2019). Furthermore, pesticides may disturb the internal metabolic activity by binding to the –NH2 and S2− groups after their uptake by bacterial cells and thus cause damage to the complete metabolic machinery of growing bacteria. Deviations take place throughout in the oxido-reduction potential of soil, and depending on the speciation and pesticides dose rate, microbes may eventually be killed or eliminated from polluted areas. Prior to our study, lethal and growth-retarding impact of pesticides on growth behaviour of other soil bacterium growing in MS medium supplemented with different doses of pesticides has formerly been reported (Alam et al. 2018; Niemi et al. 2009).
Growth pattern of M. ciceri strain BRM5 at increasing concentrations of pesticides as a function of time of incubation at 28 ± 2 °C. Data represent the means ± SD of three independent experiments done in triplicate. a–i Represent the growth behaviour of strain BRM5 grown in MSM broth supplemented/treated with 300, 600, 900 µg mL−1 glyphosates, 400, 800, 1200 µg mL−1 quizalofop, 300, 600, 900 µg mL−1 atrazine, 500, 1000, 1500 µg mL−1 kitazin, 600, 1200 1800 µg mL−1 hexaconazole, 400, 800. 1200 µg mL−1 metalaxyl, 500, 1000, 1500 µg mL−1 fipronil, 600, 1200, 1800 µg mL−1 monocrotophos and 700, 1400, 2100 µg mL−1 imidacloprid, respectively
3.4 Assessment of oxidative stress
The by-products of the reactive oxygen species such as O2−, H2O2 and OH· are formed in the cells of bacterial membrane which are greatly influenced by the xenobiotic (Ahmed et al. 2018). Also, agricultural chemicals and other organic pesticides are well-known toxicants against the bacteria cells which modulate the structural configuration and composition of the microorganisms that resulted in oxidative stress (Cao et al. 2017). Consequently, the oxidative stress produced after pesticide exposure was determined in the present investigation. The results revealed that glyphosate-, quizalofop-, atrazine-, kitazin-, hexaconazole-, metalaxyl-, fipronil-, monocrotophos- and imidacloprid-treated cells of strain BRM5 had a noteworthy negative effect on catalase activity (Fig. 3a–c). There was a significantly higher (P < 0.05) toxic impact on the bacterial cells when grown with monocrotophos (1800 μg mL−1), which led to a significant (P < 0.05) decrease in CAT activity (0.010 mM H2O2 min−1 μg−1 protein) compared to control cells (0.12 mM H2O2 min−1 μg−1 protein). Similarly, cells of BRM5 strain grown in liquid culture broth supplemented with three times of the normal doses of atrazine and imidacloprid excreted 0.012 and 0.06 mM H2O2 min−1 μg−1 protein at 900 and 2100 μg mL−1, respectively (Fig. 3a–c). Toxic behaviour of pesticides and other numerous organic solvents are well recognized against the bacteria leading to several changes in the body producing the membrane and oxidative damage (Jabłońska-Trypuć 2017). Several species of pesticides such as paraquat, bipyridylium, acetochlor, acetamiprid and synthetic auxins can also induce oxidative stress due to obstruction of electron flow through the ETC (electron transport chain) and can directly/indirectly influence the structure and function of the bacterial membranes (Lü et al. 2009; Isik et al. 2007). Exposure to pesticides alters the protein expression associated with energy metabolism and membrane transport and thus increases the oxidative stress (Bhat et al. 2015). Also, oxidative damage can activate the SOS responses, arrest cell division and promote/elongate and initiate the DNA repair process (Cooper and Lovett 2011; Kawai et al. 2003). In a follow-up study, Martins et al. (2011) have reported the noxious impacts of two herbicides (acetochlor and metolachlor) on antioxidative enzymes systems (CAT and GR) of a soil bacterium Enterobacter asburiae and concluded that these specific isoenzymes could be involved in the stress tolerance ability of bacterium. In yet another experiment, Mongkolsuk et al. (2005) recommended that bacterium Xanthomonas can respond to the oxidative damages induced by xenobiotics and environmental contaminants such as pesticides and heavy metals, through physiological adaptation to hydrogen peroxide (H2O2). Additionally, the acetamiprid-induced responses on superoxide dismutase (SOD), catalase (CAT) and ascorbate peroxidase (APX) activity of soil bacteria have been reported (Yao et al. 2006).
Catalase activity in cells of M. ciceri strain BRM5 induced by selected and treated with 300, 600, 900 µg mL−1 glyphosates, 400, 800, 1200 µg mL−1 quizalofop, 300, 600, 900 µg mL−1 atrazine, 500, 1000, 1500 µg mL−1 kitazin, 600, 1200 1800 µg mL−1 hexaconazole, 400, 800. 1200 µg mL−1 metalaxyl, 500, 1000, 1500 µg mL−1 fipronil, 600, 1200, 1800 µg mL−1 monocrotophos and 700, 1400, 2100 µg mL−1 imidacloprid, respectively. Corresponding error bars represent standard deviation (SD) of three replicates (SD, n = 3)
3.5 Structural damage and membrane permeability assessment
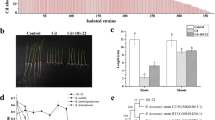
The toxic and damaging impacts of selected pesticides on root-nodulating M. ciceri strain BRM5 were perceived under SEM. It was detected that cells of strain BRM5 developed in the absence of pesticides appeared as smooth, rod-shaped cells with uninjured surface morphostructure (Fig. 4a). Contrary to this, electron micrographs of bacterium treated with glyphosate (Fig. 4b), quizalofop (Fig. 4c), atrazine (Fig. 4d), kitazin (Fig. 4e), hexaconazole (Fig. 4f), metalaxyl (Fig. 4g), fipronil (Fig. 4h) and monocrotophos (Fig. 4i) at dose rates of 900, 1200, 900, 1500, 1800, 1200, 1500 and 1800 μg mL−1, respectively, embodied a cracked/damaged, distorted, misshaped and somehow increased bacterial cell length under scanning electron microscopy.
SEM of M. ciceri strain BRM5; control (a) treated with 900 µg mL−1 glyphosate (b), 1200 µg mL−1 quizalofop (c), 900 µg mL−1 Atrazine (d), 1500 µg mL−1 kitazin (e), 1800 µg mL−1 hexaconazole (f), 1200 µg mL−1 metalaxyl (g) and 1500 µg mL−1 fipronil (h) and 1800 µg mL−1 monocrotophos (i). Yellow arrows and circles represent the distorted/damaged, broken bacterial cells due to the toxicity of pesticides
For the assessment of cellular membrane permeability of bacterial cells under CLSM, strain BRM5 was grown in MS liquid broth supplemented with different dose rates of pesticides and stained with AO and PI. Untreated cells of M. ciceri were viable and appeared as green (Fig. 5a), whereas cells treated with 900, 1200, 900, 1500, 1800, 1200, 1500 and 1800 and 2100 μg mL−1 glyphosate (Fig. 5b), quizalofop (Fig. 5c), atrazine (Fig. 5d), kitazin (Fig. 5e), hexaconazole (Fig. 5f), metalaxyl (Fig. 5g), fipronil (Fig. 5h) and monocrotophos (Fig. 5i, j) and imidacloprid (Fig. 5k, l), respectively, exhibited red-coloured small rods that are the representative of injured/dead cells, and this is the clear indication of pesticidal toxicity to bacterial cells. Among all the test pesticides, atrazine and glyphosate had more remarkable toxic impacts on cells of strain BRM5, resulting in increased cell mortality which was evident by an increase in red fluorescence.
Propidium iodide (PI) and acridine orange (AO) stained CLSM images of M. ciceri strain BRM5 untreated control cells (a), treated with 900 µg mL−1 glyphosate (b), 1200 µg mL−1 quizalofop (c), 900 µg mL−1 atrazine (d), 1500 µg mL−1 kitazin (e), 1800 µg mL−1 hexaconazole (f), 1200 µg mL−1 metalaxyl (g), 1500 µg mL−1 fipronil (h), 1800 µg mL−1 monocrotophos (i) and 2100 µg mL−1 imidacloprid (j, k). The red- and orange coloured dots depict the number of dead cells
Changes at morphological level in microorganisms under the harsh/stressed conditions are an important observable parameter of bacteriological adaptation (Chakravarty and Banerjee 2008; Jan et al. 2001). Morphological changes in microorganism as an adaptive response to stressed/adverse conditions have been reported by several workers (Ahmed et al. 2019; Saif and Khan 2018; Shahid et al. 2018; Millach et al. 2017; Gandhi and Shah 2016). But, reports on the effect of pesticidal stress on structural morphology of soil beneficial microorganisms, especially on root-nodulating symbiotic bacteria, are negligible. In this study, morphology of different groups of pesticide (fungicide, herbicide and insecticide)-treated cells of M. ciceri strain BRM5 observed under SEM provided a strong evidence that pesticides are indeed stressful to the bacterial cells, characterized by distorted, broken, mis-shaped, irregular and somehow larger cells. The increased size of bacterial cell decreased the contact surface and therefore condensed the attachable surface for synthetic and organic (pesticides) compounds. Consequently, larger cells can endure superiorly under the stressed environment than the control cells of same bacterial species. This study, therefore, clearly revealed that nodule bacteria do have an adaptive measure to counteract pesticide stress. In an identical experiment, Shahid and Khan (2018a, b) have also found some significant damages to Bacillus subtilis while growing under stress.
Cell membranes permit the cells to interconnect with their contiguous surroundings. Being selectively permeable, bacterial cells allow a controlled exchange of molecules across it. So, like any other cellular components, pesticides and other stressor molecules cause toxicity to membrane, which results in oxidative damage. To affirm this, the bacterial cells were stained with AO/PI, which is reported to interpose with the genetic material as nucleic acids (DNA and RNA) comprising of negatively charged PO4 group, sugars (ribose and deoxyribose) and H+ bonding. Of these dyes, PI is most commonly used as a marker of cell death and it is excluded by the cell membranes of living entities. Hence, the fluorescence conferred by dye is frequently correlated with cells whose membrane integrity was damaged. And hence, when observed under CLSM, a clear toxicity to bacterial membranes due to pesticides was also reported by Shahid and Khan (2018a, b) for Bacillus subtilis.
4 Conclusion
Conclusively, the inconstant doses of different pesticidal species applied individually had a clear and visible toxic impact on M. ciceri strain BRM5 with progressive decline in growth pattern, structural damage, oxidative stress and membrane disintegration. While assessing the growth kinetics, pesticides hampered the growth of BRM5 in time- and concentration-dependent manner. Pesticides, in general, enhanced the permeability of cell membrane, which consequently increased the uptake of test pesticides and reduced the cell viability. Similarly, alterations in CAT activity of bacterial cells treated with different pesticides were noticeable. The findings of the current research revealed the agricultural importance of a novel bacterium M. ciceri BRM5 under pesticide stress. Therefore, the present investigation clearly suggests that before applying the pesticides, for optimizing crop production, they must be carefully examined for toxicity aspects.
References
Abdollahi, M., Ranjbar, A., Shadnia, S., & Nikfar, S. (2004). Pesticides and oxidative stress: a review. Medical Science Monitor, 10, 141–147.
Abei, H. (1974). Methods of enzymatic analysis. Catalase, 673–684.
Ahemad, M., & Khan, M. S. (2010). Plant growth promoting activities of phosphate-solubilizing Enterobacter asburiae as influenced by fungicides. European Asian Journal of Biological Sciences, 4, 88–95.
Ahmed, B., Hashmi, A., Khan, M. S., & Musarrat, J. (2018). ROS mediated destruction of cell membrane, growth and biofilms of human bacterial pathogens by stable metallic AgNPs functionalized from bell pepper extract and quercetin. Advanced Powder Technology, 29(7), 1601–1616.
Ahmed, B., Solanki, B., Zaidi, A., Khan, M. S., & Musarrat, J. (2019). Bacterial toxicity of biomimetic green zinc oxide nanoantibiotic: insights into ZnONP uptake and nanocolloid–bacteria interface. Toxicology Research, 8(2), 246–261.
Aktar, W., Sengupta, D., & Chowdhury, A. (2009). Impact of pesticides use in agriculture: Their benefits and hazards. International Toxicology, 2, 1–12.
Alam, S., Kumar, A., Kumar, A., Prasad, S., Tiwari, A., Srivastava, D., Srivastava, S., Tiwari, P., Singh, J., & Mathur, B., (2018). Isolation and characterization of pesticide tolerant bacteria from brinjal rhizosphere. International Journal of Current Microbiology and Applied Sciences, 4849–4859.
Asad, M. A. U., Lavoie, M., Song, H., & Jin, Y. (2017). Interaction of chiral herbicides with soil microorganisms, algae and vascular plants. Science of the Total Environment, 80, 1287–1299.
Bellinaso, M., Greer, C. W., & Peralba, M. (2003). Biodegradation of the herbicide trifluralin by bacteria isolated from soil. FEMS Microbial Ecology, 43, 191–194.
Bhat, S. V., Booth, S. C., Vantomme, E. A., & Afroj, S. (2015). Oxidative stress and metabolic perturbations in Escherichia coli exposed to sublethal levels of 2, 4-dichlorophenoxyacetic acid. Chemosphere, 135, 453–461.
Cao, X., Yu, C., Wang, H., Zhou, F., & Li, X. (2017). Simultaneous degradation of refractory organic pesticide and bioelectricity generation in a soil microbial fuel cell with different conditions. Environmental Technology, 38(8), 1043–1050.
Carvalho, F. P. (2017). Pesticides, environment, and food safety. Food and Energy Security, 6, 48–60.
Chakravarty, R., & Banerjee, P. C. (2008). Morphological changes in an acidophilic bacterium induced by heavy metals. Extremophiles, 12(2), 279–284.
Chen, W. F., Guan, S. H., Zhao, C. T., Yan, X. R., Man, C. X., Wang, E. T., et al. (2008). Different Mesorhizobium species associated with Caragana carry similar symbiotic genes and have common host ranges. FEMS Microbiology Letters, 283(2), 203–209.
Cooper, D. L., & Lovett, S. T. (2011). Toxicity and tolerance mechanisms for azidothymidine, a replication gap-promoting agent. Escherichia coli DNA Repair, 10(3), 260–270.
Cycoń, M., Markowicz, A., Borymski, S., Wójcik, M., & Piotrowska-Seget, Z. (2013). Imidacloprid induces changes in the structure, genetic diversity and catabolic activity of soil microbial communities. Journal of Environmental Management, 131, 55–65.
Datta, S., Singh, J., Singh, J., & Singh, S. (2018). Assessment of genotoxic effects of pesticide and vermicompost treated soil with Allium cepa test. Sustainable Environmental Research. https://doi.org/10.1016/j.serj.2018.01.005.
Gandhi, A., & Shah, N. P. (2016). Effect of salt stress on morphology and membrane composition of Lactobacillus acidophilus, Lactobacillus casei, and Bifidobacterium bifidum, and their adhesion to human intestinal epithelial-like Caco-2 cells. Journal of Dairy Science, 99(4), 2594–2605.
Haroon, M., Zaidi, A., Ahmed, B., Rizvi, A., Khan, M. S., & Musarrat, J. (2019). Effective inhibition of phytopathogenic microbes by eco-friendly leaf extract mediated silver nanoparticles (AgNPs). Indian Journal of Microbiology, 59(3), 273–287.
Heard, M. S., Baas, J., Dorne, J. L., & Lahive, E. (2017). Comparative toxicity of pesticides and environmental contaminants in bees: are honey bees a useful proxy for wild bee species? Science of the Total Environment, 578, 357–365.
Holt, G. J., Krieg, N.R., & Sneath, P. H. A. (2009). Gram negative aerobic/microaerophilic rods and cocci. In Bergey’s manual of determinative bacteriology.
Işık, K., Kayalı, H. A., Şahin, N., Gündoğdu, E. Ö., & Tarhan, L. (2007). Antioxidant response of a novel Streptomyces sp. M3004 isolated from legume rhizosphere to H2O2 and paraquat. Process Biochemistry, 42(2), 235–243.
Jabłońska-Trypuć, A. (2017). Pesticides as inducers of oxidative stress. Reactive Oxygen Species, 3(8), 96–110.
Jahn, L. J., Munck, C., Ellabaan, M. M., & Sommer, M. O. (2017). Adaptive laboratory evolution of antibiotic resistance using different selection regimes lead to similar phenotypes and genotypes. Frontiers in Microbiology. https://doi.org/10.3389/fmicb.2017.00816.
Jan, G., Leverrier, P., Pichereau, V., & Boyaval, P. (2001). Changes in protein synthesis and morphology during acid adaptation of Propionibacterium freudenreichii. Applied Environmental Microbiology, 67(5), 2029–2036.
Johnsen, K., Jacobsen, C. S., Torsvik, V., & Sørensen, J. (2001). Pesticide effects on bacterial diversity in agricultural soils-a review. Biology and Fertility of Soils, 33, 443–453.
Kaur, H., Bhardwaj, R., Kumar, V., & Sharma, A. (2016). Effect of pesticides on leguminous plants: an overview. In Legumes under environmental stress: Yield, improvement and adaptations 91.
Kawai, Y., Moriya, S., & Ogasawara, N. (2003). Identification of a protein, YneA, responsible for cell division suppression during the SOS response in Bacillus subtilis. Molecular Microbiology, 47(4), 1113–1122.
Lindström, K., & Mousavi, S. A. (2019). Effectiveness of nitrogen fixation in rhizobia. Microbial Biotechnology. https://doi.org/10.1111/1751-7915.13517.
Lü, Z., Sang, L., Li, Z., & Min, H. (2009). Catalase and superoxide dismutase activities in a Stenotrophomonas maltophilia WZ2 resistant to herbicide pollution. Ecotoxicology and Environmental Safety, 72(1), 136–143.
Martens, M., Dawyndt, P., Coopman, R., Gillis, M., De Vos, P., & Willems, A. (2008). Advantages of multilocus sequence analysis for taxonomic studies: a case study using 10 housekeeping genes in the genus Ensifer (including former Sinorhizobium). International Journal of Systematic and Evolutionary Microbiology, 58(1), 200–214.
Martinez Condom, G. (2019). Identification and characterization of beneficial microorganisms associated with chickpea (Cicer arietinum L.) for increased productivity and drought tolerance. Bachelor’s thesis, Universitat Politècnica de Catalunya.
Martins, P. F., Carvalho, G., Gratão, P. L., & Dourado, M. N. (2011). Effects of the herbicides acetochlor and metolachlor on antioxidant enzymes in soil bacteria. Process Biochemistry, 46, 1186–1195.
Millach, L., Obiol, A., Sole, A., & Esteve, I. (2017). A novel method to analyse in vivo the physiological state and cell viability of phototrophic microorganisms by confocal laser scanning microscopy using a dual laser. Journal of Microscopy, 268(1), 53–65.
Mongkolsuk, S., Dubbs, J. M., & Vattanaviboon, P. (2005). Chemical modulation of physiological adaptation and cross-protective responses against oxidative stress in soil bacterium and phytopathogen, Xanthomonas. Journal of Industrial Microbiology and Biotechnology, 32, 687.
Niemi, R. M., Heiskanen, I., Ahtiainen, J. H., Rahkonen, A., Mäntykoski, K., Welling, L., et al. (2009). Microbial toxicity and impacts on soil enzyme activities of pesticides used in potato cultivation. Applied Soil Ecology, 41(3), 293–304.
Niewiadomska, A., & Klama, J. (2005). Pesticide side effect on the symbiotic efficiency and nitrogenase activity of Rhizobiaceae bacteria family. Polish Journal of Microbiology, 54, 43–48.
Nollet, L. M., Heinzenm, H., & Fernandez-Alba, A. R. (2017). Multi-residue methods for the analysis of pesticide residues in food 1–116.
Nour, S. M., Fernandez, M. P., Normand, P., & Cleyet-Marel, J. C. (1994). Rhizobium ciceri sp. nov., consisting of strains that nodulate chickpeas (Cicer arietinum L.). International Journal of Systematic and Evolutionary Microbiology, 44(3), 511–522.
Pandey, A., Jain, R., Sharma, A., Dhakar, K., Kaira, G. S., Rahi, P., et al. (2019). 16S rRNA gene sequencing and MALDI-TOF mass spectrometry based comparative assessment and bioprospection of psychrotolerant bacteria isolated from high altitudes under mountain ecosystem. SN Applied Sciences, 1(3), 278.
Pandey, R. P., Srivastava, A. K., Gupta, V. K., O’Donovan, A., & Ramteke, P. W. (2018). Enhanced yield of diverse varieties of chickpea (Cicer arietinum L.) by different isolates of Mesorhizobium ciceri. Environmental Sustainability, 1(4), 425–435.
Raghavendra, K. S., Gundappagol, R. C., & Santhosh, G. P. (2017). Impact of herbicide application on beneficial soil microbial community, nodulation and yield parameters of chickpea (Cicer arietinum L.). Bulletin of Environmental and Pharmaceutical Life Sciences, 6, 154–159.
Rizvi, A., Ahmed, B., Zaidi, A., & Khan, M. S. (2019). Heavy metal mediated phytotoxic impact on winter wheat: oxidative stress and microbial management of toxicity by Bacillus subtilis BM2. RSC Advances, 9(11), 6125–6142.
Rong, X., & Huang, Y. (2010). Taxonomic evaluation of the Streptomyces griseus clade using multilocus sequence analysis and DNA–DNA hybridization, with proposal to combine 29 species and three subspecies as 11 genomic species. International Journal of Systematic and Evolutionary Microbiology, 60(3), 696–703.
Rubio-Bellido, M., Morillo, E., & Villaverde, J. (2018). Assessment of soil diuron bioavailability to plants and microorganisms through non-exhaustive chemical extractions of the herbicide. Geochemistry, 312, 130–138.
Saha, J. K., Selladurai, R., Coumar, M. V., & Dotaniya, M. L. (2017). Impact of different developmental projects on soil fertility. In Soil pollution—an emerging threat to agriculture (pp. 251–269). Singapore: Springer.
Saif, S., & Khan, M. S. (2018). Assessment of toxic impact of metals on proline, antioxidant enzymes, and biological characteristics of Pseudomonas aeruginosa inoculated Cicer arietinum grown in chromium and nickel-stressed sandy clay loam soils. Environmental Monitoring and Assessment, 190(5), 290.
Sang, H., Popko, J. T., Jr., Chang, T., & Jung, G. (2016). Molecular mechanisms involved in qualitative and quantitative resistance to the dicarboximide fungicide iprodione in Sclerotinia homoeocarpa field isolates. Phytopathogen, 107, 198–207.
Sarkar, A., Ghosh, P. K., Pramanik, K., Mitra, S., & Soren, T. (2018). A halotolerant Enterobacter sp. displaying ACC deaminase activity promotes rice seedling growth under salt stress. Research in Microbiology, 169, 20–32.
Shahid, M., Ahmed, B., & Khan, M. S. (2018). Evaluation of microbiological management strategy of herbicide toxicity to greengram plants. Biocatalysis and Agricultural Biotechnology, 14, 96–108.
Shahid, M., & Khan, M. S. (2017). Assessment of glyphosate and quizalofop mediated toxicity to greengram [Vigna radiata (L.) Wilczek], stress abatement and growth promotion by herbicide tolerant Bradyrhizobium and Pseudomonas species. International Journal of Current Microbiology and Applied Sciences, 6, 3001–3016.
Shahid, M., & Khan, M. S. (2018a). Cellular destruction, phytohormones and growth modulating enzymes production by Bacillus subtilis stain BC8 impacted by fungicides. Pesticide Biochemistry and Physiology, 149, 8–19.
Shahid, M., & Khan, M. S. (2018b). Glyphosate induced toxicity to chickpea plants and stress alleviation by herbicide tolerant phosphate solubilizing Burkholderia cepacia PSBB1 carrying multifarious plant growth promoting activities. 3 Biotech, 8, 131.
Singh, R. P., Manchanda, G., Yang, Y., Singh, D., Srivastava, A. K., Dubey, R. C., et al. (2019). Deciphering the Factors for Nodulation and Symbiosis of Mesorhizobium Associated with Cicer arietinum in Northwest India. Sustainability, 11(24), 7216.
Srivastava, J., Chandra, H., Singh, N., & Kalra, S. J. (2016). Understanding the development of environmental resistance among microbes: a Review. Clean-Soil, Air, Water, 44, 901–908.
Vincent, J. M. (1970). A manual for the practical study of the root-nodule bacteria. Oxford: Blackwell Scientific Publications.
Wasi, S., Tabrez, S., & Ahmad, M. (2011). Suitability of immobilized Pseudomonas fluorescens SM1 strain for remediation of phenols, heavy metals, and pesticides from water. Water, Air, and Soil Pollution, 220(1–4), 89–99.
Wu, M., Li, G., Chen, X., & Liu, J. (2018). Rational dose of insecticide chlorantraniliprole displays a transient impact on the microbial metabolic functions and bacterial community in a silty-loam paddy soil. Science of the Total Environment, 616, 236–244.
Yao, X., Min, H., & Lv, Z. (2006). Response of superoxide dismutase, catalase, and ATPase activity in bacteria exposed to acetamiprid. Biomedicine and Environmental Sciences, 19, 309.
Zafar, M., Ahmed, N., Mustafa, G., Zahir, Z. A., & Simms, E. L. (2017). Molecular and biochemical characterization of rhizobia from chickpea (Cicer arietinum). Pakistan Journal of Agricultural Sciences, 54(2), 373–381.
Acknowledgements
The authors would like to thank National Bureau of Agriculturally Important Microorganisms (NBAIM) Mau (India), Macrogen, Seoul, Korea, for providing 16SrRNA gene sequencing analysis, University Sophisticated Instrument Facility (USIF), for providing SEM and CLSM facilities.
Funding
The author (Mohammad Shahid) would like to acknowledge the financial support received in the form of UGC Non-NET fellowship Granted by University Grants Commission, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shahid, M., Zaidi, A. & Saghir Khan, M. Modulations in growth, structure, cell viability and antioxidant enzyme of a nodule bacterium Mesorhizobium ciceri induced by pesticides. Environ Dev Sustain 23, 4103–4119 (2021). https://doi.org/10.1007/s10668-020-00758-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10668-020-00758-2