Abstract
Having been largely used in industrial and household products, perfluoroalkyl acids (PFAAs) appear in environmental and biological systems with prevalence and persistence and have raised great concern in recent years. The present study is aimed at studying concentrations and composition profiles of 16 PFAAs in surface sediments collected from 51 sampling locations in 4 main rivers of the Pearl River Delta, one of the economy-developed areas in China. The total PFAA concentrations (∑ PFAAs) were determined in a wide range of 1.89–15.1 ng g−1 dw (dry weight) with an average concentration to be 3.54 ng g−1 dw. Higher ∑ PFAAs were observed in the downstream of Dongjiang River and the Pearl River, possibly due to the discharge of industrial wastewater. Perfluoropentanoic acid (PFPeA) and perfluorooctane sulfonate (PFOS) were the dominant PFAAs, accounting for 51 to 85% of ∑ PFAAs in 27% of the samples. High PFPeA concentrations in sediments of urban river were scarcely observed in previous studies worldwide. The sources of short-chain perfluoroalkyl carboxylic acids (PFCAs) were significantly different from those of other PFAAs. Preliminary hazard assessment proved negligible for PFOS, perfluorooctanoic acid (PFOA), PFPeA, and perfluorohexanoic acid (PFHxA) concentrations in sediments from rivers of the Pearl River Delta.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Perfluoroalkyl acids (PFAAs) were subjected to extensive production and use in industrial and household products, i.e., textile coatings, firefighting foams, carpet, pesticides, stain repellents, and lubricants during the past decades (Kissa 2001) because of their special properties such as chemical and thermal stability, high surface activity (Lindstrom et al. 2011), and hydrophobic and negligible nature (Giesy and Kannan 2002). Consequently, high detection rates of PFAAs have been found in a variety of environmental matrices, i.e., surface and drinking water (Ahrens 2011; Post et al. 2013; Xiao et al. 2013; Yamashita et al. 2005), soil (Kim et al. 2014; Meng et al. 2013; Wang et al. 2013), air (Chaemfa et al. 2010; Dreyer et al. 2009; Li et al. 2011; Liu et al. 2015a), and biological tissues (Houde et al. 2006; Giesy and Kannan 2001; Pérez et al. 2013). As the degradation products of numerous precursor perfluorinated compounds, PFAAs including perfluoroalkyl carboxylic acids (PFCAs) and perfluoroalkane sulfonic acids (PFSAs) have been receiving much concerns in recent years due to their environmental persistence (Dreyer et al. 2009), bioconcentration (Higgins et al. 2007), long-distance transmission (Dreyer et al. 2009; Stock et al. 2007), and adverse environmental impact (Lau et al. 2007).
The water solubility of PFAAs decreases with the increase of the alkyl chains, so that long-chain PFAAs (C > 8) appear to be bound to sediments (Ahrens et al. 2009a, 2010). Owing to a fairly high solubility contradistinguished with other POPs (Nakata et al. 2006) and negligible vapor pressure, most short-chain PFAAs (C ≤ 8) can accumulate in surface water and transport based on a variety of hydrological parameters (Yamashita et al. 2005). Sediments are usually regarded as a sink for hydrophobic organic contaminants such as long-chain PFAAs, frequently serving on a source making them released back to waters (Prevendouros et al. 2006). PFAAs, remobilized in sediment, could be bioavailable to aquatic organism (Higgins et al. 2007). Previous studies have indicated that PFAAs tend towards bioaccumulating in living beings lying on lower trophic levels and may generate biomagnification along the food chain (Houde et al. 2006; Lescord et al. 2015; Li et al. 2008; Loi et al. 2011; Martin et al. 2004), posing potential threat to the health of people via consuming aquatic foods (Berger et al. 2009; Picó et al. 2011; Schuetze et al. 2010). PFAAs have been detected worldwide in sediments including China (Bao et al. 2009; Bao et al. 2010b; Zhao et al. 2013; Xu et al. 2013), Japan (Ahrens et al. 2009a, 2010; Senthilkumar et al. 2007), Korea (Naile et al. 2010), the USA (Lasier et al. 2011; Senthil et al. 2009), Australia (Thompson et al. 2011), and a number of EU countries (Becker et al. 2008; Beškoski et al. 2013; Campo et al. 2016; Gomez et al. 2011; Labadie and Chevreuil 2011; Picó et al. 2012). For instance, perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) have been frequently detected in environmental samples in the range of single digits to hundreds ng g−1 and recognized as emerging global pollutants (Taniyasu et al. 2007).
The factors, i.e., the movement characteristics between the liquid and solid phases (Higgins and Luthy 2006), volatilization (Dreyer et al. 2009), hydrodynamic conditions (Ahrens et al. 2009b), and carbon chain length of PFAAs (Martin et al. 2003) that influence distribution of PFAAs in sediment, have also been investigated. Sediment-associated PFAAs were mainly derived from the effluents of wastewater treatment plants (WWTPs) with low PFAA removal efficiency (Sinclair and Kannan 2006; Xiao et al. 2012; Yu et al. 2009) and the leaking of PFAA-manufacturing processes (Awad et al. 2011).
China has been undergoing rapid industrialization and is becoming one of the world’s largest and fastest growing economies. For instance, 220–240 t a−1 PFOS and PFOS-related chemicals were produced for export and domestic sale in recent years (Xie et al. 2013b). The Pearl River Delta (PRD) is known as one of high urbanization area regions in the world and one of the most rapid economic development regions in China, contributing to around 10% of China’s annual gross domestic product (GDP). The PRD has an area of approximate 56,000 km2 with Xijiang River, Dongjiang River, Beijiang River, and the Pearl River running through it. The effluents from WWTPs and industrial enterprises are the main sources of PFAAs in rivers of the PRD (Liu et al. 2015b). PFAAs have been detected in rivers of the PRD (Bao et al. 2010b; So et al. 2007; Zhao et al. 2014). However, previous studies were restricted to demonstrate the distribution of PFAA in sediment in the Pearl River flowing through Guangzhou and Foshan (Bao et al. 2010b; So et al. 2007), while the levels of PFAAs in sediments of the other rivers including Xijiang River, Dongjiang River, and Beijiang River, which are main tributaries in the PRD as well, were not monitored. This study was aimed at illustrating PFAA spatial distribution in sediments from rivers of the PRD and further assessing the potential risks of these compounds to the aquatic organisms in the rivers through the calculation of risk quotients.
Materials and methods
Chemicals and reagents
Acronyms and molecular formulas of 16 PFAAs and 8 internal standards (ISs) used in this study were described in Table S1. Methanol (SupraSolv grade) was supplied from J.T. Baker Technologies (USA). HPLC-grade methanoic acid, ammonium acetate, and ammonia hydroxide were supplied by Dikma Technologies (USA). The cartridges (6 mL, 0.15 g) used in solid phase extraction (SPE) were purchased from Waters (Milford, MA, USA).
Sample collection
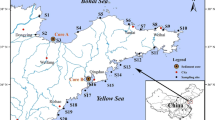
In August 2013, 51 surface sediment samples with the depth ranges of 0–5 cm were collected from rivers of the PRD (Fig. 1). Table S2 presented the geographic locations of all the sampling sites. At each sampling site, approximately 0.5 kg of surface sediment was collected and stored in polypropylene (PP) bag pre-cleaned with methanol followed by Milli-Q water with electrical resistivity being greater than 18.2 MΩ cm and frozen at −20 °C before freeze-dried. After freeze-dried at −50 °C in a stainless steel container for 72 h, the sediments were homogenized by sieving through a 100-mesh sieve. After pretreatment, all the collected samples were lastly put into PP bags until the extraction. The method of extracting sediment samples and exhaustive information about the instrumental analysis were described by Liu et al. (2015b).
Sample preparation and instrument analysis
The method of extracting sediment samples was based on the method described by Liu et al. (2015b). Briefly, 5 ng of IS mix and 10 mL of methanol were added into a 15-mL PP centrifuge tube, in which 1.0 g sample (dry weight) was previously added. Each tube was ultrasonically extracted at 60 °C and centrifuged for 10 min after shaken for 60 min. The extraction process was repeated twice and all extracts were combined in 100-mL PP centrifuge tube. The extracts were concentrated to dryness using a gentle stream of nitrogen gas, and then, 8 mL of 2% methanoic acid and 42 mL of ultra-pure water were added in each PP tube. The mixture was passed through WAX cartridge which was preconditioned with 2 mL of methanol and 2 mL of ultra-pure water. Two-milliliter of 2% methanoic acid, 2 mL of methanoic acid in methanol (v/v = 1:1), and 2 mL of methanol were used to gradiently wash each cartridge, and then, 4 mL of 9% ammonia in methanol was applied to elute PFAAs. The elutes were dried under a nitrogen stream and 1 mL of methanol was added. Each concentrated sample was filtered through a nylon membrane (pore diameter 0.22 μm; Millipore, Billerica, MA).
PFAAs were analyzed using an Agilent 1290 high-performance liquid chromatography system coupled with an API 3000 tandem triple-quadrupole mass spectrometry (HPLC-MS/MS; Agilent Technologies, CA, USA). Exhaustive information about the instrumental analysis were described by Liu et al. (2015b).
Quality assurance/quality control
To check for the possible contaminations occured in the whole experimental process, the analysis of procedural blanks were conducted every 10 samples. The instrumental background was monitored by use of performing the solvent blanks comprising cleaned Milli-Q water and methanol (1:1, v/v). The calibration curves were established by a series of gradient concentrations of external standards added by 1 ng mixed ISs with regression coefficients (r 2) for all PFAAs being from 0.9907 to 0.9997. The limit of detection (LOD) and the limit of quantification (LOQ) were determined using a signal-to-noise (S/N) ratio of 3 and 10, respectively. LODs, LOQs, and the spiked recoveries for all PFAAs were listed in Table S3. All PFAA concentrations were not recovery corrected. SPSS 19.0 (IBM Inc., USA) and Origin 8.6 (Origin Inc., USA) for Windows were used to perform statistical analyses.
Results and discussion
Levels of PFAAs in sediments of the PRD
Concentrations of ten PFAAs were higher than their LODs in surface sediments from rivers of the PRD (for details, see Table S2). An overview of PFAA concentrations in surface sediments was presented in Fig. 2. PFHpA, PFTrDA, PFTeDA, PFHxDA, PFODA, and PFDS were not detected in the collected sediment samples. The total PFAA concentrations (∑ PFAAs) ranged from 1.89 to 15.1 ng g−1 dw (dry weight), with an average value of 3.54 ng g−1 dw. Spatial distributions of ∑ PFAAs, ∑ PFCAs, and ∑ PFSAs in sediments from the PRD were shown in Fig. S1. Generally, ∑ PFAAs in the sediments increased with the direction of flowing water. For example, ∑ PFAAs in sites 40 and 36 that, respectively, were situated in the downstream of the Pearl River and Dongjiang River were approximately four times higher than the mean value of ∑ PFAAs, while lower levels of ∑ PFAAs were observed in the sediments collected from the upper reaches. The difference could be attributed to anthropogenic discharges. For instance, a large industrial park named Newton and Dongguan electronic industry located at 3 km of the upstream from site 40 had an effect on ∑ PFAAs in the sediments. In addition, high ∑ PFAAs were also observed in sites 35 and 39 situated in the river section of Guangzhou, site 18 situated in the river section of Jiangmen, and sites 46 and 47 situated in the river section of Dongguan, where the conventional industrial cities distributed in southern China. Except for the influences on ∑ PFAAs by the effluents from industries and WWTPs, the flood gates can also obstruct the sediment transport to the truck stream, which resulted in higher ∑ PFAAs in the sediments of sites 18, 39, and 46. The sediment prevented by the flood diversion gate is usually used for composting and land application, leading to a secondary PFAA contamination in soil (Busch et al. 2010). In the other sampling sites, comparable ∑ PFAAs which were measured at low levels of ∑ PFAAs were observed. Consequently, ∑ PFAAs in the sediments from downstream rivers of the PRD were higher than those of upstream rivers. Furthermore, the organic matter content of sediment was usually regarded as one of the most important factors influencing the sorption process of PFAAs to the sediments (Higgins and Luthy 2006). The contents of total organic carbon (TOC) in the upstream sediments (mean 0.91%) in this study were significantly less than those in the downstream sediments (mean 1.32%), indicating a weaker affinity for PFAAs especially for long-chain PFAAs in the upstream sediments and thus resulting in a lower concentration of PFAAs in the upstream sediments.
Among the PFAAs, PFPeA and PFOS were the two predominant contaminants with average values of 0.995 and 0.879 ng g−1 dw, respectively. PFPeA concentrations ranged from 0.126 to 11.8 ng g−1 dw (median 0.222 ng g−1 dw; coefficient of variation 219%) and accounted for 4–85% of the ∑ PFAA concentrations. High levels and frequencies (≥75%) of PFPeA were found in the sediments of sites 36, 43, 40, and 17 located in the urban rivers of Guangzhou, Dongguan, and Zhongshan, which were recognized as well-developed manufacturing cities (Liu et al. 2015b). Meanwhile, concentrations of PFOS were in the range from not detected (nd) to 2.44 ng g−1 dw (median 0.760 ng g−1 dw; coefficient of variation 48%) and occupied up to 46% of the ∑ PFAAs. It is noteworthy that PFOA was found to be the most prevalent PFAA in sediment reported in previous studies (Bao et al. 2009; Xu et al. 2013; Theobald et al. 2012; Zhao et al. 2013), as compared to lower concentrations of PFOA (mean 0.270 ng g−1 dw) in sediments from the PRD in the present study. PFBS had been generally measured in urban water and offshore oceans (Cai et al. 2012; Labadie and Chevreuil 2011; Liu et al. 2015b; Wang et al. 2011). In river sediments of the PRD, the concentrations of PFBS were below the LOD except for a few heavily contaminated sites, e.g., 46, 47, 29, 30, and 31, which were mainly distributed in Dongjiang River. The different profiles of homologous PFAAs in sediment between the PRD and other regions might be ascribed to the land utilization and the local pollution sources (Zhao et al. 2013).
For accurately understanding the possible sources of PFAAs, hierarchical cluster analysis (HCA) which was proved as a credible method for source apportionment of PFAAs in sediments (Xu et al. 2013) was employed to investigate the sources of PFAAs in sediments from rivers of the PRD. The measured concentrations of PFAAs were standardized using the following formula:
where Z ij is the standardized concentration of the ith PFAAs from the jth sampling location, C ij is the measured concentrations of the ith PFAAs from the jth sampling location, \( {C}_i^0 \) is the average concentrations of the ith PFAAs from all the sampling locations, and SD i is the standard deviation of the ith PFAAs from all the sampling locations.
Two groups of nine PFAAs were discriminated in Fig. 3. The other PFAAs including PFHpA, PFDoA PFTrDA, PFTeDA, PFHxDA, PFODA, and PFDS were not listed due to their extremely low or zero detection rates. Group A included PFPeA and PFHxA as PFPeA cluster, and PFOS, PFBS, PFHxS, PFOA, PFDA, PFNA, and PFUdA were clustered in group B as PFOS cluster. PFAAs in the identical group may be from the same source (McGregor et al. 2012; Shi et al. 2012). There were obvious different sources between short-chain PFCAs and medium/long-chain PFCAs. In group A, the high concentration of PFPeA scarcely observed in previous reports worldwide was found, which may be due to the shift of PFAA application because of the phaseout of PFOS and PFOS-related compounds since 2009. As a short-chain PFAA, PFPeA was likely a substitute for PFOS-based or PFOA-based production. In addition, the extremely uneven spatial distribution of PFPeA (coefficient of variation 219%) may illustrate the anthropogenic discharge in some areas. In group B, PFOS was the representative PFAA with coefficient of variation only being 48% (the minimum value of all PFAAs), which could explain that PFOS in most sediments of the PRD may be derived from the deposit after historical discharge. Completely as developed countries have phase out the production of PFOS, China is still a main producer and limited user of PFOS-based production. The emission of PFOS in the PRD was estimated to be 5.0–26 kg a−1, which is an intensively discharged region of China (Xie et al. 2013a). In addition, there were significantly different sources between medium/long-chain PFCAs and PFSAs (P < 0.05). The sources of PFCAs include PFCA manufacture, fluoropolymer manufacture and dispersions, aqueous firefighting foam products, consumer and industrial products, and the degradation of fluorotelomer-based products (Prevendouros et al. 2006). PFOS and other PFSAs were mainly applied to the electrochemical fluorination process (Hekster et al. 2003) and mist suppressant (Kelly and Solem 2008; Xiao et al. 2012).
Global comparison of PFOS and PFOA concentrations in sediment
A comparison of PFOS and PFOA concentrations in sediments was shown in Fig. 4 using the minimum and maximum concentrations of PFOS and PFOA, even though PFOA was not the predominant PFAA in sediments in this study. Higher concentrations of PFOS in sediments were found in a few rivers in Japan (Senthilkumar et al. 2007) and Haihe River in China (Li et al. 2011a), while lower concentrations were observed in German Roter Main River (Becker et al. 2008), Hun River (Bao et al. 2009), and Yangtze River near Shanghai in China (Bao et al. 2010b). Comparable PFOS concentrations were found in this study with other studies including the Pearl River (Bao et al. 2010b) and Dagu Drainage River (Li et al. 2011b). Furthermore, PFOS concentrations in sediments from some Japanese rivers (Senthilkumar et al. 2007) and Haihe River, China (Li et al. 2011a), located in the highly industrialized areas were about two to three times higher than those in rivers of the PRD in this study and Dagu Drainage River (Li et al. 2011b). The PFOS concentrations in Dagu Drainage River were approximately one order of magnitude higher than those in Roter River (Becker et al. 2008); Savannah River estuary, USA (Senthil et al. 2009); Hun River (Bao et al. 2009); Liao River (Yang et al. 2011); and Yangtze River near Shanghai in China (Bao et al. 2010b), suggesting the influences of geographical variations of industries on PFOS concentrations in the sediments.
Concentrations (minimum, maximum) of PFOS and PFOA in sediments collected from rivers of the Pearl River Delta in ng g−1 dw (Li et al. (2011a) (a), Bao et al. (2010b) (b), Bao et al. (2009) (c), Yang et al. (2011) (d), Zhao et al. (2013) (e), Senthil et al. (2009) (f), Becker et al. (2008) (g), Senthilkumar et al. (2007) (h), Bao et al. (2010a) (i), and Corsolini et al. (2012) (j)). Note that the concentrations of PFOS and PFOA below the limit of detection (LOD) reported in the literatures were statistically recognized as zero in the figure
Higher levels of PFOA were observed in sediments from Laizhou Bay adjacent rivers (Zhao et al. 2013) and Xi River in China (Bao et al. 2010a) with more than two orders of magnitude higher than a few investigated rivers such as Yangtze River near Shanghai and the Pearl River (Bao et al. 2010b). In addition, Ganges River in India was another PFOA heavily polluted river with the concentration of PFOA being approximately one order of magnitude higher than that in Dagu Drainage River sediments (Li et al. 2011a) and rivers of the PRD in this study. Relatively lower concentrations of PFOA were observed in Hun River (Bao et al. 2009) and Liao River in China (Yang et al. 2011), Roter River in Germany (Becker et al. 2008), and Savannah River estuary in the USA (Senthil et al. 2009).
There are differences in the distribution between different PFAAs. Generally, PFOS were detected in higher concentrations than PFOA in sediments (Fig. 2), likely due to the higher sediment/water partitioning coefficient of PFOS than that of PFOA (Ahrens 2011; Higgins and Luthy 2006; Li et al. 2011a; Liu et al. 2015b). However, a few exceptions were also reported in previous studies (Nakata et al. 2006; Yang et al. 2011). Remarkably higher concentrations of PFOA than PFOS were reported in sediments collected from the Xiaoqing River near Laizhou Bay and Xi River belonging to Daliao River system, which were all situated downstream from a polytetrafluoroethylene-manufacting plant regarded as the source of PFOA (Zhao et al. 2013; Bao et al. 2010a). In this study, higher levels of PFOS than PFOA were observed in almost all the sampling sites except sites 20 and 34, suggesting greater sediment contamination of PFOS than PFOA in the PRD. Xie et al. (2013a) reported that 93% of PFOS emission were from textile treatment and metal plating (7.4 t in 2010), ranking second in all provinces in China. On the other hand, the production and/or use of PFOA in Guangdong Province has been unknown to discharge into the environment. Further studies on source differentiation and statistics of discharge of PFOA in China should be performed for a better understanding of ecological risk of PFAAs.
Preliminary environmental risk assessment of PFAA exposure to zoobenthos
The bioaccumulation and toxicity of PFAAs in sediment-water-benthic organism system have been investigated in previous studies (Giesy et al. 2010; Higgins et al. 2007; Loi et al. 2011). PFOS and PFNA were the most bioaccumulative PFAAs for the oligochaete Lumbriculus variegatus in contaminated field sediments (Higgins et al. 2007). Trophic magnification was observed for PFOS, PFDA, PFUdDA, and PFDoDA in a subtropical food web (Loi et al. 2011). Lin et al. (2010) illustrated that PFOS with concentration up to 6.05 μg L−1 in Nanmen River would cause a considerable risk to local aquatic ecosystem. While there are many researches about the toxicities of PFOS and PFOA for the past few years (Ding and Peijnenburg 2013; Liu et al. 2013, 2014), the predicted no-effect concentration (PNEC) and environmental quality standard (EQS) in river sediment have been scarcely ever reported in the previous studies. Given this, sediment PNEC and EQS can be calculated from the partitioning coefficients due to unavailability of the toxicity data (Lepper 2002). The PNECs in freshwater for PFOA (1250 μg L−1) and PFOS (25 μg L−1) were obtained by Colombo et al. (2008) and Brooke et al. (2004), respectively. The EQS for PFOA (0.1 μg L−1) and related short-chain PFAAs including PFPeA (3 μg L−1) and PFHxA (1 μg L−1) were derived by Valsecchi et al. (2016). The organic carbon normalized partitioning coefficients (K oc) for PFOA (log K oc = 2.11 L kgoc −1) and PFOS (log K oc = 2.68 L kgoc −1) were derived by Higgins and Luthy (2006) in an experimental simulation research. Liu et al. (2015a) derived the log K oc (L kgoc −1) 4.67 for PFOA, 5.07 for PFOS, 4.94 for PFPeA, and 4.80 for PFHxA in rivers of the PRD with the same sampling locations as this study. In addition, the non-TOC-normalized PNECs were also calculated using partitioning coefficient (K d ; 0.1973 L kg−1 for PFOS and 0.0690 L kg−1 for PFOA) between sediment and dissolved phase cited by Ferrey and Wilson (2009). The relatively low K d values calculated in this study will cause the overestimation of PNECs. The TOC-normalized PNECs and non-TOC-normalized PNECs based on the above coefficients were listed in Table S5. The sediment EQSs for PFOA, PFPeA, and PFHxA based on the partitioning coefficients derived by Liu et al. (2015b) were listed in Table S6.
In this study, the risk quotients (RQs) were calculated based on the ratios of detected concentrations and PNECs/EQSs to assess the environmental risk of PFAAs in sediments from rivers of the PRD. Table S4 listed the TOC-normalized concentrations of PFOS, PFOA, PFPeA, and PFHxA. The potential risk of PFAA in sediments was higher in the estuaries than the terrestrial rivers of the PRD. For instance, the highest TOC-normalized concentration of PFOS was 2.13 ng·(g TOC%)−1 dw at site 23 near the estuary of Xijiang River, followed by 1.83 ng·(g TOC%)−1 dw at sites 39 near the Pearl River Estuary and 46 in Dongguan section of Dongjiang River. The highest TOC-normalized concentration of PFOA was detected to be 0.94 ng·(g TOC%)−1 dw at site 34 near the Pearl River Estuary. The highest TOC-normalized concentrations of PFPeA and PFHxA were, respectively, 9.42 and 1.64 ng·(g TOC%)−1 dw at site 36 near the Pearl River Estuary. All the RQs of PFOS and PFOA were less than 1 (Table S5), exhibiting a low risk to zoobenthos in rivers of the PRD, even though the results were obtained based on the overestimated non-TOC-normalized PNECs. When measured by EQSs, all the RQs of PFOA, PFPeA, and PFHxA were also less than 1 (Table S6), which reflected that the measured concentrations of PFAAs did not exceed EQSs.
Conclusions
The PRD that is vital for both industry and agriculture is a well-developed region in China. Relatively high PFAA concentrations in surface water were observed in previous studies; however, little was known about their levels, distribution, and risk in sediments. As a part of an ongoing study to determine the current level of PFAA concentrations, as well as potential environmental risk to zoobenthos, this study determined 16 PFAA concentrations in sediments from rivers of the PRD. Higher PFAA concentrations were observed in the Pearl River and the downstream of Dongjiang River, where PFPeA accounted for a very large proportion of ∑ PFAAs, which might be attributed to the local urban and industrial effluents. PFPeA and PFOS were the predominant PFAAs, and high PFPeA concentration in sediment of urban river was scarcely found in previous reports worldwide. In this study, PFOS and PFOA concentrations had negligible environmental risk to zoobenthos. Concentrations of PFOA, PFPeA, and PFHxA did not exceed the derived sediment environmental quality standard. Further study about the no-observed-adverse-effect levels or the median lethal concentrations of PFAAs in sediment should be conducted to make the environmental risk assessment of PFAAs more accurate in the future researches.
References
Ahrens, L. (2011). Polyfluoroalkyl compounds in the aquatic environment: a review of their occurrence and fate. J. Environ. Monitor., 13(1), 20–31.
Ahrens, L., Yamashita, N., Yeung, L. W. Y., Taniyasu, S., Horii, Y., Lam, P. K. S., & Ebinghaus, R. (2009a). Partitioning behavior of per- and polyfluoroalkyl compounds between pore water and sediment in two sediment cores from Tokyo Bay. Japan. Environ. Sci. Technol., 43(18), 6969–6975.
Ahrens, L., Barber, J. L., Xie, Z., & Ebinghaus, R. (2009b). Longitudinal and latitudinal distribution of perfluoroalkyl compounds in the surface water of the Atlantic Ocean. Environmental Science & Technology, 43(9), 3122–3127.
Ahrens, L., Taniyasu, S., Yeung, L. W. Y., Yamashita, N., Lam, P. K. S., & Ebinghaus, R. (2010). Distribution of polyfluoroalkyl compounds in water, suspended particulate matter and sediment from Tokyo Bay, Japan. Chemosphere, 79(3), 266–272.
Awad, E., Zhang, X., Bhavsar, S. P., Petro, S., Crozier, P. W., Reiner, E. J., Fletcher, R., Tittlemier, S. A., & Braekevelt, E. (2011). Long-term environmental fate of perfluorinated compounds after accidental release at Toronto Airport. Environmental Science & Technology, 45(19), 8081–8089.
Bao, J., Jin, Y., Liu, W., Ran, X., & Zhang, Z. (2009). Perfluorinated compounds in sediments from the Daliao River system of northeast China. Chemosphere, 77(5), 652–657.
Bao, J., Liu, W., Liu, L., Jin, Y., Dai, J., Ran, X., Zhang, Z., & Tsuda, S. (2010a). Perfluorinated compounds in the environment and the blood of residents living near fluorochemical plants in Fuxin. China. Environ. Sci. Technol., 45(19), 8075–8080.
Bao, J., Liu, W., Liu, L., Jin, Y., Ran, X., & Zhang, Z. (2010b). Perfluorinated compounds in urban river sediments from Guangzhou and Shanghai of China. Chemosphere, 80(2), 123–130.
Becker, A. M., Gerstmann, S., & Frank, H. (2008). Perfluorooctanoic acid and perfluorooctane sulfonate in the sediment of the Roter Main River, Bayreuth, Germany. Environmental Pollution, 156(3), 818–820.
Berger, U., Glynn, A., Holmstrom, K. E., Berglund, M., Ankarberg, E. H., & Tornkvist, A. (2009). Fish consumption as a source of human exposure to perfluorinated alkyl substances in Sweden—analysis of edible fish from Lake Vattern and the Baltic Sea. Chemosphere, 76(6), 799–804.
Beškoski, V. P., Takemine, S., Nakano, T., Beškoski, L. S., Gojgić-Cvijović, C., Llić, M., Miletić, S., & Vrvić, M. M. (2013). Perfluorinated compounds in sediment samples from the wastewater canal of Pančevo (Serbia) industrial area. Chemosphere, 91(10), 1408–1415.
Brooke, D., Footitt, A., & Nwaogu, T. A. (2004). Environmental risk evaluation report: perfluorooctane sulphonate (PFOS).
Busch, J., Ahrens, L., Sturm, R., & Ebinghaus, R. (2010). Polyfluoroalkyl compounds in landfill leachates. Environmental Pollution, 158(5), 1467–1471.
Cai, M., Zhao, Z., Yang, H., Yin, Z., Hong, Q., Sturm, R., Ebinghaus, R., Ahrens, L., Cai, M., He, J., & Xie, Z. (2012). Spatial distribution of per- and polyfluoroalkyl compounds in coastal waters from the East to South China Sea. Environmental Pollution, 161(1), 162–169.
Campo, J., Lorenzo, M., Pérez, F., Picó, Y., Farré, M., & Barceló, D. (2016). Analysis of the presence of perfluoroalkyl substances in water, sediment and biota of the Jucar River (E Spain). Sources, partitioning and relationships with water physical characteristics. Environmental Research, 147, 503–512.
Chaemfa, C., Barber, J. L., Huber, S., Breivik, K., & Jones, K. C. (2010). Screening for PFOS and PFOA in European air using passive samplers. Journal of Environmental Monitoring, 12(5), 1100–1109.
Colombo, I., Wolf, W. D., Thompson, R. S., Farrar, D. G., Hoke, R. A., & L’Haridon, J. (2008). Acute and chronic aquatic toxicity of ammonium perfluorooctanoate (APFO) to freshwater organisms. Ecotoxicology and Environmental Safety, 71(3), 749–756.
Corsolini, S., Sarkar, S. K., Guerranti, C., Bhattacharya, B. D., Rakshit, D., Jonathan, M. P., & Godhantaraman, N. (2012). Perfluorinated compounds in surficial sediments of the Ganges River and adjacent Sundarban mangrove wetland. India. Mar. Pollut. Bull., 64(12), 2829–2833.
Ding, G., & Peijnenburg, W. J. G. M. (2013). Physicochemical properties and aquatic toxicity of poly- and perfluorinated compounds. Crit. Rev. Env. Sci. Tec., 43(6), 598–678.
Dreyer, A., Weinberg, I., Temme, C., & Ebinghaus, R. (2009). Polyfluorinated compounds in the atmosphere of the Atlantic and Southern Oceans: evidence for a global distribution. Environmental Science & Technology, 43(17), 6507–6514.
Ferrey ML, Wilson JT, 2009. Extent of sorption and biodegradability of perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) in aquifer sediment (Maryland).
Giesy, J. P., & Kannan, K. (2001). Global distribution of perfluorooctane sulfonate in wildlife. Environmental Science & Technology, 35(7), 1339–1942.
Giesy, J. P., & Kannan, K. (2002). Peer reviewed: perfluorochemical surfactants in the environment. Environmental Science & Technology, 36(7), 146A–152A.
Giesy, J. P., Naile, J. E., Khim, J. S., Jones, P. D., & Newsted, J. L. (2010). Aquatic toxicology of perfluorinated chemicals. In: Whitacre DM, editor. Reviews of Environmental Contamination and Toxicology, 202, 1–52.
Gomez, C., Vicente, J., Echavarri-Erasun, B., Porte, C., & Lacorte, S. (2011). Occurrence of perfluorinated compounds in water, sediment and mussels from the Cantabrian Sea (North Spain). Marine Pollution Bulletin, 62(5), 948–955.
Hekster, F. M., Laane, R. W. P. M., & de Voogt, P. (2003). Environmental and toxicity effects of perfluoroalkylated substances. Rev. Environ. Contam. T., 179(2), 99–121.
Higgins, C. P., & Luthy, R. G. (2006). Sorption of perfluorinated surfactants on sediments. Environmental Science & Technology, 40(23), 7251–7256.
Higgins, C. P., Mcleod, P. A., Macmanus-Spencer, L. A., & Luthy, R. G. (2007). Bioaccumulation of perfluorochemicals in sediments by the aquatic oligochaete Lumbriculus variegatus. Environmental Science & Technology, 41(13), 4600–4606.
Houde, M., Bujas, T. A. D., Small, J., Wells, R. S., Fair, P. A., Bossart, G. D., Solomon, K. R., & Muir, D. C. G. (2006a). Biomagnification of perfluoroalkyl compounds in the bottlenose dolphin (Tursiops truncatus) food web. Environmental Science & Technology, 40(13), 4138–4144.
Houde, M., Martin, J. W., Letcher, R. J., Solomon, K. R., & Muir, D. C. (2006b). Biological monitoring of polyfluoroalkyl substances: a review. Environmental Science & Technology, 40(11), 3463–3473.
Kelly, J., & Solem, L. (2008). Identification of a major source of perfluorooctane sulfonate (PFOS) at a wastewater treatment plant in Brainerd. Minnesota. Environ. Epidemiol., 19, 303–304.
Kim, E. J., Park, Y. M., Park, J. E., & Kim, J. G. (2014). Distributions of new Stockholm convention POPs in soils across South Korea. The Science of the Total Environment, 476-477, 327–335.
Kissa E, 2001. Fluorinated surfactants and repellents. In: Dekker M, editor. New York: Marcel Dekker.
Labadie, P., & Chevreuil, M. (2011). Partitioning behaviour of perfluorinated alkyl contaminants between water, sediment and fish in the Orge River (nearby Paris, France). Environmental Pollution, 159(2), 391–397.
Lasier, P. J., Washington, J. W., Hassan, S. M., & Jenkins, T. M. (2011). Perfluorinated chemicals in surface waters and sediments from northwest Georgia, USA, and their bioaccumulation in Lumbriculus variegatus. Environmental Toxicology and Chemistry, 30(10), 2194–2201.
Lau, C., Anitole, K., Hodes, C., Lai, D., Pfahles-Hutchens, A., & Seed, J. (2007). Perfluoroalkyl acids: a review of monitoring and toxicological findings. Toxicological Sciences, 99(2), 366–394.
Lepper P, 2002. Identification of quality standards for priority substances in the field of water policy. In: Ecology F-IMBaA, editor. p. 1–124.
Lescord, G. L., Kidd, K. A., De Silva, A. O., Williamson, M., Spencer, C., Wang, X., & Muir, D. C. G. (2015). Perfluorinated and polyfluorinated compounds in lake food webs from the Canadian High Arctic. Environmental Science & Technology, 49(5), 2694–2702.
Li, F., Sun, H., Hao, Z., He, N., Zhao, L., Zhang, T., & Sun, T. (2011a). Perfluorinated compounds in Haihe River and Dagu Drainage Canal in Tianjin, China. Chemosphere, 84(2), 265–271.
Li, J., Vento, S. D., Schuster, J., Zhang, G., Chakraborty, P., Kobara, Y., & Jones, K. C. (2011b). Perfluorinated compounds in the Asian atmosphere. Environmental Science & Technology, 45(17), 7241–7248.
Li, X., Yeung, L. W. Y., Xu, M., Taniyasu, S., Lam, P. K. S., Yamashita, N., & Dai, J. (2008). Perfluorooctane sulfonate (PFOS) and other fluorochemicals in fish blood collected near the outfall of wastewater treatment plant (WWTP) in Beijing. Environmental Pollution, 156(3), 1298–1303.
Lin, A. Y. C., Panchangam, S. C., & Ciou, P. S. (2010). High levels of perfluorochemicals in Taiwan’s wastewater treatment plants and downstream rivers pose great risk to local aquatic ecosystems. Chemosphere, 80(10), 1167–1174.
Lindstrom, A. B., Strynar, M. J., & Libelo, E. L. (2011). Polyfluorinated compounds: past, present, and future. Environmental Science & Technology, 45(19), 7954–7961.
Liu, C., Chang, V. W. C., & Gin, K. Y. H. (2013). Environmental toxicity of PFCs: an enhanced integrated biomarker assessment and structure–activity analysis. Environmental Toxicology and Chemistry, 32(10), 2226–2233.
Liu, C., Chang, V. W. C., & Gin, K. Y. H. (2014). Oxidative toxicity of perfluorinated chemicals in green mussel and bioaccumulation factor dependent quantitative structure-activity relationship. Environmental Toxicology and Chemistry, 33(10), 2323–2332.
Liu, B., Zhang, H., Yao, D., Li, J., Xie, L., Wang, X., Wang, Y., Liu, G., & Yang, B. (2015a). Perfluorinated compounds (PFCs) in the atmosphere of Shenzhen, China: spatial distribution, sources and health risk assessment. Chemosphere, 138, 511–518.
Liu, B., Zhang, H., Xie, L., Li, J., Wang, X., Zhao, L., Wang, Y., & Yang, B. (2015b). Spatial distribution and partition of perfluoroalkyl acids (PFAAs) in rivers of the Pearl River Delta, southern China. The Science of the Total Environment, 524-525, 1–7.
Loi, E. I. H., Yeung, L. W. Y., Taniyasu, S., Lam, P. K. S., Kannan, K., & Yamashita, N. (2011). Trophic magnification of poly- and perfluorinated compounds in asubtropical food web. Environmental Science & Technology, 45(13), 5506–5513.
Martin, J. W., Mabury, S. A., Solomon, K. R., & Muir, D. C. G. (2003). Bioconcentration and tissue distribution of perfluorinated acids in rainbow trout (Oncorhynchus mykiss). Environmental Toxicology and Chemistry, 22(1), 196–204.
Martin, J. W., Whittle, D. M., Muir, D. C. G., & Mabury, S. A. (2004). Perfluoroalkyl contaminants in a food web from Lake Ontario. Environmental Science & Technology, 38(20), 5379–5385.
McGregor, L. A., Gauchotte-Lindsay, C., Daéid, N. N., Thomas, R., & Kalin, R. M. (2012). Multivariate statistical methods for the environmental forensic classification of coal tars from former manufactured gas plants. Environmental Science & Technology, 46(7), 3744–3752.
Meng, J., Wang, T., Wang, P., Giesy, J. P., & Lu, Y. (2013). Perfluorinated compounds and organochlorine pesticides in soils around Huaihe River: a heavily contaminated watershed in central China. Environmental Science and Pollution Research, 20(6), 3965–3974.
Naile, J. E., Khim, J. S., Wang, T., Chen, C., Luo, W., Kwon, B.-O., Park, J., Koh, C.-H., Jones, P. D., Lu, Y., & Giesy, J. P. (2010). Perfluorinated compounds in water, sediment, soil and biota from estuarine and coastal areas of Korea. Environmental Pollution, 158(5), 1237–1244.
Nakata, H., Kannan, K., Nasu, T., Cho, H., Sinclair, E., & Takemura, A. (2006). Perfluorinated contaminants in sediments and aquatic organisms collected from shallow water and tidal flat areas of the Ariake Sea, Japan: environmental fate of perfluorooctane sulfonate in aquatic ecosystems. Environmental Science & Technology, 40(40), 4916–4921.
Pérez, F., Nadal, M., Navarro-Ortega, A., Fàbrega, F., Domingo, J. L., Barceló, D., & Farŕe, M. (2013). Accumulation of perfluoroalkyl substances in human tissues. Environment International, 59(3), 354–362.
Picó, Y., Farré, M., Llorca, M., & Barceló, D. (2011). Perfluorinated compounds in food: a global perspective. Critical Reviews in Food Science and Nutrition, 51(7), 605–625.
Picó, Y., Blasco, C., Farré, M., & Barceló, D. (2012). Occurrence of perfluorinated compounds in water and sediment of L’Albufera Natural Park (València, Spain). Environmental Science and Pollution Research, 19(4), 946–957.
Post, G. B., Louis, J. B., Lee, L. R., & Procopio, N. A. (2013). Occurrence of perfluorinated compounds in raw water from New Jersey public drinking water systems. Environmental Science & Technology, 47(23), 13266–13275.
Prevendouros, K., Cousins, I. T., Buck, R. C., & Korzeniowski, S. H. (2006). Sources, fate and transport of perfluorocarboxylates. Environmental Science & Technology, 40(1), 32–44.
Schuetze, A., Heberer, T., Effkemann, S., & Juergensen, S. (2010). Occurrence and assessment of perfluorinated chemicals in wild fish from northern Germany. Chemosphere, 78(6), 647–652.
Senthil, K. K., Zushi, Y., Masunaga, S., Gilligan, M., Pride, C., & Sajwan, K. S. (2009). Perfluorinated organic contaminants in sediment and aquatic wildlife, including sharks, from Georgia. USA. Mar. Pollut. Bull., 58(4), 621–629.
Senthilkumar, K., Ohi, E., Sajwan, K., Takasuga, T., & Kannan, K. (2007). Perfluorinated compounds in river water, river sediment, market fish, and wildlife samples from Japan. Bulletin of Environmental Contamination and Toxicology, 79(4), 427–431.
Shi, G., Tian, Y., Guo, C., Feng, Y., Xu, J., & Zhang, Y. (2012). Sediment–pore water partition of PAH source contributions to the Yellow River using two receptor models. Journal of Soils and Sediments, 12(7), 1154–1163.
Sinclair, E., & Kannan, K. (2006). Mass loading and fate of perfluoroalkyl surfactants in wastewater treatment plants. Environmental Science & Technology, 40(5), 1408–1414.
So, M. K., Miyake, Y., Yeung, W. Y., Ho, Y. M., Taniyasu, S., Rostkowski, P., Yamashita, N., Zhou, B. S., Shi, X. J., Wang, J. X., Giesy, J. P., Yu, H., & Lam, P. K. S. (2007). Perfluorinated compounds in the Pearl River and Yangtze River of China. Chemosphere, 68(11), 2085–2095.
Stock, N. L., Furdui, V. I., Muir, D. C. G., & Mabury, S. A. (2007). Perfluoroalkyl contaminants in the Canadian arctic: evidence of atmospheric transport and local contamination. Environmental Science & Technology, 41(10), 3529–3536.
Taniyasu, S., Yamashita, N., Petrick, G., Gamo, T., Lam, P. K. S., & Kannan, K. (2007). The long range transportation of PFOS and PFOA by global scale water current. Organohalogen Compounds, 69, 150–152.
Theobald, N., Caliebe, C., Gerwinski, W., Hühnerfuss, H., & Lepom, P. (2012). Occurrence of perfluorinated organic acids in the North and Baltic Seas. Part 2: distribution in sediments. Environmental Science and Pollution Research, 19(2), 313–324.
Thompson, J., Roach, A., Eaglesham, G., Bartkow, M. E., Edge, K., & Mueller, J. F. (2011). Perfluorinated alkyl acids in water, sediment and wildlife from Sydney harbour and surroundings. Marine Pollution Bulletin, 62(12), 2869–2875.
Valsecchi, S., Conti, D., Crebelli, R., Polesello, S., Rusconi, M., Mazzoni, M., Preziosi, E., Carere, M., Lucentini, L., Ferretti, E., Balzamo, S., Simeone, M. G., & Aste, F. (2016). Deriving environmental quality standards for perfluorooctanoic acid (PFOA) and related short chain perfluorinated alkyl acids. Journal of Hazardous Materials, 323, 84–98.
Wang, T., Lu, Y., Chen, C., Naile, J. E., Khim, J. S., Park, J., Luo, W., Jiao, W., Hu, W., & Giesy, J. P. (2011). Perfluorinated compounds in estuarine and coastal areas of north Bohai Sea. China. Mar. Pollut. Bull., 62(8), 1905–1914.
Wang, P., Wang, T., Giesy, J. P., & Lu, Y. (2013). Perfluorinated compounds in soils from Liaodong Bay with concentrated fluorine industry parks in China. Chemosphere, 91(6), 751–757.
Xiao, F., Halbach, T. R., Simcik, M. F., & Gulliver, J. S. (2012). Input characterization of perfluoroalkyl substances in wastewater treatment plants: source discrimination by exploratory data analysis. Water Research, 46(9), 3101–3109.
Xiao, F., Simcik, M. F., & Gulliver, J. S. (2013). Mechanisms for removal of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) from drinking water by conventional and enhanced coagulation. Water Research, 47(1), 49–56.
Xie, S., Lu, Y., Wang, T., Liu, S., Jones, K., & Sweetman, A. (2013a). Estimation of PFOS emission from domestic sources in the eastern coastal region of China. Environment International, 59, 336–343.
Xie, S., Wang, T., Liu, S., Jones, K. C., Sweetman, A. J., & Lu, Y. (2013b). Industrial source identification and emission estimation of perfluorooctane sulfonate in China. Environment International, 52, 1–8.
Xu, J., Tian, Y., Zhang, Y., Guo, S., Shi, G., Zhang, C., & Feng, Y. (2013). Source apportionment of perfluorinated compounds (PFCs) in sediments: using three multivariate factor analysis receptor models. Journal of Hazardous Materials, 260(18), 483–488.
Yamashita, N., Kannan, K., Taniyasu, S., Horii, Y., Petrick, G., & Gamo, T. (2005). A global survey of perfluorinated acids in oceans. Marine Pollution Bulletin, 51(8–12), 658–668.
Yang, L., Zhu, L., & Liu, Z. (2011). Occurrence and partition of perfluorinated compounds in water and sediment from Liao River and Taihu Lake, China. Chemosphere, 83(6), 806–814.
Yu, J., Hu, J., Tanaka, S., & Fujii, S. (2009). Perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) in sewage treatment plants. Water Research, 43(9), 2399–2408.
Zhao, Z., Tang, J., Xie, Z., Chen, Y., Pan, X., Zhong, G., Sturm, R., Zhang, G., & Ebinghaus, R. (2013). Perfluoroalkyl acids (PFAAs) in riverine and coastal sediments of Laizhou Bay. North China. Sci. Total Environ., 447(1), 415–423.
Zhao, Y., Wan, H., Wong, M., & Wong, C. K. C. (2014). Partitioning behavior of perfluorinated compounds between sediment and biota in the Pearl River Delta of South China. Marine Pollution Bulletin, 83(1), 148–154.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Nos. 11275130 and 21407108); the Science and Technology Research Project of the Education Department of Jilin Province, China (No. 2014518); and the Natural Science Foundation of Changchun Normal University (No. 2015003).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 359 kb)
Rights and permissions
About this article
Cite this article
Liu, B., Zhang, H., Li, J. et al. Perfluoroalkyl acids (PFAAs) in sediments from rivers of the Pearl River Delta, southern China. Environ Monit Assess 189, 213 (2017). https://doi.org/10.1007/s10661-017-5921-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-5921-7