Abstract
For nearly a decade, the majority of the world’s population has been living in cities, including a considerable percentage of people suffering from pollen allergy. The increasing concentration of people in cities results in larger populations being exposed to allergenic pollen at the same time. There is almost no information about spatial distribution of pollen within cities as well as a lack of information about the possible impact to human health. To obtain this increasing need for pollen exposure studies on an intra-urban scale, a novelty screening network of 14 weekly changed pollen traps was established within a large metropolitan area—Berlin, Germany. Gravimetric pollen traps were placed at a uniform street-level height from March until October 2014. Three important allergenic pollen types for Central Europe—birch (Betula), grasses (Poaceae), and mugwort (Artemisia)—were monitored. Remarkable spatial and temporal variations of pollen sedimentation within the city and the influences by urban local sources are shown. The observed differences between the trap with the overall highest and the trap with the overall lowest amount of pollen sedimentation were in the case of birch pollen 245%, grass pollen 306%, and mugwort pollen 1962%. Differences of this magnitude can probably lead to different health impacts on allergy sufferers in one city. Therefore, pollen should be monitored preferably in two or more appropriate locations within large cities and as a part of natural air quality regulations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Airborne pollen is an important aeroallergen with remarkable impacts to human health and the quality of life. Worldwide, approximately 10 to 30% of adults and 40% of children suffer from allergic rhinitis, commonly caused by a reaction to pollen (Pawankar et al. 2013). An annual cost of up to 151 billion € within European Union countries is caused by ongoing insufficient treatment of allergy and a lack of understanding of environmental risk factors and causes (Zuberbier et al. 2014).
In 2014, 54% of the world’s population was already residing in urban areas (UN 2015). As a consequence, larger parts of the population can be exposed to a specific amount of pollen at the same moment. Moreover, urban areas increase the vulnerability of pollen allergy sufferers toward environmental conditions typically of cities.
Elevated temperatures together with increased CO2 levels affect pollen production of plants in cities and subsequently the amount of pollen in the air (Ziska et al. 2003; Ziello et al. 2012). Increased levels of ozone may cause a higher content of allergens in pollen (Eckl-Dorna et al. 2010). Other pollutants associated specifically with a town environment, such as particle matter (PM), diesel exhaust particles (DEPs), nitrogen dioxide (NO2), or sulfur dioxide (SO2) can lead to a stronger allergic response (Steerenberg et al. 2003; Feo Brito et al. 2007; Cakmak et al. 2012; Sousa et al. 2012; D’Amato et al. 2014).
Nevertheless, in many cities worldwide, there have already been existing strategies to reduce air pollution and prevent potentially negative effects of climate changes such as planting urban vegetation—as for example, in Los Angeles, USA (McPherson et al. 2011); in Berlin, Germany (Senate Department for Urban Development and the Environment, SDUDE, 2013); or in Strasbourg, France (Selmi et al. 2016). Urban greens have also important and considerable cultural and recreational value. But concurrently, the planting of non-native and/or allergenic plant species and an inappropriate management of urban vegetation represents another allergic threatening condition of urban environments (Bergmann et al. 2012; Dadvand et al. 2014; Cariñanos et al. 2016).
Sufficient representative data is available for many chemical and gaseous pollutants as well as for meteorological conditions, regularly collected at several sites within one city, representing the complexity of urban environments. However, little attention is paid to the complexity of pollen distribution in cities. To provide basic information about daily changes of airborne allergenic pollen, there are pollen observation stations running in many countries worldwide. Nevertheless, large cities are usually equipped with only one pollen trap (often a 7-day volumetric spore trap; Hirst 1952), placed at roof-top level (10–20 m above ground level), representative for the whole area. There are a few studies which compare results of multiple pollen traps per city—mostly of two, three, or four traps (Fornaciari et al. 1996; Arroba et al. 2000; Barnes et al. 2001; Cariñanos et al. 2002; Alcázar et al. 2004; Katelaris et al. 2004; Puc and Puc 2004; Gonzalo-Garijo et al. 2006; Celenk et al. 2010; Rodríguez-Rajo et al. 2010; Nowak et al. 2012; Skjøth et al. 2013; Velesco et al. 2013; Fernández-Rodríguez et al. 2014; Mücke et al. 2014; Simoleit et al. 2017). These aforementioned studies ran for different periods of time (from hours or days to continuously measuring for five consecutive years), with different numbers of observed pollen types (from one pollen type up to the full pollen spectrum). Taken together, the results of all these studies indicate similarities in pollen spectrum and the seasonal courses throughout the city but inconsistencies in spatial distribution of pollen within cities, which are often connected with the proximity to pollen sources. In the vast majority of these studies, the inconsistency of sampling heights restricted the comparability between the different sampling sites.
To our knowledge, there are eight studies with five or more pollen traps per city. In Yowa City (USA), Shapiro and Rooks (1951) created a monitoring network of 11 gravimetric pollen traps at a height of 1.8 m placed above ground level and changed daily, which were running when ragweed pollen season in 1950 (August–September) was established. In New York (USA), Raynor et al. (1975) established transects with eight rotoslide samplers, placed approximately at breathing level, across three localities during tree and ragweed pollen season in 1971 and in 1972. In North London (UK), Emberlin and Noris Hill (1991) observed the spatial distribution of birch (Betula), grasses (Poaceae), and sycamore (Platanus) pollen for two consecutive years by 12 weekly changed gravimetric pollen traps placed at rooftop level in 1987 and 14 traps in 1988. In Tokyo (Japan), Ishibashi et al. (2008) concentrated on pollen of Japanese cedar (Cryptomeria japonica) and Japanese cypress (Chamaecyparis obtusa) trapped on vaseline-coated glass slides at 32 sampling sites for 24 h in 2005 and at 78 sampling sites for 24 h in 2006. In Humpolec (Czech Republic), Obstová (2012) monitored the full pollen spectrum via a network of six weekly changed gravimetric traps placed at a height of between 2 and 6 m above ground level within the town and in a nearby village during the pollen season in 2011. In Detroit (USA), ragweed pollen sedimentation of 6 days during August 2016 was monitored by a network of 34 gravimetric pollen samplers placed at uniform street level height (Katz and Carey 2014). In the Helsinki metropolitan area (Finland), Hjort et al. (2016) observed grass pollen by a transect of 16 volumetric traps placed at the height of 1.5 m above ground level during a three and half week period in 2016. In New York (USA), Weinberger (2016) established tree pollen monitoring in 2013 via a network of 45 yearly changed gravimetric pollen traps placed at a uniform height of approximately 2.4 m above ground level. In all the abovementioned studies, there were found to be differences in spatial distribution of pollen types often linked with influences of local vegetation. The main limitations of these studies were (1) using only a few pollen traps per a city, (2) pollen sampling only within a part of a city, (3) non-uniform heights of pollen traps, and (4) long interval of time between changing traps and/or any combination of these limitations.
The main aim of our study was to widen the knowledge about the distribution of allergenic pollen within a large metropolitan area. The novelty of our screening network is (1) the usage of 14 pollen traps (2) within the complete city of Berlin (3) all placed at a uniform street-level height, relevant to human exposure, (4) with pollen data collected on a weekly basis. We assume that our network was the biggest city network of pollen traps worldwide which was characterized by these features.
Materials and methods
Study area and its characteristics
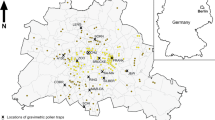
Berlin is situated in the northeast of Germany, in the North German Plain with a mainly flat topography (Fig. 1) and a maritime temperate climate (Cfb) according to the Köppen-Geiger climate classification (Kottek et al. 2006). In 2014, the annual precipitation was 477 mm with the highest amounts occurring in April and the mean annual temperature was 11.0 °C (Amt für Statistik Berlin-Brandenburg 2015). Berlin is the most populated city in Germany, with 3.5 million inhabitants and is the largest in area (892 km2). Approximately 40% of the surface area consists of open spaces such as bodies of water, forests, parks, and agricultural areas (Amt für Statistik Berlin-Brandenburg 2016).
Map of Berlin with locations of gravimetric pollen traps and the geographical center (N 52° 30.092 E 13° 24.137) of the city of Berlin. Colors: white—built-up areas; light green—meadows, fields and gardens; dark green—forests and tree-covered urban greens; blue—bodies of water. TE Tegel, TH Templehof, SF Schönefeld
Gravimetric trap
Self-constructed gravity samplers, inspired by the Durham trap (Durham 1946), were used in this study. The trap consists of two disks placed above each other at a distance of 13.5 cm. The lower disk is flat, 18 cm in diameter, and is made of stainless steel. The upper disk is flat with a downward sloping rim, is 22 cm in diameter, and is made of white plastic material. The slide holder is situated 4 cm above the lower disk. The microscopic slide measuring 76 mm × 26 mm was covered with petroleum jelly and placed on the holder. Due to low sampling efficiency by Durham samplers, we worked with a longer monitoring period of a week rather than a day.
The network of gravimetric traps
Fourteen gravimetric pollen traps were placed within the city (Fig. 1) by taking into account the following criteria: (1) close to the ground at a uniform height where people breathe, (2) with respect to various urban environments ranging from the city center to the outskirts, (3) the time management for changing the slides, (4) on stands protected from vandalism as well as (5) at sites where an easy and continuous access to locations could be ensured, and (6) in those areas that are highly frequented by people during the day. The traps were situated on exposed rooftops of small houses or containers. A brief description of each station including coordinates, predominant land use, and possible sources of the relevant pollen types around each trap are given in Table 1.
Analyzed pollen types
In Germany, 14.8% of adults (Langen et al. 2013) and 10.7% of minors under 18 years old (Schmitz et al. 2012) suffer from physician-diagnosed hay fever. As in the case of Berlin, 15 to 18% of inhabitants suffer from pollen allergy which equals an approximate number of 500,000 to 615,000 people (Bergmann et al. 2012).
Pollen season, as for the whole of Central Europe, is divided here into three main temporal periods: the spring period (February–May) with dominance of tree pollen in the air, the summer (May–August) period with predominant grass pollen, and the late summer/autumn (August–September) period with dominant pollen of weeds.
For our study, three important allergenic pollen types, each representing one main pollen period were chosen—birch (Betula) pollen as the most important allergenic tree genus and the main cause of spring pollen allergies in Central Europe (D’Amato et al. 2007); grass (Poaceae) pollen as a representative of summer pollen season and the most common reason of seasonal inhalant allergies worldwide (Kleine-Tebbe and Davies 2014); and mugwort (Artemisia) pollen as an important cause of pollinosis during late summer in Central Europe (Ferreira et al. 2001), affecting about 3–10% of European pollinotics (D’Amato et al. 1998).
Analyses
The pollen observation took place over a period of 33 weeks, from 11 March till 28 October 2014. The slides were changed weekly. A safranin-stained mixture of Mowiol®, glycerol, and water was applied on exposure slides and covered with a coverslip. The slides were examined under a light microscope at a magnification of ×400. Pollen types according to Beug (2004) were determined from 447 slides from 14 traps. Pollen analysis was derived by identifying and counting pollen in a whole circular area of 48 mm2 in the middle of the slide, which should guarantee the most isotropic place on the slide for the case of being affected by rainfall and wind.
Model pollen data for weeks with missing data was generated based upon multiple imputations. Interpolated data points were based on data from the trap corresponding to the missing data time frame. The model of complete conditional specification which relies on Markov Chain Monte Carlo (MCMC) methods was used.
The possible presence of residual spatial autocorrelation in pollen sedimentation were explored by calculating Moran’s I coefficient (Moran, 1950). Numerous criteria and methods are proposed to weight and analyze spatial dependences (Koenig 1998; Legendre and Legendre 1998; Fortin and Dale 2005). In preparation for the calculation of Moran’s I, we used the Delaunay Triangulation among the sampling sites to defining connections between neighboring sites. In our study, we calculated Kendall’s Tau B (Kendall 1955) coefficient in pair correlations of the seasonal pollen sedimentations of birch, grass, and mugwort between all possible combinations of traps. Every correlation coefficient was plotted in a correlogram against the distance between corresponding traps. The town-wide pollen season for each pollen type for the year 2014 is determined as the time in which at least 1% and maximal 95% of the pollen of a specific type occurred. Meteorological data (valid for the height of 2 m above ground level) originates from the meteorological stations Berlin-Tegel, Berlin-Tempelhof, and Berlin-Schönefeld of the German Weather Service (DWD).
To obtain basic information about the potential overestimation of pollen loads caused by pollen emissions of local sources, we inspected the relevant corresponding vegetation in a radius of 100 m of each trap—for Betula as the number of fertile trees; for Poaceae as the size of irregularly or uncut grass areas, estimated as areas of 100% cover-abundance; and for Artemisia as the number of individual fertile stems. Areas with monocultures of low pollen productive grass species (as for example Hordeum murinum or Bromus sterilis) were not included as well as areas with vegetation occurring on private land (Table 1).
The statistical and graphical analyses of the data were processed in Microsoft® Office Excel, CorelDRAW, Adobe Photoshop CS2, and SPSS 23.
Results
Birch (Betula)
The highest overall amounts of birch pollen were observed in traps situated closer to the outskirts of Berlin; the lowest amounts were found nearer the inner city (Fig. 2). The highest total deposition of birch pollen (“LENG”) was approximately 245% higher than in the trap with the lowest total sum (“BRÜCKE”). The seasonal courses of birch pollen sedimentation were evidently very similar for all 14 traps (Figs. 3 and 4a). The week of 1 to 8 April 2014 was the week with the maximum load of pollen, uniformly in all 14 stations (Fig. 3), with the highest pollen sedimentations in traps situated within the suburbs of Berlin, and the lowest counts in the traps situated close to the city center. During this week, the differences between the highest and the lowest counts totaled 283%.
a Total sum of Betula pollen during the whole investigation period from 11 March until 28 October 2014; the circle size corresponds to the observed pollen sum; the darker part of the circle color represents the number of fertile birch trees in a radius of 100 m around a trap: “SCHI” = 25 fertile trees = 100%. b Graphic illustration of weekly birch pollen sedimentations; traps are ordered by their distance from the geographical center of the city (N 52° 30.092 E 13° 24.137); imputed imputed model data in weeks with missing/lost slides
Grasses (Poaceae)
By far, the highest total of grass pollen sedimentation was detected in trap coded “SCHI” and the lowest in trap coded “CORR” (Fig. 5). The total difference between these two traps was 306%. The seasonal courses of grass pollen sedimentation between all 14 traps are less uniform than for birch (Figs. 4b, 5b, and 6). Differences occur among individual traps and varying week by week in the magnitudes of their differences. Over the course of the season, three peaks of grass pollen sedimentations are visibly represented by peak sedimentations for individual traps (Fig. 6).
a Total sum of Poaceae pollen during the whole investigation period from 11 March until 28 October 2014; the circle size corresponds to the observed pollen sum; the darker part of the circle color represents area of non-cut or irregularly cut grasses in a radius of 100 m around a trap: “SCHI” = 3020 m2 = 100%. b Graphic illustration of weekly grass pollen sedimentations; traps are ordered by their distance from the geographical center of the city (N 52° 30.092 E 13° 24.137); imputed imputed model data in weeks with missing/lost slides
Mugwort (Artemisia)
The highest total and peak deposition of mugwort pollen was measured in the inner city trap “BRÜCKE” and the lowest deposition in the inner city trap “KA-MA” (Fig. 7). The total difference between these traps amounted to 1962%.
a Total sum of Artemisia pollen during the whole investigation period from 11 March until 28 October 2014; the circle size corresponds to the observed pollen sum; the darker part of the circle color represents the number of fertile stems in a radius of 100 m around a trap: A100 = 520 fertile stems = 100%. b Graphic illustration of weekly mugwort pollen sedimentations; traps are ordered by their distance from the geographical center of the city (N 52° 30.092 E 13° 24.137); imputed imputed model data in weeks with missing/lost slides
The seasonal courses of mugwort pollen developed very disparately both spatially and temporarily for each individual trap (Figs. 4c, 7b, and 8). The week with the maximum pollen load differs between individual traps partially over a 5-week period (Fig. 8).
Statistical results
The Moran’s I coefficient measures the degree of autocorrelation within a single quantitative variable—here pollen sedimentation at the different traps. The positive value of I = 0.23 for the Morans I of birch pollen sedimentation between neighboring sampling sites exhibits a clustered pattern of pollen deposition and a weak spatial autocorrelation. With Morans I of −0.21 for mugwort and of I = −0.17 for grass pollen sedimentation, a more dispersed pattern is exhibited.
Plotting the relationship of the distances between each trap and the correlation (Kendall’s Tau B) of their seasonal pollen courses for each pollen type, high positive correlations between adjacent as well as more distant traps are revealed for birch (mostly around t = 0.8 significant at 95% level) equally for grass (mostly between t = 0.7 and t = 0.8 significant at 95% level). For mugwort, the seasonal courses of pollen deposition between the sampling sites are more uneven than those for grasses or birch and show only low, non-significant positive correlation (around t = 0.5). Rising distances between the traps are only of minor influence on the seasonal course of pollen sedimentations for all of the three pollen types. While the correlation coefficient for birch and grass show a slight downward trend with rising distances between traps, the coefficient for mugwort even show a general tendency for stronger correlations in more distant traps (Fig. 4).
Discussion
The use of the gravimetric method is an efficient way to collate information in regard to airborne pollen in the air and is quite often applied in scientific studies (Emberlin and Noris-Hill 1991; Piotrowska and Weryszko-Chmielewska 2003; Teranishi et al. 2006; Kishikawa et al. 2009; Crispen 2010; Obstová 2012; Tosunoğlu et al. 2015; Weinberger 2016).
In the “Introduction” section, we have listed publications in which pollen distribution within cities was observed by five or more pollen traps. Therefore, based on our knowledge, a study on screening the spatial distribution of pollen by 14 weekly changed gravimetric pollen traps, placed at uniform height, within a whole city, and during one pollen season has not yet been undertaken.
Birch (Betula)
There are almost no temporal differences in birch seasonal curves of all 14 traps (Figs. 2b and 4a). Birch pollen occurs abundantly over all stations, but certain spatial differences in distribution within the city are discernible (Fig. 2a).
Higher sedimentations of birch pollen were detected in suburban traps (18,663 pollen grains) in comparison to inner city traps (13,910 pollen grains) (Fig. 2b) which has also been observed in Poznan by Rodríguez-Rajo et al. (2010). The decreasing number of pollen in the direction toward the city center may be caused by a gradual sedimentation of pollen, released by sources outside the city as well as a filtration process caused by buildings and vegetation (Fig. 2a). Major nearby external sources of birch pollen are woodlands in the region Brandenburg around Berlin. Birch is the third most common tree species after pine (Pinus) and oak (Quercus), covering 3.5% of the total forest area of this region (Ministerium für Ländliche Entwicklung, Umwelt und Landwirtschaft des Landes Brandenburg, MLUL Brandenburg, 2015). Additionally, notable sources of locally existing birch pollen within the Berlin city area: birch trees occupy a share of 11% of the forest area and constitute 3% of regulated planted street trees (Senate Department for Urban Development and the Environment, SDUDE, 2013) and frequently occur inside the spontaneous vegetation of inner-city fallow areas or poorly maintained urban green zones (Kowarik et al. 2013). Possible influences of birch pollen sources within a city were discussed in Skjøth et al. (2008) resulting in the conception that urban local sources act as a significant source of birch pollen measured in the city Copenhagen, Denmark. Corden et al. (2000) highlighted parks and gardens in London (UK) as important pollen sources not only for the city but also for the surrounding region.
We found more birch pollen often in the abundant presence of birch trees within a 100-m radius of trap (Fig. 2a and Table 1). This was often related to traps that were closer to the city periphery, whereas the built-up areas were less dense and left more space for urban vegetation, such as birch trees. In our study, a possible influence of birch pollen spectrum by local sources of birch trees could be demonstrated by the two inner city traps “ZOO” and “SILBER” and by the suburban traps “J&W” and “BORN”. Despite the fact that the traps “ZOO” and “SILBER” were situated further away from external pollen sources outside the city, their birch pollen sedimentations were higher than in the traps “BORN” and “J&W”, which were located closer to the outside sources. As a significant source of birch pollen, 18 fertile birch trees located inside the 100-m radius of “SILBER” and 12 trees located inside the radius of “ZOO” can be taken into consideration. Less than 100 m from the traps, “J&W” and “BORN” were respectively only three and two fertile birch trees which could act as such local pollen sources. Therefore, it can be assumed that the pollen sedimentation in “SILBER” and “ZOO” were higher than in “J&W” and “BORN” because of the close proximity to a higher number of local birch pollen sources. Calculation of a local-scale concentration profile of birch pollen released from a source tree resulted in highest near-ground concentration within a distance of 50 to 100 m from the source Skjøth et al. (2013).
Grasses (Poaceae)
The explanation of grass pollen origins seems to be a difficult and challenging topic mainly due to the uniform morphology of grass pollen grains (expect of Cerealia). The Poaceae pollen type includes many different grass species (Beug 2004), with different dispersion characteristic of their pollen grains, with specific variances of flowering periods (Léon-Ruiz et al. 2011, Kmenta et al. 2016), with different pollen production rates (Prieto-Baena et al. 2003; Aboulaich et al. 2009), and in addition different ecology and distribution.
By far, the highest overall grass pollen sedimentation appeared in the trap “SCHI” (Figs. 5 and 6), probably as the direct result of the close proximity of the trap to the adjacent agricultural land (approx. 250 m southern) with permanent pastures and grasslands, which cover approx. 22% of the total agricultural land synonymously with ca. 11% of the total surface area of the region of Brandenburg surrounding Berlin (Amt für Statistik Berlin-Brandenburg 2015). Results of the studies from Emberlin and Noris-Hill (1991), Gonzalo-Garijo et al. (2006), or Obstová (2012) indicate that grass pollen inside the town originates mostly from areas outside of the town. In this case, the rate of pollen sedimentation should be gradually reduced in the direction toward the city center, which is not valid for our study where the grass pollen loads of the remaining 13 traps showed in contrast to “SCHI” a considerably lower and more even overall grass pollen sedimentation (Fig. 5a). In comparison to birch pollen, grass pollen grains are in general larger (Beug 2004). As a consequence, grass pollen shows an approximately two to four times higher settling velocity than birch pollen, resulting in a shorter presence of grass pollen grains in the air (Durham 1946; Skjøth et al. 2013). Considering the surface roughness which serves as a momentum sink for the atmospheric stream and which is in dense built up areas approximately ten times higher than those of open landscapes (Wieringa 1993) with crop fields and meadows may hinder the transport of higher quantities of grass pollen inside the city of Berlin. Therefore, local grass pollen sources within the city area have to have a certain influence and enrich the airborne grass pollen loads within the city of Berlin. The importance of urban sources have been reported from Poznan by Rodrígez-Rajo et al. (2010), and in addition, the results of a study by Skjøth et al. (2013) indicate that urban grass pollen sources can be expected to elevate grass pollen concentrations significantly within urban areas, albeit not exclusively, the relevant sources for the peak pollen concentrations in one observed pollen trap were found first at a distance of 20 to 60 km. During particular events, grass pollen may even cover distances of up to 100 km or more (Smith et al. 2005) but in general short distance transportations are more crucial for grass pollen (Skjøth et al. 2013).
The seasonal pollen curves of all 14 traps were similar to each other (Fig. 6) but were less clear than for birch (Fig. 4b). There were three peaks with high grass pollen sedimentation detected (Fig. 6) similar to those in studies from Basel in Switzerland (Leuschner et al. 2000); Bajadoz in Spain (Tormo et al. 2011); Vienna, Austria; or Berlin, Germany (Kmenta et al. 2016). The following considerations may provide possible explanations for these peaks: (1) every peak comprises the maxima of pollen sedimentation from a number of the 14 pollen traps (Fig. 6) and is possibly influenced by the individual grass species composition around each trap in which start, end, and length of the flowering seasons differs from species to species (Knox 1979; Subba Redi et al. 1988; Léon-Ruiz et al. 2011; Kmenta et al. 2016; Kmenta et al. in prep.). (2) Increased grass pollen loads may also be caused by a re-blooming of some grass species due to their ability of a continuous development of new flowers (Jäger et al. 2013). Observations by the first two authors of this paper in 2015 in Berlin revealed a re-blooming for example for Lolium perenne, Trisetum flavescens, Arrhentaherum elatius, and Poa annua. (3) Higher amounts of grass pollen during the first two peaks were connected with higher temperatures, a longer sunshine duration, and with lower relative air humidity and precipitation (Fig. 9). Similar results from other cities have been reported for grass pollen loads and for temperature by Puc and Puc (2004), Peternel et al. (2007), or Ianovici (2015); for sunshine duration by Ianovici (2015) or Simoleit et al. (2016); for relative humidity by Puc and Puc (2004) or Simoleit et al. (2016); and for rainfall by Peternel et al. (2007), Ianovici (2015), or Sabit et al. (2016). For the third peak, a higher wind speed seems to be the determining factor since lower air temperature, a shorter sunshine duration, and relative high air humidity did not reflect the meteorological conditions of the first two peaks. A predominate western wind during the week with this third peak (Fig. 9) was reflected in higher pollen counts especially in traps located in the western parts of Berlin—“ZOO”, “CORR”, “A100”, and “SCHI” (Fig. 6). Abundant amounts of pollen may be transported from outside into the city.
Weekly averaged meteorological data (from Tuesday 12.00 midday until Tuesday 12.00 midday) from the meteorological stations Berlin-Tegel, Berlin-Tempelhof, and Berlin-Schönefeld including one standard deviation and weekly grass pollen sum for all pollen traps (columns); the week from 17.06 until 24.06 shows specifically the grass pollen distribution divided in traps from west (n = 4), middle (n = 6), and east (n = 4). Gray circles show the weekly percentage of the prevailing wind direction (as black bars); big circle is equal to 100%, small circle to 50%
Mugwort (Artemisia)
Seasonal courses of mugwort pollen are still positively correlated at all sites (except the correlation between “J&W” and “KA-MA”), but with remarkable differences (Fig. 4c). Evident spatial variations in Artemisia pollen sedimentation have occurred throughout the city of Berlin (Fig. 7) following the direct influence of abundant local sources (Fig. 7a). The mostly early-morning release of mugwort pollen from plants—before air turbulences and convections are able to lift pollen grains in upper layers of air—together with the relatively high weight of Artemisia pollen grains seems to be the crucial factors of the higher occurrence of the pollen in closer proximity to its sources than further away from the sources (von Wahl and Puls 1991; Rantio-Lehtimäki et al. 1991; de Weger et al. 2013). During our study, a volumetric pollen trap was running adjacent to the gravimetric trap at “A100” (Simoleit et al. 2017). The highest mugwort concentration was detected there between 6 and 8 a.m. CET, which responds to the other already mentioned studies. In Spieksma et al. (2000), two traps installed at street-level and roof-level were compared: “After cutting mugwort plants in an area of 100 m around the street-level trap, the ratio dropped from 4.8 to 2.7, averagely for the season.”
There are some exceptions in our study. In the case of results from the traps “CORR” and “SILBER”, where vegetation with mugwort was presented within 100-m radius of the trap, lower amounts of pollen were detected and in case of “SCHI”, where only low abundance of plants occurred near the trap, higher pollen sedimentations appeared (Fig. 7a and Table 1). The aforementioned specifics of Artemisia plants and pollen and the presence of high buildings acting as barriers between traps in addition to the vegetation with mugwort plants could be an explanation for the lower pollen counts detected in the traps CORR and SILBER. For the relatively high counts of mugwort pollen in the trap SCHI, the close proximity of extended rural areas bordering fields and meadows just beyond the southern border of Berlin and no tall barriers, which could potentially block the pollen transmission from the rural areas to this sampling site, seems to be relevant.
Spatial autocorrelation
The seasonal synchrony in pollen loads of grass and even more of birch within the small geographical area of Berlin that are expressed in the most often close correlation between every trap of the network could be explained by the large influence of pollen sources outside of Berlin and the impact of meso (or large)-scale variability in pollen production, release, and dispersal. In order to investigate the effects of spatial autocorrelation, we calculated the coefficient Moran’s I for each pollen type. The relatively weak tendency for spatial patterns in birch pollen loads (I = 0.226) as well as the weak negative spatial autocorrelation in grass (I = −0.169) in contrast to the close positive non-parametric correlation of the seasonal courses which may be explained by small-scale variations in pollen sources or other explanatory variables throughout the city which can have certain effects of instability in broad-scale parameter estimates (Hawkins et al. 2007). The weak signs of a more dispersed pattern of mugwort pollen (I = −0.210) are related to the noticeable weaker positive correlations of the seasonal courses between the traps. It supports the relevance of local sources for the pollen distribution within a city together with the low influence of distant sources outside of Berlin for the pollen sedimentations in the city center.
Limitations
The reliability of pollen data is influenced by the kind of trap used. Gravimetric traps are affected by meteorological conditions, especially wind and rain and do not reflect the concentration of pollen in the air but rather weekly deposition rates. Turbulent eddies occurring at the edges of buildings or with passing cars will have an influence on the pollen counts and limit the general informative value. Therefore, it is recommended to verify the results by additional measurements at the same places during another pollen season or by parallel measurements with two adjacent samplers.
Trajectory analyses from pollen sources of the urban area of Berlin may help to understand, e.g., the enormous differences in the amount of mugwort pollen (>1900%) between the adjacent sampling sites “BRÜCKE” and “KAMA”. Essential elements in making calculations on this very local scale are in addition to the existence of detailed pollen data (e.g., as bi-hourly concentrations), a detailed mapping of vegetation and/or information about land usage and vegetation management (Skjøth et al. 2013). Our data, comprising pollen data on a weekly basis and vegetation data within a radius of 100 m do not appear to be sufficient for a full description of the differences of pollen sedimentations on such a fine scale. The first approach towards the goal of estimation of pollen concentrations on intra-urban scales have been made by Hjort et al. (2016). They combined in a statistical model multi-sourced geospatial data on different scales (from local- to regional- and long-distance transport scales) with daily grass pollen concentrations at 16 sites in Helsinki to predict intra-urban variability of grass pollen concentrations at street-level heights. However, even the number of 16 sampling sites is low to evaluate and calibrate a model for intra-urban scales (Hjort et al. 2011), especially when considering for example the immediate local influence of the presence and absence of mugwort plants within a city.
Postscript
The presented differences in pollen sedimentation throughout the city are in parts so high that they may influence the severity of clinical symptoms in pollen-allergic subjects. Based on our results, there should be an increased awareness of the negative impact and subsequent consequences for public health imposed by the potentially increasing pollen amount cause by climate changes and by a higher presence of high pollen-allergenic trees and plants which result from urban landscape gardening and insufficient care of the urban environment. Evidently, these human activities have an impact on the pollen composition and the amount of airborne pollen.
For the assessment of the distribution of pollen in towns, it is necessary to consider that every city has its own unique urban structure, size, type of urban environment, and characteristics such as architecture and/or vegetation within the city as well as in its surrounding areas, its specific meteorological conditions, and/or a combination of any of these factors. Therefore, in large cities such as Berlin, a certain number of suitable traps would be required in order to collate reliable information regarding pollen occurrence/concentrations as it is in the case of measurement stations for air pollutant monitoring within Berlin (16 stations) or within other towns.
In our study, we have observed less birch pollen nearer to the city center and more pollen on the outskirts of the city. Birch pollen information from an inner city trap will not serve as an adequate or true source of information for allergy sufferers living within the suburbs. As a consequence of this, a second trap should be placed within the suburban area, preferably in the southern part of the city, in which an earlier detection of pollen from the southern regions/countries, in which birch is already flowering, could be ensured. Another possible monitoring site could be placed near and/or in a large urban source area, e.g., parks with planted birch trees or areas with a higher occurrence of birch trees within spontaneous vegetation.
For grass pollen, the use of at least two pollen traps would be appropriate—within the city center and within the suburban/outskirts. Another possible location might be in and/or near an extended grass area within the city.
Appropriate information about airborne mugwort pollen cannot be determined for a wider area due to the high influence of local plant sources. However, a possible monitoring design would include one pollen trap in the city center as a reference and/or background station and another trap in an area where no mugwort plants are detectable, with a third trap in or nearby a location with a higher occurrence of mugwort. Taking this all into consideration, often higher occurrence of mugwort pollen at street-level height in comparison to a roof-top height should be considered (see the “Mugwort [Artemisia]” section).
Besides the necessity of monitoring allergenic pollen to provide allergy sufferers with reliable information on individual and personal allergens, another important action would be a closer management of areas where this allergenic species are known to occur. For example, in studies by Staffolani et al. (2011) or by Cariñanos and Casares-Porcel (2011), frequently allergenic plant species used as ornamental plants in urban environments with specification of their allergenic properties are discussed. A list of non-allergenic native trees and shrubs, suitable for a gradual replacement of highly allergenic woodland plant species within the city of Berlin as an example, is stated in Bergmann et al. (2012).
It should be kept in mind that our study and recommendations are performed on a scale of a population or part of a population. Pollen expositions of individual persons are usually constantly changing, due to human spatial mobility. To provide allergy sufferers with individualized pollen information, the use of a personal pollen sampler could make more accurate results available (Werchan et al. 2016), as well as a standardized pollen exposure setting, for example in a pollen chamber (Bergmann et al. 2015), will render more realistic data. Personalized methods are still in ongoing development and are not yet widely available. Therefore, it is crucial to improve the already existing pollen information systems.
Due to the existing high number of pollen allergy sufferers, in combination with the growing importance of cities as centers of population, the high socio-economic impact of allergies, potential increase of pollen amounts, and changes in pollen seasons caused by climate change should be paid special attention to further the research of pollen distribution within various urban environments.
While allergenic pollen has a remarkable impact on human health, human activities have a remarkable impact on the composition of airborne pollen, the production rate of the source plants together with the pollen load in the air as well as on the allergenicity of pollen. Despite these circumstances, the authors are not aware of any country worldwide which has implemented legislations regarding pollen as a part of regulated/monitored biogenic air pollution up to now.
Conclusions and practical implications of the study
Betula, Poaceae, and Artemisia airborne pollen show spatial and partly temporal variations within the city of Berlin. The highest amounts of Betula pollen were detected in traps closer to the outskirts of the city and/or in traps with higher number of birch trees nearby. In particular, high counts of Poaceae pollen were related to the proximity of extended outside sources in the form of meadows and fields with many different grass species. Higher depositions of Artemisia pollen were closely connected to the nearby presence of mugwort plants. Ratio and importance of external and local pollen sources distributed through the city are individual for each pollen type and for each specific region.
-
In large cities with a similar infrastructure and character to Berlin, significant differences in the distribution of airborne pollen are to be expected.
-
These differences would probably lead to different pollen load intensities on the population with a difference in the impact on the health of individual allergy sufferers.
-
Therefore, a similar network, as presented in this study and other studies dealing with spatial distribution of pollen within cities, is appropriate.
-
With respect to the high number of inhabitants suffering from pollen allergy, an interdisciplinary cooperation of urban planners, architects, botanists, palynologists, and allergologists should be considered when planning land use of towns and projects for urban green areas.
-
Birch trees should not be used as ornamental or street trees. Grasses should be cut regularly before flowering. Urban areas with mugwort vegetation should be maintained to prevent overgrowth and not become dilapidated.
-
The results of this study highlight the need to include allergenic pollen as a health-related factor of air quality monitoring regulations. As a result, more detailed and solid information regarding pollen exposure would be more widely available which in turn would allow for improved healthcare and improved individual information for allergy sufferers.
References
Aboulaich, N., Bouziane, H., Kadiri, M., et al. (2009). Pollen production in anemophilous species of the Poaceae Family in Tetouan (NW Marocco). Aerobiologia, 25(1), 27–38.
Alcázar, P., Cariñanos, P., De Castro, C., Guerra, F., Moreno, C., Domínguez-Vilches, E., & Galán, C. (2004). Airborne plane-tree (Platanus Hispanica) pollen distribution in the city of Córdoba, South-Western Spain, and possible implications on pollen allergy. Journal of Investigational Allergology and Clinical Immunology, 14(3), 238–243.
Amt für Statistik Berlin-Brandenburg (2015). Statistisches Jahrbuch 2015. Resource document. Amt für Statistik Berlin-Brandenburg. https://www.statistik-berlin-brandenburg.de/produkte/Jahrbuch/BE_Kap_2015.asp Accessed 20 February 2017.
Amt für Statistik Berlin-Brandenburg (2016). Statistisches Jahrbuch 2016. Resource document. Amt für Statistik Berlin-Brandenburg. https://www.statistik-berlin-brandenburg.de/produkte/Jahrbuch/BE_Kap_2016.asp Accessed 20 February 2017.
Arroba, D., Guido, M. A., Minale, P., et al. (2000). Airborne pollen in Genoa (NW-Italy): a comparison between two pollen-sampling stations. Aerobiologia, 16(2), 233–243.
Barnes, C., Pacheco, F., Landuyt, J., Hu, F., & Portnoy, J. (2001). Hourly variation of airborne ragweed pollen in Kansas City. Annals of Allergy, Asthma & Immunology, 86(2), 166–171.
Bergmann, K.-C., Sehlinger, T., Boelke, G. & Zuberbier, T. (2015). Clinical validation of a mobile allergen exposure chamber. Resource document. http://mcxperts.com/wp-content/uploads/2015/06/Clinical-Poster-EAACI-2015-small-_-reduced.pdf Accessed 11 May 2016.
Bergmann, K.-C., Zuberbier, T., Augustin, J., Mücke, H.-G., & Straff, W. (2012). Climate change and pollen allergy: cities and municipalities should take people suffering from pollen allergy into account when planting in public spaces. Allergo Journal, 21(2), 103–107.
Beug, H. J. (2004). Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete. München: Verlag Dr. Friedrich Pfeil.
Cakmak, S., Dales, R. E., & Coates, R. (2012). Does air pollution increase the effect of aeroallergens on hospitalization for asthma? Journal of Allergy and Clinical Immunology, 129(1), 228–231.
Cariñanos, P., Adinolfi, C., Díaz de la Guardia, C., De Linares, C., & Casares-Porcel, M. (2016). Characterization of allergen emission sources in urban areas. Journal of Environmental Quality, 45(1), 244–252.
Cariñanos, P., & Casares-Porcel, M. (2011). Urban green zones and related pollen allergies: a review. Guidelines for designing spaces of low allergy impact. Landscape and Urban Planning, 101(3), 205–214.
Cariñanos, P., S’nchez-Mesa, J. A., Prieto-Baena, J. C., et al. (2002). Pollen allergy related to the area of residence in the city of Córdoba, south-west Spain. Journal of Environmental Monitoring, 4(5), 734–739.
Celenk, S., Bicakci, A., Tamay, Z., et al. (2010). Airborne pollen in European and Asian parts of Istanbul. Environmental Monitoring and Assessment, 164(1), 391–402.
Corden, J., Millington, W., Bailey, J., et al. (2000). UK regional variations in Betula pollen (1993–1997). Aerobiologia, 16(2), 227–232.
Crispen, K. L., Gillespie, D. N., Weiler, E. C., Noonan, C. W., Hamilton, R. F., & Ward, T. J. (2010). A comparison of 1978 and 2006 peak pollen seasons and sampling methods in Missoula, Montana. Grana, 49(2), 128–133.
Dadvand, P., Villanueva, C. M., Font-Ribera, L., et al. (2014). Risks and benefits of green spaces for children: a cross-sectional study of associations with sedentary behavior, obesity, asthma, and allergy. Environmental Health Perspectives. doi:10.1289/ehp.1308038.
D'Amato, G., Bergmann, K. C., Cecchi, L., et al. (2014). Climate change and air pollution. Effects on pollen allergy and other allergic respiratory diseases. Allergo Journal International, 23(132), 17–23.
D'Amato, G., Cecchi, L., Bonini, S., Nunes, C., Annesi-Maesano, I., Behrendt, H., Liccardi, G., Popov, T., & van Cauwenberge, P. (2007). Allergenic pollen and pollen allergy in Europe. Allergy, 62(9), 976–990.
D'Amato, G., Spieksma, F. T. M., Liccardi, G., Jäger, S., Russo, M., Kontou-Fili, K., Nikkels, H., Wüthrich, B., & Bonini, S. (1998). Pollen-related allergy in Europe. Allergy, 53(6), 567–578.
de Weger, L. A., Bergmann, K. C., Rantio-Lehtimäki, A., et al. (2013). Impact of pollen. In M. Sofiev & K.-C. Bergmann (Eds.), Allergenic pollen: a review of the production, release, distribution and health impacts (pp. 161–215). Dordrecht: Springer.
Durham, O. C. (1946). The volumetric incidence of atmospheric allergens. IV. A proposed standard method of gravity sampling, counting, and volumetric interpolation of results. Journal of Allergy, 17(2), 79–86.
Eckl-Dorna, J., Klein, B., Reichenauer, T. G., Niederberger, V., & Valenta, R. (2010). Exposure of rye (Secale cereale) to elevated ozone levels increases the allergen content in pollen. Journal of Allergy and Clinical Immunology, 126(6), 1315–1317.
Emberlin, J., & Norris-Hill, J. (1991). Spatial variation of pollen deposition in north London. Grana, 30(1), 190–195.
Feo Brito, F., Mur Gimeno, P., Martínez, C., Tobías, A., Suárez, L., Guerra, F., Borja, J. M., & Alonso, A. M. (2007). Air pollution and seasonal asthma during the pollen season. A cohort study in Puertollano and Ciudad Real (Spain). Allergy, 62(10), 1152–1157.
Fernández-Rodríguez, S., Tormo-Molina, R., Maya-Manzano, J. M., Silva-Palacios, I., & Gonzalo-Garijo, A. (2014). Comparative study of the effect of distance on the daily and hourly pollen counts in a city in the south-western Iberian peninsula. Aerobiologia, 30(2), 173–187.
Ferreira, F., Himly, M., Hebenstreit, D., Dabrowska, M., Vollmann, U., Kraft, D., Obermeyer, G., & Richter, K. (2001). Isolation and characterization of cDNA clones coding for mugwort (Artemisia vulgaris) pollen allergens. International Archives of Allergy and Immunology, 124(1–3), 77–79.
Fornaciari, M., Bricchi, E., Frenguelli, G., & Romano, B. (1996). The results of 2-year pollen monitoring of an urban network in Perugia, Central Italy. Aerobiologia, 12(1), 219–227.
Fortin, M.-J., & Dale, M. R. T. (2005). Spatial analysis: a guide for ecologists. Cambridge: Cambridge University Press.
Gonzalo-Garijo, M. A., Tormo-Molina, R., Muñoz-Rodríguez, A. F., & Silva-Palacios, I. (2006). Differences in the spatial distribution of airborne pollen concentrations at different urban locations within a city. Journal of Investigational Allergology and Clinical Immunology, 16(1), 37–43.
Hawkins, B. A., Diniz-Filho, J. A. F., Mauricio Bini, L., De Marco, P., & Blackburn, T. M. (2007). Red herrings revisited: spatial autocorrelation and parameter estimation in geographical ecology. Ecography, 30(3), 375–384.
Hirst, J. M. (1952). An automatic volumetric spore trap. Annals of Applied Biology, 39(2), 257–265.
Hjort, J., Hugg, T. T., Antikainen, H., et al. (2016). Fine-scale exposure to allergenic pollen in the urban environment: evaluation of land use regression approach. Environmental Health Perspectives, 124(5), 619–626.
Hjort, J., Suomi, J., & Käyhkö, J. (2011). Spatial prediction of urban–rural temperatures using statistical methods. Theoretical and Applied Climatology, 106(1), 139–152.
Ianovici, N. (2015). Relation between Poaceae pollen concentrations and meteorological factors during 2000–2010 in Timisoara, Romania. Acta Agrobotanica, 68(4), 373–381.
Ishibashi, Y., Ohno, H., Oh-ishi, S., Matsuoka, T., Kizaki, T., & Yoshizumi, K. (2008). Characterization of pollen dispersion in the neighborhood of Tokyo, Japan in the spring of 2005 and 2006. International Journal of Environmental Research and Public Health, 5(1), 76–85.
Jäger, E. J., Müller, F., Ritz, C. M., Welk, E., & Wesche, K. (Eds.). (2013). Rothmaler: Exkursionsflora von Deutschland, Gefäßpflanzen: Atlasband (12. Auflage ed.). Berlin & Heidelberg: Springer Verlag.
Katelaris, C. H., Burke, T. V., & Byth, K. (2004). Spatial variability in the pollen count in Sydney, Australia: can one sampling site accurately reflect the pollen count for a region? Annals of Allergy. Asthma & Immunology, 93(2), 131–136.
Katz, D. S. W., & Carey, T. S. (2014). Heterogeneity in ragweed pollen exposure is determined by plant composition at small spatial scales. Science of the Total Environment, 485-486, 435–440.
Kendall, M. G. (1955). Rank correlation methods. New York: Hafner Publishing Co..
Kishikawa, R., Sahashi, N., Saitoh, A., Kotoh, E., Shimoda, T., Shoji, S., Akiyama, K., & Nishima, S. (2009). Japanese cedar airborne pollen monitoring by Durham’s and Burkard samplers in Japan—estimation of the usefulness of Durham’s sampler on Japanese cedar pollinosis. Global Environmental Research, 13(1), 55–62.
Kleine-Tebbe, J., & Davis, J. M. (2014). Grass pollen allergens. In C. A. Akdis & I. Agache (Eds.), Global atlas of allergy (pp. 22–26). Zurich: European Academy of Allergy and Clinical Immunology (EAACI).
Kmenta, M., Bastl, K., Bergman, K.-C., et al. (2016). Grass pollen season 2015 in Vienna (Austria), Berlin (Germany) and Turku (Finland): Spatial and temporal variation in pollination of different grass species and their impact on pollen allergy sufferers. Allergy, 71(Suppl. 102), 217.
Knox, R. B. (1979). Pollen and allergy. In Studies in biology no. 107. London: Edward Arnold.
Koenig, W. (1998). Spatial autocorrelation of ecological phenomena. Trees, 14(1), 22–26.
Kottek, M., Grieser, J., Beck, C., Rudolf, B., & Rubel, F. (2006). World map of the Köppen–Geiger climate classification updated. Meteorologische Zeitschrift, 15(3), 259–263.
Kowarik, I., von der Lippe, M., & Cierjacks, A. (2013). Prevalence of alien versus native species of woody plants in Berlin differs between habitats and at different scales. Preslia, 85(2), 113–132.
Langen, U., Schmitz, R., & Steppuhn, H. (2013). Häufigkeit allergischer Erkrankungen in Deutschland - Ergebnisse der Studie zur Gesundheit Erwachsener in Deutschland (DEGS1). Bundesgesundheitsblatt, 56(5/6), 698–706.
Legendre, P., & Legendre, L. (1998). Numerical ecology. Amsterdam: Elsevier.
León-Ruiz, E., Alcázar, P., Domínguez-Vilches, E., & Galán, C. (2011). Study of Poaceae phenology in a Mediterranean climate. Which species contribute most to airborne pollen counts? Aerobiologia, 27(1), 37–50.
Leuschner, R. M., Christen, H., Jordan, P., & Vonthein, R. (2000). 30 years of studies of grass pollen in Basel (Switzerland). Aerobiologia, 16(3–4), 381–391.
McPherson, E. G., Simpson, J. R., Xiao, Q., & Wu, C. (2011). Million tress Los Angeles canopy cover and benefit assessment. Landscape and Urban Planning, 99(1), 40–50.
Ministerium für Ländliche Entwicklung, Umwelt und Landwirtschaft des Landes Brandenburg (MLUL Brandenburg) 2015. Ergebnisse der ersten landesweiten Waldinventur in Brandenburg. Resource document. MLUL Brandenburg. http://forst.brandenburg.de/cms/media.php/lbm1.a.3310.de/inv2015.pdf Accessed 17 April 2016.
Moran, P. A. P. (1950). Notes on continuous stochastic phenomena. Biometrika, 37(1–2), 17–23.
Mücke, H.-G., Wagener, S., Werchan, M., & Bergmann, K.-C. (2014). Measurements of particulate matter and pollen in the city of Berlin. Urban Climate, 10(4), 621–629.
Nowak, M., Szymańska, A., & Grewling, Ł. (2012). Allergenic risk zones of plane tree pollen (Platanus sp.) in Poznan. Postępy Dermatologii i Alergologii, 29(3), 156–160.
Obstová, B. (2012). Pylová spektra v ovzduší různých typů městské zástavby: sezonní dynamika a význam pro alergologii. Resource document. Masaryk University. http://is.muni.cz/th/223092/prif_m/ Accessed 19 February 2017.
Pawankar, R., Canonica, G. W., Holgate, S. T., Lockey, R. F. & Blaiss, M. S. (2013). World Allergy Organization (WAO) white book on allergy: Update 2013. Resource document. World Allergy Organization. http://www.worldallergy.org/UserFiles/file/WhiteBook2-2013-v8.pdf Accessed 19 February 2017.
Peternel, R., Milanović, S. M., Hrga, I., Mileta, T., & Culig, J. (2007). Incidence of Betulaceae pollen and pollinosis in Zagreb, Croatia, 2002-2005. Annals of Agricultural and Environmental Medicine, 14(1), 87–91.
Piotrowska, K., & Weryszko-Chmielewska, E. (2003). Pollen count of selected taxa in the atmosphere of Lublin using two monitoring methods. Annals of Agricultural and Environmental Medicine, 10(1), 79–85.
Prieto-Baena, J. C., Hidalgo, P. J., Domínguez, E., & Galán, C. (2003). Pollen production in the Poaceae Family. Grana, 42(3), 153–160.
Puc, M., & Puc, M. I. (2004). Allergenic airborne grass pollen in Szczecin, Poland. Annals of Agricultural and Environmental Medicine, 11(2), 237–244.
Rantio-Lehtimäki, A., Koivikko, A., Kupias, R., Mäkinen, Y., & Pohjola, A. (1991). Significance of sampling height of airborne particles for aerobiological information. Allergy, 46(1), 68–76.
Raynor, G. S., Ogden, E. C., & Hayes, J. V. (1975). Spatial variabilty in airborne pollen concentrations. Journal of Allergy and Clinical Immunology, 55(3), 195–202.
Rodríguez-Rajo, F. J., Fdez-Sevilla, D., Stach, A., & Jato, V. (2010). Assessment between pollen seasons in areas with different urbanization level related to local vegetation sources and differences in allergen exposure. Aerobiologia, 26(1), 1–14.
Sabit, M., Ramos, J. D., Alejandro, G. J., & Galán, C. (2016). Seasonal distribution of airborne pollen in Manila, Philippines, and the effect of meteorological factors to its daily concentrations. Aerobiologia, 32(3), 375–383.
Schmitz, R., Atzpodien, K., & Schlaud, M. (2012). Prevalence and risk factors of atopic diseases in German children and adolescents. Pediatric Allergy and Immunology, 23(2), 716–723.
Selmi, W., Weber, C., Rivière, E., Blond, N., Mehdi, L., & Nowak, D. (2016). Air pollution removal by trees in public green spaces in Strasbourg city, France. Urban Forestry & Urban Greening. doi:10.1016/j.ufug.2016.04.010.
Senate Department for Urban Development and the Environment (SDUDE) (2013). Less pollutants. More quality of life. The air quality plan 2011–2017 of Berlin. Resource document. Senate Department for Urban Development and the Environment. http://www.stadtentwicklung.berlin.de/umwelt/stadtgruen/stadtbaeume/en/daten_fakten/uebersichten/index.shtml Accessed 13 April 2016.
Shapiro, R. S., & Rooks, R. (1951). The accuracy of the reported ragweed pollen count as a measure of the actual pollen exposure of individuals in that community. Journal of Allergy, 22(5), 450–460.
Simoleit, A., Gauger, U., Mücke, H.-G., Werchan, M., Obstová, B., Zuberbier, T., & Bergmann, K.-C. (2016). Intradiurnal patterns of allergenic airborne pollen near a city motorway in Berlin, Germany. Aerobiologia, 32(2), 199–209.
Simoleit, A., Werchan, M., Werchan, B., Mücke, H.-G., Gauger, U., Zuberbier, T., & Bergmann, K.-C. (2017). Birch, grass, and mugwort pollen concentrations and intradiurnal patterns at two different urban sites in Berlin. Germany: Allergo Journal International.
Skjøth, C. A., Sommer, J., Brandt, J., Hvidberg, M., Geels, C., Hansen, K., Hertel, O., Frohn, L., & Christensen, J. (2008). Copenhagen—a significant source of birch (Betula) pollen? International Journal of Biometeorology, 52(6), 453–462.
Skjøth, C. A., Ørby, P. V., Becker, T., et al. (2013). Identifying urban sources as cause of elevated grass pollen concentrations using GIS and remote sensing. Biogeosciences, 10(1), 541–554.
Smith, M., Emberlin, J., & Kress, A. (2005). Examining high magnitude grass pollen episodes at Worcester, United Kingdom, using backtrajectory analysis. Aerobiologia, 21(2), 85–94.
Spieksma, F. T. M., van Noort, P., & Nikkels, H. (2000). Influence of nearby stands of Artemisia on street-level versus roof-top-level ratio’s of airborne pollen quantities. Aerobiologia, 16(1), 21–24.
Sousa, R., Duque, L., Duarte, A. J., Gomes, C. R., Ribeiro, H., Cruz, A., Esteves da Silva, J. C., & Abreu, I. (2012). In vitro exposure of Acer negundo pollen to atmospheric levels of SO2 and NO2: effects on allergenicity and germination. Environmental Science & Technology, 46(4), 2406–2412.
Staffolani, L., Velasco-Jiménez, M. J., Galán, C., & Hruska, K. (2011). Allergenicity of the ornamental urban flora: ecological and aerobiological analyses in Córdoba (Spain) and Ascoli Piceno (Italy). Aerobiologia, 27(3), 239–246.
Steerenberg, P. A., Withagen, C. E., Dormans, J. A., van Dalen, W. J., van Loveren, H., & Casee, F. R. (2003). Adjuvant activity of various diesel exhaust and ambient particles in two allergic models. Journal of Toxicology and Environmental Health, 66(15), 1421–1439.
Subba Reddi, C., Reddi, N. S., & Atluri Janaki, B. (1988). Circadian patterns of pollen release in some species of Poaceae. Review of Palaeobotany and Palynology, 55(1–2), 11–42.
Teranishi, H., Katoh, T., Kenda, K., & Hayashi, S. (2006). Global warming and the earlier start of the Japanese-cedar (Cryptomeria japonica) pollen season in Toyama, Japan. Aerobiologia, 22(2), 91–95.
Tormo, R., Silva, I., Gonzalo, A., Moreno, A., Pérez, R., & Fernández, S. (2011). Phenological records as a complement to aerobiological data. International Journal of Biometeorology, 55(1), 51–65.
Tosunoğlu, A., Babayiğit, S., & Bıçakçı, A. (2015). Aeropalynological survey in Büyükorhan, Bursa. Turkish Journal of Botany, 39(1), 40–47.
United Nations, Department of Economic and Social Affairs, Population Division (2015). World Urbanization Prospects: The 2014 Revision, (ST/ESA/SER.A/366). Resource Document. United Nations. https://esa.un.org/unpd/wup/Publications/Files/WUP2014-Report.pdf Accessed 19 February 2017.
Velasco-Jiménez, M. J., Alcázar, P., Domínguez-Vilches, E., & Galán, C. (2013). Comparative study of airborne pollen counts located in different areas of the city of Córdoba (south-western Spain). Aerobiologia, 29(1), 113–120.
von Wahl, P. G., & Puls, K. E. (1991). Pollenemission und Pollenflug von Kräuterpollen. Grana, 30(1), 260–264.
Weinberger, K. R., Kinney, P. L., Robinson, G. S., Sheehan, D., Kheirbek, I., Matte, T. D., & Lovasi, G. S. (2016). Levels and determinants of tree pollen in New York City. Journal of Exposure Science and Environmental Epidemiology. doi:10.1038/jes.2016.72.
Werchan, M., Sehlinger, T., Werchan, B. & Bergmann, K. C. (2016). Klein und handlich—Das persönliche Pollenmessgerät. 11. Deutscher Allergiekongress, Berlin, Germany: ePoster P47.
Wieringa, J. (1993). Representative roughness parameters for homogeneous terrain. Boundary-Layer Meteorology, 6(4), 323–363.
Ziello, C., Sparks, T. H., Estrella, N., Belmonte, J., Bergmann, K.-C., Bucher, E., et al. (2012). Changes to airborne pollen counts across Europe. PloS One. doi:10.1371/journal.pone.0034076.
Ziska, L. H., Gebhard, D. E., Frenz, D. A., Faulkner, S., & Singer, B. D. (2003). Cities as harbingers of climate change: common ragweed, urbanization, and public health. Journal of Allergy and Clinical Immunology, 111(2), 290–295.
Zuberbier, T., Lötvall, J., Simoens, S., Subramanian, S. V., & Church, M. K. (2014). Economic burden of inadequate management of allergic diseases in the European Union: a GA2LEN review. Allergy, 69(10), 1275–1279.
Acknowledgments
The main author thanks and dedicates this paper to her dear father, geologist RNDr. Petr Obst. The authors would like to thank the anonymous reviewers for their valuable comments. The study was supported by the German Environment Agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Werchan, B., Werchan, M., Mücke, HG. et al. Spatial distribution of allergenic pollen through a large metropolitan area. Environ Monit Assess 189, 169 (2017). https://doi.org/10.1007/s10661-017-5876-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-5876-8